Abstract
Introduction
Liver cancer is the sixth most common cancer and the second leading cause of cancer-related deaths worldwide.1,2 Hepatocellular carcinoma (HCC), the most common primary liver cancer, accounts for approximately 90% of cases. 3 Angiogenesis, the process by which new capillaries grow out of preexisting blood vessels, is 1 of the 10 hallmarks of cancer and has a vital role in the progression of cancer.4,5 Some important signaling molecules associated with angiogenesis, such as vascular endothelial growth factor, platelet-derived growth factor, and other related molecules, have been described. 6 In HCC, angiogenesis is regarded as a potential therapeutic target, and drugs that inhibit angiogenesis have been developed.7–9 However, the internal mechanism of HCC angiogenesis remains unclear.
Chloride intracellular channel protein 1 (CLIC1), a member of the CLIC family, is a transmembrane protein that directly forms part of the ion channel and is widely distributed in mammalian tissues and cells.10,11 As a membrane channel protein, CLIC1 plays an important role in transmembrane transport, cell membrane stabilization, and the maintenance of intracellular pH. 11 In addition, CLIC1 has been reported to have a main role in diseases associated with oxidative stress, including tumors and neurodegenerative diseases. 12 Blockade of CLIC1 was shown to arrest the cell cycle in the G2/M phase. 13 CLIC1 is upregulated in several tumors, such as oral squamous cell carcinoma, pancreatic ductal adenocarcinoma, and gastric cancer.14–20 By analyzing the relationship between the expression of CLIC1 in tumor tissues and clinical data, CLIC1 has been shown to be a predictive tumor biomarker for the prognosis of HCC patients. CLIC1 also plays a vital role in tumor migration and invasion. In our previous study, we found that CLIC1 promotes migration and invasion by regulating maspin, MMP2, and MMP9. 21
In the present study, we aimed to investigate the effect and mechanism of CLIC1 in HCC angiogenesis. The research data in this article are drawn from immunohistochemistry (IHC) staining of HCC clinical samples, tube formation experiments of endothelial cells, and subcutaneous tumor models in nude mice. For the first time, we showed that CLIC1 is positively correlated with microvascular density (MVD) and contributes to HCC angiogenesis by regulating the secretion of vascular endothelial growth factor A (VEGFA) in tumor cells.
Materials and Methods
Clinical Samples
A tissue microarray was constructed by Shanghai Zhuoli Biotechnology Co., Ltd. Sixty-seven pairs of HCC tissues and paracarcinoma tissues were fixed in formaldehyde and embedded in paraffin for further pathological analysis.
IHC Analysis
IHC was performed in 67 pairs of HCC tissues and paracarcinoma tissues. Formalin-fixed paraffin-embedded tumor tissues of 4 μm thickness were dewaxed in xylene and rehydrated using decreasing concentrations of ethanol. Endogenous peroxidase activity was blocked with 3% H2O2 methanol solution for 10 min at room temperature. The slides were further placed in EDTA buffer for antigen retrieval and blocked with 200 μL of 5% fetal bovine serum (FBS) for 5 min followed by overnight incubation of primary antibodies at 4°C. The primary antibodies used were against CLIC1 (1:100, 14545-1-AP, Proteintech), CD34 (1:1000, GB13013, Servicebio), and VEGFA (1:300, GB11034, Servicebio). After incubation, the slides were washed in phosphate-buffered saline and treated with the corresponding biotinylated secondary antibody for 60 min at room temperature. 3′-Diaminobenzidine was used as the chromogenic substrate. After counterstaining with Mayer's hematoxylin, the slides were dehydrated, cleared, and mounted. The IHC score of CLIC1 was calculated as P Score × I Score (P Score: 0: positive cells <10%; 1: positive cells <25%; 2: positive cells <50%; 3: positive cells <75%; 4: positive cells >75%; and I Score: 1: weak; 2: moderate; 3: strong).22,23 Patients with IHC scores ≥4 were defined as having high CLIC1 expression, and patients with IHC scores <4 were defined as having low CLIC1 expression.
Cell Culture
HCC cell lines including BEL7402 (Accession number: CBP60192) and Huh7 (Accession number: CBP60202) were obtained from Cobioer Biosciences. And the vascular endothelial cell line EA.hy926 (Accession number: GNHu39) was obtained from the bank of the Chinese Academy of Sciences. The BEL7402 cell line was cultured in Roswell Park Memorial Institute medium 1640 supplemented with 10% FBS. The Huh7 and EA.hy926 cell lines were cultured in Dulbecco's modified Eagle medium supplemented with 10% FBS. All cells were cultured in an incubator at 37°C with 5% CO2.
Cell Transfection
The BEL7402 cell line and Huh7 cell line used for coculture with EA.hy926 and preparation of conditioned media (CMs) were infected with control/CLIC1 overexpression plasmids and control/CLIC1 siRNA (Genechem Company). For xenograft assay in nude mice, the BEL7402 cell line was infected with negative control (NC)/CLIC1 overexpression lentivirus (GV492, Genechem Company). And the Huh7 cell line for xenograft assay was infected with NC/CLIC1 RNA interface (RNAi) lentivirus (GV248, Genechem Company). The RNAi sequence targeting CLIC1 was 5′-TCGGTACTTGAGCAATGCCTA-3′. Infected cells were incubated with media with puromycin (6 μg/mL) for 1 week, and then the expression of CLIC1 was verified by western blot.
EA.hy926 Migration Assay
To examine the role of vascular endothelial growth factor (VEGF) in angiogenic activity in CMs of BEL7402/Huh7 cells, we collected the CMs from different groups as follows: (1) Huh7 NC/CLIC1-knockdown, (2) Huh7 NC/CLIC1 overexpression, (3) Huh7 CLIC1 overexpression + AntiVEGF (Bevacizumab) (A2006, Selleck Chemicals), (4) BEL7402 NC/CLIC1-knockdown, (5) BEL7402 NC/CLIC1 overexpression, and (6) BEL7402 CLIC1 overexpression + AntiVEGF. The levels of VEGFA were measured using an ELISA kit (ELH-VEGF-CL, Raybiotech) according to the manufacturer's instructions.
For the EA.hy926 migration assay, 4 × 104 EA.hy926 cells were seeded in the upper chamber with FBS-free media, and 4 × 104 HCC cells (Huh7/BEL7402) or CMs of HCC cells were placed in the lower chamber of a 24-well transwell plate (8 μm, Corning). After 24 h of incubation at 37°C and 5% CO2, the cells were fixed with methanol and stained with 0.5% crystal violet. Cells on the upper surface were removed with cotton swabs, and cells that migrated to the lower surface were counted under an optical microscope. The experiment was repeated 3 times.
Animal Models
Twenty 4- to 6-week-old male nude mice were randomly divided into 4 groups and maintained at the Zhejiang Academy of Medical Sciences. A total of 5 × 106 HCC cells (Huh7/BEL7402) infected with lentivirus were subcutaneously injected into the right side of the mouse dorsal flanks. 24 The xenograft tumors were measured by a Vernier caliper once a week. Tumor volume was estimated by the following formula: V = 1/2 × length × width × width. The mice were sacrificed when the tumor volumes reached 1500 mm3 or in the fourth week. The reporting of this study conforms to ARRIVE 2.0 guidelines. 25 In animal experiments, the authors fully follow the requirements of animal ethics and comply with the “Guide for the Care and Use of Laboratory Animals, Eighth Edition.” 26 The authors have made their efforts to minimize the number of animals utilized and to decrease their suffering.
Western Blot
Western blotting was performed according to the directions described in our previous research. 27 The primary antibodies used were as follows: CLIC1 (1:1000, 14545-1-AP, Proteintech) and β-actin (1:1000, A5441, Sigma).
Statistical Analysis
Unpaired Student's
Results
High Expression of CLIC1 is Correlated with Higher MVD
IHC was performed on 67 pairs of HCC and paracarcinoma tissues to evaluate the expression levels of CLIC1 and CD34. We analyzed the MVD of slices stained with CD34. The results indicated that CLIC1 expression was higher in HCC tumor tissues than in paracarcinoma tissues (Figure 1A). Patients with increased CLIC1 expression showed a higher MVD (Figure 1B) (
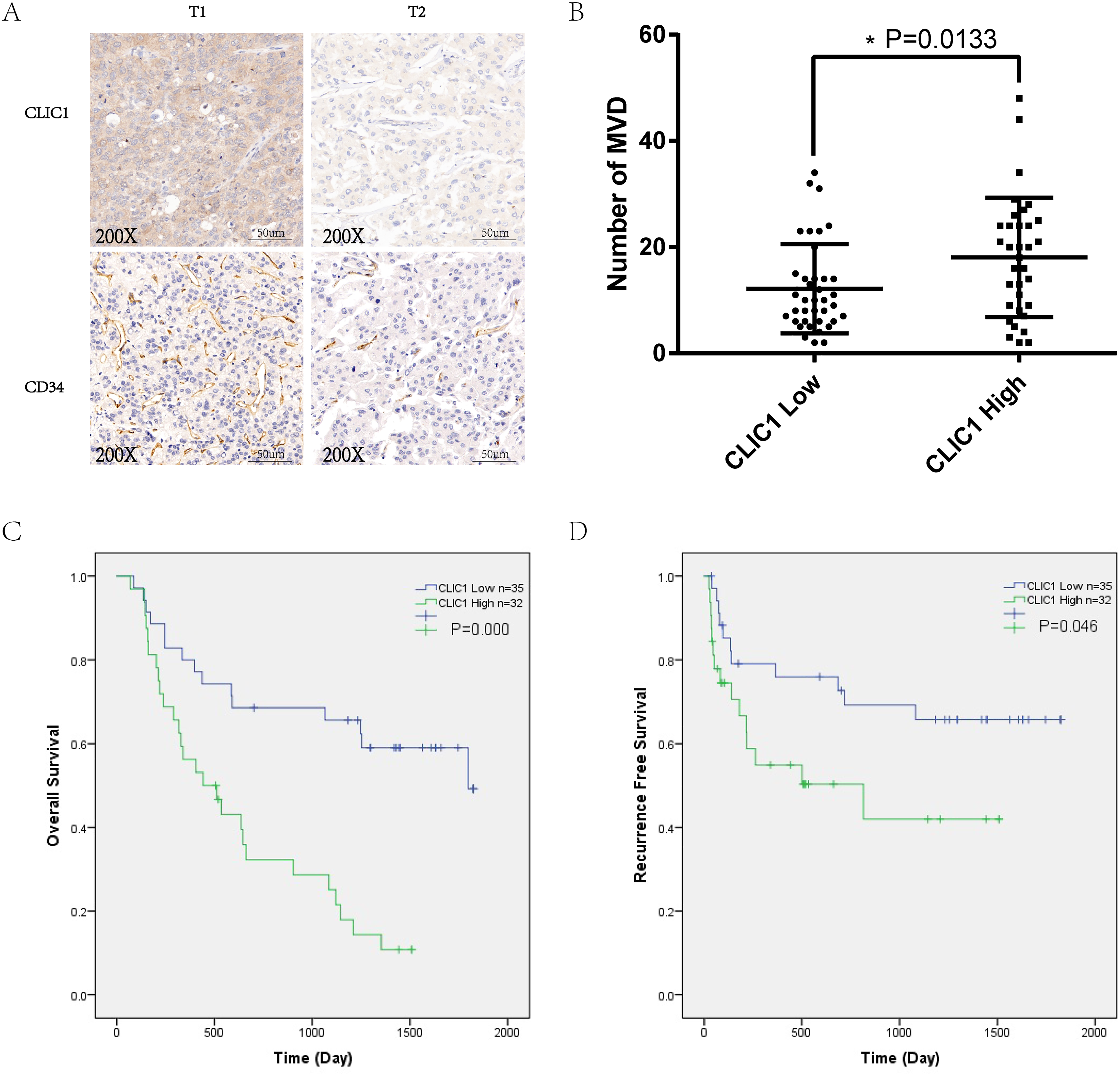
High expression of CLIC1 was associated with higher MVD and worse survival. (A) IHC of CLIC1 and CD34 in HCC tissues. (B) MVD in HCC tissues with different CLIC1 expression levels. (C) The relationship between CLIC1 expression and overall survival. (D) The relationship between CLIC1 expression and recurrence-free survival.
High Expression of CLIC1 is Associated with Poor Survival
Sixty-seven HCC patients were divided into 2 groups according to CLIC1 expression. The number of CLIC1 low-expression patients was 35, and the number of CLIC1 high-expression patients was 32. The results showed that the expression of CLIC1 was correlated with TNM staging, pathological differentiation, and vascular invasion (Table 1). Kaplan-Meier curves indicated that patients with lower expression of CLIC1 had better overall survival (
Relationship between CLIC1 expression and clinical-pathological features of HCC.

Abbreviations: CLIC1, chloride intracellular channel protein 1; HCC, hepatocellular carcinoma; MVD, microvascular density; AFP, alpha-fetoprotein.
*
Univariate and multivariate analyses of factors associated with overall survival of HCC patients (n = 67).
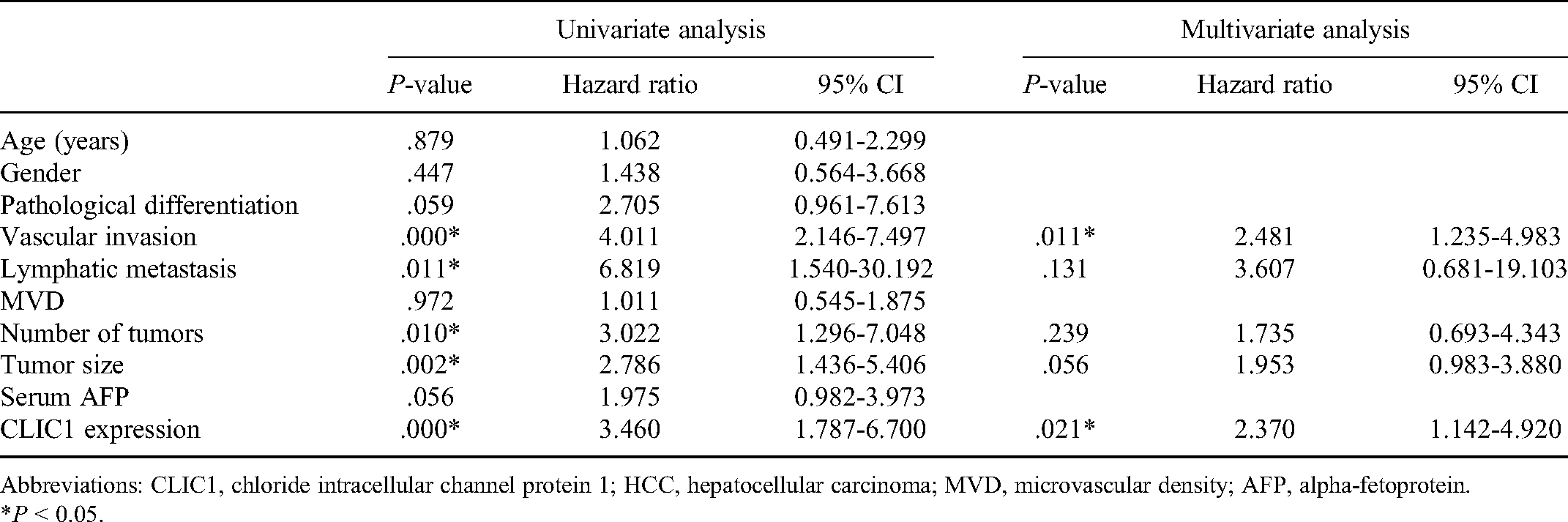
Abbreviations: CLIC1, chloride intracellular channel protein 1; HCC, hepatocellular carcinoma; MVD, microvascular density; AFP, alpha-fetoprotein.
*
CLIC1 Induces VEGFA Secretion in HCC Cell Lines
CMs were collected from CLIC1 low-expression and CLIC1 overexpression HCC cell lines. The concentration of VEGFA was measured in the CM of the different groups. VEGFA in CM from CLIC1-knockdown Huh7 cells was approximately 2652 pg/mL, compared with 2497 pg/mL of the control group (Figure 2C,

CLIC1 expression in HCC cells affected VEGFA secretion. (A) Validation of stable CLIC1-knockdown and -overexpressing in Huh7 cell lines. (B) Validation of stable CLIC1-knockdown and -overexpressing in BEL 7402 cell lines. (C) The concentrations of VEGFA in Huh7 cell lines. (D) The concentrations of VEGFA in BEL 7402 cell lines.
Coculture with CLIC1 Overexpression Cell Lines Facilitates the in Vitro Migration of EA.hy926 Cells
To investigate the migration of endothelial cells under different culture conditions with HCC cells, we cocultured EA.hy926 cells with CLIC1 low-expression and CLIC1 overexpression HCC cell lines in Transwells (8 μm). In a coculture system with CLIC1-knockdown Huh7 cells and BEL7402 cells, the migration ability of EA.hy926 cells was significantly repressed (Figure 3A,

CLIC1 expression in HCC cells affected the migration ability of endothelial cells. (A) EA.hy926 cells were coculture with HCC cells with different expression levels of CLIC1. (B) EA.hy926 cells were treated with CM from HCC cells with different expression levels of CLIC1. (C) The number of EA.hy926 cells was measured using ImageJ software.
CM from CLIC1 Overexpression Cell Lines Promotes the in Vitro Migration of EA.hy926 Cells
We then analyzed the impact of the CM derived from CLIC1 low-expression and CLIC1 overexpression HCC cells on the migration ability of endothelial cells. The results were consistent with those achieved in the HCC cell-endothelial cell coculture system. CM from CLIC1 overexpression Huh7 cells and BEL7402 cells promoted the migration of EA.hy926 cells (Figure 3B,
CLIC1 Promotes Angiogenesis in Vivo
To determine whether CLIC1 could regulate angiogenesis in vivo, we developed xenograft tumors using CLIC1-knockdown Huh7 cells, CLIC1-overexpressing BEL7402 cells, and HCC cells transfected with control lentivirus. The results showed that the growth of xenograft tumors derived from CLIC1-knockdown Huh7 cells was restrained compared to that of the control group. The growth of CLIC1-overexpressing BEL7402 cells showed was slightly different from the control group, but the difference was not statistically significant. IHC staining of the xenograft showed results similar to those obtained in human HCC tissues. MVD was higher in tumors with CLIC1 overexpression. In the tumor tissues derived from CLIC1-knockdown cells, microvessels could rarely be seen (Figure 4).

CLIC1 promotes angiogenesis in vivo. (A, B) The gross morphology and tumor growth of xenografts. (C) IHC staining of CLIC1 and CD34 in xenografts. Arrows point to microvessels.
Discussion
CLIC1 is a member of the chloride intracellular channel gene family. 13 It is considered a potential tumor biomarker among several tumors, such as oral squamous cell carcinoma, epithelial ovarian cancer, pancreatic ductal adenocarcinoma, human glioma, gastric cancer, and clear cell renal cell carcinoma.14–20 Studies have suggested that CLIC1 plays an important role in tumor migration and invasion, mainly by regulating reactive oxygen species/mitogen-activated protein kinase/extracellular-signal regulated kinase signaling.28–30 In addition, our previous work revealed that CLIC1 contributes to HCC migration and invasion by targeting maspin. 21 However, the relationship between CLIC1 and angiogenesis has rarely been reported, and the role of CLIC1 in angiogenesis remains unclear. In this study, we found that higher expression of CLIC1 is correlated with angiogenesis in HCC; hence, CLIC1 is a promising biomarker for predicting the prognosis of HCC patients.
In primary human endothelial cells, CLIC1 has been reported to regulate angiogenesis in vitro by targeting various integrins. 31 In our study, we conducted IHC analyses of CLIC1 and CD34 in 67 pairs of HCC tissues and paracarcinoma tissues. In the analysis of IHC staining of clinical samples and corresponding clinical information, our study provides evidence that is consistent with other studies of different tumor types. First, higher expression of CLIC1 was detected in HCC tissues than in paracarcinoma tissues. Patients with higher CLIC1 expression had worse survival, indicating that CLIC1 is a promising biomarker for predicting prognosis. Moreover, we also observed that tumor tissues with higher CLIC1 expression had a higher MVD.
To validate and explain this phenomenon, we further established CLIC1-knockdown and CLIC1-overexpressing HCC cell lines, and the results indicated that coculture with CLIC1-knockdown Huh7 cells inhibited the migration ability of endothelial cells compared to the control group. In our previous study, we found that CLIC1 regulates maspin to influence tumor migration and invasion, 21 and another study indicated a relationship between maspin and VEGF. 32 We assessed the concentration of VEGFA in the CM from HCC cells with different CLIC1 expression levels and found it to be lower in CMs from CLIC1-knockdown cells. The CM from CLIC1-overexpressing cells also had a higher concentration of VEGFA. Vascular endothelial cells treated with various CM showed diverse migration abilities, which confirm the effect of CLIC1 in regulating the excretion of VEGFA.
In vivo experiments showed that the MVD in CLIC1-overexpressing tumors was significantly higher than that in control tumors. This finding indicates a correlation between CLIC1 expression and MVD. However, tumors in the CLIC1-knockdown group were too small to allow for sufficient IHC staining. Tumor growth showed an obvious difference between the CLIC1-knockdown and control groups, indicating that CLIC1 gene silencing inhibits tumor proliferation, which is consistent with the results of other studies. 33 There was a tendency for rapid tumor growth in the CLIC1 overexpression group, but this difference was not statistically significant and could be due to the heterogeneity of the group. This study has some other limitations. First, the samples used were collected from a single-center population. And the sample size is still not large enough. Besides, the mechanism of how CLIC1 regulates VEGFA secretion requires further study.
Conclusion
In summary, our study found that CLIC1 is a promising biomarker for predicting the prognosis of HCC patients, and expression of CLIC1 correlates with angiogenesis in HCC through regulating VEGFA.
Footnotes
Ethics Statement
The study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University. And the Tab of Animal Experimental Ethical Inspection of the First Affiliated Hospital, College of Medicine, Zhejiang University. And the authors have followed the principles outlined in the Declaration of Helsinki for all human and animal experimental investigations. Reference Number: 2020-196 and 2020-271. Date: March 12, 2020.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Youth Program of the National Natural Science Foundation of China, National S&T Major Project, Zhejiang Province Medicine, and Health General Research Program, the China Postdoctoral Science Foundation, (grant numbers 81702858, 2017ZX10203205, 2016KYB087, 2018KY375, and 2016M600470).
