Abstract
Purpose:
Magnetic resonance imaging–guided radiation therapy has entered clinical practice at several major treatment centers. Treatment of early-stage non-small cell lung cancer with stereotactic body radiation therapy is one potential application of this modality, as some form of respiratory motion management is important to address. We hypothesize that magnetic resonance imaging–guided tri-cobalt-60 radiation therapy can be used to generate clinically acceptable stereotactic body radiation therapy treatment plans. Here, we report on a dosimetric comparison between magnetic resonance imaging–guided radiation therapy plans and internal target volume–based plans utilizing volumetric-modulated arc therapy.
Materials and Methods:
Ten patients with early-stage non-small cell lung cancer who underwent radiation therapy planning and treatment were studied. Following 4-dimensional computed tomography, patient images were used to generate clinically deliverable plans. For volumetric-modulated arc therapy plans, the planning tumor volume was defined as an internal target volume + 0.5 cm. For magnetic resonance imaging–guided plans, a single mid-inspiratory cycle was used to define a gross tumor volume, then expanded 0.3 cm to the planning tumor volume. Treatment plan parameters were compared.
Results:
Planning tumor volumes trended larger for volumetric-modulated arc therapy–based plans, with a mean planning tumor volume of 47.4 mL versus 24.8 mL for magnetic resonance imaging–guided plans (P = .08). Clinically acceptable plans were achievable via both methods, with bilateral lung V20, 3.9% versus 4.8% (P = .62). The volume of chest wall receiving greater than 30 Gy was also similar, 22.1 versus 19.8 mL (P = .78), as were all other parameters commonly used for lung stereotactic body radiation therapy. The ratio of the 50% isodose volume to planning tumor volume was lower in volumetric-modulated arc therapy plans, 4.19 versus 10.0 (P < .001). Heterogeneity index was comparable between plans, 1.25 versus 1.25 (P = .98).
Conclusion:
Magnetic resonance imaging–guided tri-cobalt-60 radiation therapy is capable of delivering lung high-quality stereotactic body radiation therapy plans that are clinically acceptable as compared to volumetric-modulated arc therapy–based plans. Real-time magnetic resonance imaging provides the unique capacity to directly observe tumor motion during treatment for purposes of motion management.
Introduction
Magnetic resonance imaging (MRI)–guided radiation therapy (RT) has entered into clinical use in several centers in the United States as well as abroad. 1,2 Currently, the only commercially available system in use combines on-board 0.35-T MRI with 3-cobalt-60 sources in order to provide real-time imaging during treatment. Real-time imaging during treatment has an obvious theoretical benefit in allowing clinicians to know the exact position of a tumor during treatment, which may potentially prevent geometric tumor miss and allow for a smaller planning target volume (PTV) to be used. Lung cancer, and specifically, early-stage lung cancer, is a clear potential indication for this treatment modality, as respiratory tumor motion must be taken into account. Although alternatives exist for real-time tumor tracking, these approaches typically require radio-opaque, implanted fiducial markers that involve invasive procedures, which have the potential for morbidity such as the development of pneumothorax, marker migration, and arrhythmias. 3 –5 Although noninvasive tracking methods have also emerged, these use surrogates for tumor movement, which may not be a representative of true tumor motion and position during treatment. 5 –7
Stereotactic body radiation therapy (SBRT) is a standard-of-care treatment for inoperable, early-stage non-small cell lung cancer (NSCLC). By delivering a high ablative dose of highly conformal RT, excellent local control rates are feasible. 8,9 For purposes of treatment planning, a 4-dimensional computed tomography (4DCT) scan is often utilized in order to generate an internal target volume (ITV) to account for tumor motion during treatment. Nevertheless, there is evidence that an ITV-based approach can underestimate true respiratory tumor excursion and lead to geometric miss. 7 Real-time imaging during treatment is one potential solution to this problem.
We hypothesize that MRI-guided tri-cobalt-60 SBRT can be used to generate clinically deliverable RT treatment plans while providing increased certainty in tumor motion and normal structure location. Here, we report on a dosimetric comparison between MRI-guided tri-cobalt-60 SBRT plans and ITV-based plans delivered by a linear accelerator (LINAC) utilizing volumetric-modulated arc therapy (VMAT).
Materials and Methods
Simulation
Ten patients with early-stage NSCLC cancer who underwent RT planning and treatment with LINAC-based SBRT were used for this study. Tumor locations were varied, with superior, inferior, central, and peripheral tumors included. Patients were simulated in the supine position using a 4-dimensional free-breathing computed tomography (GE Discovery STE; GE Healthcare, Little Chalfont, United Kingdom) scan. A stereotactic BodyFix (Elekta, Stockholm, Sweden) immobilization system was utilized during computed tomography simulation and treatment to minimize respiratory tumor motion and for reproducibility during treatment.
Tumor Volumes
Images were exported to MIM (MIM Software, Cleveland, Ohio) for target and organ at risk (OAR) delineation. Normal OARs were contoured for each patient including the spinal cord, lungs, heart, trachea, proximal bronchial tree, esophagus, and chest wall. For LINAC-based plans, an ITV was generated by contouring the gross tumor volume (GTV) on each phase of the 4DCT treatment planning scan. A 0.5-cm expansion in all directions from the ITV to the PTV was used. For tri-cobalt-60 plans, a GTV was contoured on a single mid-inspiratory phase from the 4DCT scan. A 0.3-cm expansion from the GTV to the PTV was used as is common for MRI-guided RT at our center, accounting for the submillimeter spatial resolution of the 3-D True Fast Imaging sequence used for real-time imaging as measured with end-to-end testing using an MRI-compatible motion phantom and the ability to directly visualize tumors during RT.
Planning
For LINAC-based plans, contours from MIM were imported into the Pinnacle3 treatment planning system (Philips Healthcare, Andover, Massachusetts). The VMAT plans were generated for patient treatment using a multileaf collimator (MLC) with thirty-two 2.5-mm leaves located centrally and twenty-eight 5-mm leaves located peripherally using noncoplanar 6 MV photon beams. Doses were calculated with a convolution superposition algorithm on the Pinnacle treatment planning system. For MRI-guided tri-cobalt-60 treatment plans, patient contours were imported into the ViewRay treatment planning system (ViewRay, Oakwood Village, Ohio) to generate treatment plans using the ViewRay treatment planning system’s Monte Carlo algorithm, which uses a deterministic optimizer. Plans were generated and optimized using 10 to 12 intensity-modulated beams from 3 cobalt sources using 1.05-cm MLC leaves, equally spaced around the patient angularly and located to avoid entrance through the treatment couch edges.
A dose of 50 Gy in 5 fractions was prescribed to the 80% to 90% isodose line for both treatment plans, with a planning target goal of 95% of the PTV to be covered by the prescription. Dose constraints, based on RTOG 0813, RTOG 0915, and TG-101, included total lung volume minus PTV V20 Gy < 10%, V5 Gy < 50%, spinal cord maximum dose < 30 Gy, chest wall V30 Gy < 30 cm3, trachea and proximal bronchial tree maximum dose of 37 Gy, and esophagus maximum dose of 32.5 Gy.
Analysis
Dosimetric outcomes for normal tissue toxicity as well as planning target coverage were compared between LINAC-based and MRI-guided tri-cobalt-60 plans using a 2-sided t test with a statistical significance threshold of P < .05, and normal distribution was confirmed using the Kolmogorov-Smirnov normality method. Metrics evaluated included organ-specific dose constraints, 50 Gy and 25 Gy isodose volumes, and ratios of prescription isodose volume and 50% isodose volume to PTV volume (R50). A heterogeneity index (HI) as defined by Wang et al 10 and used by Wooten et al 11 was used to describe the extent of dose variability within the PTV. The HI is defined as the ratio of the D5%/D95%, where D5% and D95% represent the minimum dose covering 5% and 95% of the PTV volume, respectively. The minimum value of HI is 1.0, which represents a perfectly homogeneous PTV dose.
The conformity number (CN), defined as: (TVRI/TV) × (TVRI/VRI), was also computed to determine the dose distribution relative to the PTV that accounts for the volume of the target and the volume of healthy tissue receiving the reference dose or greater. 12 TVRI is defined as the target volume covered by the reference isodose lines (100% and 95%), TV is the volume of the PTV, and VRI is the volume of the reference isodose. The CN ranges between 0 and 1.0, with 0 implying a greater patient volume receiving the reference dose than the target volume and 1.0 implying that exactly the target volume receiving the reference dose. Finally, D2cm, the maximum dose at 2 cm from the PTV as a percentage of the prescription dose, was calculated as per recent RTOG protocols, and the dose covering 99% of the GTV was calculated.
Results
Plans delivering 50 Gy in five 10 Gy fractions were generated for both LINAC-based VMAT and MRI-guided SBRT tri-cobalt-60 delivery. Plan characteristics are noted in Table 1. As expected, PTV volumes trended to be larger in the LINAC-based plans, with a mean LINAC-based PTV of 47.4 mL compared to 24.8 mL in the MRI-guided PTV (P = .08). Clinically acceptable plans were achievable via both LINAC and MRI-guided planning methods, with lung V5 being 15.8% and 19.3% (P = .42) in LINAC and MRI-guided tri-cobalt-60 plans, respectively (Figure 1). This was also true for bilateral lung V20, 3.9% versus 4.8% (P = .62), and mean lung dose, 3.3 versus 3.9 Gy (P = .48; Figure 2). The volume of chest wall receiving greater than 30 Gy was also similar, 22.1 versus 19.8 mL (P = .78), as were all other parameters listed in Table 1, which are commonly used for planning early-stage NSCLC SBRT.
Normal Structure Dosimetry.
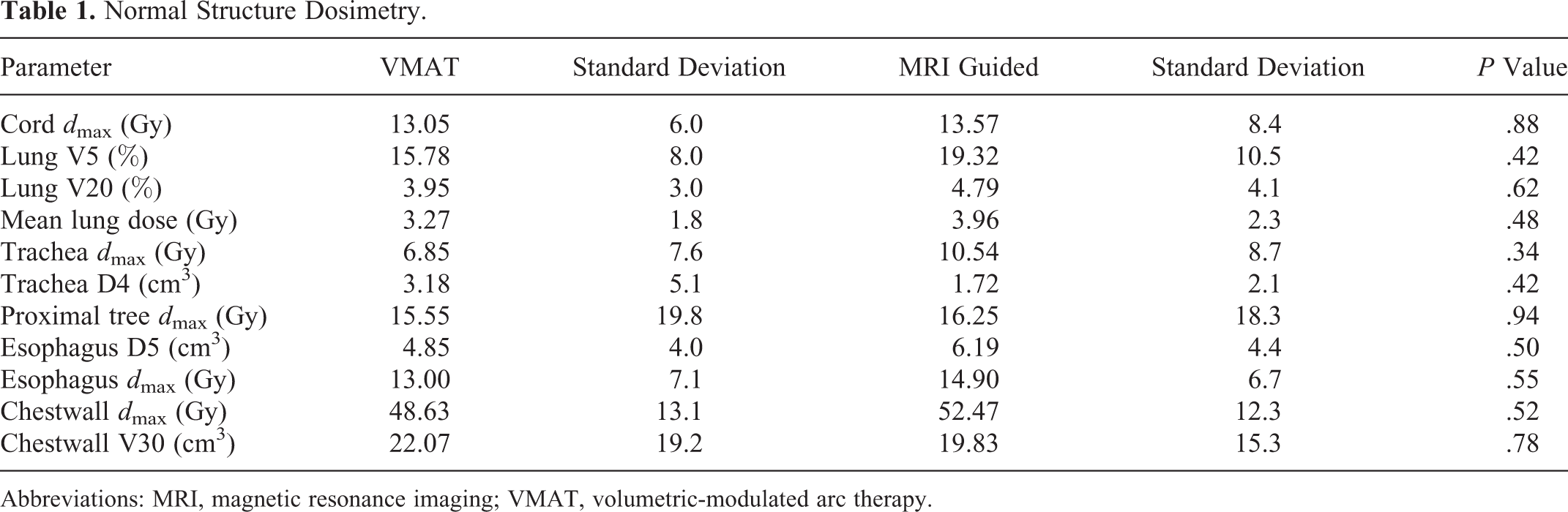
Abbreviations: MRI, magnetic resonance imaging; VMAT, volumetric-modulated arc therapy.

A, Sample VMAT plan. B, Sample magnetic resonance imaging (MRI)–guided tri-cobalt-60 plan. MRI indicates magnetic resonance imaging; VMAT, volumetric-modulated arc therapy.

Dose–volume histogram comparison. Solid lines represent VMAT, dashed lines represent MRI-guided therapy. ITV/GTV: yellow, PTV: red, chest wall: green, lungs–ITV: purple, spinal cord: blue. MRI indicates magnetic resonance imaging; GTV, gross tumor volume; ITV, internal target volume; PTV, planning tumor volume; VMAT, volumetric-modulated arc therapy.
Plan parameters related to dose distribution are listed in Table 2. Although the mean ratio of prescription isodose volume to PTV volume was similar between VMAT and MRI-guided plans, 1.14 versus 1.17 (P = .67), the ratio of the 50% isodose volume to PTV volume was lower in VMAT plans, 4.19 versus 10.0 (P < .001). Absolute 25 Gy isodose volumes were similar between plans, 178.7 mL versus 232.1 (P = .54). Heterogeneity index was comparable between plans, 1.25 versus 1.25 (P = .98), although CN for both the 100% isodose line, 0.86 versus 0.74 (P < .001), and the 95% isodose line, 0.83 versus 0.61 (P < .001), was higher in VMAT-based plans (P < .001). Mean D2cm was slightly lower in in the VMAT plans, 0.57 versus 0.63 (P = .06). Mean D99 to the GTV between plans was similar between VMAT and MRI-guided plans, 53.1 versus 53.4 Gy (P = .72).
Plan Evaluation.

Abbreviations: GTV, gross tumor volume; MRI, magnetic resonance imaging; PTV, planning tumor volume; VMAT, volumetric-modulated arc therapy.
Discussion
The increased availability of real-time MRI-guided tri-cobalt-60 RT gives physicians a new tool to ensure that treatment is delivered accurately and with a decreased chance of geometric miss. Stereotactic body radiation therapy for lung cancer, specifically for early-stage NSCLC, is a natural application of real-time guidance due to the presence of respiratory motion and the potential to decrease required treatment volumes through the use of real-time gating instead of ITV-based approaches. We have shown that it is feasible to generate MRI-guided tri-cobalt-60 RT plans that are comparable to VMAT-based LINAC plans. In the above treatment plans, almost all clinically relevant treatment parameters were not statistically significantly different between LINAC VMAT and tri-cobalt-60 SBRT-based plans, including all metrics commonly used to avoid normal tissue toxicity. Nevertheless, there were several differences between plans. Despite a smaller PTV, the MRI-guided plans had a larger R50, lesser conformity, and slightly increased D2cm as compared to the LINAC VMAT-based plans. This difference can be largely attributed to the increased geometric penumbrae of the cobalt-60 source as well as the larger MLC size in the cobalt-60 unit as compared to a 6 MV LINAC. This difference may not be clinically relevant as determinants of normal tissue toxicity such as mean lung dose and the percentage of lung receiving 5 and 20 Gy did not significantly change between the 2 treatment plans.
The R50, which is required in RTOG SBRT protocols, is more commonly used as a measure of plan quality and has not been associated with pneumonitis. 12 Metrics that have been associated with increased pneumonitis risk in lung SBRT patients include the lung volume receiving 20 Gy (V20) and increased PTV size. 13 Our plans did not show any difference in the dose received by the bilateral lungs. Furthermore, our current plans may be slightly overestimating lung volume radiated, since these were planned in a mid-inspiratory breath-hold position and not in a maximum inspiratory breath-hold position. A maximum inspiratory breath hold is not commonly used with traditional LINAC-based treatment due to concerns regarding reproducibility and the potential for geometric miss. Real-time imaging during treatment allows for direct visualization of the tumor during treatment, ensuring that the breath hold is adequately reproduced repeatedly throughout. As a result, our current practice when using MRI-guided treatment is to treat patients with a maximum breath hold. By employing this technique with maximum breath hold, patient lung volumes are increased and mean lung dose and total lung V20 would be decreased.
The CN for both the 100% and 95% isodose lines was also inferior in the MRI-guided plans compared to the LINAC VMAT-based plans. Although the VMAT-based plans did show superior conformity compared to the tri-cobalt-60 plans, the clinical relevance of this is unknown and is likely minimal. Although dose conformity is desirable in traditional intensity-modulated RT plans, this is not the case in SBRT plans where more variation may be acceptable due to the relatively small target volume, lack of critical structures within the PTV, and potential advantage of a higher dose gradient between the center of the tumor and the periphery. Conformity could potentially be improved by increasing permissible beam modulation; however, this would increase the treatment time without necessarily providing increased clinical benefit. In any case, our MRI-guided tri-cobalt-60-based plans have R50 and conformality metrics that were deemed clinically acceptable per the treating physician.
Treatment time is a significant factor in delivering MRI-guided plans, due to both the radioactive decay of the cobalt-60 sources over time and the increased treatment duration that respiratory gating requires. In our center, we attempt to ensure that MRI-guided radiation plans are deliverable in a patient’s time of less than 1 hour on the treatment table.
It is likely that if we were to use identical PTV expansions in our MRI-guided plans as those in the VMAT plans, our normal structure dosimetry and plan evaluation parameters would be further degraded. Nevertheless, one immediate benefit of real-time imaging is the increased confidence with regard to tumor position during treatment, which then allows for a non-ITV approach and allows for a smaller PTV expansion (0.3 cm) to be used in MRI-guided plans as compared to the commonly employed 0.5-cm expansion. This is observed in the lack of difference between the absolute volume of lung receiving 20 Gy. The disadvantage of the larger penumbrae of cobalt-60 is offset by the ability to use a smaller PTV expansion as compared to VMAT plans. Furthermore, as described above, 7 ITV-based approaches can potentially underestimate true tumor motion and excursion during treatment due to breathing variation that may not be fully captured at the time of a 4-D simulation scan. This may be especially true for tumors with a decreased volume and those with a large motion range such as those at the lung bases. 14 It is conceivable that in these situations, a tumor may not receive the true planned dose using an ITV-based approach, adversely impacting local control rates. Real-time MRI guidance allows for visualization of true tumor motion during treatment and thus a decreased chance of geometric miss.
Conclusion
Magnetic resonance imaging–guided tri-cobalt-60 RT is now in clinical use at our facility with expanding use at several sites across the world. Although the clinical benefit of real-time MRI-guided RT is yet to be fully defined in patient outcome studies, it does provide the powerful ability to directly observe tumor motion for purposes of motion management and determine the dose delivered during treatment with the option to adapt for future fractions. Additional studies are warranted to further validate and expand the optimum use of this promising technology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
