Abstract
Background/Purpose:
Delivering stereotactic body radiotherapy for liver metastases remains a challenge because of respiratory motion and poor visibility without intravenous contrast. The purpose of this article is to describe a novel and simple computed tomography (CT) simulation process of integrating timed intravenous contrast that could overcome the uncertainty of target delineation.
Methods and Results:
The simulation involves two 4-dimensional CT (4DCT) scans. The first scan only encompasses the immediate region of the tumor and surrounding tissue, which reduces the 4DCT scan time so that it can be optimally timed with intravenous contrast injection. The second 4DCT scan covers a larger volume and is used as the primary CT data set for dose calculation, as well as patient setup verification on the treatment unit. The combination of the two 4DCT scans allows us to optimally visualize liver metastases over all phases of the breathing cycle while simultaneously acquiring a long enough 4DCT data set that is suitable for planning and patient setup verification.
Conclusion:
This simulation technique allows for a better target definition when treating liver metastases, without being invasive.
Introduction
Surgery is the standard of care for resectable liver metastases. However, less invasive local options like radiofrequency ablation and stereotactic body radiation therapy (SBRT) are emerging alternatives with promising results. 1 -4 With new innovations in technique and technology, radiotherapy is challenging the dogma of what can and cannot be treated for liver metastases. Historically, radiotherapy for liver tumors was limited because of the lack of tumor dose-shaping possibilities (eg, intensity-modulated radiotherapy), techniques to address respiratory motion and liver radiosensitivity. 1,5 Stereotactic body radiation therapy allows for very high doses of radiation to be delivered with high conformality to the target lesion, while maintaining a steep dose gradient toward the surrounding normal tissue and thus minimizing radiation to normal surrounding liver and reducing the risk of radiation-induced liver disease. 1,6 However, with increased conformality, accurate delineation of the target volume becomes more critical.
For SBRT planning in the treatment of liver metastases, the main problems are 2-fold. First, the intrafraction position of liver tumors usually undergoes significant motion due to respiration. 7,8 Although the respiratory motion can be reduced by abdominal compression, 9 residual tumor motion can still be significant. 10 Second, liver metastases are often relatively similar in density with the surrounding tissue when using noncontrast computed tomography (CT) for imaging, 10,11 making it difficult to identify and delineate. Although the respiratory motion-related positioning error can be accounted for by using a 4-dimensional CT (4DCT), the poor visibility could be improved with a timed intravenous contrast injection, similar to diagnostic CT scans. However, a planning 4DCT scan, which extends 5 cm cranial to the top of the diaphragm to the bottom of L5, takes approximately 2 minutes, whereas intravenous injection of contrast in a 70-kg patient will remain in the patient for only 28 seconds followed by a fast washout. Thus, only a fraction of the 4DCT simulation scan will have proper contrast enhancement of the metastases, making it easy to miss them. 12 To overcome this problem, we describe a novel approach developed at our center whereby 2 separate coregistered 4DCT scans are performed, with optimally timed intravenous contrast used to improve the target(s) delineation over all the phases of a breathing cycle.
Methods
Computed Tomography Simulation
Patients are simulated in the supine position with the thorax BlueBAG BodyFIX (Medical Intelligence, Schwabmuenchen, Germany) vacuum cushion that creates a comfortable, stable, and precise mold of the patient’s position, enabling reproducible patient positioning from imaging through the entire treatment process. In addition, abdominal compression, consisting of a support frame and plate affixed with a screw, is used to reduce the organ motion related to respiratory excursion (Figure 1A). In the event that the abdominal compression system is not feasible (eg, when the patient is too large or if the screws on the compression plate would interfere with SBRT), the double-vacuum sandwiched BodyFIX system (consisting of a clear plastic foil wrapping on a patient’s lower part of the body up to the thorax in addition to the BlueBAG) is used (Figure 1B). 9,13
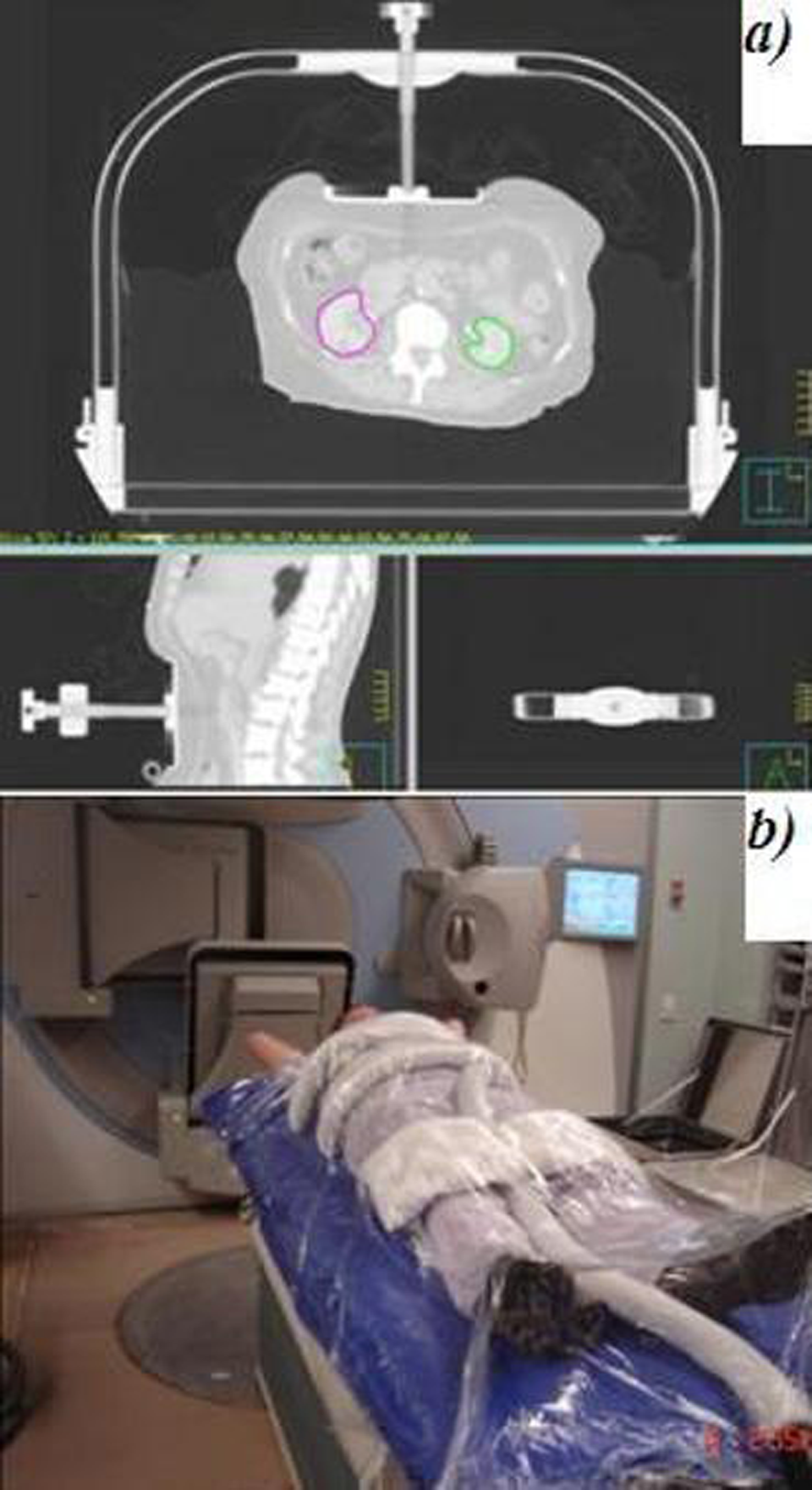
The patient setup and immobilization system. A, Abdominal compression consisting of a support frame and plate affixed with a screw used to reduce the organ motion related to respiratory excursion. B, The double-vacuum sandwiched BodyFIX system (consisting of a clear plastic foil wrapping on a patient’s lower part of the body up to the thorax in addition to the BlueBAG used if the abdominal plate is not feasible (eg, when the patient is too large) or when the lesion is close to the screw.
Intravenous contrast is used to improve tumor visibility. Visipaque 270 (GE Healthcare Canada, Mississauga, Ontario, Canada), 2 cm3/kg, is given at 5 cm3/s to a maximum of 200 cm3 using a CT contrast injection system (Medrad Stellant CT Injection System, Indianola, Pennsylvania). A 4DCT simulation scan (Brilliance CT Big Bore; Philips, Cleveland, Ohio) is used to acquire respiratory-correlated images of the patient anatomy as it moves with breathing. An air bellows sensor, which contracts and expands with respiratory motion, is placed on the lower chest or upper abdomen, depending on the location of the greatest breathing motion. Following stabilization of the patient’s breathing, the patient is injected with contrast, which is followed by the 4DCT scans. Two separate coregistered 4DCT scans are performed:
The first 4DCT scan, “short 4DCT,” encompasses only the target(s) volume (solitary or multiple lesions) as well as immediately surrounding tissue (slice thickness = 3 mm). The location of the target lesion(s) (eg, upper third of liver) is indicated by the radiation oncologist based on the diagnostic images. Note that it is imperative that the short 4DCT captures the full extent of the target(s)’s respiratory motion. Therefore, a reasonable margin should be added inferior and superior of the target’s position. The typical extent of the short 4DCT scan encompasses about 1/2 of the maximal liver cranial-caudal dimensions. However, if there are multiple spatially diverse targets, the short 4DCT scan can include the entire liver. Since the scanned length is shorter than a regular 4DCT planning scan, the scan time is significantly reduced to approximately 40 to 60 seconds. If multiple lesions are targeted, the short 4DCT scan is synchronized with the contrast injection such that contrast enhancement of the lesion(s) is optimized. The target(s) is presumed to be approximately in the middle of the short 4DCT scan. Therefore, the following formula is used to calculate the scan delay time, The second 4DCT scan, “long 4DCT,” is done immediately after the short 4DCT. The long 4DCT covers a much larger volume, extending from L5 to 5 cm cranial to the diaphragm (slice thickness = 3 mm). The estimated radiation dose from both CT scans corresponding to the volume computed tomography dose index (CTDI volume) is ≈17 cGy.
14,15
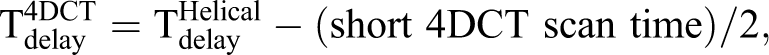
Planning and Contouring
Following the acquisition of both 4DCT data sets, an average image data set as well as 0% phase and 50% phase from both the short and the long 4DCT scans are fused together. The fusion of all 6 CT data sets is very straightforward, since both 4DCT scans are done one after another and the patient’s position is not altered. Thus, the fusion registration parameters, translation and rotation, are set to zero for all the scans. The average image data set derived from the long 4DCT is the primary image on which the treatment plan is created.
Target Definition
Images derived from the short 4DCT (0%, 50%, and average) are used for contouring the gross tumor volume(s) (GTVs) and generating internal target volume (ITV), which is defined as the summation of all GTVs in all respiratory phases. The planning target volume is an isotropic 0.5 cm expansion around the ITV to compensate for the uncertainties of set-up and organ motion. 16 Note that since the long 4DCT is not used for target contouring, the time delay between the contrast injection and the long 4DCT scan is not important.
Organs at Risk Delineation
Images derived from the average long 4DCT are used to contour organs at risk (OARs): heart, stomach, duodenum, bowel, chest wall, spinal cord, kidneys, and skin.
Liver: images derived from the long 4DCT (0%, 50% phase, and average) are used to contour the whole liver at the maximum inhale and maximum exhale positions.
Setup and Treatment Delivery
For treatment delivery, the patient is set up under the same conditions as that used for radiotherapy planning. The planning CT (average) as well as the liver contours (liver 0% and liver 50%) are exported to the on-board cone-beam CT on the treatment unit for patient setup verification.
Results
Figure 2 illustrates an example of 4DCT images obtained during CT simulation of a patient with a solitary liver metastasis. The images on the left were acquired during the short 4DCT, whereas images on the right were acquired during the long 4DCT scan. The arrows indicate position of the disease. Axial view of maximum inhale (Figure 2A and B) and maximum exhale (Figure 2C and D) phases of the breathing cycle is shown in Figure 2. In addition, axial (Figure 2E and F) and coronal (Figure 2G and H) views of the average CT data set are shown in Figure 2. The metastatic deposit exhibits low contrast relative to surrounding liver parenchyma. An absolute difference of 2 Hounsfield units (HU) between the tumor (76 HU) and the normal liver parenchyma (78 HU) was found on the long scan without contrast (Figure 2B, D, F, and H). The use of an optimally timed injection enhances the tumor visibility by increasing the difference to 35 HU (65 vs 100 HU, respectively; Figure 2A, C, E, and G). The long 4DCT scan is performed just a few minutes after the short 4DCT scan, and it is clear that the tumor visibility is reduced dramatically due to fast contrast washout. Note that the short 4DCT scan covers significantly smaller volume than the long 4DCT scan (Figure 2G and H), making synchronization of the contrast injection and 4DCT scan possible.

The images corresponding to the short and long 4DCT are on the left and right, respectively; (A) maximum inhale phase, short 4DCT; (B) maximum inhale phase, long 4DCT; (C) maximum exhale phase, short 4DCT; (D) maximum exhale phase, long 4DCT; (E) average image data set derived from short 4DCT; (F) average image data set derived from long 4DCT; (G) coronal view of the average image data set, short 4DCT; and (H) coronal view of the average image data set, long 4DCT. The arrows on the left hand side indicate location of the disease. On the long scans (B, D, F, and H), the normal liver parenchyma has a density of 78 Hounsfield units (HU) and the tumor 76 HU (difference of 2 HU), whereas with the use of an optimally timed injection on the short scans (A, C, E, and G), the normal liver parenchyma density is 100 HU and the tumor 65 HU (difference of 35 HU). 4DCT, 4-dimensional computed tomography.
Discussion
Delivering SBRT to liver lesions remains a challenge. Large intrafraction motion from respiration, organ deformation, and poor visibility on standard noncontrast CT simulation scans are major concerns when treating liver lesions and contribute significantly to the uncertainty of the target contouring done by radiation oncologist. With highly conformal radiation treatments like SBRT becoming more popular, the resulting contouring uncertainty may potentially lead to geographic miss during treatment.
There are multiple methods for imaging liver in diagnostic settings. 10,11 Radiation oncologists often use diagnostic images as a guide to locate the tumors on the planning CT images and delineation of the target volume. Multimodal imaging with contrast-enhanced magnetic resonance imaging (MRI) and/or positron emission tomography (PET) is useful for better target definition. However, they do not provide tumor motion information in the diagnostic images, cannot be used for radiotherapy planning, and are not widely available. Hence, accurate delineation of the target based on diagnostic images is suboptimal.
For target visualization with diagnostic CT scans, timed injection of intravenous contrast is critical. 11,12 The time delay between contrast injection and initiation of the CT scan varies depending on the tumor histology. With the liver blood supply divided approximately 20%/80% between hepatic artery and portal venous system, there are 3 distinct phases of liver imaging: hepatic arterial phase (15-20 seconds time delay), late arterial phase (25-30 seconds time delay), and hepatic venous phase (60-70 seconds time delay). 10 The majority of liver metastases are hypovascular and as such, they are most prominent during the hepatic venous phase, when liver parenchyma is strongly enhanced by the contrast. This includes lung, colon, lymphoma, and genitourinary tumors. Hypervascular tumors, like neuroendocrine tumors, melanoma, sarcoma, carcinoid, renal cell, thyroid, and choriocarcinoma, are best observed during arterial phase. In some cases, scans performed in the equilibrium phase, that is, several minutes after contrast injection, can be beneficial. Due to the complexity of the CT contrast protocols used for liver imaging, we recommend that individual centers consult with their radiologist.
To account for liver motion, an abdominal compression device is one of the options available to reduce organ motion related to respiratory excursion. 13 The latter can be evaluated by a 4DCT scan. During a 4DCT scan, several CT data sets are acquired, corresponding to various phases of the breathing cycle. When combined, the resulting data sets provide for accurate representation of the breathing-induced motion. Therefore, they can be used for delineation of the ITV. To capture the full extent of the motion, the image acquisition for any particular volume should be done at least over 1 complete breathing cycle. Hence, the image acquisition time of a 4DCT (∼2 minutes) significantly exceeds that of a helical CT. When combined with intravenous contrast injection, the ability to obtain optimal visualization of the target lesion in the proper phase (eg, hepatic venous phase) is problematic. Although significantly reducing the volume to be imaged on the 4DCT would reduce scan time (40-60 seconds), it would not be practical as the surrounding OARs (eg, kidneys, liver, heart, and bowel) may not be included in the scan. Calculating proper dose volume histograms for the OARs would not be feasible without including the entire organ in the scan. In addition, the scan volume should be large enough to allow for potential noncoplanar radiation beams. It should also include the full extent of the diaphragm motion. Since liver metastases are usually not visible on cone beam images, one approach is to use the whole liver or the right dome of the diaphragm in its maximum inhale and maximum exhale phases as a surrogate for patient setup verification. Narrowing the gap between diagnostic CT images and 4DCT simulation images, the use of two 4DCT scans, short and long, as described in this article allows for proper synchronization of the contrast injection and better definition of the target lesion, as compared to a single 4DCT without contrast or a helical CT scan with timed contrast but no motion information.
An alternative would be to use fiducial markers for target localization. Percutaneous fiducial implantation is a mildly invasive procedure with a related risk of seeding and migration. 17 Other emerging options of better identifying the target lesion include functional imaging (eg, functional MRI, respiratory-gated PET-based target volume delineation). 18 -20 However, these options have not been extensively studied for radiation planning and thus remain investigational. Furthermore, these new imaging tools are costly and thus remain limited in use. In contrast, our “less” sophisticated approach to contrast-enhanced CT simulation scans for SBRT of the liver is simple, relatively inexpensive, and can easily be adopted in many radiotherapy centers globally.
The main limitation of our approach is that the optimal contrast enhancement of the tumor in the short scan is highly dependent on the calculation of the optimal timing delay from contrast injection to initiating the CT scan. 4 Therefore, a discussion with the radiologist is warranted. In our experience, with an optimally timed intravenous contrast injection, it is unusual not to be able to visualize the liver lesion even as small as 0.5 cm. In the rare cases where despite everything, the lesion is too small to be seen, delaying the treatment of the specific lesion is our standard approach.
Conclusion
Stereotactic body radiation therapy for liver metastases is challenging due to poor target visibility and significant breathing motion. This presents a considerable hurdle for radiation oncologists in target delineation. The simulation technique presented here combines two 4DCT scans and accurately timed contrast injection. This approach represents a simple and effective method to allow for better target definition when treating liver metastasis, without being invasive.
With current advances of liver imaging and the use of functional imaging for simulation, 14 it would be of considerable interest to compare the impact of these emerging imaging tools to our proposed method in terms of patient outcomes and relative costs of these techniques.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
