Abstract
Introduction
This study aimed to perform a rigorous dosimetric comparison of three advanced radiotherapy techniques—Volumetric Modulated Arc Therapy(VMAT), Helical Tomotherapy (TOMO), and Intensity-Modulated Radiation Therapy(IMRT)—for nasopharyngeal carcinoma (NPC), with particular emphasis on target coverage homogeneity, organ-at-risk (OARs) sparing, and the impact of using a unified treatment planning system (TPS) to eliminate inter-platform biases.
Methods
A total of 62 non-metastatic NPC patients were included in this retrospective study. All plans were re-optimized and calculated using a single TPS (RayStation 10B) with identical dose calculation algorithms to ensure comparability. Each patient underwent planning with dual-arc VMAT, helical TOMO, and 9-field IMRT techniques. Plans were evaluated based on homogeneity index (HI), conformity index (CI), and dose-volume parameters for multiple serial and parallel OARs. Statistical analyses included ANOVA with post-hoc Tukey tests.
Results
All techniques achieved adequate target coverage. VMAT yielded significantly superior homogeneity across all target volumes (HI = 0.06 ± 0.01 for PGTV70, p < 0.05), with no significant differences in CI among techniques. Both VMAT and TOMO significantly reduced doses to serial OARs such as the brainstem and spinal cord compared to IMRT (p < 0.05). TOMO provided the best parotid glands sparing (mean dose 26.13 ± 1.76 Gy), outperforming both VMAT and IMRT (p < 0.01). VMAT achieved the lowest brainstem mean dose (21.43 ± 4.47 Gy), while TOMO excelled in reducing high-dose volumes for most parallel OARs.
Conclusion
VMAT offers superior dose homogeneity and reduced delivery time, making it a highly efficient and dosimetrically favorable option for NPC radiotherapy. TOMO demonstrates advantages in sparing critical OARs, particularly in high-dose regions. The use of a unified TPS platform confirms that observed differences are technique-derived rather than planning-system artifacts. These findings support technique selection based on institutional resources and clinical priorities.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) demonstrates a distinctive geographical distribution, being highly endemic in Southern China and Southeast Asia. Radiotherapy constitutes the cornerstone of definitive treatment for this malignancy. The evolution from conventional techniques to intensity-modulated radiation therapy (IMRT) has marked a paradigm shift, significantly improving local-regional control and overall survival while concurrently reducing the severity of radiation-induced toxicities through superior dose conformity.1,2 The pursuit of optimal dosimetric outcomes and treatment efficiency has propelled the development of advanced IMRT delivery techniques. Among these, Volumetric Modulated Arc Therapy (VMAT) achieves highly conformal dose distributions through the continuous rotation of the gantry, synchronized with dynamic multileaf collimator (MLC) motion, variable dose rates, and gantry speed modulation.3,4 Compared to traditional fixed-field IMRT, VMAT offers comparable or improved plan quality with a substantial reduction in monitor units (MUs) and treatment delivery time, potentially enhancing patient comfort and minimizing intrafractional motion.5,6 Helical tomotherapy (TOMO), another advanced form of rotational IMRT, utilizes a fan beam and binary MLC to deliver highly conformal doses with integrated image-guidance capabilities. 7 Numerous dosimetric studies have compared fixed-field IMRT, VMAT, and TOMO for NPC.8,9 However, a critical, often overlooked confounding factor in such comparisons is the heterogeneity of treatment planning system(TPS) and dose calculation algorithms used. Inconsistent optimization engines and dose calculation models (eg, Pencil Beam vs Monte Carlo) across different TPS platforms can introduce significant variations in dose reporting, making it challenging to attribute dosimetric differences solely to the delivery technique itself.10–12 Consequently, the question of whether the reported dosimetric advantages of one technique over another are genuine or merely artifacts of disparate planning and calculation methodologies remains inadequately addressed.
This single-institutional, retrospective study was therefore designed to conduct a rigorous and unbiased dosimetric comparison of three advanced techniques: step-and-shoot IMRT (using a 9-field beam arrangement), dual-arc VMAT, and helical TOMO for the radiotherapy of NPC. The principal strength and novelty of our investigation lie in the fact that all treatment plans for 62 patients were meticulously re-optimized and calculated within a single, unified TPS platform (RayStation 10B, RaySearch Laboratories, Sweden) using an identical dose calculation algorithm. 13 This stringent methodology effectively eliminates inter-platform discrepancies, ensuring that any observed dosimetric differences are intrinsically attributable to the technical characteristics of each delivery modality rather than variations in TPS performance. Our analysis provides a clear, equitable evaluation of the capabilities of each technique in achieving target coverage and sparing critical OARs, thereby offering valuable insights for clinicians in selecting the most appropriate radiotherapy technique for NPC.
Material and Methods
Patient Enrollment and Target Delineation
This retrospective study enrolled 62 patients with histologically confirmed, non-metastatic nasopharyngeal carcinoma, all of whom were treated at West China Hospital of Sichuan University between October 2024 and February 2025. The study protocol was approved by the Institutional Review Board of the West China Hospital Biomedical Research Ethics Committee (Approval No.: 20241535). The requirement for informed consent was waived due to the use of anonymized treatment planning data. To ensure planning consistency and eliminate inter-planner variability, all treatment plans for each technique were developed by a single, highly experienced medical dosimetrist specializing in head and neck radiotherapy. Patient disease staging was classified according to the American Joint Committee on Cancer (AJCC) Staging System, ninth Edition. 14 In terms of treatment regimen, the majority of patients (88%) received three cycles of induction chemotherapy followed by concurrent chemoradiotherapy, while a minority with early-stage disease (12%) underwent concurrent chemoradiotherapy alone. The clinical characteristics of the 62 enrolled patients and the corresponding planning irradiation volumes are summarized in Table 1.
Clinical Finding of 62 Patients with Nasopharyngeal Carcinoma.

AJCC, American Joint Committee on Cancer 14
Prior to simulation, all patients were immobilized in the supine position using a customized thermoplastic head-neck-shoulder mask. Planning computed tomography (CT) simulation was performed using a GE Revolution CT scanner (GE Healthcare, USA). Images were acquired from the cranial vertex to the sternoclavicular joint with a slice thickness of 3 mm. For superior soft-tissue delineation, diagnostic magnetic resonance imaging (MRI) was acquired on a GE Discovery 750 3.0 T scanner. The MRI protocol included contrast-enhanced T1-weighted sequences and T2-weighted sequences, which were utilized for improved visualization of the primary tumor and involved nodes. MR-CT image fusion was performed within the RayStation 10B TPS using a rigid registration algorithm 15 supplemented by manual refinement based on anatomical landmarks. Registration accuracy was verified by a qualified medical physicist and radiation oncologist, ensuring a residual error of less than 2 mm in all translational and rotational dimensions. All target volumes were contoured on the fused imaging dataset, with the contrast-enhanced T1-weighted MRI series serving as the primary reference for soft-tissue definition.
Gross Tumor Volume (GTV) delineation was performed following the RTOG 0225 and 0615 guidelines,16,17 encompassing both the primary nasopharyngeal tumor (GTVnx) and any metastatic regional lymph nodes (GTVnd) identified either by a diameter greater than 10 mm or by the presence of central necrosis on CT imaging. The GTV and involved lymph nodes were expanded isotropically by 5 mm to generate the high-dose clinical target volume, designated GTV70. High-risk regions, including the entire nasopharynx, clivus, skull base, pterygoid fossae, parapharyngeal space, inferior sphenoid sinus, posterior third of the nasal cavity and maxillary sinuses, as well as the retropharyngeal and upper deep jugular nodal areas, were encompassed within CTV60. The low-risk elective volume, CTV56, included uninvolved lower jugular and supraclavicular nodal regions. To account for inter-fractional setup variations, each clinical target volume was expanded by a 3 mm margin to create the corresponding planning target volume (ie, PGTV70, PCTV60, PCTV56). A simultaneous integrated boost (SIB) technique was employed. The actual delivered doses were 69.96 Gy, 60.06 Gy, and 56.10 Gy in 33 fractions to PGTV70, PCTV60, and PCTV56, respectively, ensuring an integer dose per fraction, which resulted in a minor discrepancy between the nominal target volume names and the prescribed doses. 18 Figure 1 illustrates the delineation of three graded target volumes in a representative patient, as well as their spatial relationships relative to surrounding organs at risk.

Spatial configuration and delineation of graded target volumes and spatial relationship between target volumes and critical organs at risk.
In accordance with ICRU 83 recommendations, 19 the planning objectives stipulated that D98% (near-minimum dose) for PGTV70, PCTV60, and PCTV56 should be no less than 90% of the prescribed dose for each respective volume. Additionally, D97% for all three planning target volumes was required to reach at least the prescribed dose. For PGTV70, the D2% (near-maximum dose) was constrained to a maximum of 108% of its prescribed dose. (Note: D2%, D98%, and D95% refer to the minimum dose absorbed by the highest 2%, 98%, and 95% of the target volume, respectively.) Regarding OARs, dose constraints were set as Table 2.
Target Dose Cover and Dose Constrains for the Critical Structures.

Abbreviation: OARs – Organs at risk, Dx% represents the dose delivered to x% of the target volume, Max dose-Maximum dose, Dmean – mean dose, Vx represents the % volume receiving x Gy, PRV – planning organ at risk volume, dose limit values for OARs PRV are indicated in parentheses.
Planning Technique
All treatment plans were optimized and calculated using the RayStation 10B. The system employs a gradient-based inverse planning algorithm with direct machine parameter optimization (DMPO) for dose calculation. 20 Key planning objectives were as follows: to ensure that 97% of the PTV received 100% of the prescribed dose; to restrict the maximum dose to serial critical OARs within established guideline limits; and to minimize the radiation exposure to all other OARs as much as possible. The optimization tolerance was set to 1 × 10−5. The maximum number of iterations per optimization run was 120, with 15 iterations allowed before conversion. Optimization constraint weighting priorities were assigned as follows: highest for target volumes, followed by OARs, and finally for auxiliary structures. In clinical practice, however, absolute priority was consistently given to meeting the hard dose constraints for serial organs such as the brainstem and spinal cord. This commitment to critical organ sparing could, in certain cases, necessitate slight trade-offs—such as accepting minor compromises in target coverage homogeneity or elevated doses to certain lower-priority OARs—in order to ensure patient safety and adherence to established clinical limits.
Final dose calculation was performed using the Collapsed Cone Convolution (CCC) algorithm
21
with a high-resolution calculation grid of 3.0
For the IMRT technique employed in this study, a nine-field step-and-shoot approach was utilized with a total of 100 segments. A coplanar beam arrangement was adopted with the following fixed gantry angles: 181°, 220°, 260°, 300°, 340°, 20°, 60°, 100°, and 140°. These angles were strategically selected to avoid traversing critical OARs and to enhance dose conformity. The collimator angles were set between 10° and 30° to minimize interleaf leakage. For the dual-arc VMAT technique, the following configuration was applied: Arc 1 rotated clockwise from 181° to 180° with a collimator angle of 10°, while Arc 2 rotated counterclockwise from 180° to 181° with a collimator angle of 350°. A 4° spacing between arcs was maintained to reduce interleaf transmission, and leaf motion was constrained to 5 mm per degree of gantry rotation. In the case of TOMO, treatment plans were generated using a continuous 360° rotation with a field width of 2.5 cm and a pitch of 0.287. The delivery time factor was set to 1.5. To ensure planning consistency and minimize inter-planner variability, all plans were designed by a single clinical medical physicist and subsequently reviewed independently by two senior medical physicists.
Dose-Volume Histograms (DVH) and Dose Comparisons
Dose distribution parameters were systematically evaluated using validated indices for target coverage and OARs sparing. For planning target volumes (PGTV70, PCTV60, PCTV56), dose homogeneity was quantified via the Homogeneity Index (HI) defined as
23
:

Vx% represents the relative volume of the target irradiated by x% prescription dose; AVx% represents the absolute volume; AVtarget represents the target's geometric volume; TAV100% is the absolute volume receiving 100% prescription dose.
Radiation-sensitive OARs exhibit distinct functional architectures that determine their dose-response behaviors. Serial OARs, such as the Brain Stem (including its planning risk volume, PRV), Lens L/R (with PRVs), Optic Nerve L/R, Optic Chiasm, Spinal Cord (with PRV), and Temporal Lobe L/R, are characterized by threshold-dependent toxicity. Localized overdose—even to small volumes—beyond tolerance thresholds can lead to severe functional impairment. In contrast, parallel OARs tolerate irradiation through functional redundancy, preserving global organ function via compensatory mechanisms when subvolumes receive subcritical damage. 24
For serial OARs, dosimetric evaluation included mean dose (Dmean) and near-maximum doses (D0.01cc), along with the median dose (D50%). 25 The metric D0.01cc (dose to the hottest 0.01 cm3) was adopted instead of Dmax to standardize reporting across TPS and minimize system-specific artifacts, while maintaining clinical relevance to serial tissue tolerance as recommended by ICRU Report 91. 26
Parallel OARs assessed in this study comprised the Brain, Ear L/R, Larynx, Oral Cavity, Mandible L/R, Parotid L/R, temporomandibular joints (TMJ L/R), and Thyroid. Dose-volume parameters assessed for these structures included the Dmean and volumetric parameters V5, V20, V30, V40, V50, V60, and V70, where Vx denotes the volume percentage (%) receiving at least x Gy.
Statistical Analysis
All statistical analyses were conducted with SPSS version 26.0 (IBM SPSS Statistics). Continuous variables were presented as mean
Results
Target Coverage
Dose distributions for all treatment plans across the three techniques—IMRT, VMAT, and TOMO—met all clinical constraints outlined in Table 2 for the 62 enrolled NPC patients. Figure 2A presents the volumetric dose data of the target regions for the three treatment techniques. A quantitative evaluation of plan quality, including the CI and HI for each technique, is provided in Table 3. No significant differences were observed in the CI values among the three techniques for all three target volumes (p < 0.05). Regarding the HI, both VMAT and TOMO demonstrated significantly better dose homogeneity than IMRT across all target volumes (p < 0.05). For the PGTV70, VMAT demonstrated the lowest homogeneity index (HI) of 0.06 ± 0.01, along with a significantly lower median dose (D50%) of 72.18 ± 0.15 Gy compared to both IMRT and TOMO (p < 0.05).

Dose-volume metrics and distributions of the HI and CI for the three gradient target volumes across the three radiotherapy plan groups.
Comparison of Target Volume Homogeneity and Conformity in all Three Radiotherapy Plan Groups, Mean ± SD (Gy).

Abbreviation: SD: standard deviation; D2%: the minimum dose received by the hottest 2% of the target volume, D50%: the median dose to the target volume, D98%: the minimum dose received by the hottest 98% of the target volume, HI: homogeneity index; CI: conformity index; ANOVA: one-way analysis of variance, HSD: Tukey honestly significant difference.
Serial OARs
The Dmean and Dmax to serial organs for the three irradiation techniques are summarized in Table 4. All three planning techniques achieved clinically acceptable dose constraints. No significant differences were observed among the three techniques in doses to the Lenses, Optic Chiasm, Optic Nerve L, Optic Nerve R, or Temporal Lobe L/R. However, both VMAT and TOMO yielded significantly lower Dmean and Dmax to the Brain Stem and Spinal Cord compared to IMRT (p < 0.05), as showed in Figure 3. For the Brain Stem PRV, TOMO yielded the lowest Dmax of 49.49 ± 7.03 Gy, which was significantly lower than that of IMRT (p < 0.05), while no significant difference was observed between TOMO and VMAT. For the Spinal Cord PRV, both VMAT and TOMO resulted in significantly reduced Dmax values compared to IMRT(p < 0.05), with no statistically significant difference between the two techniques.

Dmean and dmax to brain stem and spinal cord for the three techniques.
Dosimetric Comparison of Serial OARs in Three Radiotherapy Plan Groups, Mean ± SD (Gy).

Abbreviation: Dmax: maximum dose, Dmean: mean dose.
Parallel OARs
The mean doses and volumetric parameters (Vx) for parallel organs are provided in Table 5. Dose-volume metrics for selected parallel organs are further depicted in Figure 4. For the left and right ears and oral cavity, no statistically significant differences were found in Dmean, V30, or V40 between IMRT and VMAT. In contrast, TOMO delivered significantly lower values for these parameters (p < 0.05). Additionally, TOMO achieved the lowest Dmean values for the Parotid L and R (26.30 ± 1.87 Gy and 25.97 ± 1.64 Gy, respectively) and the Thyroid (42.11 ± 3.95Gy), with statistically significant differences compared to both IMRT and VMAT. No significant dose differences were observed among the techniques for the TMJ L/R.

Dose-volume histograms for selected parallel organs across the three treatment techniques.
Dosimetric Comparison of Parallel OARs in Three Radiotherapy Plan Groups, Mean ± SD (Gy).
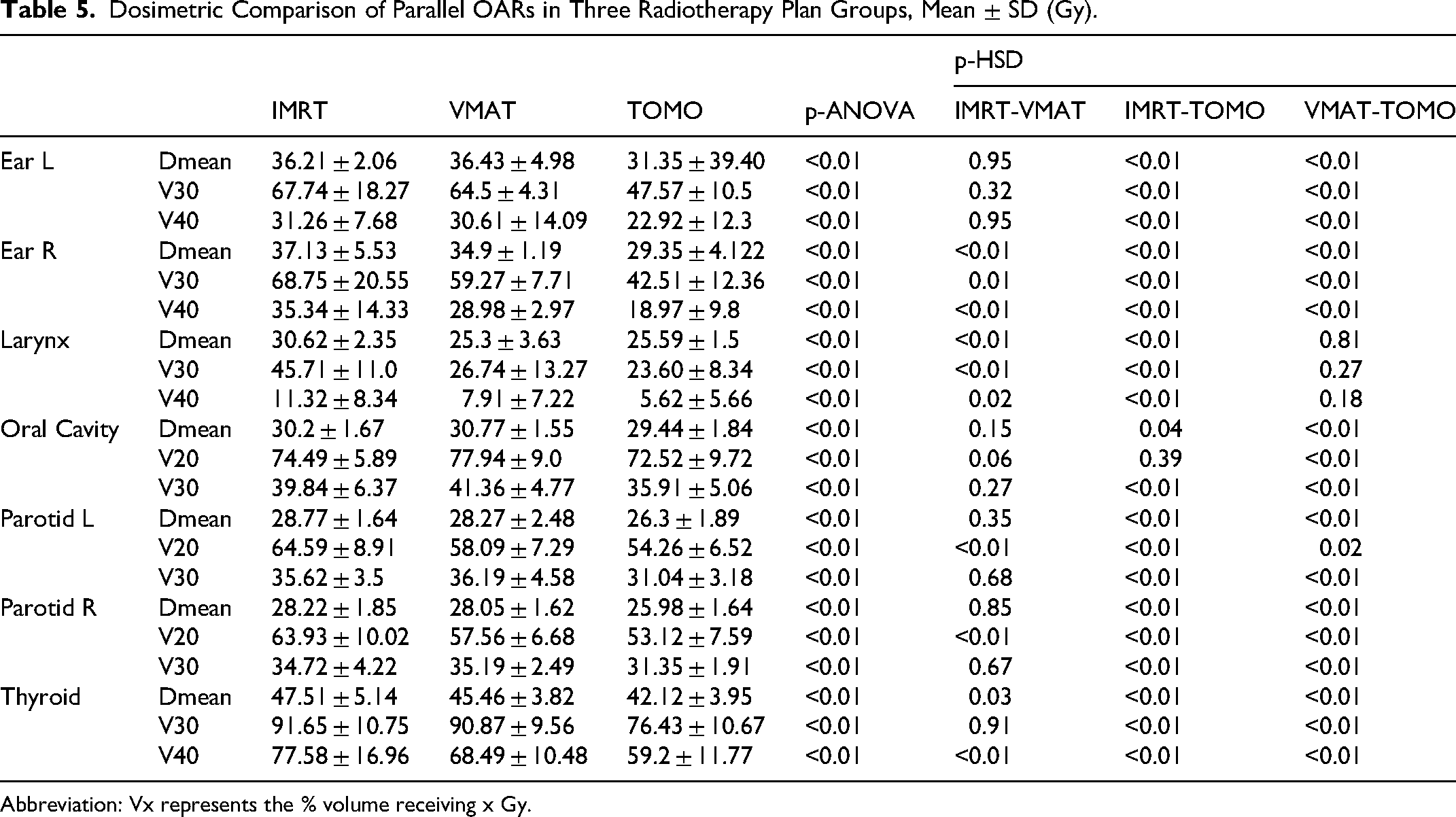
Abbreviation: Vx represents the % volume receiving x Gy.
Discussion
NPC demonstrates a distinct geographic distribution, with high incidence in southern China, Southeast Asia, and other endemic regions. 27 Radiotherapy serves as the primary treatment modality, offering high rates of local control. However, the high frequency of contralateral lymphatic metastasis often necessitates comprehensive bilateral neck irradiation. The typical target volume extends from the skull base to the supraclavicular fossa, resulting in larger treatment fields compared to other head and neck cancers. The proximity of multiple OARs further complicates planning, emphasizing the need for techniques that maximize target coverage while minimizing doses to adjacent normal tissues. Over 70% of patients present with stage III–IV disease, often with skull base invasion or intracranial extension, further increasing planning complexity.28,29 Despite these challenges, radiotherapy yields favorable survival outcomes, with 5-year rates reaching 85%–97% for early-stage disease and approximately 75% for advanced stages.30–32
Advanced techniques such as IMRT, VMAT, and TOMO have significantly improved dose conformity and reduced toxicity, thereby enhancing the therapeutic ratio and quality of life. In this study, we conducted a comprehensive dosimetric comparison of IMRT, VMAT, and TOMO in NPC radiotherapy. Although all techniques provided clinically acceptable target coverage, significant dosimetric differences were observed. VMAT consistently demonstrated superior dose homogeneity, with significantly improved HI values compared to IMRT and TOMO (p < 0.05). While CI values were comparable across techniques, VMAT showed advantageous dose coverage metrics, including lower D2% and higher D98% values Significant differences in D50% further supported the favorable dose distribution achieved with VMAT.
Notably, our study required at least 97% of the target volume to receive the prescription dose, exceeding the commonly adopted 95% threshold in many protocols. This stricter criterion may explain the higher D98% value for the PGTV70 target in our VMAT plans compared to results reported by Radhakrishnan et al 33 In terms of OARs sparing, both VMAT and TOMO outperformed IMRT, consistent with previous studies. Parotid L/R protection was notably improved, with V20 reduced by 6%–11% (p < 0.001) using arc-based techniques. TOMO achieved the lowest mean parotid dose (26.13 ± 1.76 Gy) and V30 (31.0 ± 2.5%), lower than both IMRT and VMAT, and also below values reported by Radhakrishnan et al. 33 Although Lu et al 8 reported that TOMO excelled in high-dose sparing (V30) and VMAT in intermediate doses (V20), our unified TPS platform showed TOMO outperforming VMAT in both V20 and V30 for parotid glands.
VMAT also significantly reduced the Dmax to the Spinal Cord by 15%, compared to a 6.4% reduction with TOMO. These findings align with reports by Vanetti et al 34 in other head and neck cancers, reinforcing the value of arc therapies in complex anatomical scenarios. Interestingly, VMAT achieved the lowest mean Brain Stem dose (21.43 ± 4.47 Gy), a 14% reduction over IMRT. TOMO showed a modest but significant reduction in Brain Stem Dmax versus IMRT (9.7%, p < 0.01). TOMO also yielded lower mean doses and V30/V40 values for the Ear L/R and Thyroid compared to IMRT and VMAT (p < 0.01). In contrast, VMAT provided better sparing of the Eye L/R, with mean and maximum doses reduced by 11.6–14.8% and 5.2–9.5%, respectively (p < 0.01 vs IMRT). No significant differences were observed among the three techniques in mean or maximum doses to the Lens L/R, Optic Chiasm, Optic Nerve L/R, or TMJ L/R suggesting that technical advantages are organ-specific—an important consideration in clinical decision-making. An interesting finding was that IMRT achieved lower mean doses to the Brain tissue and Temporal Lobe L/R (7.54 ± 1.67 Gy and 11.44 ± 3.11 Gy, respectively) compared to VMAT (8.62 ± 1.94 Gy and 13.66 ± 3.27 Gy) and TOMO (8.26 ± 1.98 Gy and 13.08 ± 3.40 Gy). This may be attributable to the limited beam angles used in IMRT, resulting in reduced exposure to these structures. In terms of overall plan quality, VMAT provided superior homogeneity within the high-dose target volume (PGTV70), while TOMO offered better protection for several OARs, particularly parallel organs.
Given that not all institutions have access to TOMO, VMAT represents a more universally applicable technique with robust dosimetric performance in NPC radiotherapy. Figure 5 summarizes selected OARs dose-volume metrics from this study, which are generally lower than those reported in previous publications.8,33–34 Although international guidelines provide dose constraints for OARs in NPC radiotherapy (Table 2), these should be considered minimum standards. Clinicians and physicists should strive to further reduce OARs doses without compromising target coverage. The high plan quality achieved in this study—even under stricter coverage criteria—reflects our institution's commitment to excellence in NPC radiotherapy planning. For example, the mean and maximum Brain Stem doses in IMRT plans (25.07 ± 3.4 Gy and 45.59 ± 5.18 Gy) were lower than those in prior studies. Similarly, VMAT achieved lower mean and maximum optic apparatus doses (11.92 ± 2.03 Gy and 28.9 ± 4.83 Gy) compared to Ning et al 9 (23.46 ± 2.74 Gy and 32.59 ± 2.91 Gy). All VMAT plans demonstrated efficient beam delivery performance, achieving a mean total MUs of 920 ± 30 per fraction. This result is significantly lower than the previously reported value of 1232.1 ± 146.2 MU in the literature, 9 confirming the superior delivery efficiency of our planning strategy.

Volume Fractions (V5–V70) of all parallel OARs in VMAT for 62 NPC patients. Each subplot ranks OARs in descending order of volume fraction at the respective x Gy dose level.
An important consideration in interpreting our dosimetric results is their potential translation into clinical benefits. While all evaluated techniques met the institutional dose constraints for OARs (Table 1), the observed differences in dose distribution may still hold clinical significance. According to established NTCP models, the probability of complications for serial organs such as the spinal cord and brainstem is predominantly determined by the maximum dose (Dmax), where even incremental reductions below tolerance limits can substantially decrease toxicity risks. 35 Similarly, for parallel organs including the parotid glands, the mean dose (Dmean) serves as a robust predictor of functional impairment, with well-documented dose-response relationships demonstrating reduced complication rates at lower dose levels. 36
In our study, techniques that achieved lower doses to critical structures—such as VMAT's superior sparing of the brainstem and spinal cord, and TOMO's advantage in parotid protection—are therefore expected to yield clinical benefits through reduced complication probabilities. Although direct correlation with clinical endpoints was beyond the scope of this dosimetric comparison, the consistency of our findings with established radiobiological principles supports their potential clinical relevance. Future studies incorporating patient-reported outcomes and toxicity assessments will be valuable to validate these dosimetric advantages in clinical practice.
While this study identified statistically significant differences in dosimetric parameters among the three techniques, it is imperative to interpret these findings within the context of the study's scope. It must be emphasized that this was a retrospective planning study, and its primary objective was to compare physical dose distributions. Consequently, the analysis does not, and cannot, extend to demonstrating differences in clinical endpoints such as toxicity rates or tumor control. The “significance” referred to throughout our results pertains to statistical significance in dose measurement, not necessarily clinical significance in patient experience. The relationship between a reduction in physical dose (eg, a few Gray in Dmax/Dmean) and a tangible clinical benefit is complex and modulated by numerous patient-specific biological factors.37–40 Factors such as individual tissue radiosensitivity, genetic predisposition, and functional reserve mean that a statistically significant dosimetric improvement may not manifest identically across a patient population. Therefore, the dosimetric advantages reported here for certain techniques should be viewed as creating a favorable condition for potentially improved clinical outcomes, which must be validated through future prospective studies incorporating direct clinical endpoint assessment.
This study has several limitations. Its single-center design may limit generalizability; future multi-center studies are needed to establish more universally applicable findings. Furthermore, while the use of a single, experienced dosimetrist for all planning ensured internal consistency and eliminated inter-planner variability as a confounding factor, it may not fully represent the range of plan qualities achievable across multiple planners in a clinical setting. Additionally, the analysis focused solely on dosimetric endpoints without clinical follow-up. Prospective studies correlating these dosimetric advantages with clinical outcomes and toxicity profiles are warranted. Finally, the use of a specific step-and-shoot IMRT technique and coplanar beams for all modalities may not fully represent the capabilities of other technical approaches, which could limit the generalizability of our results to all IMRT and VMAT implementations.
Conclusions
Both VMAT and TOMO offer significant dosimetric advantages over IMRT in NPC radiotherapy, including improved OARs sparing and treatment efficiency. The choice between techniques should be guided by institutional capabilities, tumor characteristics, and specific OAR priorities. This study provides a single-center reference for refined dose constraints in NPC radiotherapy, supporting the ongoing effort to optimize treatment quality and patient outcomes.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251413154 - Supplemental material for A Unified Planning Platform Comparison of VMAT, Helical Tomotherapy, and IMRT in Nasopharyngeal Carcinoma: Dosimetric Superiority and Organ Sparing Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338251413154 for A Unified Planning Platform Comparison of VMAT, Helical Tomotherapy, and IMRT in Nasopharyngeal Carcinoma: Dosimetric Superiority and Organ Sparing Analysis by Birong Hu, Qianqian Meng, Xiaodong Peng, Sheng Han, Yansen Liao, Shan Ren, Nuo Xu, Tao Lin, Yuying Jiang and Renming Zhong in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338251413154 - Supplemental material for A Unified Planning Platform Comparison of VMAT, Helical Tomotherapy, and IMRT in Nasopharyngeal Carcinoma: Dosimetric Superiority and Organ Sparing Analysis
Supplemental material, sj-docx-2-tct-10.1177_15330338251413154 for A Unified Planning Platform Comparison of VMAT, Helical Tomotherapy, and IMRT in Nasopharyngeal Carcinoma: Dosimetric Superiority and Organ Sparing Analysis by Birong Hu, Qianqian Meng, Xiaodong Peng, Sheng Han, Yansen Liao, Shan Ren, Nuo Xu, Tao Lin, Yuying Jiang and Renming Zhong in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
This research work was supported by grants from Science and Technology Department of Sichuan Province of China (grant No.2024YFFK0147.).
Ethical Approval Statement
This retrospective study was approved by the Institutional Review Board of the West China Hospital of Sichuan University Biomedical Research Ethics Committee (Approval No.: 20241535). Given the retrospective nature of the study and the use of de-identified patient data, the requirement for informed consent was waived by the IRB. The study was conducted in accordance with the ethical standards of the Declaration of Helsinki and its later amendments.
Author Contribution Statement
Birong Hu: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing - original draft.
Qianqian Meng: Data curation, Formal analysis, Investigation, Validation, Writing -review & editing.
Xiaodong Peng: Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing - review & editing.
Sheng Han: Investigation, Methodology, Software, Validation.
Yansen Liao: Investigation, Methodology, Resources.
Shan Ren: Data curation, Formal analysis, Visualization.
Nuo Xu: Data curation, Validation.
Tao Lin: Resources, Software.
Yuying Jiang: Resources, Data curation.
Renming Zhong: Conceptualization, Funding acquisition, Project administration, Supervision, Writing - review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Department of Sichuan Province of China, (grant number 2024YFFK0147).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Unmasked Statement
The authors confirm that all patient identifiers have been removed from the data presented in this manuscript. All images, tables, and text have been anonymized to protect patient privacy in compliance with journal policies.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
