Abstract
Objective:
To explore the expressions of annexin A2 in bladder cancer cell lines and bladder cancer tissues, we want to find the relationship among annexin A2, drug resistance, and recurrence of bladder cancer.
Methods:
Our laboratory established the PUMC-91 bladder cancer cell line against gradient concentration of Adriamycin (0.3, 0.6, and 1.0 μg/mL), and we also collected 60 cases of surgically resected bladder cancer recurrent tissue samples. The tissues were classified into 2 groups according to the frequency of recurrence (<6 months and >2 years) after initial surgery. The method of immunohistochemistry was used to examine the differences in the expression of annexin A2.
Results:
There were statistical differences in annexin A2 among normal bladder epithelial cell line SV-HUC-1, PUMC-91, PUMC-91 against 0.3 μg/mL Adriamycin, and PUMC-91 against 1.0 μg/mL Adriamycin (P < .05). The expressions of Annexin A2 were found to be higher than those that recurred at >2 years (P = .002) in the bladder cancer tissues and that recurred at <6 months after initial surgery. It was also associated with invasion depth (stage) of bladder cancer, such as higher expression in T2 (invasive muscular) group than Tis (carcinoma in situ) and T1 (invasive mucosa lamina propria) groups (P = .003 and P = .000, respectively). But, it did not correlate with the differentiation (grade) of cancer cells in bladder cancer tissues (P = .593).
Conclusion:
Annexin A2 can act as a valuable biomarker for predicting the drug resistance and recurrence of bladder cancer.
Introduction
Bladder cancer is the ninth most common malignant tumor in the world 1 and is the fourth in men and the tenth in women in China. As we all know, bladder cancer has a tendency for metastases and recurrence after initial surgery. About 15% of patients with bladder cancer develop muscle invasive tumor after initial diagnosis. 2 Currently, drug resistance is the main obstacle for the successful chemotherapy in clinical human bladder cancer therapy. Drug resistance after surgical operations significantly increases the rate of cancer recurrence.
Multiple genetic mutations cause the development and progression of bladder cancer. 3 There have been many molecular biomarkers that are associated with the tumorigenesis and the development of bladder cancer, for example, genes CCNB2, KIF20A, and TOP2A, which overregulated in bladder cancer. Compared with normal human tissue samples and body fluids, there exists abnormal expression of certain protein biomarkers due to mutation of genes in the bodies of patients with cancer. So, finding a suitable tumor biomarker to predict, diagnose, and treat bladder cancer is becoming necessary and vital.
Annexin A2 (ANXA2) is a member of annexin family, which binds to negatively charged phospholipids in a Ca2+-dependent manner. 4 Accumulating evidence suggest ANXA2 is a multifunctional protein 5 which participates in many cellular activities such as cell proliferation, 6 cell cycle regulation, 7 and DNA replication. 8,9 Annexin A2 normally combines with p11 to form a heterotetramer. It is important to note that the percentages of heterotetramers present are not the same in different organ cells. 10 Heterotetramers localize with cytoskeletons, 11 and monomers are expressed in the cytoplasm, nuclei, and cellular membranes. 12 -14 According to the many published reports, ANXA2 is expressed differentially in many human tumors. But the relationship between ANXA2 expressions and human bladder cancer has not been reported.
In this study, we cultivated a normal bladder epithelial cell line (SV-HUC-1), a bladder cancer cell line (PUMC-91), and 3 bladder cancer cell lines PUMC-91 against gradient dose of Adriamycin (ADM; 0.3, 0.6, and 1.0 μg/mL), and we also collected human bladder cancer tissues that had recurred at 2 different intervals (<6 months and >2 years). We used immunocytochemistry to verify the expression of ANXA2 on cultured epithelial cell lines (SV-HUC-1, PUMC-91, and PUMC-91/ADM [0.3, 0.6, and 1.0 μg/mL]) in order to determine whether ANXA2 was a key regulator in drug resistance. We further used immunohistochemistry, exploring factors of frequency of recurrence, invasion depth, and differentiation on resected bladder cancer tissues to verify the role of ANXA2 in the recurrence of human bladder cancer.
Methods and Materials
Cell Culture
Normal human bladder epithelium cell line SV-HUC-1 was bought from the cell bank of the Chinese Academy of the Sciences. Bladder cancer cell line PUMC-91 was granted from Peking Union Medical College Hospital. Our laboratory has established PUMC-91 cell lines against gradient dose of ADM (0.3, 0.6, and 1.0 μg/mL). We induced and established the drug-resistant bladder cancer cell lines PUMC-91/ADM by increasing the use of ADM (Dingguo, China). To preserve the biologic characteristics of multidrug resistant, PUMC-91/ADM cell lines were cultured in RPMI 1640 medium with 18% fetal calf serum and 0.3, 0.6, and 1.0 μg/mL ADM. The ability of drug resistance was evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. The cell cycle and the dose of ADM were determined by fluorescence flow cytometry. The details of PUMC-91 cell line established could be seen in the article that was published in the Journal of Medical Research. 15 The SV-HUC-1 cell line was cultured in F12K supplemented with 10% fetal bovine serum (Hyclone, Utah). The PUMC-91 cell line was cultured in RPMI 1640 supplemented with 10% fetal bovine serum (Dingguo, China). The PUMC-91/ADM cell lines were cultured in RPMI 1640 medium with 18% fetal bovine serum. All the cell lines were supplemented with 100 U/mL streptomycin at 37°C in a humidified atmosphere of 5% CO2.
Patient Tissue Samples
A total of 60 tissue section samples were analyzed. All the samples were bought from Beijing University Health Science Center. The tissue samples were all bladder cancer tissues that recurred after initial surgery. All the tissue samples were first grouped by the frequency of recurrence (<6 months and >2 years), then regrouped by invasion depth (stage), and regrouped once again by differentiation (grade). The details are listed in Table 1. Clinical parameters of invasion depth were divided into Tis (carcinoma in situ), T1 (invasive mucosa lamina propria), and T2 (invasive muscular). Clinical parameters of differentiation were divided into high differentiation and low differentiation. This study had gained approval from the institutional review board for the protection of human subjects, and all participants were given informed consent.
Tissue Characteristics of Patients With Bladder Cancer Recurrence.

Abbreviations: HG, high differentiation; LG, low differentiation; SD, standard deviation; T1, invasive mucosa lamina propria; T2, invasive muscular; Tis, carcinoma in situ.
a Age is presented as mean ± SD.
Immunohistochemistry
All the cell lines were cultured on glass coverslips in the corresponding medium for 3 to 4 days. Then, cells were rinsed twice with phosphate-buffered saline (PBS). The paraffin-embedded tissue samples were sectioned into 5-mm slices. The tissue sections must be dewaxed 30 minutes by xylene and then dehydrated by 100%, 100%, 95%, and 75% alcohol, each 4 minutes. All the cell lines and tissue sections were fixed by 4% paraformaldehyde for 30 minutes. After 3 rinses using PBS for 15 minutes, endogenous peroxidase (Dingguo Biotechnology, China) was blocked for 20 minutes at room temperature to deprive endogenous peroxidase activity. After antigen retrieval, the samples were incubated at 4°C overnight with rabbit polyclonal ANXA2 antibody (Abcam [USA]) at the dilution of 1:300 in PBS. Then, the samples were rinsed with PBS, incubated in horseradish peroxidase-conjugated secondary antibody (Dako [USA]) for 30 minutes at 37°C, and were again rinsed with PBS for 15 minutes; samples were stained using chromogen 3,3′-diaminobenzidine solution (Dako, USA) for 2 minutes. Then, the samples were counterstained with hematoxylin for 2 minutes, dehydrated with 75%, 95%, 100%, 100% alcohol, and cleaned with xylene and sealed with the natural gum.
Assessment of ANXA2 in Immunohistochemistry
According to the report by Hendricks and Milkinson, 16 the result of immunostaining could be quantified by image software. We used the image software of KODAK MI SE (Fujisiao, China) to quantify the staining results of ANXA2 in different cell lines. The results were shown by calculating the gray value. Semiquantitative immunohistochemical detection was used to determine the ANXA2 protein level of tissue samples. All the scores were decided under double-blind conditions by 3 independent pathologists. Cytoplasm immunoreactivity for ANXA2 protein was scored by evaluating the percentage and the intensity of positive cancer cells. The information recorded included the subcellular location of ANXA2 staining, the intensity of staining (grade 0 = negative, 1 = weak, 2 = moderate, and 3 = strong), and the percentage of staining cells: “0” (0%), “1” (1%-10%), “2” (11%-50%), “3” (51%-80%), and “4” (81%-100%). A score was acquired for each tissue section using the formula: the intensity of staining × the percentage of cells stained. Then, the scores were then classified as follow: “−” (score 0), “+” (score 1-4), “++” (score 5-8), and “+++” (score 9-12). To do the statistical analysis, the tissues were grouped into low expression group (“−” and “+”) and high expression group (“++” and “+++”).
Statistical Analysis
The software of SPSS 16.0 was used to do statistical analysis. Student t test (2-tailed) was used to analyze the differential expression of ANXA2 between different recurrence groups. The expression of ANXA2 among different cell lines was tested by 1-way analysis of variance. P < .05 was accepted as statistically significant difference.
Results
Expression of ANXA2 Correlated With Drug Resistance in Different Cell Lines
In this study, our focus on a gradient of ADM showed that ANXA2 was mostly expressed in the cell membrane and cytoplasm of bladder cancer cell lines by immunocytochemistry, the same as the normal bladder epithelial cell line. Annexin A2 had differential expressions among SV-HUC-1, PUMC-91, and PUMC-91/ADM (0.3, 0.6, and 1.0 μg/mL). The results are presented in Figure 1.

Annexin A2 was differentially expressed in above cell lines by immunocytochemistry. Annexin A2 immunostaining of different localizations and intensities was expressed in above cell lines (A, positive control; B, SV-HUC-1; C, PUMC-91; D, PUMC-91/0.3 ADM [0.3 μg/mL]; E, PUMC-91/0.6 ADM [0.6 μg/mL]; F, PUMC-91/1.0 ADM [1.0 μg/mL]). Annexin A2 was localized in the cell membrane and cytoplasm in bladder cancer cell (×1000 magnifications). Here, β actin acted as positive control.
The results were quantified by the standard of gray value, using KODAK MI SE software. In the SV-HUC-1, PUMC-91, PUMC-91/0.3 ADM, PUMC-91/0.6 ADM, and PUMC-91/1.0 ADM cell lines, the gray values were 63.44 ± 6.99, 89.48 ± 9.52, 104.56 ± 15.26, 105.59 ± 19.49, and 119.24 ± 17.61, respectively. The differences among SV-HUC-1, PUMC-91, PUMC-91/0.3 ADM, and PUMC-91/1.0 ADM had statistical significance, and the statistical analyses are shown in Figure 2. Meanwhile, PUMC-91/0.3 ADM and PUMC-91/0.6 ADM had no statistical significance (P = .890), the same as PUMC-91/0.6 ADM and PUMC-91/1.0 ADM (P = .066).

The differential expressions of annexin A2 among SV-HUC-1, PUMC-91, PUMC-91/0.3 ADM (0.3 μg/mL), and PUMC-91/1.0 ADM (1.0 μg/mL) had statistical significance by immunocytochemistry. The software of SPSS 16.0 was used to analyze the statistical results. Expression of ANXA2 among different cell lines was tested by 1-way analysis of variance (ANOVA). P < .05 was accepted as statistically significant difference.
Relationship of ANXA2 Expressions With Recurrence of Bladder Cancer
The expressions of ANXA2 were higher in bladder cancer tissues than normal bladder epithelial tissue (Figure 3). In the bladder cancer tissues which recurred <6 months, expressions of ANXA2 were found to be higher than those which recurred >2 years (Figure 4; P = .002). The expression levels of ANXA2 in <6 months recurrence tissue samples were 1.39 times higher than the samples which recurred >2 years. The results suggested ANXA2 functions had a critical role in the tumorigenesis and recurrence of bladder cancer.

The differential expression of annexin A2 between the normal bladder epithelial tissue (Figure A) and bladder cancer tissue (Figure B) was detected by immunohistochemistry (×1000 magnifications). Compared with normal bladder epithelial tissue, annexin A2 was highly expressed in bladder cancer tissue.

Annexin A2 was highly expressed in patients with bladder cancer recurred <6 months (A) compared to >2 years (B) by immunohistochemistry. There was very strong staining of annexin A2 in cell membrane and cytoplasm of bladder cancer tissue samples that recurred <6 months. The difference was tested by Student t test (2 tailed; P = .002). Annexin A2 was associated with the frequency of the bladder cancer recurrence (×1000 magnification).
Expression of ANXA2 in Bladder Cancer Tissues With Clinical Parameters
The expression of ANXA2 was further explored to investigate whether it is associated with clinical parameters, specifically invasion depth and cell differentiation. Significant statistical differences were found for invasion depth. Figure 5 showed the expressions of ANXA2 were higher in the T2 group than the Tis group and the T1 group (P = .000, P = .003, respectively). The results of ANXA2 expression in bladder cancer tissues did not correlate with the grading of tumor (Figure 6). The differences were tested by the t test analysis.
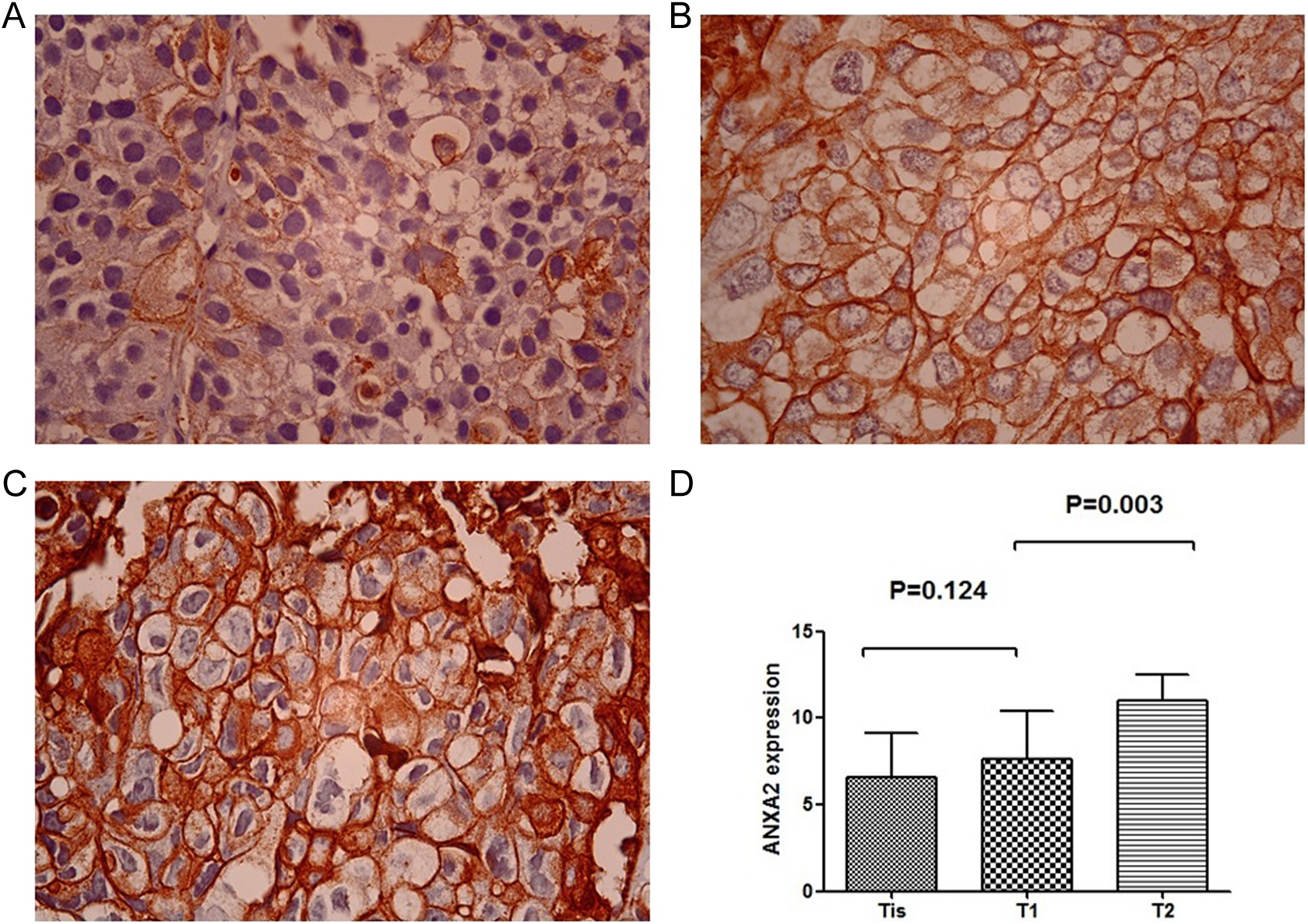
Annexin A2 expression was detected higher in the T2 human bladder cancer tissue samples (Figure C) compared with the Tis (Figure A) and T1 groups (Figure B). Annexin A2 had differential expressions between Tis and T2 groups (P = .000) as well as T1 and T2 groups (P = .003). The expression of annexin A2 was higher in the T2 group than the Tis and T1 groups. The difference had statistical significance (×1000 magnifications). Annexin A2 was associated with the stage of bladder cancer.

Annexin A2 expression was observed in different differentiation (high differentiation (Figure A) and low differentiation (Figure B)) of human bladder cancer tissue samples by immunohistochemistry (×1000 magnification). The difference was verified by Student t test (2 tailed; P = .593). The difference had no statistical significance (P > .05). Annexin A2 did not correlate with the grade of bladder cancer.
Discussion
Bladder cancer is a frequently occurring tumor, and more than 50% of superficial cancer recurs within 5 years. 17 The 5-year survival rates of bladder cancer range from 40% to 80%. 18 The survival rates correlate closely with the invasion, cancer cell differentiation, and recurrence of bladder cancer. It is thus important to detect the cancer early before cancer cells become invasive. Clinical monitoring after initial surgery uses cystoscopy which is complex and brings psychological burden to the patients. The clinical therapy of bladder cancer after initial surgery is always chemotherapy, and the formation of drug resistance is the most troublesome problem. Further more, we use the recurrence to evaluate the curative effect of the therapy. The progression of bladder cancer is a multistep outcome, and it has been studied for clinical parameters that could assist to predict recurrence and progression of bladder cancer. 19 Tada et al have studied the frequency of recurrence, and they found patients with bladder cancer having recurrence at 3 months have frequent recurrences. 20 That suggests the frequency of recurrence has a significant impact in the progression of bladder cancer.
Recent reports show ANXA2 plays a crucial role in carcinogenesis and correlates with clinical parameters in human cancers. Ponlapat (Yonglitthipagon) and colleagues found ANXA2 could act as a prognostic marker in cholangiocarcinoma that was caused by opisthorchis viverrini. In the cholangiocarcinoma cells, ANXA2 was upregulated. 21 Maxwell et al also studied ANXA2 associated with oxidative processes and inflammation in stage 1 endometrial cancer tissues based on proteomic analysis. 22 The expressions of ANXA2 also correlated with cervical cancer 23 and pancreatic cancer. 24 Annexin A2 also played a critical role in the drug resistance model. Zhang et al found ANXA2 was a key regulator in the formation and progression in MCF-7/ADR cell line. 25 Gong and colleagues also suggested the spliced segment of ANXA2 participated in pancreatic cancer cells which resisted against gemcitabine. 26 In bladder cancer, Meng et al studied a bladder cancer cell line PUMC-91/ADM. Annexin A2 was expressed higher than its parental cell line. 27 Yu et al reported annexin A1 was correlated with the drug resistance and recurrence of bladder cancer. 28 However, they did not study the relationship between ANXA2 and gradient drug resistance or the relationship between ANXA2 and bladder cancer clinical parameters, and the relationship between recurrence frequency and ANXA2 had not been studied.
Our laboratory’s previously published article on PUMC-91 reported that ANXA2 was more highly expressed in PUMC-91/ADM (1.0 μg/mL) cell line compared with the PUMC-91 cell line without exposure to ADM, based on 2-dimensional polyacrylamide gel electrophoresis (2D-PAGE) proteomic approaches. All the analyses were repeated 3 times. The optical density value of ANXA2 protein spots and statistical graph of ANXA2 between PUMC-91 and PUMC-91/1.0 ADM cell lines were also analyzed. The whole 2D gel image and the condition (such as pH range and gel concentration) of the 2D-PAGE experiment by our laboratory are described in the article by Meng et al. 27
In the present study, we showed the expression of ANXA2 was increased on a gradient in the SV-HUC-1, PUMC-91, PUMC-91/0.3 ADM, and PUMC-91/1.0 ADM cell lines. These results suggested that ANXA2 was associated with the drug resistance of bladder cancer. In bladder cancer recurrence tissue samples, expression of ANXA2 was correlated with the invasion depth and the recurrence frequency of bladder cancer. In the samples that recurred at <6 months, ANXA2 was expressed more highly than those that recurred at >2 years. The data indicate a role for ANXA2 as a potential biomarker in bladder cancer progression. There is a strong relationship between drug resistance and recurrence after initial surgery. The expression of ANXA2 should help to solve the problem. This study confirms the crucial role of ANXA2 in the pathogenesis and drug resistance of bladder cancer. But, why differentiation was not associated with ANXA2 expression yet drug resistance, recurrence, and invasiveness, that might be the exact mechanism of ANXA2 participating in cancer development and progress is not unclear. Annexin A2 is independently correlated with the frequency of recurrence in bladder cancer tissue samples. However, it is noteworthy that further investigation should be required as to the mechanism of ANXA2 participating in bladder cancer drug resistance and recurrence. These discoveries demonstrate that ANXA2 has a key role in both drug resistance and recurrence of bladder cancer. Annexin A2 may offer better guidance and diagnosis in clinical bladder cancer therapy.
Footnotes
Acknowledgments
All authors would like to thank all the staffs in clinical laboratory of Beijing Shijitan Hospital for enthusiastic assistance.
Authors’ Note
The authors alone are responsible for the content of this article. This study had gained approval from the institutional review board for the protection of human subjects, and all the participants were given informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project has received funding from Beijing Key Laboratory of Urinary Cellular Molecular Diagnostics (Z151100001615066) and Capital Health Research and Development of Special (2011-2008-02).
