Abstract
There is a significant correlation between the degree of tumor differentiation and the survival of patients with gastric cancers. In this report, we compared proteomic differences between poorly differentiated gastric adenocarcinoma tissues and well-differentiated gastric adenocarcinoma tissues in order to identify differentiation-related proteins that may be closely correlated with differentiation of gastric cancer pathogenesis. We identified 7 proteins, of which calreticulin precursor, tapasinERP57 heterodimer, pyruvate kinase isozymes M1/M2 isoform M2, class Pi glutathione S-transferase, and chain A crystal structure of human enolase 1 were upregulated in poorly differentiated gastric adenocarcinoma compared with well-differentiated gastric adenocarcinoma, while myosin-11 isoform SM2A and actin alpha cardiac were downregulated. Two of them, pyruvate kinase isozymes M1/M2 isoform M2 and enolase 1 are enzymes involved in glycolytic pathway. The upregulation of pyruvate kinase isozymes M1/M2 isoform M2 and enolase 1 in poorly differentiated gastric adenocarcinoma was confirmed by Western blotting and immunohistochemistry. Furthermore, we observed 107 cases with gastric adenocarcinoma and found that the high expression of pyruvate kinase isozymes M1/M2 isoform M2 and enolase 1 correlates with tumor size (P = .0001 and P = .0017, respectively), depth of invasion (P = .0024 and P = .0261, respectively), and poor prognosis of patients. In conclusion, with this proteomic analysis, pyruvate kinase isozymes M1/M2 isoform M2 and enolase 1 were identified upregulated in poorly differentiated gastric adenocarcinoma comparing with well-differentiated gastric adenocarcinoma. The expression level of pyruvate kinase isozymes M1/M2 isoform M2 and enolase 1 was significantly correlated with overall survival. Some of them would be differentiation-related cancer biomarkers and are associated with tumor metastasis, invasion, and prognosis.
Keywords
Introduction
Gastric cancer is one of the 4 most common cancers and the second cancer-related cause of death in the world, which is only surpassed by lung cancer. World Health Organization (WHO) estimated that there were 1 400 000 new gastric cancer cases and 820 000 cancer deaths occurred in 2012. More than 40% of newly diagnosed cases and deaths caused by gastric cancer occurred in China. 1
Current experimental data strongly support that there is a significant correlation between the degree of tumor differentiation and the survival rate of patients with gastric cancers. The 5-year survival rate for patients with poorly differentiated carcinomas is less than 10%, while patients with well-differentiated carcinomas have a better chance to be cured, with a 5-year survival rate of over 90%. 2 -4
It has been shown that poorly differentiated tumors show preferential overexpression of genes normally enriched in embryonic stem cells and overexpression of activation targets of Nanog, OCT4, SOX2, and c-Myc. 5 However, genomics-based techniques have a major limitation that the detection of high levels of messenger RNA (mRNA) transcripts of a specific gene may not necessarily mean that high levels of the corresponding protein product will be present. Now, proteomics-based techniques can help us find differentiation-related cancer biomarkers in protein level.
Several groups have carried out proteomics studies on gastric carcinoma, and a number of proteins with altered expression levels have been identified. 6 -8 Most of them reflect the distinction between tumor and nontumor tissues. In this article, we try to look for proteins that are related to tumor differentiation. First, patients with gastric cancer were classified into poorly differentiated group and a well-differentiated group based on the extension of glandular differentiation in the tumor mass and cell anomalies. Then, proteins that are expressed in poorly differentiated gastric adenocarcinoma (PDGA) tissues and well-differentiated gastric adenocarcinoma (WDGA) were identified and some of them were confirmed by Western blotting and immunohistochemistry. These proteins might be closely correlated with the differentiation of gastric cancer and will provide useful information for us to understand the mechanisms of tumor development.
Materials and Methods
Materials
The power supply, the 2-dimensional (2D) gel electrophoresis (2DE) system (PROTEAN IEF cell, PROTEAN II Xi cell), the Trans-Blot Cell, immobilized pH gradient (IPG) strip (pH 3-10, nonlinear, 17 cm), Bio-Lyte (pH 3-10), urea, CHAPS, sodium dodecyl sulfate (SDS), acrylamide, N,N′-methylenebisacrylamide, glycine, and Tris-base were purchased from Bio-Rad (Richmond, California). Iodoacetamide was purchased from Merck (Darmstadt, Germany). Thiourea was purchased from Sigma (St Louis, Missouri). Complete Protease Inhibitor Cocktail (EDTA free) was purchased from Roche (Mannheim, Germany). Ammonium persulfate and dithiothreitol (DTT) were purchased from Sangon (Shanghai, China).
Tissue Specimens
Primary gastric adenocarcinomas were collected on gastrectomy and provided by the Department of Surgical Oncology, the First Hospital of Lanzhou University, Lanzhou, China, between June 2009 and May 2010. The research was approved by the ethical committee of the First Hospital of Lanzhou University. Six patients, 5 males and 1 female, with an average age of 56 years old (range, 34-77 years), who provided the clinical samples, were involved in this experiment. None of the patients received antineoplastic anticancer therapy prior to surgery. Detailed information, such as age, sex, tumor location, histodifferentiation, and surgical–pathologic stage, of the patients is listed in Table 1. Resections were examined by a pathologist, and the margin tissue samples were replicated from noncancerous regions, 5 cm apart from the primary tumor, and were verified to be without cancer cells by pathology. The degree of cellular differentiation was classified according to the WHO classifications. 9 All tissues were immediately snap-frozen in individual vials using liquid nitrogen after necrotic tissues were excluded soon as possible and were washed with isotonic Na chloride 3 times. Frozen specimens were stored in a deep freezer (at −80°C) before use.
Clinical and Pathological Characters of 6 Patients With Gastric Cancer.

Abbreviations: F, female; M, male.
Preparation of Tissue Protein Samples
For proteomic analysis, 100 mg of tissue sample was cut into pieces to about 2 mm3, homogenized in liquid nitrogen, and lysed in 0.5 mL lysis buffer (8 mol/L urea, 2 mol/L thiourea, 4% CHAPS, 65 mmol/L DTT, 0.2% Bio-Lyte 3/10 ampholyte, and 40 mmol/L Tris-HCl, pH 7.2) together with 4% complete protease inhibitor cocktail (EDTA free). Samples were then kept on ice, sonicated in 60 cycles each consisting of 3-second sonication followed by a 7-second break, finally held for 40 minutes on ice with occasional vortex mixing. Insoluble materials were removed by centrifugation at 21 000 rpm (approx 62 000g) for 1 hour at 4°C. Total protein amount in each sample was assayed using the technique described by Bradford. 10 Individual sample concentrations were adjusted by dilution in the same lysis buffer. Samples were either applied immediately to IEF or stored at −80°C in aliquots before analysis.
Two-Dimensional Electrophoresis
The first-dimensional gel separation was carried out with 17 cm IPG strips (pH 3-10, nonlinear) following the manufacturers’ protocol with minor modifications. Samples containing up to 800 μg protein were diluted to 350 μL with rehydration solution (7 mol/L urea, 2 mol/L thiourea, 4% CHAPS, 65 mmol/L DTT, 0.2%, w/v Bio-Lyte, pH 3-10) and applied to strips for 16 hours rehydration at 50 V. Proteins were focused succeedingly for 30 minutes at 250 V, 30 minutes at 500 V, 2 hours at 1000 V, then a gradient was applied from 1000 to 10 000 V in 5 hours, and focusing was continued at 10 000 V for 6.5 hours to give a total of 95 kVh on an IPGphor. All IEF steps were carried out at 20°C. Once IEF was completed, the strips were equilibrated in equilibration buffer (37.5 mmol/L Tris-HCl, pH 8.8, 6 mol/L urea, 20% glycerol, 2% SDS, and 2% DTT) for 15 minutes followed by the same buffer containing 2.5% iodoacetamide instead of DTT for another 15 minutes. Equilibrated IPG strips were transferred into 12% uniform polyacrylamide gels and then were run in PROTEAN II Xi cell tank at a voltage setting of 80 V per gel for the initial 30 minutes and 220 V for 8 hours thereafter. The second-dimensional SDS polyacrylamide gel electrophoresis (SDS-PAGE) was developed until the bromophenol blue dye marker had reached the bottom of the gel. The gels were visualized using the Coomassie Brilliant Blue G250 staining method. After the staining, 2D gels were imaged using Powerlook III (UMAX, Fremont, California), and the images were analyzed using PDQuest 8.0 (Bio-Rad).
Protein Identification
Protein spots of interest were cut out manually from the gel and washed 3 times with ultrapure water and were put into siliconized microcentrifuge tubes. In-gel digestion, matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF-MS) or MS/MS and database searching were performed by Shanghai Boyuan Biological Technology Co Ltd.
Western Blotting
Total cell lysates from tumors were prepared, and the protein concentration was determined by the method described by Bradford. 10 Western blot analysis was performed as described previously. 11 Briefly, equal amounts of protein (30 μg) per well were fractioned via SDS-PAGE on a 10% gel and electrophoretically transferred onto polyvinylidene difluoride membrane by wet blotting at 80 mA for 8 to 12 hours. Primary antibodies were diluted in Tris-buffered saline and Tween 20 with 5% nonfat dry milk at the following concentrations: rabbit antipyruvate kinase type M2 (anti-PKM2, BS1259; Bios) diluted 1:300, rabbit anti-pyruvate kinase type R (anti-PKLR, GTX107546 S; Gene Tex) diluted 1:1000, rabbit antienolase1 (anti-ENO1, GTX101803 S; Gene Tex) diluted 1:1000, ENO2/neuron-specific enolase (NSE; BS1259; Bioworld) diluted 1:400, rabbit anti-aldolase A (BS1912; Bioworld) diluted 1:500, and rabbit antiglyceraldehyde 3-phosphate dehydrogenase (anti-GAPDH; AB-P-R001; GOOD HERE) diluted 1:500. After blocking in 5% nonfat dry milk in TBS containing 0.05% Tween 20 for 2 hours, membranes were incubated with primary antibodies at 4°C overnight with gentle rocking and were then washed. Detection was performed with horseradish peroxidase linked secondary antibodies using enhanced chemiluminescence detection kit. The membranes were stripped and reprobed with GAPDH to monitor for equal loading.
Immunohistochemistry
There are 107 paraffin-embedded tissue samples from patients with gastric cancer collected. These patients had gastric adenocarcinoma and underwent surgery at the First Hospital of Lanzhou University from January 2010 to December 2012. None of these patients received chemotherapy or radiation therapy before surgery (Table 2).
Characters of 107 Patients With Gastric Cancer.

Paraffin-embedded tissues were cut into 4 μm sections, deparaffinized with xylene, and washed with PBS. After blocking with 1% goat serum in PBS for 15 minutes, 10 mmol/L (pH 6.0) citrate buffer was added to the tissue sections and heated in microwave for 3 to 5 minutes, followed by incubation overnight at 4°C with mouse ENO1 antibody (Santa Cruz, California; dilution: 1:1000) or rabbit anti-M2-PK (Cell Signaling, Giessen, German; dilution: 1:100). Under the room temperature after washing with PBS, sections were incubated with secondary biotinylated antibody for 30 minutes at 37°C. The sections were then incubated with streptavidin–peroxidase (ZYMED), followed by incubation with 3,3′-diaminobenzidine (ZYMED), and were counterstained for 2 minutes in hematoxylin, separated using hydrochloric acid. Sections were then dehydrated in graded ethanols and followed with neutral resin. The image system comprised a camera (Olympus BX51, Japan). The ENO1 and PKM2 was identified by digital image analysis with the Image Pro-Plus 6.0 software. To quantify the areas of PKM2 and ENO1, 6 random fields per slide were selected to measure mean optical density. SPSS 17.0 software was used for the statistical analysis. P < .05 was considered as statistically significant (Figure 1).

A, Kaplan-Meier survival analysis of pyruvate kinase type M2 (PKM2) expression in 107 patients with gastric cancer. B, Kaplan-Meier survival analysis of enolase 1 (ENO1) expression in 107 patients with gastric cancer.
Results
The 2DE Profiling of Differentially Expressed Proteins in Gastric Cancer
The 2DE using nonlinear IPG ranging from pH 3 to 10 was performed to separate the proteins extracted from PDGA and WDGA. Figure 2 shows the representative proteome profiling profiles for PDGA (A) versus WDGA (B) tissues. Approximately 600 to 700 protein spots were detected by CBB G-250 staining in a single 2DE gel. Through 2DE profile (Figure 2), 7 differentially expressed proteins, including 5 upregulated and 2 downregulated in PDGA samples compared with WDGA, were defined as statistically meaningful (P < .05) based on the following 2 criteria: (1) intensity alterations >2.0-fold and (2) recurrence more than 3 times in the 3 pairs of samples examined. These proteins and their corresponding spots are boxed and enlarged in the surrounding area (Figure 3).

Representation of 2-dimensional gel electrophoresis (2DE) analysis of proteome profiles of gastric cancer. Total protein (800 μg) extracted from (A) PDGA tissues and (B) WDGA tissues was separated on nonlinear strips with a pH range of 3 to 10, followed by protein separation by 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). In all, 6 spots (1, 2, 3, 4, 5, and 6) were upregulated and 2 spots (7 and 8) were downregulated in PDGA tissues compared with WDGA. PDGA indicates poorly differentiated gastric adenocarcinoma; WDGA, well-differentiated gastric adenocarcinoma.

Enlarged 2-dimensional gel electrophoresis (2DE) images of altered proteins. Proteins upregulated in PDGA compared with well-differentiated gastric adenocarcinoma (WDGA) included calreticulin precursor (spot 1), tapasinERP57 heterodimer (spot 2), pyruvate kinase type M2 (PKM2; spot 3), class Pi glutathione S-transferase (GST-Pi; spot 4), and enolase 1 (ENO1; spots 5 and 6). Proteins were downregulated in PDGA included myosin-11 isoform SM2A (spot 7) and actin alpha cardiac (spot 8). Each spot number is the same as those in Figure 2. PDGA indicates poorly differentiated gastric adenocarcinoma.
Protein Identification by MALDI-TOF-MS or MS/MS
All the 7 proteins were identified successfully by MALDI-TOF-MS or MS/MS. Corresponding amino acid residue numbers are indicated on peaks that were matched to the identified protein based on using the search algorithm MASCOT against the National Center for Biotechnology Information nonredundant protein sequence database. Table 3 summarizes the identification information including isoelectric point (pI), molecular weight, number of matched peptides, sequence coverage, and MOWSE score. Proteins that were upregulated in PDGA included calreticulin precursor (spot 1), tapasinERP57 heterodimer (spot 2), pyruvate kinase isozymes M1/M2 isoform M2 (PKM2; spot 3), class Pi glutathione S-transferase (GST-Pi; spot 4), and chain A crystal structure of human ENO1 (spot 5 and spot 6). Proteins that were downregulated in PDGA included myosin-11 isoform SM2A (spot 7) and actin alpha cardiac (spot 8). The ENO1 protein was observed as multiple spots with slightly different pIs or molecular weights. These 2 spots may be explained by posttranslational modifications of the proteins.
Identification of Spots From the 2DE Maps by MALDI-TOF-MS or MALDI-TOF/TOF Tandem Mass Spectrometry.
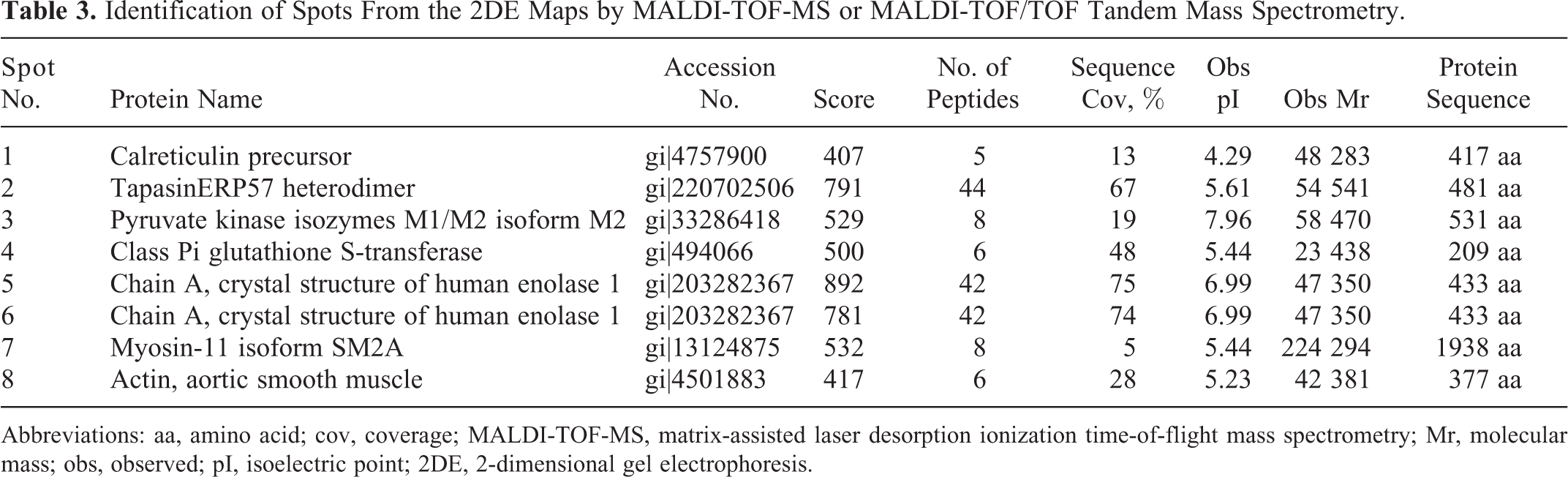
Abbreviations: aa, amino acid; cov, coverage; MALDI-TOF-MS, matrix-assisted laser desorption ionization time-of-flight mass spectrometry; Mr, molecular mass; obs, observed; pI, isoelectric point; 2DE, 2-dimensional gel electrophoresis.
Validation of the Differentially Expressed Proteins by Western Blot
To validate the protein quantification and identification results by proteomic approach, PKM2 and ENO1 were further detected by Western blotting (Figure 4). The changes in PKM2 and ENO1 in Western blots demonstrated similar trends as shown by 2DE.

Western blot assays for enzymes involved in the glycolytic pathway in gastric cancer tissues. Aldolase A, pyruvate kinase type M2 (PKM2), enolase 1 (ENO1), and enolase 2 (ENO2) were upregulated in PDGA (P). Pyruvate kinase type L/R (PKLR) was downregulated in PDGA, in comparison with well-differentiated gastric adenocarcinoma (WDGA; W). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used to monitor equal loading. PDGA indicates poorly differentiated gastric adenocarcinoma.
In addition, since PKM2 and ENO1 are 2 important enzymes of glycolysis, we investigated the expression of another significant enzyme in glucose metabolism, aldolase A. In different differentiated gastric adenocarcinomas, the expression of aldolase A had a similar trend as PKM2 and ENO1 (Figure 4). Moreover, we also examined pyruvate kinase type L/R (PKLR), which was isoenzyme of PKM2, and NSE/ENO2, which was isoenzyme of ENO1, to observe whether PKLR and ENO2/NSE had the same trend as their isoenzyme in PDGA and WDGA. The results showed that the expression of NSE/ENO2 was consistent with the ENO1 but PKLR had the opposite trend with PKM2 (Figure 4).
Furthermore, ENO1 protein, as being of concern, was tested in 3 pairs of WDGA, moderately differentiated gastric adenocarcinoma, and PDGA and corresponding noncancerous gastric tissues to evaluate the correlation of ENO1 expression with the differentiation of gastric cancer. The result indicated that the ENO1 expression was closely correlated with the tumor differentiation as the expression of ENO1 was linearly decreased from poorly differentiated tumor, moderately differentiated tumor, to well-differentiated tumor. In addition, there is significant difference between gastric tumors and their adjacent normal mucosae (Figure 5).

Correlation analysis of ENO1 expression with grade of tumor differentiation and compared with matched noncancerous adjacent mucosa. The expression of ENO1 was linearly decreased from poorly differentiated tumor (P), moderately differentiated tumor (M), to well-differentiated tumor (W). In addition, significant difference existed between gastric tumor and matched noncancerous adjacent mucosa (N). ENO1 indicates enolase 1.
Furthermore, we detected the expression of PKM2 and ENO1 in gastric tissues by immunohistochemical method. From the univariate analysis between the clinicopathological characteristics (Figure 6). The results show that there were significant differences in ENO1 expression in different differentiation grades of tumor tissues (well vs middle: P = .014 and P = .002; well vs poorly: P = .000 and P = .000; and middle vs poorly: P = .000 and P = .000; Table 4). The differences at expression of ENO1 and PKM2 were not significantly correlated with age (P = .396 and P = .160, respectively) and gender (P = .860 and P = .962, respectively). Analysis of variance of tumor size (<3 vs 3-5 cm: P = .05 and P = .538; <3 vs >5 cm: P = .032 and P = .104; 3-5 vs >5 cm: P = .470 and P = .151), <3 vs >5 cm and <3 vs 3 to 5 cm, P < .05 ENO1 showed statistical difference and then, the overall survival (OS) was further analyzed by Kaplan-Meier analysis using SPSS 13 software. Results showed that mean OS time for patients with gastric cancer expressing low levels of glypican 3 (GPC3) was 29.6 months, compared with 50.6 months for those expressing high levels of GPC3 (P = .006, log-rank test; Figure 1A). The mean TTR for patients with hepatocellular carcinoma expressing low levels of GPC3 was 15.2 months compared with those expressing high levels of GPC3 (P = .001, log-rank test; Figure 1B).

Expression of ENO1 and PKM2 in differentiation status of gastric cancer tissues (×200). Immunohistochemical detection showed that the expression of ENO1 and PKM2 was linearly decreased from poorly differentiated tumor, moderately differentiated tumor, to well-differentiated tumor. ENO1 indicates enolase 1; PKM2, pyruvate kinase type M2.
Immunohistochemical Analysis of ENO1 and PKM2 Expression in the Samples From the 107 Patients With Gastric Cancer.

Abbreviations: ENO1, enolase 1; PKM2, pyruvate kinase type M2.
a P < .05.
Kaplan-Meier analysis revealed that patients with gastric cancer having high levels of PKM2 (P = .001, log-rank test) or ENO1 (P = .010, log-rank test) expression appeared to have increased risk of lower survival rate compared with the patients with low expression (Figure 1).
Discussion
The mortality of gastric cancer has been maintained at high levels. The degree of gastric tumor differentiation affects a variety of biological behavior of cancer and prognosis of patients. 12 Li et al investigated the differentiation-related proteins in human gastric carcinoma cell lines by comparative proteomics. 13 However, the study employed cultured cell lines as a research model, which cannot reflect in vivo microenvironment and lack of the influence of tumor–host interaction. In this study, we collected several fresh gastric cancer tumor blocks from patients as analytic materials, which is reserved as the real condition of disease development. Comparative proteomic studies on poorly differentiated tumor tissues and well-differentiated tumor tissues identified 7 differently expressed proteins in the tumor tissues from 6 gastric cancer cases by 2DE and MALDI-TOF-MS or MS/MS. Five proteins that were upregulated in PDGA included calreticulin precursor, tapasinERP57 heterodimer, PKM2, GST-Pi, and ENO1. After annotation, they were classified into 2 categories: molecular chaperones (calreticulin precursor and tapasinERP57 heterodimer) and metabolism-associated proteins (GST-Pi, PKM2, and ENO1). Two proteins that were downregulated in PDGA included myosin-11 isoform SM2A and actin alpha cardiac, which belong to cytoskeleton system-associated protein.
Calreticulin and tapasinERP57 heterodimer have an important role in the peptide-loading complex, which is essential for the stability and transport of MHC class I molecules. 14 Wu et al investigated key regulatory molecules involved in prostate cancer (PCA) metastasis in 2 human androgen-independent PCA cell lines, highly metastatic 1E8-H, and lowly metastatic 2B4-L cells by proteomics analyses. They found that calreticulin precursor was upregulated in highly metastatic cells. 15 In this study, calreticulin precursor was found upregulated in PDGA. These phenomena suggest that calreticulin precursor is closely related to malignant phenotype of cancer.
It was reported that overexpression of GST-pi in human tumors was associated with malignancy, poor prognosis, and the development of drug resistance. 16 Increased levels of GST-pi are associated with resistance to alkylating agents such as melphalan, chlorambucil, and cyclophosphamide. 17 Toffoli and Cecchin have found that GST-pi has a role in the development of pharmacoresistance to platinum derivatives, which are the main chemotherapy for gastric cancer. 18 In this study, the GST-pi protein was upregulated in PDGA and downregulated in WDGA, which suggests that high expression of GST-pi might be one of the mechanisms that poor differentiated stomach tumor is eligible to get pharmacoresistance.
Moreover, among 5 proteins whose expression increased in human PDGA tissues, 2 of them, PKM2 and ENO1, were enzymes involved in the glycolytic pathway. Glycolysis has been shown to be elevated in almost all cancer types, even under normoxia, which is termed Warburg effect. 19 Metabolic adaptation to tumor microenvironmental conditions is a crucial step in tumor progression. It is well know that the expression of many key glycolytic enzymes (including phosphoglycerate kinase 1, lactate dehydrogenase A, pyruvate kinase M, glyceraldehyde-3-phosphate dehydrogenase, α-enolase, and aldolase A) is coordinately upregulated under hypoxia. 20 In this study, we found that PKM2 and ENO1 were significantly overexpressed in PDGA tissue compared with WDGA (Figure 4).
The pyruvate kinase has 4 isoenzymes. Pyruvate kinase type L is the characteristic pyruvate kinase isoenzyme of tissues with gluconeogenesis such as liver and kidney. Erythrocytes express the pyruvate kinase isoenzyme type R. Pyruvate kinase type M1 is present in the tissues in which large amounts of energy have to be rapidly provided such as muscle and brain. The PKM2 is characteristic of lung tissues as well as all cells with high rates of nucleic acid synthesis, including all proliferating cells such as embryonic cells, adult stem cells, and especially tumor cells. 21 The PKL and PKR isoenzymes are encoded by the same gene and are expressed under the control of different tissue-specific promoters. 22 Pyruvate kinase isoenzymes type M1 and M2 are different splicing products of the same mRNA transcript and differ in 21 amino acids. 23 During embryogenesis, the PKM2 isoenzyme is progressively replaced by the respective tissue-specific isoenzyme. Conversely, during tumorigenesis, the tissue-specific isoenzymes of pyruvate kinase, that is, PKL in the liver or PKM1 in the brain, disappear and the PKM2 isoenzyme is expressed. 24 Within the tumor metabolome, PKM2 regulates the proportions of glucose carbons that are channeled to synthetic processes (inactive dimeric form) or used for glycolytic energy production (highly active tetrameric form, a component of the glycolytic enzyme complex). In tumor cells, the dimeric form of PKM2 (tumor PKM2) is always predominant. The switch between the tetrameric and dimeric forms of PKM2 allows tumor cells to survive in environments with varying oxygen and nutrient supply. 25 In this study, the Western blot result showed that PKLR had the opposite trend with PKM2 between WDGA and PDGA (Figure 4). This further confirmed that more energy was needed from glycolysis in PDGAs, which might be supported by PKM2.
The ENO1 (α-enolase isoenzyme) protein has been reported to be upregulated in several cancer tissues in previous proteomic studies using 2DE. 26 -29 This study showed that ENO1 was upregulated in PDGA compared with WDGA. The ENO1 expression was closely correlated with the tumor differentiation since the expression of ENO1 was linearly decreased from poorly differentiated tumor, moderately differentiated tumor, well-differentiated tumor to adjacent normal mucosa (Figure 5). The ENO1 is a multifunctional protein As a glycolytic enzyme, ENO1 have important function in glucose metabolism and growth regulation; as a c-myc binding protein, ENO1 may play a central role in regulating the abnormal proliferative characteristics of transformed cells 30 ; as a plasminogen receptor, the enolase/plasminogen (plasmin) system is one of the mechanisms facilitating the invasiveness of pathogens in the human organism and it plays an important role in the processes of myogenesis and in the development of tumor tissues 31 ; and as a hypoxia-inducible genes, ENO1 perhaps assists hypoxia-inducible factor 1α to increase aggressiveness of tumors, metastatic spread, and poor response to therapy. 32,33 Neuron-specific enolase (NSE/ENO2, αγ, and γγ isoforms) is another isoform of enolase and it is known to be a useful marker for monitoring the progression of disease and response to treatment in small cell lung carcinoma and neuroblastoma. 34,35 Oskam et al studied the expression of enolase isoenzymes in rat medullary thyroid carcinomas and reported that the αγ and γγ isoforms isoenzyme levels were relatively high in well-differentiated rat tumors, whereas αα isoform was the majority of enolase isoenzymes in undifferentiated and anaplastic tumors. 36 This study showed that ENO1 and NSE/ENO2 are being upregulated in PDGAs (Figure 4). Baudin et al also reported that NSE/ENO2 level was exclusively associated with poor tumor differentiation. 37 In summary, our proteomics analysis revealed that the differentiation of gastric cancer is a complex process involving multiple factors such as molecular chaperone (calreticulin precursor and tapasinERP57 heterodimer) and metabolism-associated protein (GST-Pi, PKM2, and ENO1). Overexpression of glycolytic enzymes such as PKM2 and ENO1 in PDGA compared with WDGA provided a detailed profiling of the relationship between glycolysis and degree of tumor differentiation at the molecular level. These differentially expressed proteins could be potential differentiation-related cancer markers and will contribute to rationally improve diagnosis and treatment to gastric cancer.
Footnotes
Acknowledgment
The authors are very grateful to Dr Peng Zhao and Wen-Zhen Yuan for offering gastric adenocarcinoma tissue samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Changjiang Scholar fund (YZ), the talent supporting fund from the Ministry of Education of China (NCET-07-0403) (BZ), and the Fundamental Research Funds for the Central Universities (lzujbky-2010-143) (QG).
