Abstract
Introduction
Immune checkpoint inhibitors (ICIs) are extensively utilized in lung cancer patients, with documented instances of ICIs-associated acute kidney injury (ICIs-AKI). This study aims to explore a model for early recognition of ICIs-AKI.
Methods
The study involved 413 adult lung cancer patients treated with ICIs at Ningbo No.2 Hospital between Sept. 1, 2021, and June 30, 2023. Patients were followed until death or Dec. 31, 2023, and categorized into ICIs-AKI or non-AKI groups. We employed univariate and multivariate logistic regression to identify risk factors, developed both logistic regression and Multilayer Perceptron (MLP) prediction models, and used Kaplan-Meier survival analysis to assess prognosis.
Results
The study included 381 lung cancer patients receiving ICIs treatment after excluding 32 patients. ICIs-AKI occurred in 13.39% of cases, with a median onset time of [123 (63, 303)] days. Multivariable logistic analysis identified diabetes, proteinuria, extrarenal irAEs, diuretic use, and chemotherapy as significant risk factors (all P < 0.05), while higher baseline eGFR levels were protective (P < 0.05). Two prediction models were developed: logistic regression (AUC=0.877, sensitivity=0.922, specificity=0.726) and MLP (AUC=0.950, accuracy=0.843, precision=0.847). Survival analysis showed no difference in overall survival between ICIs-AKI and non-AKI groups (HR = 1.021, 95% CI = 0.629-1.659, P = 0.932; adjusted HR = 0.950, 95% CI = 0.558-1.616, P = 0.849). AKI to CKD progression incidence was 58.82%, with no significant difference in overall survival between CKD and non-CKD groups (P = 0.157).
Conclusion
This study offers detailed insights into ICIs-AKI, including its rate, onset timing, risk factors, and clinical features. Approximately half of the affected patients experienced spontaneous renal function recovery. Both logistic regression and MLP models effectively predicted ICIs-AKI. Importantly, neither ICIs-AKI incidence nor renal function restoration correlated with patient mortality. These findings underscore the importance of early detection and management strategies.
Introduction
Lung cancer emerged as the most commonly diagnosed cancer and the leading cause of cancer-related deaths in 2022, representing approximately one in 8 (12.4%) of all cancer diagnoses and 18.7% of cancer-related deaths. 1 Oncologists face an urgent need to develop novel treatments that effectively target tumor cells to address this widespread challenge. Immune checkpoint inhibitors (ICIs) have emerged as promising interventions that enhance immune responses against tumors by activating T cells to eliminate malignant cells, leading to significant improvements in survival rates across various cancer types, particularly melanoma and lung cancer. 2 With a growing body of clinical evidence, treatment regimens based on ICIs have gained approval as frontline therapies for lung cancer. 3
Unfortunately, ICIs have been shown to trigger autoimmune toxicities, leading to immune-related adverse events (irAEs) that can affect nearly any organ. 4 The incidence rates of irAEs in patients undergoing ICIs treatment vary widely, ranging from 15% to 90%, with the most commonly observed affecting the skin, gastrointestinal tract, endocrine system, and liver. 5 Despite the relatively low frequency of renal-associated irAEs, the expanding literature on the topic has facilitated a deeper understanding and recognition of immune checkpoint inhibitor-related acute kidney injury (ICIs-AKI). Acute interstitial nephritis (AIN) has been identified as the predominant pathology associated with ICIs, accounting for approximately 90% of biopsied patients. 6 Additionally, cases of lupus nephritis, IgA nephropathy, thrombotic microangiopathy, minimal change nephrosis, membranous nephropathy, and focal segmental glomerulosclerosis have been reported. 7
Previous studies have examined the incidence and risk factors associated with ICIs-AKI. The study by Tan and Sprangers highlighted that exposure to proton pump inhibitors (PPIs) correlated with the emergence of clinically significant short- and long-term kidney-related adverse effects in ICIs-treated patients. However, establishing causality remains a challenge. 8 Additionally, exposure to PPIs and nonsteroidal anti-inflammatory drugs (NSAIDs) was associated with an increased odds ratio (OR) for the risk of ICIs-AKI. 9 While several authors have noted an association between low baseline estimated glomerular filtration rates (eGFR) and ICIs-AKI, this finding has not been consistent across studies. 10 Recently, some studies have described significantly higher risks of irAEs occurrence in patients who received pembrolizumab, did not have central nervous system metastases, had a history of autoimmune disorders, and underwent chemotherapy in combination with ICIs. Race, socioeconomic status, prior radiation therapy, and comorbidity burden were identified as factors associated with the development of specific types of irAEs. 11 Nevertheless, reliable biomarkers for predicting the occurrence of ICIs-AKI are still lacking.
In summary, this study conducted a retrospective analysis of lung cancer patients undergoing ICIs treatment, aiming to elucidate the incidence and clinical characteristics of ICIs-AKI in real-world settings. The study seeks to identify effective predictors of ICIs-AKI and develop a predictive model for its occurrence. Additionally, long-term clinical outcomes and renal recovery after AKI were evaluated through follow-up assessments.
Materials and Methods
The reporting of this study conforms to the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) guideline. 12
Study Subjects
A total of 413 adult patients diagnosed with lung cancer who underwent ICIs treatment were recruited from Sept. 1, 2021 to June 30, 2023 at Ningbo No.2 Hospital. Inclusion criteria for this study were as follows: (1) age ≥ 18 years; (2) diagnosis of primary lung cancer according to the Guidelines for the Diagnosis and Treatment of Primary Lung Cancer in 2023 3 ; (3) receipt of at least one course of ICIs treatment. Exclusion criteria were as follows: (1) presence of end-stage renal disease, renal transplant, or undergoing renal replacement therapy; (2) incomplete clinical data; and (3) acute kidney injury attributed to other causes such as hypovolemia, urinary obstruction, or other factors. The enrollment process is illustrated in Figure 1. This study was approved by the Ethics Committee of Ningbo No.2 Hospital (YJ-NBEY-KY-2023-013-01).

Enrollment scheme in this study.
All included patients were diagnosed with primary lung cancer, with pathological subtypes including adenocarcinoma, squamous cell carcinoma, small cell lung cancer, and others. The line of immunotherapy (first-line, second-line, or beyond) at the time of study enrollment was recorded based on treatment history. For patients receiving ICIs in combination with chemotherapy or as sequential therapy, detailed timelines of each treatment modality were collected.
Definition and Diagnostic Criteria
AKI was defined in accordance with KDIGO guidelines 13 as meeting any of the following criteria (not graded): an increase in sCr by ≥0.3 mg/dl (≥26.5 μmol/L) within 48 h; or an increase in sCr to ≥1.5 times the baseline, which is known or presumed to have occurred within the prior 7 days; or urine volume <0.5 ml/kg/hour for 6 h. In this study, a diagnosis of ICIs-AKI required: (1) meeting the KDIGO AKI criteria during or after ICI therapy; (2) exclusion of other predominant causes of AKI (eg, urinary obstruction, severe hypotension, sepsis) through clinical assessment; and (3) a temporal association with ICI administration. For patients receiving concurrent chemotherapy (eg, cisplatin), careful adjudication was performed to distinguish between chemotherapy-induced nephrotoxicity and ICIs-AKI, based on the timing of AKI onset relative to chemotherapy cycles, the presence of extrarenal irAEs supporting an immune-mediated mechanism, and, when available, renal biopsy findings.
Staging of AKI according to KDIGO 13 : (1) Stage 1: absolute increase in sCr ≥ 0.3 mg/dL (≥26.5 μmol/L) or ≥ 1.5 to 2.0 fold from baseline; (2) Stage 2: increase in sCr > 2.0 to 3.0 fold from baseline; (3) Stage 3: increase in sCr > 3 fold from baseline or increase of sCr to ≥4.0 mg/dL (≥ 354 μmol/L), or initiation of renal replacement therapy.
Chronic kidney disease (CKD) was defined as renal structural or functional abnormalities persisting for ≥3 months due to various causes according to KDIGO. 13
Staging of CKD: (1) Stage 1: eGFR≥90 ml/min/1.73m2; (2) Stage 2: 60 ≤ eGFR<90 ml/min/1.73m2; (3) Stage 3: 30 ≤ eGFR<60 ml/min/1.73m2; (4) Stage 4: 15 ≤ eGFR<30 ml/min/1.73m2; (5) Stage 5: eGFR<15 ml/min/1.73m2.
Follow up
The patients were followed up until death or Dec. 31, 2023. Based on disease progression, participants were categorized into two groups: (1) ICIs-AKI group; (2) non-AKI group. The primary outcome assessed was overall survival, defined as the duration from the initiation of ICIs therapy to death from any cause. CKD staging was determined based on eGFR levels three months after AKI onset. Patients were classified into CKD stages 3–5 as one subgroup and CKD stages 1–2 as another subgroup, further divided into: (1) CKD group; (2) non-CKD group.
Data Collection
Basic information on study subjects was collected, including age, gender, height, weight, duration of ICIs treatment, history of hypertension, history of diabetes, and medication history such as PPIs, NSAIDs, antibiotics, chemotherapy, and radiation therapy. Laboratory data included white blood cell count (WBC), hemoglobin (Hb), lymphocyte ratio, triglycerides (TC), albumin (ALB), creatinine (Cr), blood urea nitrogen (BUN), uric acid (UA), and liver function parameters such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Besides, electrolyte levels, proteinuria, and medical history of extrarenal irAEs were also recorded. Additionally, history of radiotherapy (including treatment site, dose, and timing relative to ICIs initiation) was recorded to assess its potential confounding effect.
BMI was calculated as weight in kilograms divided by the square of height in meters. Residual renal function (RRF), represented by the glomerular filtration rate (GFR), was assessed using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation (CKD-EPI). For males with sCr≤0.9 mg/dL, the equation is 141 × (sCr/0.9)−0.411 × 0.993Age; for sCr > 0.9 mg/dl, it is 141 × (sCr/0.9)−1.209 × 0.993Age. For females with sCr≤0.7 mg/dL, the equation is 144 × (sCr/0.7)−0.329 × 0.993Age; for sCr > 0.7 mg/dL, it is 144 × (sCr/0.7)−1.209 × 0.993Age. IrAEs were defined as encephalitis, myocarditis, pneumonitis, hepatitis, thyroiditis, colitis, etc Baseline laboratory tests were conducted at Ningbo No. 2 Hospital's laboratory within two weeks before initiating ICIs treatment to evaluate the occurrence of ICIs-AKI. For patients with multiple laboratory measurements during this period, the value closest to the initiation of ICIs therapy (but prior to it) was selected as the baseline value.
All data items specified in the protocol were collected retrospectively from electronic medical records. Among the 381 included patients, a small proportion of data points were missing for certain laboratory variables (eg, lymphocyte ratio, triglycerides). For these variables, missing values were imputed using the median value of the non-missing data from the entire cohort. This method was chosen for its robustness against outliers. Crucially, the outcome variable (ICIs-AKI status) and all key clinical predictors used in the final multivariate model (eg, diabetes, proteinuria, eGFR, extrarenal irAEs) had complete data with no missing values. The overall rate of missing data for imputed variables was less than 5%, which is unlikely to introduce substantial bias.
Construction and Evaluation of MLP Predictive Models
The Multilayer Perceptron (MLP) stands as a classic form of feedforward neural network, esteemed for its reliability, flexibility, nonlinearity, and widespread applicability in neural networks. Comprising multiple layers of neurons, including input, hidden, and output layers, the MLP operates by receiving raw data through its input layer, which is then processed through hidden layers, serving as abstract interfaces. Ultimately, the output layer generates predictions or class labels based on the given problem. Renowned for its self-learning and modeling capabilities, the MLP is adept at handling nonlinear and complex problem domains while exhibiting strong generalization abilities. Upon completion of training, the MLP model can discern correlations within unseen real-world data, rendering it invaluable for predictive analysis and adeptly managing large datasets. Through adjustments to network complexity and weight values, it can effectively navigates parameter complexity. The non-parametric nature of the MLP enables it to minimizing errors in parameter estimation. Furthermore, the utilization of the MLP classifier imposes no constraints on the overall distribution of input data, presenting a notable advantage in its application.
After selecting pertinent factors from all independent variables, patients were partitioned into a 70% training set and a 30% testing set. The MLP was employed as the classifier in our research study. Initially, the classification model underwent training, followed by evaluation using the testing set, utilizing specific performance metrics such as accuracy rate, precision, recall, F1-score, and more. Model evaluation was conducted based on the receiver operating characteristic area under the curve (ROC-AUC) and precision-recall area under the curve (PR-AUC) metrics. ROC-AUC was utilized to characterize the overall classification performance of the model, while PR-AUC was employed to assess the predictive performance of the model.
ROC-AUC assesses the overall classification performance of the model, whereas PR-AUC evaluates the predictive performance specifically concerning positive samples.
Model development details: candidate predictors with a univariate association of P < 0.1 were considered for inclusion in the multivariate logistic regression model. The final logistic model was built using backward stepwise selection. For the MLP model, the scikit-learn library (version 1.0.2) in Python 3.7.6 was employed. The MLP architecture comprised an input layer, two hidden layers with 100 and 50 neurons respectively (both using ReLU activation functions), and a single-neuron output layer with a sigmoid activation function for binary classification. The model was trained using the Adam optimization algorithm (solver='adam’, initial learning rate=0.001) to minimize the binary cross-entropy loss (log loss). To mitigate overfitting, early stopping was implemented (early_stopping = True, n_iter_no_change=10) with a validation set comprising 20% of the training data (validation_fraction=0.2).
Statistical Methods
Descriptive statistics were utilized to outline the baseline characteristics of patients. Categorical variables were depicted as absolute values and percentages, while normally distributed continuous variables were represented as means ± standard deviations (SD). Continuous variables conforming to a normal distribution were reported as mean ± standard deviation (SD), and group comparisons were performed using independent sample t-tests. Conversely, variables not adhering to a normal distribution were predominantly described as median (Q1, Q3), with group comparisons conducted using the Mann-Whitney U test. Categorical variables were expressed as proportions or rates, and group comparisons were carried out using the chi-square test.
Clinical data underwent correlation and regression analyses, including logistic regression models and MLP predictive models. The accuracy of predictive models was assessed using the area under the receiver operating characteristic curve (AUC). Survival curves were generated using the Kaplan-Meier method. Covariates with P < 0.1 in univariate regression analysis were included in multivariate regression analysis. Statistical significance was defined as P < 0.05. All analyses were conducted using SPSS 24.0 and Python 3.7.6.
Results
Baseline Characteristic
A total of 381 patients with lung cancer who underwent ICIs treatment were included in the study, following the exclusion of 32 patients. Among them, 51 patients were classified into the ICIs-AKI group, while 330 patients were in the non-AKI group. The incidence of ICIs-AKI was 13.39%. Among them, 32 patients (62.75%) experienced mild acute kidney injury (KDIGO stage 1), 10 patients (19.61%) experienced moderate acute kidney injury (KDIGO stage 2), and 9 patients experienced severe acute kidney injury (KDIGO stage 3). The median time to onset of ICIs-AKI was [123 (63, 303)] days, with 27.45% of patients experiencing AKI within 3 months of initiating ICIs and 41.18% within 6 months.
The ICIs-AKI group demonstrated a higher prevalence of hypertension, diabetes, proteinuria, and extrarenal irAEs compared to the non-AKI group (all P < 0.05). Regarding combination therapy, the ICIs-AKI group had a higher frequency of diuretic, chemotherapy, and radiotherapy usage compared to the non-AKI group (P < 0.05, respectively). Additionally, the ICIs-AKI group exhibited significantly lower concentrations of Hb and eGFR, along with higher concentrations of sCr, BUN, and uric acid compared to the non-AKI group (P < 0.05, respectively). Detailed data are presented in Table 1.
Baseline Characteristics of Subjects in the Study.
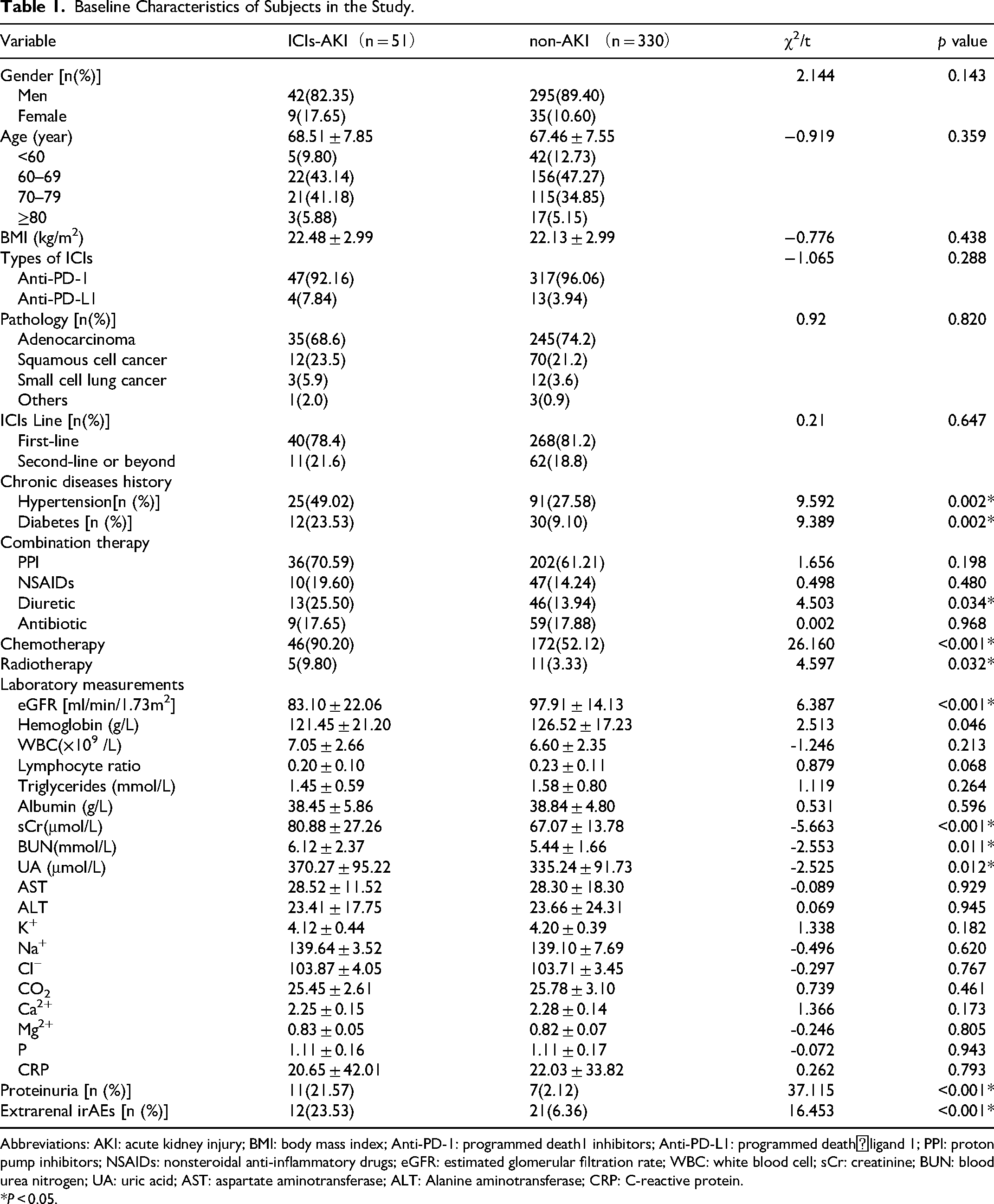
Abbreviations: AKI: acute kidney injury; BMI: body mass index; Anti-PD-1: programmed death1 inhibitors; Anti-PD-L1: programmed death⁃ligand 1; PPI: proton pump inhibitors; NSAIDs: nonsteroidal anti-inflammatory drugs; eGFR: estimated glomerular filtration rate; WBC: white blood cell; sCr: creatinine; BUN: blood urea nitrogen; UA: uric acid; AST: aspartate aminotransferase; ALT: Alanine aminotransferase; CRP: C-reactive protein.
*P < 0.05.
The distribution of lung cancer pathological subtypes and the line of ICI therapy were balanced between the ICIs-AKI and non-AKI groups (all P > 0.05, Table 1). Furthermore, to ensure the robustness of the prediction models, the allocation of pathological subtypes and treatment lines between the 70% training set and the 30% validation set was also balanced (all P > 0.05, data not shown).
Logistic Regression
In the univariable logistic analysis, significant associations were observed between the history of hypertension, concurrent proteinuria, concurrent extrarenal irAEs, diuretic usage, chemotherapy combination, and the occurrence of AKI in lung cancer patients undergoing ICIs therapy (all P < 0.05). Furthermore, the occurrence of ICIs-AKI demonstrated positive correlations with baseline blood urea nitrogen and uric acid levels, while exhibiting negative correlations with baseline hemoglobin and eGFR levels. Detailed results of this analysis are provided in Table 2.
Univariable Logistic Regression Analysis of Factors Associated with ICIs-AKI.

Abbreviations: BMI: body mass index; PPI: proton pump inhibitors; NSAIDs: nonsteroidal anti-inflammatory drugs; eGFR: estimated glomerular filtration rate; WBC: white blood cell; sCr: creatinine; BUN: blood urea nitrogen; UA: uric acid; AST: aspartate aminotransferase; ALT: Alanine aminotransferase; CRP: C-reactive protein.
*P < 0.05.
The indicators that showed statistical significance in the univariable logistic analysis were utilized as covariates in a multivariable logistic analysis. Although the history of diabetes showed a trend (P = 0.052) and lymphocyte ratio showed a borderline trend (P = 0.068) in univariable analysis, they were retained in the multivariable model due to their established clinical relevance to kidney injury and immune function, and to allow for comprehensive adjustment of potential confounders. The results of the multivariable logistic regression analysis indicated that a history of diabetes, concurrent proteinuria, concurrent extrarenal irAEs, diuretic use, and chemotherapy combination remained significantly associated with the occurrence of AKI in lung cancer patients undergoing ICIs therapy (all P < 0.05). Furthermore, higher eGFR was identified as a protective factor against ICIs-AKI, as demonstrated in Table 3.
Multivariable Logistic Regression Analysis of ICIs-AKI.

*P < 0.05.
Predictive Models
Logistic Regression Model
After conducting comparative analyses of factors between the ICIs-AKI and non-AKI groups, the predictive factors identified from multifactor analysis were employed to develop a logistic regression model for predicting ICIs-AKI. These factors included hypertension, diabetes, diuretic use, chemotherapy, eGFR, hemoglobin levels, lymphocyte ratio, creatinine levels, blood urea nitrogen levels, uric acid levels, presence of proteinuria, and occurrence of extrarenal irAEs.
The Hosmer-Lemeshow test resulted in χ2 = 10.596, P = 0.226 > 0.05. The area under the ROC curve (AUC) was calculated as 0.877 [95% CI (0.831, 0.923)], with a sensitivity of 0.922 and a specificity of 0.726, demonstrating a high level of accuracy for the predictive model. These results are depicted in Figure 2.

The ROC curve of multivariable for prediction of ICIs-AKI.
MLP Predictive Model
The dataset was divided into a training set comprising 70% and a validation set comprising 30%, utilizing an MLP classifier. The results demonstrated the model's accurate predictive performance, with an accuracy of 0.843, precision of 0.847, recall of 0.989, F1 score of 0.913, and AUC of 0.950. The area under the PR curve (AP) was calculated as 0.762, indicating good model performance. These findings suggest that the MLP model is well-suited for predicting the occurrence of ICIs-AKI, as detailed in Table 4 and depicted in Figure 3.
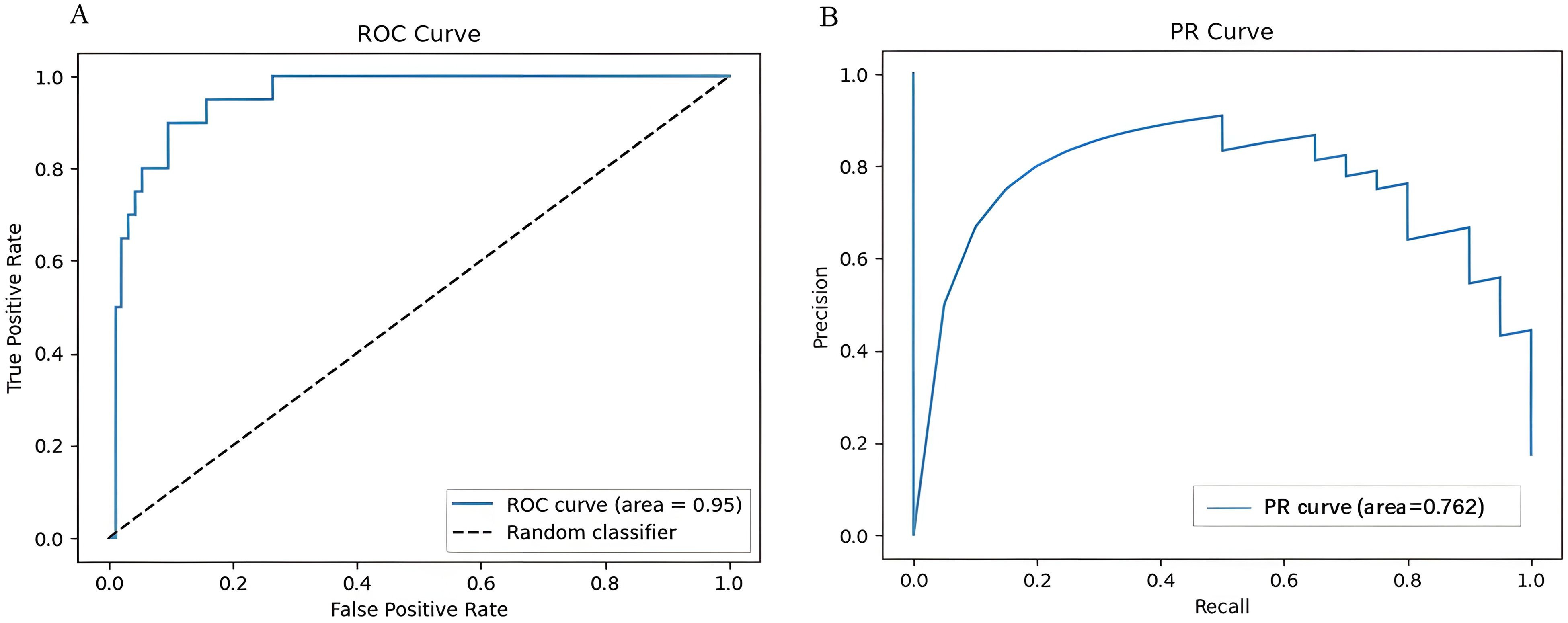
The ROC and PR curves of the validation sets for the MLP model.
Confusion Matrices of MLP Model.

Abbreviations: MLP: multilayer perceptron model; TP: true positives; FP: false positives; FN: false negatives; TN: true negatives; ACC: accuracy; Pre: precision; Recall: recall rate; F-Measure: F1 score; AUC: area under the ROC curve.
Survival Analysis
The unadjusted Cox regression analysis revealed no statistically significant difference in overall survival between the ICIs-AKI group and the non-AKI group [HR = 1.021, 95% CI (0.629, 1.659), P = 0.932]. Upon adjustment for diabetes, NSAIDs and antibiotic usage, eGFR levels, hemoglobin, lymphocyte ratio, albumin, serum creatinine, uric acid, Cl−, Mg2+ and CRP concentration, the absence of a statistically significant difference in overall survival between the ICIs-AKI group and the non-AKI group persisted [HR = 0.950, 95% CI (0.558, 1.616), P = 0.849]. Notably, the independent association of NSAIDs usage, Cl−, and Mg2+ concentration with mortality was observed. Detailed results of this analysis are provided in Table 5 and illustrated in Figure 4.

(A) Unadjusted Kaplan–Meier survival plot between ICIs-AKI group and non-AKI group; (B) Adjusted Kaplan–Meier survival plot between ICIs-AKI group and non-AKI group.
Unadjusted and Adjusted cox Regression for Overall Survival Following ICIs Treatment.

Abbreviations: BMI: body mass index; PPI: proton pump inhibitors; NSAIDs: nonsteroidal anti-inflammatory drugs; eGFR: estimated glomerular filtration rate; WBC: white blood cell; sCr: creatinine; BUN: blood urea nitrogen; UA: uric acid; AST: aspartate aminotransferase; ALT: Alanine aminotransferase; CRP: C-reactive protein.
*P < 0.05.
The Renal Outcome
Among the initial 51 patients diagnosed with ICIs-AKI, 21 patients ultimately experienced renal function recovery, resulting in a recovery rate of 41.18%. The incidence of AKI to CKD progression was observed to be 58.82%. In the CKD group, there were 25 males and 5 females, with a mean age of (70.87 ± 6.10) years and a median eGFR of [56.33 (46.0,78.19)] ml/min/1.73m2. Within this group, 28 patients were classified as CKD stage 3, while 1 patient each was categorized into CKD stage 4 and CKD stage 5. Moreover, no statistically significant difference in overall survival was found between the CKD group and the non-CKD group (P = 0.157). These results are depicted in Figure 5.

Kaplan–Meier survival plot between CKD group and non-CKD group.
Discussion
This retrospective study revealed an incidence of ICIs-AKI at 13.39%. We identified several risk factors potentially contributing to ICIs-AKI, including diabetes, proteinuria, extrarenal irAEs, diuretic use, combination chemotherapy, and low baseline eGFR levels. A noteworthy aspect of this study was the development of prediction models using logistic regression and MLP classifier, demonstrating robust predictive capabilities. These models aid in early clinical diagnosis of ICIs-AKI, thereby potentially mitigating the occurrence of severe adverse reactions. Surprisingly, we found no association between the occurrence of ICIs-AKI and primary or renal outcomes.
In this cohort of lung cancer patients treated with ICIs therapy, we observed an incidence of ICIs-AKI at 13.39%. This rate slightly surpasses findings from other studies, which typically report incidences ranging from 2% to 5%.2,5,11,14 A single-center retrospective study conducted in China documented a 12.8% incidence of renal-associated irAEs. 15 Furthermore, projections suggest that the occurrence rate of renal-associated irAEs may rise to approximately 9.9%-19% in the future. 16 Discrepancies in AKI incidence rates among studies stem from variations in inclusion criteria for AKI diagnosis. Additionally, while renal biopsy serves as the gold standard for diagnosing ICIs-AKI, its utilization in clinical practice is limited among cancer patients experiencing renal dysfunction, resulting in a low detection rate of ICIs-AKI.
In our study, we found that the median onset time for ICIs-AKI was [123 (63, 303)] days, with 27.45% of patients experiencing AKI within 3 months of initiating ICIs treatment and 41.18% within 6 months. These findings are generally consistent with results from several foreign studies.9,11 The duration of onset for ICIs-AKI is typically influenced by multiple factors. Onset times vary among different irAEs, with ICIs-AKI often exhibiting a longer latent period compared to extrarenal irAEs. For example, dermatitis typically occurs within 4 weeks after initiation of ICIs treatment, colitis usually develops within 6 weeks, immune-related hypophysitis manifests as neurologic symptoms at 6 weeks post-ICIs treatment, while skin toxicity typically arises after 3.6 weeks. 17 The onset time of ICIs-AKI also depends on the type of ICIs used for treating malignant tumors. Furthermore, compared to the use of PD-1 inhibitors alone, the combination of CTLA-4 inhibitors and PD-1 inhibitors leads to a higher incidence of kidney-related irAEs. 18 The onset time for AKI with CTLA-4 inhibitors is typically 6–12 weeks, whereas with PD-1 inhibitors, it ranges from 6–8 months. 19
The significant risk factors identified in our study, including the presence of diabetes, proteinuria, extrarenal irAEs, and low baseline eGFR levels, emerged as independent risk factors for ICIs-AKI, consistent with findings from studies conducted by Seethapathy and Liu.18,20 Additionally, some studies have reported associations of ICIs-AKI with certain medications, such as PPI and NSAIDs usage.8,21,22 In our cohort, we observed that the use of diuretics increased the risk of AKI in lung cancer patients receiving ICIs therapy. This association may be explained by several potential mechanisms. Diuretics can reduce intravascular volume and renal perfusion, potentially predisposing the kidneys to ischemic injury, especially in the context of other nephrotoxic insults. They may also cause electrolyte imbalances (eg, hypovolemia, hypokalemia) which can impair renal function. Importantly, diuretic use might be a marker for underlying conditions such as heart failure or edema, which themselves are associated with compromised renal hemodynamics and increased AKI risk. This underscores the importance of vigilance regarding drug combinations with ICIs in clinical practice and suggests that patients requiring diuretics may benefit from closer renal monitoring during ICIs therapy.
In our cohort, combination chemotherapy emerged as an independent risk factor for ICIs-AKI. Similar and divergent findings have been reported. A meta-analysis encompassing all cancer types did find an association with combining ICIs and chemotherapy treatments, 23 aligning with our results. However, a recent large cohort study focusing on NSCLC did not find an increased risk with combining ICIs and other agents. 24 Nevertheless, the discordance observed in these findings may be attributed to challenges in diagnosis.
Other pertinent findings in our study include a 41.18% complete kidney recovery rate at 90 days and an incidence of AKI to CKD progression of 58.82%. The majority of patients presented at CKD stage 3, which generally carries a favorable prognosis. Previous studies have indicated that renal function recovers in more than half of ICIs-AKI patients, which may be attributed to the mild to moderate injury observed in most cases of ICIs-AKI. 25 Moreover, the recovery of renal function has been associated with the early usage of corticosteroids, 25 with some cases achieving over 90% recovery. Thus, early recognition of ICIs-AKI is crucial for improving prognosis.
In our study, two clinical prediction models were developed for the early recognition of ICIs-AKI using logistic regression and MLP classifiers. By integrating multidimensional indicators through multiple fitting, these models achieved good predictive sensitivity and specificity. Particularly, the MLP prediction model exhibited superior overall performance with an AUC of 0.950, accuracy of 0.843, precision of 0.847, recall of 0.990, and F1 score of 0.913. These results outperformed previous AKI prediction models reported in other studies. For instance, Yu et al 26 employed a neural networks (NNs) model as an AKI prediction model in patients undergoing ICIs treatment, achieving a competitive AUC of 0.8167, accuracy of 0.7703, recall of 0.7, precision of 0.56, and F1 value of 0.6222. Compared to the neural network model by Yu et al (AUC=0.8167), 26 our MLP model demonstrated superior discriminative ability (AUC=0.950). This may be attributed to our inclusion of composite clinical features like proteinuria and extrarenal irAEs. While our identified risk factors align with prior studies,18,20 the novelty of our work lies in the direct comparison and validation of both a parsimonious logistic model and a high-performance MLP model within a dedicated lung cancer cohort, providing clinicians with flexible tools based on interpretability or predictive power needs. In future clinical practice, these prediction models may be utilized for baseline risk assessment of ICIs-AKI in patients, enabling evaluation of the risk, adjustment of treatment plans, or rigorous monitoring of ICIs-AKI, thereby holding significant clinical significance.
There have been relatively few studies investigating the primary outcome of ICIs-AKI. In our study, we found no association between ICIs-AKI and increased mortality regardless of disease status, a finding supported by other reports.20,27,28 Additionally, we observed no statistically significant difference in overall survival between the CKD group and the non-CKD group. This could be attributed to the relatively mild AKI observed in most patients, with many having recovered renal function or being at an early stage of chronic kidney disease. An alternative theory suggests that the occurrence of irAEs, including ICIs-AKI, may indicate an effective antitumor response and therefore could potentially improve survival. 29 Recent studies have indicated a positive correlation between the occurrence of irAEs and the efficacy of ICIs, 30 supporting this hypothesis. Another explanation could be that the follow-up period might not have been long enough to establish a link between CKD and mortality. The overall survival of ICIs-AKI appears to be closely linked to the malignant tumor status and varies depending on the underlying cause of AKI. Increased mortality risk was not observed in patients with ICIs-AKI, but rather in those with AKI attributed to other causes, where complications such as tumor progression or infections may contribute to a higher mortality rate. 29 In another study, the mortality rate of ICIs-AKI was reported to be as high as 93.2%. 31 Therefore, further studies with larger samples and longer follow-up durations may be warranted to elucidate the prognosis of ICIs-AKI.
There were several limitations in this study. Firstly, as a single-center study, the sample size was relatively small, and external validation was lacking, thus the predictive efficacy of the models remains uncertain. Future steps should involve constructing prospective study cohorts for model validation. Secondly, the sample size, particularly the number of ICIs-AKI events (n = 51), while sufficient for initial model development, may limit the stability of the estimates, especially for the more complex MLP model. External validation in larger cohorts is essential. Thirdly, the majority of patients included in this study were treated with PD-1 inhibitors, with a smaller portion receiving PD-L1 inhibitors, and data on CTLA-4 inhibitors were lacking. Finally, only a few patients underwent renal biopsy, which limited our ability to supplement information on renal pathology and investigate the mechanism of ICIs-AKI.
Addressing Potential Confounding Factors
Several important confounding factors were considered in our study design and interpretation. First, the pathological subtype of lung cancer and the line of ICI therapy were balanced between the ICIs-AKI and non-AKI groups, reducing their potential bias in survival analysis and model development. Specifically, the proportion of patients with small cell lung cancer, known for its aggressive nature, was low and similar between groups (5.9% vs 3.6%, P = 0.820). Similarly, the distribution of first-line versus later-line ICIs therapy was comparable. This balance strengthens the inference that the lack of observed survival difference is more likely attributable to the nature of ICIs-AKI itself rather than underlying tumor biology or treatment sequence disparities.
Second, the high rate of chemotherapy use in the ICIs-AKI group (90.2%) necessitates careful consideration. While combination chemotherapy was identified as a risk factor in our multivariate model, it is crucial to clarify that our endpoint was ICIs-AKI, not all-cause AKI. We employed strict clinical adjudication to differentiate AKI primarily related to ICI from that likely caused by chemotoxic agents (eg, cisplatin) or other factors, based on onset timing, clinical context, and supportive features like extrarenal irAEs. This diagnostic rigor supports the interpretation that chemotherapy may potentiate the immune-mediated renal injury of ICIs, rather than the observed AKI being solely chemotherapy-induced. Nonetheless, the overlapping nephrotoxic potential remains a significant clinical challenge, underscoring the need for vigilant monitoring in patients receiving combination regimens.
Third, regarding radiotherapy, only a small subset of patients (16/381, 4.2%) received it, and details were analyzed. Most radiotherapy was targeted to thoracic lesions (lung/mediastinum) and administered either before or concurrently with ICI, with no direct renal irradiation reported. While systemic effects of radiotherapy cannot be entirely ruled out, its limited use and non-renal targeting minimize its likely impact as a major confounder for ICIs-AKI in this cohort.
Conclusion
In this retrospective study, we explored the rate and onset timing, risk factors, and clinical features associated with ICIs-AKI. Notably, renal function was observed to recover in about half of the patients with ICIs-AKI. Importantly, our logistic regression and MLP models demonstrated robust predictive capabilities for the early recognition of ICIs-AKI. Moreover, neither the occurrence of ICIs-AKI nor the recovery of renal function was correlated with patient mortality. Future studies incorporating longitudinal biospecimen collection are needed to provide additional insight into the prognosis and mechanisms of ICIs-AKI, and to aid clinicians in differentiating it from other causes of AKI.
Footnotes
Abbreviations
Ethics Approval and Consent to Participate
This study adhered to the principles outlined in the Declaration of Helsinki and was approved by the Ethics Committee of the Ningbo No. 2 Hospital (YJ-NBEY-KY-2023-013-01).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Medical Scientific Research Foundation of Zhejiang Province, China (2020KY853, 2023KY283).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Tracked Changes
We have removed all author-related information, including but not limited to affiliation, address and ethics approval.
