Abstract
Introduction
Body mass index (BMI) is a common clinical parameter associated with cancer prognosis, but its association with survival outcomes in lung cancer patients receiving immune checkpoint inhibitors (ICIs) remains unclear. This study aimed to clarify the prognostic value of BMI in ICI-treated lung cancer patients.
Methods
A systematic review and meta-analysis were conducted based on online databases including PubMed, Embase, Web of Science, Cochrane Library, and ClinicalTrials.gov up to December 1, 2024. Eligible studies included lung cancer patients treated with ICIs and reported hazard ratios (HRs) for overall survival (OS) and/or progression-free survival (PFS) stratified by BMI. Random-effects models were used to determine HRs with 95% confidence intervals (CIs).
Results
A total of 30 studies involving 5987 patients were included. High BMI was significantly associated with better OS (HR = 0.69, 95%CI = 0.60-0.80) and PFS (HR = 0.82, 95%CI = 0.72-0.93). The subgroup analysis showed improved survival outcomes particularly in patients with BMI ≥ 30 kg/m2 as compared with others. However, this association was not statistically significant in small-cell lung cancer.
Conclusion
High BMI was associated with a better prognosis than low BMI in ICI-treated patients with lung cancer. Due to study limitations, the prognostic impact of BMI still requires further clarification with additional evidence.
Introduction
Lung cancer is the most common human malignancy worldwide. 1 Despite improved tobacco control and treatment strategy, the five-year survival rate of this disease remains to be below 20% in majority of countries. 2 For patients with unresectable lung cancer, targeted drugs or chemotherapy are recommended, but limited efficacy is observed in a considerable proportion.3,4 The introduction of immune checkpoint inhibitors (ICIs) holds promise for them which have been proved to durably control disease with acceptable adverse effects in numerous clinical trials. 5 However, actually only a few of patients may benefit from ICI therapy, and there is a need to understand prognostic factors that associate with patient outcomes after receiving ICI therapy. Currently, several clinical and biological parameters, such as PD-L1 expression, tumor mutation burden (TMB), bacteria and microsatellite instability (MSI), have been associated with outcomes in ICI-treated populations. 6 In addition, certain concomitant drugs, such as antibiotics and proton pump inhibitors may compromise the efficacy of ICIs.7,8 A deeper understanding of the prognostic factors in ICI-treated patients will facilitate more precise therapy decisions and patient management.
During anti-cancer therapy, individual nutritional status plays a crucial role and related markers have been proved as reliable prognostic predictors such as body mass index (BMI). 9 BMI, which is calculated based on body weight and height, serves as a traditional nutritional indicator and has been extensively utilized for predicting the risk and clinical outcomes of cancers. For instance, a recent meta-analysis comprising 66 cohort studies has shown that high BMI (overweight or obesity) was linked to a higher likelihood of endometrial, kidney and liver cancers, but a lower likelihood of prostate and lung cancers. 10 An another meta-analysis including 73 cohort studies has demonstrated that high BMI was inversely associated with overall survival (OS) in patients with breast cancer, but positively associated with that of patients with gastrointestinal tumors. 11 In Asian patients with small-cell lung cancer (SCLC), low BMI (underweight) was proved as an independent unfavorable predictor for OS. 12 With its impact on immunotherapy, a recent preclinical study reported that compared with lean mice, obese mice receiving anti-PD-1 immunotherapy had worse therapeutic effect, implying obesity may reduce the efficacy of ICI drugs. 13 Several clinical investigations have explored the potential relationship between BMI and ICI efficacy, but conflicting findings have been reported. For example, higher BMI was linked with better progression-free survival (PFS) in lung cancer patients receiving ICIs. 14 However, a worldwide multicenter study demonstrated no significant link between BMI and clinical outcome in non-small-cell lung cancer (NSCLC) patients who received first-line chemoimmunotherapy. 15 A single center retrospective analysis showed a nonlinear association between BMI and OS in NSCLC patients undergoing ICI treatment, where BMI over 30 kg/m2 appeared to be a favorable prognostic factor. 16 Taken together, the association of BMI with ICI efficacy in lung cancer patients remains unclear, and more validations are essential.
Here, a systematic literature review followed by a meta-analysis was performed to evaluate the prognostic significance of BMI in lung cancer patients who received ICI-based anti-cancer therapy. This study will contribute to utilizing BMI as a noninvasive prognostic predictor for therapy decision, which may finally benefit more precise patient management.
Methods and Materials
Literature Search Strategy
The literature search strategy was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, and the corresponding flowchart is shown in Figure 1. 17 The systematic review was registered in the International Prospective Register of Systematic Reviews (PROSPERO): CRD42025640765. The comprehensive literature search was performed on Embase, PubMed, Web of Science, Cochrane Library and Clinicaltrials.gov from inception till December 1, 2024. The keywords associated with lung cancer (neoplasm, oncology, tumor or carcinoma) were paired with immunotherapy-related heading terms (immune checkpoint inhibitor, cytotoxic T lymphocyte antigen-4 inhibitor, programmed death-ligand 1 inhibitor or programmed death receptor 1 inhibitor) and body mass index (BMI). The full search strategies for each database, including all keywords and Boolean operators used, are provided in Supplementary Appendix A. The reference lists of the literatures were also carefully examined to avoid missing potential relevant studies. Ethics board approval is not applicable to this work as it constitutes a literature review with no human/animal subjects involvement.

Flowchart of the Literature Search for Meta-analysis.
Eligibility Criteria
The eligible studies were determined according to the PICO guidelines: P (Population): patients pathologically diagnosed as lung cancer; I (Intervention): ICI therapy alone or combination therapy; C (Comparison): high-BMI and low-BMI groups; O (Outcome): hazard ratios (HRs) for OS and/or PFS. Two independent reviewers screened titles, abstracts, and full texts for inclusion. Disagreements were resolved through discussion or adjudication by a third reviewer. Animal/cellular study, review, case report, meta-analysis, and non-English literatures were excluded. BMI was calculated by weight (kg) / height² (m).
Data Extraction and Quality Evaluation
The collected data encompassed the following: (1) study information (author, year, region, study design and sample size); (2) participant information; (3) interventions (ICI type); (4) primary outcome (the HRs and 95% CIs of PFS or OS). Besides, we recorded whether the reported hazard ratio was adjusted or unadjusted, and when adjusted, the covariates included in the model were extracted where available. When both univariate and multivariate analyses were available, the latter were chosen in preference. BMI was analyzed categorically based on thresholds reported in the original studies. Most studies dichotomized patients into high and low BMI groups using cut-off values derived from WHO guidelines or ROC curve–based methods, with continuous variable analysis precluded due to individual patient data unavailability. We confirmed that there were none overlapping cohorts. The study quality was evaluated based on the Newcastle-Ottawa Scale (NOS). The studies with NOS scores of at least 6 were considered having high-quality. 18 All the data were extracted by two authors and data accuracy was checked by the third author.
Statistical Analysis
Stata SE 15.0 software was utilized to conduct the statistical analysis. The Cochran Q test was used to assess the study heterogeneity. Heterogeneity was assessed using Cochran's Q test and the I 2 statistic. Given the expected clinical and methodological heterogeneity among studies, a random-effects model was applied for all pooled estimates. Sensitivity analysis was conducted to assess the result stability. Begg's and Egger's tests were conducted to assess publication bias. The lack of sufficient data not only made a consistent multi-category or dose-response analysis unfeasible due to few studies reporting more than two BMI categories, but also prevented us from conducting meta-regression analyses; consequently, a threshold-specific leave-one-threshold-out sensitivity analysis was performed to test the robustness of the findings. A p < .05 was considered to indicate statistical significance.
Results
Baseline Characteristics of the Included Studies
As presented in Figure 1, 495 relevant literatures from the preliminary search were identified. After removing repetitive studies (n = 57), reviews and case reports (n = 39), meta-analysis (n = 6), non-clinical work (n = 11), irrelevant topics (n = 343), studies published in non-English language (n = 2) or without sufficient posted results (n = 7), 30 studies were finally concluded in the meta-analysis.19–46 The baseline clinical features of the eligible studies were detailed in Table 1, while the prognostic information and quality assessment results were displayed in Table 2. A total of 5987 patients were included in the studies, among which 13 studies were conducted in Japan, followed by China (n = 7), Europe (n = 5), United States (n = 4) and Canada (n = 1). 29 studies were retrospective analyses, with only one prospective work. Most patients received a diagnosis of NSCLC. In terms of evaluated outcomes, 21 trials examined both OS and PFS. The selected studies had BMI cut-off values in the range of 18.5 to 30 kg/m2. The NOS score of each included study was at least 6 points, indicating high quality of data sources.
Baseline Characteristics of the Included Studies in the Meta-Analysis.

Abbreviations: NSCLC, non-small-cell lung cancer; non-sq NSCLC, non-squamous non-small cell lung cancer; SCLC, small-cell lung cancer; LSCC, lung squamous cell cancer; ICIs, immune checkpoint inhibitors; PD-1, programmed cell death protein-1; PD-L1, programmed cell Death-Ligand 1; NA, not available.
Baseline Characteristics of the Included Studies in the Meta-Analysis.

Abbreviations: U, univariate analysis; M, multivariate analysis; OS, overall survival; PFS, progression-free survival; HR, Hazard ratio; CI, confidence interval; NA, not available; NOS, Newcastle-Ottawa Scale.
BMI and OS
To assess the connection between BMI and OS, a pooled prognostic analysis was conducted involving 5589 patients across 27 studies. Due to heterogeneity (I2 = 74.8%, p < .001), a random-effects model was utilized. The findings demonstrated that high BMI was significantly associated with better OS in patients who received ICI-based anti-cancer therapy (HR = 0.69, 95%CI = 0.60-0.80, Figure 2).

Forest Plots of the Hazard Ratios for the Association of Body Mass Index (BMI) With Overall Survival (OS) in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors.
Then, the subgroup analyses were performed based on the cut-off values of BMI. In general, there were 16 studies using the cut-off values between 18.5 and 24.9 kg/m2, 7 studies using that of 25 kg/m2. As shown in Figure S1, for those using the cut-off values ranging from 18.5 to 24.9 kg/m2, high BMI was still associated with better OS (HR = 0.64, 95%CI = 0.53-0.76). For the studies using a cut-off value of 25 kg/m2, overweight patients were found to have no better OS than other patients (HR = 0.86, 95%CI = 0.70-1.06). For the three studies utilizing a cut-off value of 30 kg/m2, obese patients were found to have significantly better OS than other patients (HR = 0.59, 95%CI = 0.42-0.82).
Finally, the subgroups were further stratified according to age, region, sample size, histology classification, therapeutic approach, ICI type and statistical analysis using adjusted or unadjusted HRs (Table 3, Figure S2). As a result, in most subgroups of patients with ICI-treated lung cancer, high BMI was significantly linked with better OS, except for SCLC patients and studies using univariate analysis.
Subgroup Analyses of the Correlation Between BMI and Overall Survival.

Abbreviations: NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer; ICI, immune checkpoint inhibitor; PD-1, programmed cell death protein-1; PD-L1, programmed cell Death-Ligand 1; M, multivariate analysis; U, univariate analysis; CI, confidence interval.
BMI and PFS
The association between BMI and PFS was investigated among 4196 patients across 24 studies. Using a random-effects model (I2 = 67.5%, p < .001), the pooled analysis indicated that high BMI was associated with better PFS in patients who received ICI-based anti-cancer therapy (HR = 0.82, 95%CI = 0.72-0.93, Figure 3).
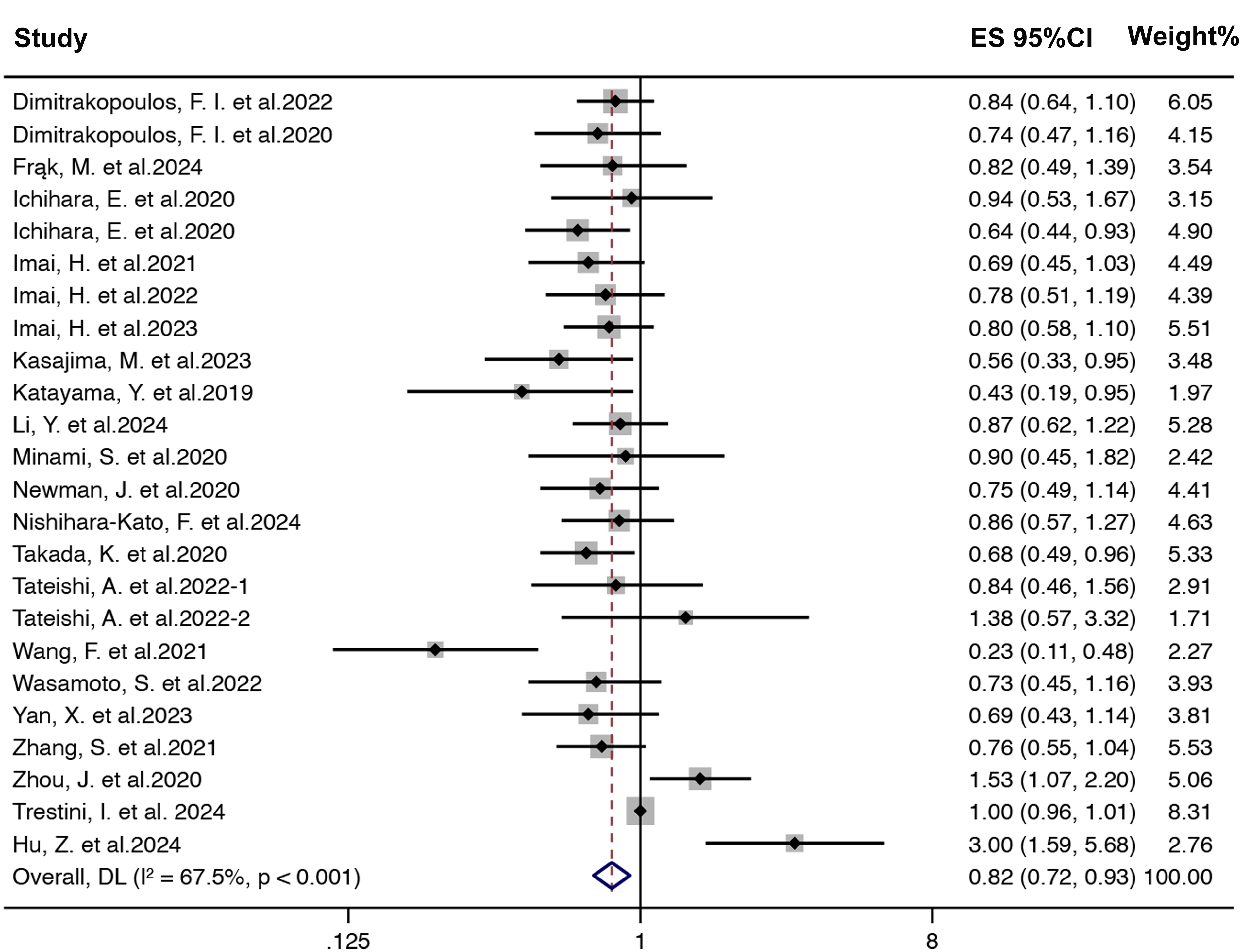
Forest Plots of the Hazard Ratios for the Association of Body Mass Index (BMI) With Progression-free Survival (PFS) in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors.
Next, the subgroup analyses were conducted based on the cut-off values of BMI. As demonstrated in Figure S3, a significantly positive association between BMI and PFS was found in studies using cut-off values between 18.5 and 24.9 kg/m2 (n = 14, HR = 0.73, 95%CI = 0.64-0.83). For the studies using 25 kg/m2 (n = 8) as the cut-off value, no apparent difference was observed in PFS between patients with BMI ≥ 25 kg/m2 and those with BMI < 25 kg/m2 (HR = 1.04, 95%CI = 0.82-1.32). There was only one study using 30 kg/m2 as the cut-off value (HR = 0.82, 95%CI = 0.49-1.38).
Finally, the association of BMI with PFS was also investigated in other subgroups stratified by region, sample size, age, tumor type, treatment strategy, ICI type and statistical analysis using adjusted or unadjusted HRs (Table 4, Figure S4). The pooled analysis demonstrated that high BMI remained linked to better PFS of cancer patients in some subgroups, with the exception of patients from China and other regions, studies with median age less than 68.7 years, patients treated with combined therapies or anti-PD-(L)1 therapy, and statistical analysis using adjusted HRs.
Subgroup Analyses of the Correlation Between BMI and Progression-Free Survival.

Abbreviations: NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer; ICI, immune checkpoint inhibitor; PD-1, programmed cell death protein-1; PD-L1, programmed cell Death-Ligand 1; M, multivariate analysis; U, univariate analysis; CI, confidence interval.
Sensitivity and Publication Bias Analysis
The sensitivity analysis demonstrated that neither the pooled HR value of OS (Figure 4A) nor that of PFS (Figure 4B) was evidently affected by excluding any single publication, confirming the result stability. The Begg's test indicated no publication bias for OS (p = .835, Figure 5A) and PFS (p = .602, Figure 5B). However, the Egger's test revealed potential publication bias in the meta-analysis (p < .01 for both OS and PFS). The trim-and-fill analysis confirmed the publication bias did not substantially impact the results.

Sensitivity Analyses of the Studies Assessing Overall Survival (OS) (A) and Progression-free Survival (PFS) (B).

Begg’s Funnel Plots for Evaluating Publication Bias of Survival (OS) (A) and Progression-free Survival (PFS) (B).
Adjusted Hazard Ratios (HRs)
To account for the non-collapsibility of HRs and potential confounding, we performed subgroup analyses stratified by adjustment status. Studies reporting adjusted HRs (clinical covariates presented in Table 5) were analyzed separately from those providing unadjusted HRs. For OS, the pooled adjusted HR demonstrated that patients receiving ICI-based anti-cancer therapy with higher BMI had significantly improved survival (HR = 0.59, 95%CI = 0.50-0.70, Figure S2G). A similar pattern was observed for PFS, where adjusted HRs showed a favorable effect of higher BMI in patients who received ICI-based anti-cancer therapy (HR = 0.79, 95%CI = 0.63-1.01, Figure S4G), although no statistically difference was found. We further stratified the adjusted HR studies by BMI thresholds, and the findings were largely consistent with those of all included studies, with the exception of the studies using a cut-off value of 25 kg/m2 for PFS (Figure S5 and Figure S6).
Adjusted Factors of the Studies Utilized Multivariate Analyses.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; PS, Performance Status; BMI, body mass index; NLR, neutrophil-to-lymphocyte ratio; PNI, prognostic nutritional index; irAEs, immune-related adverse events; PD-L1, programmed cell Death-Ligand 1; LDL, low density lipoprotein; HDL, high density lipoprotein; PLR, platelet-lymphocyte ratio; LIPI, Lung Immune Prognostic Index; GPS, Glasgow prognostic score; CRP, C-reactive protein; LMR, Lymphocyte-to-monocyte ratio; EGFR, Epidermal Growth Factor Receptor; ALK, Anaplastic Lymphoma Kinase; ANC, Absolute Neutrophil Count; LDH, Lactate Dehydrogenase; ACCI, Age-adjusted Charlson comorbidity index; KPS, Karnofsky Performance Status; WBC, white blood cell; NA, not available.
To evaluate the robustness of these associations, we conducted a threshold-specific leave-one-threshold-out sensitivity analysis by sequentially excluding each BMI cut-off definition from the adjusted HR studies as shown in Figure S7. The pooled HRs for both OS and PFS remained stable across iterations, indicating a consistent and generalizable association.
Discussion
Although introduction of ICI drugs has revolutionized the current therapeutic paradigm for lung cancer, identification of therapy related biomarkers is challenging to clinicians. The advancing sequencing techniques have suggested several powerful biomarkers such as TMB, but its value may vary greatly with different platforms and parameters. 47 The immunohistochemistry has been widely used to detect the intratumoral expression of PD-L1, while the cut-off criteria (50% or 1%) for determining ICI monotherapy is controversial. 48 Increasing studies have suggested some noninvasive blood biomarkers such as prognostic nutritional index (PNI) and neutrophil-to-lymphocyte ratio, but their actual prognostic value may be limited and there is a lack of standardized cut-off criteria. 49 BMI, as a accessible clinical standardized parameter, has been identified as a favorable prognostic predictor for lung cancer in numerous previous studies.50–52 With regard to its role in ICI therapy, several meta-analyses have been already performed and their results collectively demonstrated high BMI is linked to better outcome but increased risk of immune-related adverse events (irAEs) in solid cancers.53–55 However, these studies either included limited lung cancer data or combined diverse cancer types, limiting tumor-specific conclusions. Therefore, our study provides an updated and lung cancer-focused meta-analysis based on a significantly larger dataset to evaluate the prognostic impact of BMI specifically in lung cancer patients receiving ICI-based therapies, with 30 studies and nearly 6000 patients included. We also conducted detailed subgroup analyses stratified by BMI cut-off values, geography, histological subtype, and therapeutic strategy, offering new insights into how BMI affects clinical outcomes in this population.
In the global meta-analysis, a significant association was observed between high BMI and better OS and PFS in ICI-treated patients with lung cancer. The result stability and reliability were further validated through sensitivity analysis and assessment of publication bias. Imai et al discovered that high BMI was associated with better therapy response and identified as an independent favorable predictor for the OS of NSCLC patients with high PD-L1 expression who received first-line pembrolizumab monotherapy. 37 The researchers also found high BMI was associated with improved OS of NSCLC patients receiving second-line nivolumab monotherapy. 38 This association remains statistically significant in non-squamous NSCLC patients receiving pembrolizumab. 39 In addition to its association with OS and PFS, BMI was identified as a promising predictor for overall response and disease control in NSCLC patients who received anti-PD-1 therapy. 27 In contrast, Tateishi et al found no significant difference of therapy response and survival between patients with BMI > 25 kg/m2 and BMI < 25 kg/m2. 26 The secondary analysis from IMpower150 and IMpower130 clinical trials demonstrated high BMI was not associated with prognostic benefit in patients with advanced non-squamous NSCLC receiving front-line chemoimmunotherapy. 56 A recent multicenter study demonstrated obesity was notably associated with prolonged OS in NSCLC patients who received chemotherapy instead of those receiving immunotherapy or chemoimmunotherapy. 57 The further investigation in this work revealed that obese patients had lower level of TMB, similar PD-L1 expression and immune scores as compared with those with normal weight or overweight, partly explaining the result. With regard to its role in other cancers, the controversy remains existing. Wang et al found BMI could independently predict the OS in patients with advanced hepatocellular carcinoma who received lenvatinib plus camrelizumab. 58 Obesity is associated with better disease control and PFS in patients with microsatellite-instability-high endometrial cancer who received pembrolizumab therapy. 59 However, a recent comprehensive review suggested BMI should not be currently recommended as a prognostic biomarker for ICI-treated melanoma patients due to limited evidences. 60 Therefore, despite the positive result of the meta-analysis, the actual predictive role of BMI still needs more validation. A recent retrospective study has demonstrated BMI was not an independent predictor for the prognosis of lung cancer patients who received ICIs, but its combination with other clinical markers such as PD-L1 expression contributed to a more accurate prognostic stratification. 61 This finding suggested BMI alone may not be as a powerful prognostic biomarker and selecting suitable clinical parameters to combine with BMI may be more beneficial in outcome prediction. In conclusion, BMI is associated with prognosis in ICI-treated lung cancer, but it should be viewed primarily as a risk marker rather than a therapeutic target. BMI reflects nutritional and metabolic status and is strongly correlated with other prognostic variables which may confound its apparent effect. Thus, BMI alone is insufficient for patient stratification but may contribute to a more pragmatic prognostic framework when integrated with PD-L1, ECOG, weight-loss trajectory, and sarcopenia.
Given the substantial variation in BMI cut-off values among the included studies, ranging from 18.5 kg/m2 to 30 kg/m2, a subgroup analysis was conducted to elucidate their influence on the results. Specifically, for studies with cut-off values between 18.5 kg/m2 and 24.9 kg/m2, high BMI was substantially linked to better OS and PFS. However, for the cut-off value defined as 25 kg/m2 (overweight plus obesity vs normal weight plus underweight), this association failed to reach statistical significance. This finding indicated that single BMI > 25 kg/m2 may not be a suitable cut-off value for identifying patients who could benefit from ICI therapy. For patients with BMI < 25 kg/m2, there are individuals with normal weight or with underweight, whose nutritional status and immune microenvironment are highly heterogeneous. Therefore, the therapy response to ICI drugs may vary dramatically among these patients and should be further accurately classified. A recent dose-response analysis demonstrated a below normal BMI was linked to shorter OS while OS benefit was only observed in patients with BMI > 25 kg/m2 but less certain in those with BMI > 30 kg/m2. 62 In our subgroup analyses, although OS benefit is observed in patients with BMI > 30 kg/m2, there are only three eligible studies included and more validations are urgently needed. On the other hand, to our knowledge, there are two methods currently used for determining the cut-off values of BMI in the included studies. One is the criterion defined by World Health Organization, where a BMI greater than or equal to 25 or 30 kg/m2 indicates overweight or obesity respectively.19–21,25,26,40–42,44,45 The other is the receiver operator characteristic curve analysis based on individual BMI data and survival data.23,24,2736–38 It is difficult to determine which method is better, but it is reasonable to use multiple cut-off values (such as 18, 25 and 30 kg/m2) instead of a single one (such as 25 kg/m2) to stratify the patients who may benefit from or insensitive to ICI therapy, due to the complexity of the association between BMI and clinical outcome.
There are some reasons used to explain the relationship between BMI and the prognosis of the patients who received ICI therapy. Firstly, individuals with high BMI generally have better performance status than those with low BMI, and therefore are more likely to receive more durable ICI therapy even coupled with other anti-therapies including chemoradiotherapy or targeted therapy. Secondly, high BMI is commonly linked with better dietary intake and nutritional status. Various nutrients such as amino acids, lipids and carbohydrates are crucial for the proliferation and activation of T cells, subsequently enhancing immune surveillance. 63 In extensive-stage SCLC patients who received immunochemotherapy, low BMI was significantly associated with sarcopenia and worse therapy response. 64 Thirdly, high BMI are found to associate with increased serum levels of CD4+ and CD8+ T cells in patients with lung squamous cell carcinoma, which creates a favorable immune microenvironment for ICI therapy. 24 Finally, a mechanism investigation has revealed increased PD-1 expression in the immune cells of obese mice as compared with non-obese mice, contributing more effective anti-cancer efficacy of PD-1 inhibitors. 65 However, this study also found obesity also promoted tumor growth and T cell exhaustion in mice, adding complexity into this issue. Due to limited evidence, the association between BMI and irAEs in lung cancer patients was not investigated in this meta-analysis. One study suggested BMI < 21.26 kg/m2 is a predictive factor for increased risk of Grades 3-4 ICI related pneumonia in lung cancer patients. 66 In a retrospective Asian cohort of NSCLC patients who received anti-PD-L1 therapy, increased BMI was associated with increased risk of any grade irAEs. 67 A Mendelian randomization analysis has closely linked high BMI with increased incidence of ICI related thyroid dysfunction. 68 However, a retrospective single center study found no difference regarding irAEs incidence between patients with BMI < 30 kg/m2 and ≥30 kg/m2. 69 Another retrospective study has also found a similar result between patients with overweight and normal BMI, but those with overweight and irAEs appeared to have improved PFS than others. 70 The occurrence of some irAEs such as endocrine dysfunction are associated with enhanced ICI efficacy, while others such as pulmonary and hepatic adverse events may lead to therapy discontinuity and even death. 71 Therefore, the association among BMI, prognosis and irAEs is complicated and requires further clinical investigation.
Our study has several limitations. Firstly, there are various heterogeneous factors that potentially affect the result, such as patient selection, therapy strategy and ICI type. In addition, the publication bias exists although the trim-and-fill analysis confirmed it failed to significantly affect the result. Secondly, the association of obesity (≥30 kg/m2) with OS or PFS was not sufficiently evaluated due to the limited studies (n = 3). Thirdly, the included studies mainly focus on NSCLC, and the prognostic role of BMI on SCLC is poorly investigated. Furthermore, due to the absence of individual patient data and inconsistent reporting formats, we did not use BMI as a categorical variable based on a consistent reference group. While threshold-specific subgroup and leave-one-threshold -out sensitivity analyses helped mitigate this, the lack of uniform BMI thresholds across studies still exists. Utilizing normal weight as a consistent reference is recommended in following studies. Finally, more attention should be paid to the potential association between BMI and irAEs.
Conclusion
Our findings demonstrate that high BMI is linked to improved OS and PFS in patients with lung cancer who received ICI-based anti-cancer therapy. Due to study limitations, more multicenter validations are necessary for verifying its prognostic value. Meanwhile, it is an urgent task to develop novel nutritional intervention strategies to modulate the optimal BMI for ICI therapy.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251394573 - Supplemental material for Prognostic Impact of Body Mass index in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors: An Updated Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338251394573 for Prognostic Impact of Body Mass index in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors: An Updated Systematic Review and Meta-Analysis by Zhe Wang, Mengxue Yang, Yanlin Zhao, Jiaxin Wang, Mingyang Tao, Jingjing Yang, Jingxian Mao, Huaijuan Guo, Guohui Zhu and Xuebing Yan in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338251394573 - Supplemental material for Prognostic Impact of Body Mass index in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors: An Updated Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-tct-10.1177_15330338251394573 for Prognostic Impact of Body Mass index in Lung Cancer Patients Receiving Immune Checkpoint Inhibitors: An Updated Systematic Review and Meta-Analysis by Zhe Wang, Mengxue Yang, Yanlin Zhao, Jiaxin Wang, Mingyang Tao, Jingjing Yang, Jingxian Mao, Huaijuan Guo, Guohui Zhu and Xuebing Yan in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the program of Natural Science Foundation of Jiangsu Province, National Natural Science Foundation of China, China Postdoctoral Science Foundation, Jiangsu Traditional Chinese Medicine Science and Technology Development, the Program of Yangzhou Commission of Health, (grant number SBK2023022610, 81902422, No.MS2024140, No. 2023-2-01, 2024-2-08).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
