Abstract
Introduction
Thoracic SMARCA4-deficient tumors, which are rare and aggressive malignancies found in the lung or thoracic cavity, present a challenge in treatment standardization. This challenge arises from their resistance to chemotherapy and the absence of targeted therapy options.
Methods
Thoracic SMARCA4-deficient tumors were identified retrospectively using pathology databases. The clinicopathological characteristics of these tumors are outlined, and the clinical outcomes of advanced patients treated with immune checkpoint inhibitors (ICIs) in combination with chemotherapy and chemotherapy alone are reviewed.
Results
Thirty-nine patients had thoracic SMARCA4-deficient tumors, with a median age of 62 years. The cohort consisted of 92.3% males, and 89.7% had a history of smoking. Within this group, 94.9% had stage III/IV disease at diagnosis. SMARCA4-deficient non-small cell lung cancer (SMARCA4-DNSCLC) and SMARCA4-deficient undifferentiated tumors (SMARCA4-DUT) display distinct histological and immunohistochemical features. Thirty-five patients underwent systemic therapy, achieving an ORR of 51.4%, a DCR of 82.9%, and a median OS of 20.9 months. Patients were categorized into chemotherapy (28.6%) and ICIs plus chemotherapy (71.4%) groups. The ICIs plus chemotherapy group exhibited an ORR of 64.0% and a DCR of 96.0%, while the chemotherapy group had an ORR of 20.0% and 50.0%, respectively (P < .0001 for ORR and DCR). The median OS for ICIs plus chemotherapy and chemotherapy groups were 20.9 months and 6.5 months, and median PFS were 9.6 months and 3.5 months, respectively, all statistically significant (P < .05). Multivariate COX regression analysis indicated that treatment was an independent prognostic factor for OS.
Conclusion
Thoracic SMARCA4-deficient tumors exhibit a lack of SMARCA4 expression, displaying high malignancy and aggressiveness while exhibiting poor response to standard chemotherapy. The combination of ICIs with chemotherapy could potentially serve as an effective treatment approach for thoracic SMARCA4-deficient tumors.
Keywords
Introduction
SMARCA4-deficient tumors are a distinct type of tumors linked to high aggressiveness and poor prognosis. SMARCA4, a member of the SWI1/SNF1 family, plays a crucial role in tumor suppression. The SWI/SNF complex is implicated in developmental processes, transcriptional regulation, DNA repair, cell cycle control, and cancer development. 1 A SMARCA4 mutation can lead to the loss of SMARCA4/BRG1 protein expression, resulting in the development of cancers and sarcomas in various sites and organs, including the nasal cavity, thoracic cavity, ovary, uterus, and gastrointestinal tract.2,3
SMARCA4-deficient tumors are found in approximately 5%–10% of thoracic tumor patients, 4 which include SMARCA4-deficient non-small-cell lung cancer (SMARCA4-DNSCLC) and SMARCA4-deficient undifferentiated tumors (SMARCA4-DUT). 5 These tumors exhibit distinct histological, immunohistochemical, clinical, and prognostic features. They are more prevalent in male smokers and typically present as masses in the lungs, pleura, hilum, and/or mediastinum. SMARCA4-deficient thoracic tumors are highly aggressive, with approximately 83% of patients diagnosed with SMARCA4-DNSCLC or SMARCA-DUT at stage IV. 6 The prognosis for these tumors is extremely poor, with a median survival of approximately 4 to 7 months. 7 SMARCA4-DUT is identified by an undifferentiated or rhabdoid phenotype. SMARCA4-dNSCLC may display glandular structures with positive expression of epithelial markers. Immunohistochemical analyses of both SMARCA4-DUT and SMARCA4-dNSCLC have revealed a loss or significant reduction in SMARCA4 (BRG1) expression. 8 Currently, there are no standard treatment options for thoracic SMARCA4-deficient tumors owing to their poor response to common chemotherapy and the absence of sensitive sites for targeted therapy.
With the ongoing advances in immunotherapy, immune checkpoint inhibitors (ICIs) have become pivotal in improving treatment efficacy and prognosis in patients with non-small cell lung cancer (NSCLC). However, limited research has explored the role of ICIs in thoracic SMARCA4-deficient tumors.9,10 While some case reports.11–14 and small-scale studies15,16 suggest that immunotherapy may improve outcomes, other studies have suggested that SMARCA4-deficient tumors have a desert-like immune microenvironment, which may hinder the effectiveness of ICIs. 17 The efficacy of immunotherapy in thoracic SMARCA4-deficient tumors remains controversial owing to the lack of validation in large samples.
In this study, we retrospectively described the clinicopathological features of 39 thoracic SMARCA4-deficient tumor patients and analyzed the factors affecting their prognosis. Additionally, we investigated the response of advanced thoracic SMARCA4-deficient tumors to treatment with ICIs.
Patients and Methods
General Information
A consecutive cohort of 595 patients diagnosed with NSCLC at our hospital between September 2021 and May 2024 was retrospectively analyzed. BRG-1 immunohistochemical staining was used to identify cases with loss of Brg-1 expression, which was subsequently diagnosed as thoracic SMARCA4-deficient tumors. Inclusion criteria were: age ≥ 18 years; evidence of lung or thoracic tumors confirmed by computed tomography (CT) scans and/or radiological reports; and histopathological diagnosis of SMARCA4-DNSCLC or SMARCA4-DUT, with immunohistochemical confirmation of SMARCA4 (BRG1) protein deficiency. Exclusion criteria included the following: incomplete or inconclusive diagnostic data, patients with unavailable clinical characteristics and follow-up data, patients with SMARCA4 (BRG1)-retained lung or thoracic tumors, patients without BRG1 immunohistochemical staining results, patients with a history of other primary malignancies within the past five years; and presence of severe comorbidities, such as uncontrolled hypertension, diabetes, heart failure, and severe infections. The patient selection process is illustrated in Figure 1. All patient details have been de-identified. This retrospective study was approved by the Ethics Committee of Quanzhou First Hospital Affiliated to Fujian Medical University (approval number: [2025] K051). Given the retrospective nature of the study and the use of anonymized patient data, the requirement for informed consent was waived by the ethics committee. This study conformed to the STROBE guidelines. 18

The Selection Process of Eligible Patients.
Data Collection
General clinical data of the patients were gathered from the electronic medical record system, encompassing sex, age, smoking history, treatment regimen, and Eastern Cooperative Oncology Group performance status (ECOG PS) scores. The data also covered programmed death ligand 1 (PD-L1) expression status, tumor markers, Ki-67, tumor node metastasis stage, site of distant metastasis, pathological and immunohistochemical features, and gene test results.
Comprehensive Evaluation and Follow-up
PD-L1 expression was evaluated with the 22C3 antibody (Dako, Agilent Technologies) on an automated immunohistochemical staining platform (Dako Autostainer Link 48). Evaluation of PD-L1 expression in tumor cells was based on the tumor proportion score (TPS). A tumor was classified as having PD-L1 expression if TPS was ≥1% and as having high PD-L1 expression if TPS was ≥50%.
To assess primary tumor size, local invasion, and regional lymph node, contrast-enhanced CT scans of the chest and abdomen were conducted. CT was also utilized to evaluate distant metastases. Magnetic resonance imaging (MRI) was employed for a detailed assessment of central nervous system involvement and improved characterization of soft tissue masses. Positron emission tomography (PET) was utilized to assess the metabolic activity of the tumors and detect occult metastases.
Follow-ups were conducted using electronic medical records or telephone consultations, and the last follow-up was performed on December 1, 2024. Imaging examinations, including CT, MRI, and PET/CT, were utilized to assess treatment efficacy based on the Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1). The efficacy evaluation criteria for solid tumors version 1.1 were adopted. Patients were categorized into four categories: complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The disease control rate (DCR) was calculated as (CR + PR + SD) / total cases × 100%. The objective response rate (ORR) was calculated as (CR + PR) / total cases × 100%. Progression-free survival (PFS) was defined as the duration between the initiation of treatment and the detection of disease progression or death from any cause. Recurrence-free survival (RFS) was defined as the duration between the patient receiving surgical treatment and disease recurrence or death from any cause.
Statistical Analysis
SPSS 26.0 and GraphPad Prism 9.5.1 were utilized for analysis and mapping. Measurement data following a normal distribution were presented as mean ± standard deviation and analyzed using the t-test. Measurement data that did not follow a normal distribution were presented as medians and ranges and analyzed using the Mann–Whitney U test. Count data were expressed as cases or percentages and compared using the chi-square test or Fisher's exact test, as appropriate. Survival analysis employed the Kaplan–Meier method, with group comparisons made using the log-rank test. Cox proportional hazard models were used for univariate and multivariate analyses. Statistical significance was defined as P < .05, with all P-values reflecting bilateral tests.
Result
A total of 41 thoracic SMARCA4-deficient tumors were identified in 595 NSCLC cases, representing 6.9% of the total. Two cases were excluded due to incomplete clinical characteristics and follow-up records, yielding a final analytic cohort of 39 patients. The median age of the patients was 62 years (range, 39-77 years), with males comprising 92.3% (36/39) of the cohort. Among these patients, 89.7% (35/39) had a history of smoking, all of whom were male. Additionally, 94.9% (37/39) of the patients had an ECOG PS score of 0–1, and 92.3% (36/39) had Stage III/IV disease. Four patients underwent surgical treatment, while the remaining received systemic therapy. Based on the treatment regimen, the 35 patients were categorized into a chemotherapy group (10 patients, 28.6%) and a group receiving PD-1 monoclonal antibody in combination with chemotherapy (25 patients, 71.4%). All 35 patients received chemotherapy with PD-1/PD-L1 inhibitors as first-line therapy, with the chemotherapy group receiving platinum-based chemotherapy. Following systemic therapy, none of the patients achieved CR, while 51.4% (18/35) achieved PR, 31.4% (11/35) achieved SD, and 17.1% (6/35) achieved PD. At the end of the observation period, 54.3% (19/35) of the patients had deceased, and 45.7% (16/35) had survived. The baseline data of the 39 enrolled patients are detailed in Table 1.
Clinicopathological Characteristics of Patients with SMARCA4-Deficient Thoracic Tumors.

Histologically, SMARCA4-DNSCLC exhibits various morphological features, such as poorly differentiated solid, mucinous, and pleomorphic sarcomatoid carcinomas (Figure 2). SMARCA4-DUT cells consist of poorly differentiated, non-adherent epithelioid cells with eccentric nuclei and rhabdoid cytoplasm, along with observed mitotic activity and necrosis (Figure 3). SMARCA4-DUT cells show focal expression of CK while being negative for CK7, TTF1, P63, and P40 and exhibit overexpression of CD34, SOX2, SALL4, Syn, and p53. In contrast, SMARCA4-DNSCLC demonstrates diffuse expression of CK and CK7 but lacks expression of CD34, S0X2, or SALL4. TTF1, P63, and P40 may be positive in SMARCA4-DNSCLC. Molecular pathological testing was conducted on 23 out of 39 patients, all of whom exhibited SMARCA4 deficiency. Among these patients, 19 had gene co-mutations, with six having multiple co-mutations. The identified co-mutations included TP53 (9 cases), KRAS (4 cases), BRAF (1 case), PI3KCA (2 cases), STK11 (5 cases), KEAP1 (2 cases), JAK1 (1 case), PALB2 (1 case), KMT2D (1 case), and RET fusion (1 case) (Figure 4).
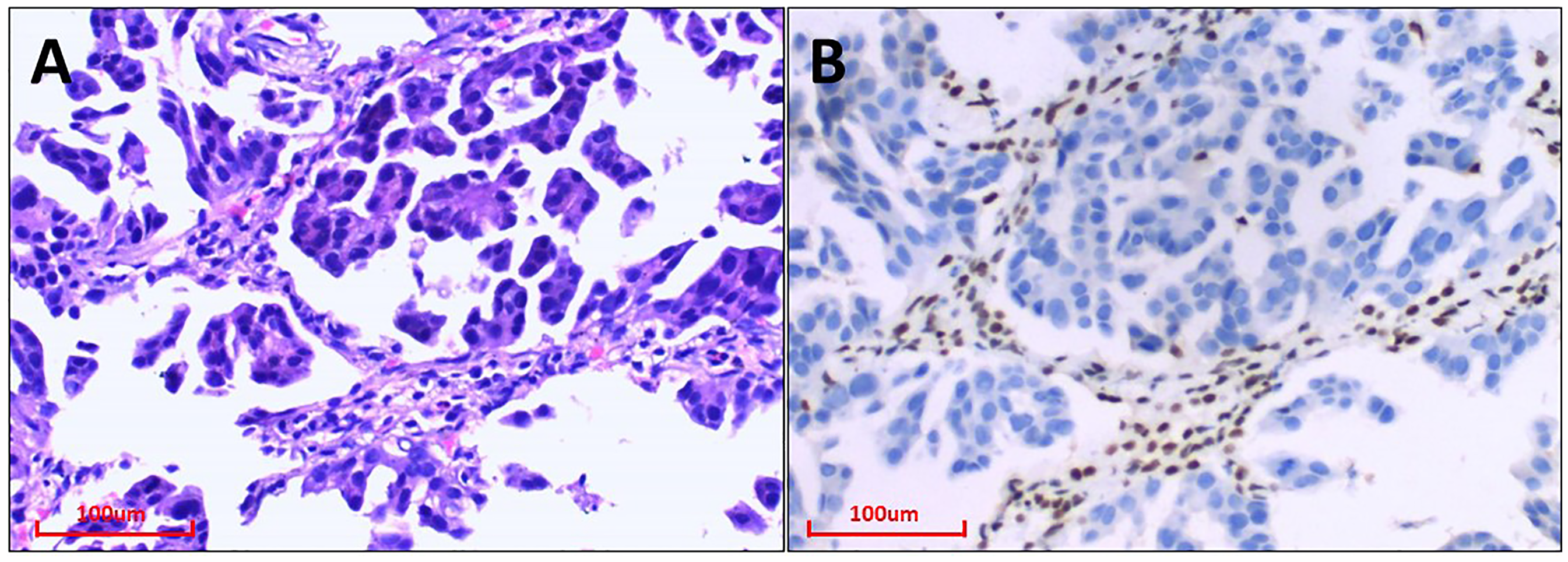
A. Classic Lung Adenocarcinoma, Acinar Structure. (HE staining, × 200) B. IHC Staining of BRG1 Revealed Complete Deficiency of the Thoracic SMARCA4-DNSCLC Region. (IHC staining, × 200).

Pathological Features of Thoracic SMARCA4-DUT. A. SMARCA4-DUT was Composed of Poorly Differentiated, Nonadherent Epithelioid Cells with Eccentric Nuclei and Rhabdoid Cytoplasm. Mitotic Figures and Necrosis Were Common. (HE Staining, × 200) B. IHC Staining of BRG1 Revealed Complete Deficiency of the Thoracic SMARCA4-DUT Region. (IHC Staining, × 200).

Gene Co-mutation in Thoracic SMARCA4-null Tumors.
Up to the follow-up time, two of the four patients who underwent surgery experienced tumor recurrence, with RFS durations of 10.6 and 7.7 months, respectively. Among 35 patients with SMARCA4-deficient NSCLC who underwent systemic therapy, the ORR was 51.4%, and the DCR was 82.9%. In the group receiving ICIs combined with chemotherapy, 64.0% (16/25) of patients achieved PR, 32.0% (8/25) achieved SD, and 4.0% (1/25) achieved PD; the ORR was 64%, and the DCR was 96.0%. Conversely, in the chemotherapy-only group, 20.0% (2/10) of patients achieved PR, 30% (3/10) achieved SD, and 50.0% (5/10) achieved PD; the ORR was 20.0%, and the DCR was 50.0%. A significant difference in the ORR and DCR (P < .0001) was observed between the two groups (Figure 5). Long-term remission was achieved by two patients receiving ICIs combined with chemotherapy. The imaging data are presented in Figures 6 and 7.

Treatment Response in the Chemotherapy Group and ICIs Plus Chemotherapy Group.
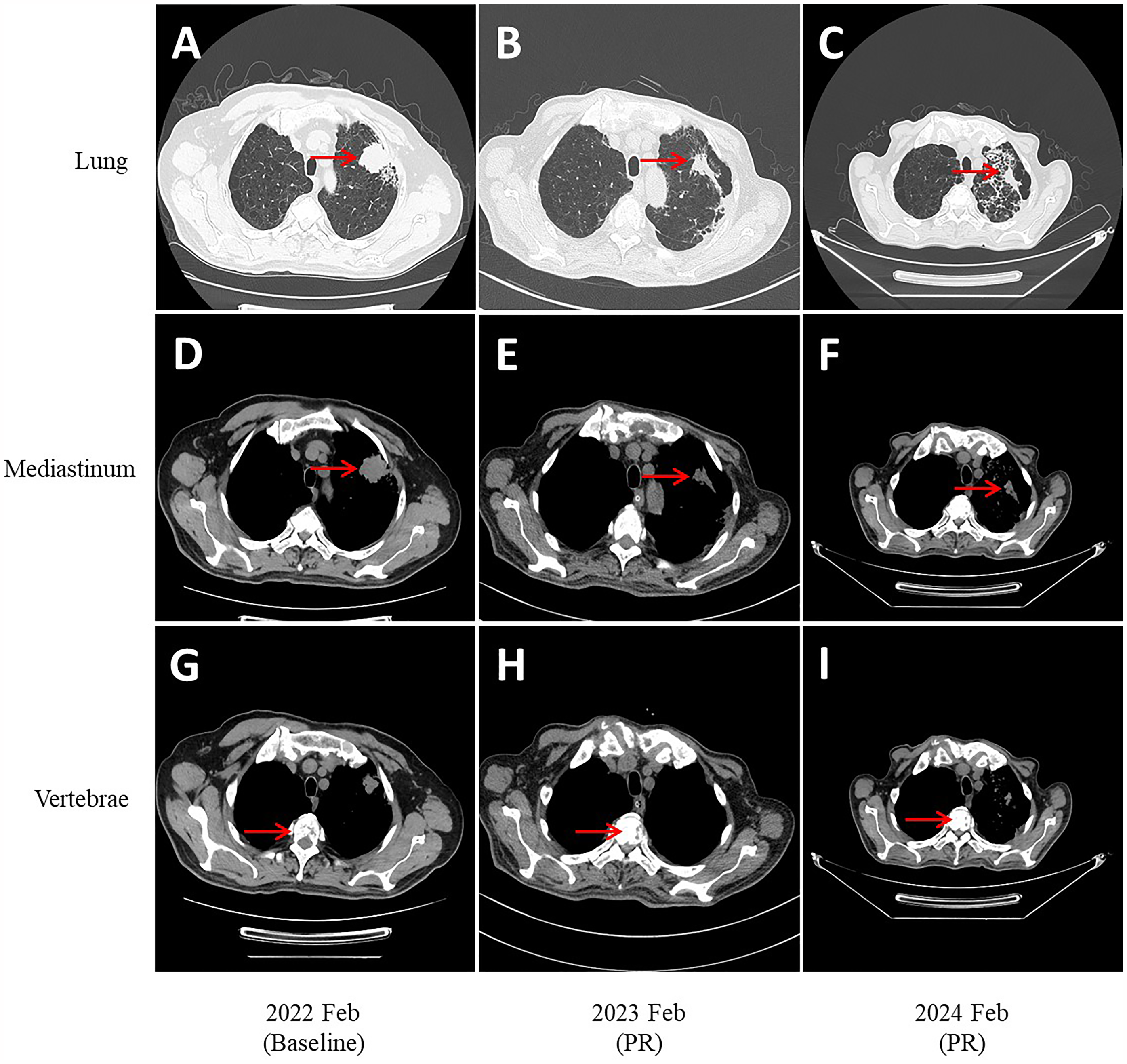
A 66-year-old Male Patient Diagnosed with SMARCA4-DNSCLC Presented with Bone Metastases. He Began Receiving ICIs (pembrolizumab) in Combination with Chemotherapy (paclitaxel + carboplatin) on February 11, 2022. As of the Last Follow-up (December 2024), both PFS and OS had Exceeded 34 Months.

A 47-Year-old Male Patient Diagnosed with SMARCA4-DUT, Presenting with Left Adrenal Metastasis, Initiated Treatment with ICIs (Toripalimab) Combined with Chemotherapy (Paclitaxel + Carboplatin) on September 27, 2021. At the Last Follow-up (December 2024), he Maintained a PFS of 15.5 Months and an OS Exceeding 36 Months.
At the data cut-off of December 1, 2024, the median follow-up time was 10.2 months (range, 2.5-38.7 months). The median OS was 10.2 months (95% CI, 2.5 months–not achieved (NA)) for all patients, and the median PFS was 7.3 months (95% CI, 0.8 months–NA) for all patients (Figures 8 and 9). The median OS of the chemotherapy group was 6.5 months (95% CI, 2.5 months–13.0 months), and that of ICIs combined with the chemotherapy group was 20.9 months (95% CI, 4.8 months–NA), with a statistically significant difference (P < .0001). The 1-year OS rates in the ICIs combined with chemotherapy and chemotherapy groups were 74.0% and 20%, respectively, with a statistically significant difference (P = .008). The median PFS of the ICIs combined with the chemotherapy group was 9.6 months, while that of the chemotherapy group was 3.5 months, showing a statistically significant difference (P < .0001) (Figures 10 and 11).

Kaplan-Meier Curves of OS in 35 Thoracic SMARCA4-deficient Tumor Patients.

Kaplan-Meier Curves of PFS in 35 Thoracic SMARCA4-Deficient Tumor Patients.

Kaplan-Meier Curves of OS in the Chemotherapy Group and ICIs Plus Chemotherapy Group.

Kaplan-Meier Curves of PFS in the Chemotherapy Group and ICIs Plus Chemotherapy Group.
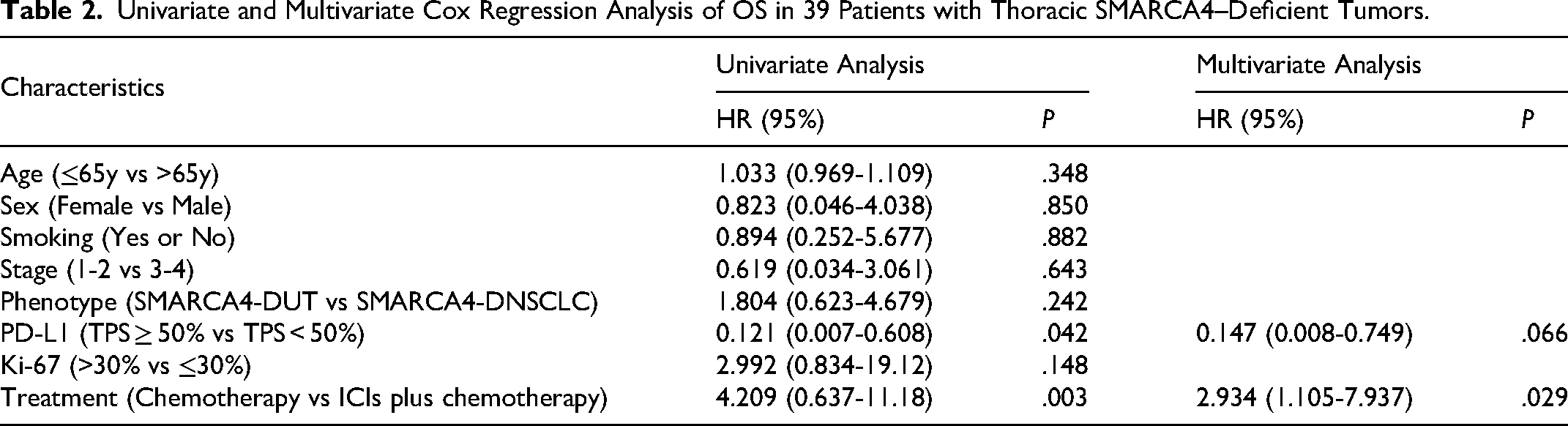
Univariate Cox proportional hazards model analysis of OS revealed that PD-L1 expression and treatment were statistically significant (P < .05), while age, sex, smoking, stage, phenotype, and ki-67 expression were not associated with OS (P > .05). Multivariate Cox regression analysis demonstrated that treatment independently influenced the OS of patients with thoracic SMARCA4-deficient tumors (hazard ratio (HR) = 0.110, 95% CI: 0.023–0.425, P = .002). Patients receiving ICIs in combination with chemotherapy exhibited a more favorable prognosis (Table 2).
Univariate and Multivariate Cox Regression Analysis of OS in 39 Patients with Thoracic SMARCA4–Deficient Tumors.
Discussion
Owing to the low incidence of thoracic SMARCA4-deficient tumors and the lack of prospective trials, clear standard treatment guidelines have not been established, relying instead on retrospective data. Given the highly aggressive nature of the disease and poor prognosis, treatment recommendations typically support combination regimens. This study describes the clinicopathological characteristics of thoracic SMARCA4-deficient tumors. Additionally, the study suggests that combining ICIs with chemotherapy may enhance the prognosis of patients with advanced thoracic SMARCA4-deficient tumors compared to chemotherapy alone.
SMARCA4 is among the most extensively researched tumor suppressor genes. Loss of SMARCA4/BRG1 leads to the deactivation of the ATPase in the SWI/SNF chromatin remodeling complex, resulting in the dedifferentiation of tumor cells. This process includes the transition from normal to low-grade neoplastic states and subsequently to highly aggressive neoplastic states with rhabdoid features. 19 Despite similarities, SMARCA4-DNSCLC and SMARCA4-DUT exhibit distinct histological and immunohistochemical characteristics. Studies have indicated the loss of TTF-1 expression in poorly differentiated and sarcomatoid areas of tumor tissue. Concurrently, the expression of SMARCA4 in poorly differentiated areas gradually diminishes or is lost, while TTF-1 and SMARCA4 expression remain normal in moderately differentiated areas. 20 This observation suggests that an increased loss of SMARCA4 may promote SMARCA4-DNSCLC toward dedifferentiation into SMARCA4-DUT. There is no established standard treatment for these two tumor phenotypes, with common chemotherapy regimens involving paclitaxel/pemetrexed combined with platinum. Previous studies with small sample sizes have not identified differences in efficacy and prognosis between these phenotypes, 21 necessitating further samples to validate these findings. Our research revealed that thoracic SMARCA4-deficient tumors often exhibit gene co-mutations, with TP53 mutations being the most prevalent, followed by STK11 and KRAS mutations. Additionally, we identified a case of combined RET fusion that had not been previously reported. 22
Although immunotherapy has made significant advancements in lung cancer treatment, its efficacy remains uncertain for certain lung cancers with specific driver genes. This study revealed that patients with thoracic SMARCA4-deficient tumors who received only chemotherapy had a median OS of 6.5 months. This was significantly shorter than the OS observed in patients with lung cancer lacking driver gene alterations, indicating a poor prognosis associated with this tumor subtype. The combination of ICIs with chemotherapy markedly improved PFS and OS in these patients, consistent with previous reports.9,23 Among patients with known baseline PD-L1 expression, 62.9% (22/35) were PD-L1-positive (PD-L1 TPS ≥ 1%), with 17.1% (6/35) exhibiting high PD-L1 expression (PD-L1 TPS ≥50%). This may explain the favorable response of thoracic SMARCA4-deficient tumors to immunotherapy. However, the prognosis of patients in the ICIs plus chemotherapy group was also highly variable, with the shortest OS being 4.8 months and only a few patients experiencing sustained benefits from immunotherapy. Therefore, identifying biomarkers that predict immunotherapy response in thoracic SMARCA4-deficient tumors warrants further investigation. Univariate COX regression analysis demonstrated that PD-L1 expression and treatment type affected OS in these patients. Subsequent multivariate COX regression analysis identified the treatment regimen as an independent predictor of OS, consistent with the findings of Liang et al 6 Conversely, these results contradicted those of Wang et al, 15 possibly due to the limited sample size. Some studies suggest that chemotherapy-induced tumor cell death can enhance immune recognition and sensitize tumor cells to ICIs.24 Moreover, ICIs can reduce PD-L1 expression in tumor cells and enhance tumor sensitivity to platinum-based chemotherapy. 25 Therefore, combining ICIs with chemotherapy may represent a promising therapeutic approach for these rare tumors.
This study retrospectively analyzed 39 patients with thoracic SMARCA4-deficient tumors, detailing their distinctive clinical characteristics. The study revealed that combining immunotherapy with chemotherapy may enhance prognosis. These results offer insights into clinical management strategies for thoracic SMARCA4-deficient tumors with poor prognoses. Additionally, the study indicates that investigating the effect of SMARCA4 deficiency on the tumor immune response could be a promising avenue for future research.
However, this study has several limitations. First, the small sample size highlights the inherent challenges in investigating rare malignancies, potentially compromising statistical power. The retrospective nature of the study and limited cases make the calculation and justification of the sample size particularly challenging. Additionally, the restricted patient pool introduces potential biases in the baseline characteristics of each subgroup. Unmeasured confounders in the retrospective analysis might impact the outcomes. In clinical practice, conducting molecular testing on all patients with thoracic SMARCA4-deficient tumors is often impractical, leading to a limited number of test cases that may not fully capture the disease's molecular alterations.
Conclusion
In summary, thoracic SMARCA4-deficient tumors are rare malignant tumors characterized by the absence of SMARCA4 expression. Multiple metastases are often present at the time of clinical diagnosis, indicating high malignancy and aggressiveness. Traditional chemotherapy demonstrates limited efficacy and poor prognosis. Combining ICIs with chemotherapy could represent a promising treatment approach for thoracic SMARCA4-deficient tumors. Large-scale multicenter clinical trials are necessary to validate these findings. Additionally, further basic research is required to elucidate the molecular biological mechanisms.
Footnotes
Abbreviation
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Ethical Considerations
This retrospective study was approved by the Ethics Committee of Quanzhou First Hospital Affiliated to Fujian Medical University (Approval Number: 【2025】K051). Given the retrospective nature of the study and the use of de-identified patient data, the requirement for informed consent was waived by the Ethics Committee. The study was conducted in accordance with the ethical standards of the Declaration of Helsinki and its later amendments.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
