Abstract
Background
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin-lymphoma. Although it can be cured in many patients, a significant proportion of patients fail the primary treatment and require second-line treatment. Currently, only limited data on real-world outcomes with standard therapies in Austrian patients with DLBCL are available, and while novel therapies are emerging, no historical benchmarks have been established to serve as a reference for these novel treatments.
Methods
We performed a retrospective, single-center analysis of patients with DLBCL diagnosed between 2010 and 2018 who had been treated with standard therapies. To establish efficacy benchmarks for novel therapies, we applied both clinical-trial and real-world-derived criteria to analyze the outcomes of patients potentially eligible for novel or future treatments.
Results
Although many patients can be cured with frontline therapy, outcomes are poor, especially in high-risk patients. Patients failing frontline therapy, especially those fulfilling the chimeric antigen-receptor (CAR) T-cell eligibility criteria, had dismal outcomes, and very few patients achieved long-term remission. Our data provide benchmark outcomes for patients eligible for novel treatments such as antibody-drug-conjugate (ADC) or CAR T-cell therapy-based treatments for potential future comparative analyses.
Conclusions
Patients with DLBCL treated in Austria showed comparable outcomes to those reported in other real-world studies. Overall, standard chemotherapy-based approaches provide unsatisfactory outcomes in high-risk patients and patients in whom frontline therapy fails. Because many patients are now eligible for alternative first- and second-line treatments, such as ADC-based or CAR T-cell therapy, our efficacy benchmarks can serve for the future evaluation of these therapies in the Austrian healthcare environment.
Keywords
Introduction
Diffuse large B-cell lymphoma (DLBCL) is an aggressive type of cancer with heterogeneous genetics and clinical presentation, and represents the most common subtype of non-Hodgkin lymphoma (NHL). 1 It accounts for one-third of all lymphoma cases with an estimated 556 new cases in Austria in 2021. 2 More than two-thirds of patients are diagnosed with advanced-stage disease, but a large proportion can be cured with standard immuno-chemotherapy consisting of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) combined with an anti-CD20 monoclonal antibody, such as rituximab (R-CHOP). With this regimen, 5-year overall survival (OS) rates of 60%–70% are achieved. 1 Accordingly, R-CHOP-based therapy is considered the current standard of care (SOC), with slight modifications according to the International Prognostic Index (IPI). 3 Efforts to further improve therapy outcomes through either (I) application of higher doses of rituximab, (II) addition of other chemotherapeutics, (III) shortening R-CHOP intervals, or (IV) increasing their cycle number conferred no additional benefit or only at the cost of enhanced toxicity and adverse events. 4 More recently, however, the introduction of polatuzumab-vedotin (Pola), a CD79b targeting antibody-drug conjugate (ADC), into the R-CHOP regimen was shown to be superior to R-CHOP in newly diagnosed patients with DLBCL in the global, randomized, phase 3 POLARIX trial. 5 Since the trial showed that the Pola-R-CHP combination was especially active in patients with high-risk features, this combination has been recently approved in the EU and US and is the recommended new SOC for patients with high IPI in Austria. 6
Approximately 30%–40% of patients with DLBCL show either therapy-refractory disease or have disease recurrence with a 2-year OS rate of only 20%–30%. 1 In refractory or relapsed (R/R) patients, intensive chemotherapy has been the SOC approach for many years until the recent rise of novel therapies. The most common procedure, if responsive to intensive platinum-based salvage regimens, is high-dose chemotherapy (HDT), followed by autologous stem cell transplantation (ASCT) due to favorable outcomes. However, less than half of R/R patients with DLBCL are fit for HDT + ASCT, while less than half of the fit patients are likely to respond to salvage treatment and outcomes in non-responding patients are generally poor. 7 Outcomes were especially poor with second-line therapy in less-fit patients treated with less intensive treatments outside clinical trials. 8
Recently, CD-19 targeting chimeric antigen receptor (CAR) T-cell therapy has become available for the treatment of relapsed or refractory DLBCL in a third-line setting, resulting in high and, more importantly, long-lasting responses that have also been confirmed in real-world settings. 9 Furthermore, randomized phase 3 trials have also demonstrated the superiority of anti-CD19 CAR T cell therapy over SOC therapy in patients with early frontline treatment failure,10,11 leading to its approval in the EU. Other novel and effective therapies, such as the combination of polatuzumab plus rituximab and bendamustine (Pola-RB), the combination of the anti-CD19 monoclonal antibody (mAB) tafasitamab plus lenalidomide (Tafa-Len) or the anti-CD19 ADC loncastuximab-teserine (Lonca), are now approved in Europe for less fit patients after the first disease recurrence based on phase 2 trials.12-14 Finally, the anti CD3xCD20 bispecific antibody (bsAB) glofitamab (Glofi) combined with gemcitabine and oxaliplatin (GemOx) has been approved very recently by the EMA for R/R DLBCL patients based on the positive phase 3 STARGLO trial. 15
As such, it may be crucial to establish benchmarks for the outcomes of historic SOC therapies to properly compare and evaluate the real-world merits of novel and often expensive substances. 16 Based on these considerations, analyses of real-world outcomes in both frontline and R/R settings have been conducted to establish realistic estimations of treatment outcomes with historic SOC therapies in patients with DLBCL have been conducted, especially to estimate the potential benefits of newly introduced and expensive therapies such as CAR T-cell therapy in the second-line setting in some countries. 17 The rationale for this study was to evaluate the outcomes in patients with DLBCL treated at a large hospital in Austria in real-world first- and second-line settings prior to 2018. The data should reflect the Austrian healthcare setting in the post-rituximab but pre-CAR T-cell era. As such, it could serve as a benchmark for future real-world DLBCL analyses, especially as novel therapies become increasingly available, and for assessing the impact of novel agents on outcomes in the Austrian healthcare system. This study should also highlight unmet needs and areas for improvement, and how and where novel therapies in the first- and second-line, such as CAR T cells, could potentially improve outcomes.
Patient Selection
This study was evaluated by the regional ethics committee “Ethikkommission für das Bundesland Salzburg” via proposal EK Nr: 415-EP/73/127-2012 and has been approved via ethics committee vote EK Nr: 1046/2022 on 18 May 2022. The study has been conducted in accordance with the Declaration of Helsinki. All patients included in this analysis provided written informed consent. The study was performed following the Equator guidelines by adhering to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement 18 (see SI for checklist).
This was a single-center, retrospective, observational study of adult patients diagnosed with DLBCL at our institution between 2010 and 2018. Accordingly, all consecutive patients diagnosed with histologically confirmed DLBCL according to the WHO 2016 classification 19 during this period were analyzed. The registry database was managed using the REDCAP electronic data capture tools 20 and patient characteristics, response to therapy, and time-to-event data were retrieved from medical records. Details entered in REDCAP for data analysis have been de-identified to ensure that the identity of any person cannot be ascertained. Patient details documented in this retrospective analysis included demographic characteristics and diagnostic assessments, including histological and laboratory data such as ECOG status, Ann-Arbor staging, and IPI assessment at diagnosis. Cell-of-origin (COO) subtypes were determined using the Hans algorithm.
Time-to-event outcome assessments were based on the Lugano criteria. 21 Primary refractory disease was defined as stable disease (SD) or progressive disease (PD) after frontline treatment. Relapsed disease was confirmed pathologically (biopsy-proven). Progression-free survival (PFS) was defined as the time from diagnosis to refractory disease, relapse, or death due to any cause, whereas OS was defined as the time from diagnosis to death due to any cause. Early failure of frontline therapy was defined as primary refractory disease or disease relapse within 12 months after the completion of first-line therapy (RR <12 months) whereas late failure of frontline therapy was defined as disease relapse occurring later than 12 months after the completion of first-line therapy (RR > 12 months).
Curative frontline therapy was defined as anthracycline- and anti-CD20 antibody-containing polychemo-immunotherapy, whereas curative second-line therapy was defined as high-intensity, platinum-containing salvage CIT with the intention of proceeding to high-dose therapy (HDT) followed by peripheral autologous stem-cell transplantation (ASCT) in responding patients.
For patients receiving frontline therapy, PFS and OS analyses were stratified based on the IPI scores (low = 0-1 vs low-intermediate = 2, vs high-intermediate =3 vs high = 4-5). Additionally, the outcomes of patients receiving frontline therapy were assessed based on their hypothetical eligibility (yes vs no) for POLARIX 5 as a recent trial for patients with IPI≥2, and ZUMA-23 22 for high-risk patients with IPI 4 or 5. For patients receiving second-line therapy, the outcomes were stratified based on the time point of disease recurrence (<12 months vs > 12 months from the end of frontline therapy failure) and intention (yes vs no) of proceeding to HDT + ASCT.
Second-line CAR-T eligibility was retrospectively determined based on the timepoint of disease recurrence (RR < 12 months) and ECOG PS (0–2). These criteria most closely mirror clinical practice at the treating center, as age, isolated laboratory, organ function, or bone marrow reserve parameters, such as low ALC, ANC, and platelet counts, are rarely considered as reasons for patient rejection, as each patient will be evaluated individually, and treatment decisions will be based on a risk/benefit assessment.
Statistical Analysis
All statistical analyses were performed retrospectively. The Kaplan-Meier method was used to calculate the PFS and OS estimates at 1, 2, and 5 years. The global effect of each variable was evaluated using the likelihood ratio test and the differences between the cohorts were compared using the log-rank test. In case of multiple comparisons, pairwise log-rank tests were conducted. The resulting p-values were adjusted for multiple comparisons using the Bonferroni-Holm correction. Effect sizes (hazard ratios (HR)) were estimated by applying a Cox proportional hazards regression model. The proportional hazards assumption was verified by Schoenfeld residuals 23 and found to be satisfied. All statistical tests were two-sided, and a significance level of <0.05 was set to determine statistical significance. All analyses were performed using R software (R version 4.4.1 24 ), and figures were created using Survival and Survminer packages. 25 For the event comparisons, we performed a post-hoc power analysis using the Schoenfeld approximation. 23 For the comparison of PFS of patients receiving first-line treatment between <61 years and >80 years, the statistical power reached 0.8, whereas the comparison between patients aged <61 years and 61–80 years was underpowered (power = 0.44). For all OS comparisons in first-line treatment, the power also reached 0.8. In addition, only OS analyses in R/R patients receiving second-line therapy reached sufficient power, while PFS remained underpowered due to lower event counts.
Results
Patient Characteristics of the Overall Study Population and Patient Trajectories Along First- and Second-Line Treatment
A total of 278 patients diagnosed between 2010 and 2018 were available for this analysis. As depicted in the flow diagram in Figure 1, 247 of 278 (88.8%) patients with newly diagnosed DLBCL were treated with SOC therapy, whereas 25 of 278 (8.9%) patients were enrolled in a clinical trial. Most patients received SOC frontline therapy with curative intent (n = 235 patients, 84.2%), while n = 18 (6.4%) received either a non-curative/palliative treatment or no treatment/best-supportive care. Fifty-nine patients (21.2% of all newly diagnosed patients with DLBCL) also received any second-line therapy. Again, most patients were treated with SOC options and three patients (5.1%) were enrolled in a clinical trial. Of patients receiving SOC second-line treatment, n = 43 (72.8%) were treated with curative intent (intensive salvage therapy) and n = 16 (27.2%) received a less-intensive/palliative second-line treatment. Of all second-line patients undergoing treatment with curative intent for relapsed/refractory disease, n = 11 patients (18.6%) were also able to undergo a consolidative high-dose therapy (HDT) followed by autologous stem-cell transplantation (ASCT).

Flow diagram of DLBCL patient trajectories along first- and second-line treatment. overview of patient trajectories for patients with DLBCL diagnosed at the institution within the defined observation period. BSC = best-supportive-care; SOC = standard-of-care therapy; HDT = high-dosetherapy; ASCT = autologous stem-cell therapy; ‡ includes regimens containing anti-CD20 plus anthracycline; * includes all patients with documented refractory or relapsed disease within the observation period; § includes salvage regimens (R-GDP, R-ICE, or R-DHAP) with intent of proceeding to HDT; $ all patients treated within the clinical trial were treated with curative intent.
In terms of treatment intent, n = 253 (91.0%) of patients with newly diagnosed DLBCL were treated with curative intent, whereas this rate dropped to 72.8% (n = 43/59) in patients needing second-line therapy.
The median age at diagnosis was 69 years (range, 22-93), and 25.8% of the patients were >75 years, respectively (Table 1). The Ann-Arbor stage at diagnosis was localized (stage I + II) in 43.5% and advanced (stage III + IV) in 50.2% of patients, respectively. The IPI at diagnosis was low or low-intermediate for 16.8% and 28.7% of the patients and high-intermediate or high for 29.4% and 10%, respectively. The baseline characteristics are depicted in Table 1. GCB- and non-GCB-type cells of origin were diagnosed in 34.1% and 33.3%, respectively, whereas data were missing for 32.6% of the patients.
Baseline Patient Characteristics at Diagnosis for the Full Analysis Cohort.

Demographic and disease characteristics of all patients with DLBCL diagnosed and treated at the institution between 2010 and 2018. IPI = International Prognostic Index; GCB = Germinal Center B-Cell-like; ABC = Activated B-Cell-like; ULN = upper limit of normal.
At the time of data cutoff and analysis, survival status was available for n = 221 (79%) of the patients, while n = 52 (18%) were lost to follow-up or had missing information (n = 5). For the n = 57 patients that were either lost to follow-up or had missing information, the median follow-time was 70 months. Among patients with available data, n = 106 (38%) were still alive and n = 115 (41%) patients were deceased.
Types and Outcomes of Frontline Treatment
Of the 278 patients diagnosed and treated between 2010 and 2018, n = 6 (2.2%) did not receive any therapy and/or only best supportive care, and n = 12 (7.0%) were not treated with curative intent (Table 2). A total of n = 253 (91.0%) patients received frontline therapy with curative intent. Among these, the most frequently used frontline therapies were R-CHOP in n = 151 (54.5%) patients and R-COMP in n = 89 (31.9%) patients, respectively. R-DA-EPOCH was used in n = 13 (4.8%) of patients. Five patients received HDT followed by ASCT as consolidative therapy after frontline immuno-chemotherapy as part of an individualized treatment plan for high-risk disease (SI Table 1).
Treatment Types and Efficacy Outcome in Newly Diagnosed DLBCL Patients.

Overview of frontline therapy strategies, treatment types and survival outcomes. *Curative intend treatment was defined as anti-CD20 and anthracycline containing regimens. NE = Not estimable.
PFS of all treated newly diagnosed patients (SI Figure 1a) differed significantly across age categories (<61, 61-80, >80 years; log-rank p < 0.001). Median PFS was not reached (NR) for patients <61 years, whereas median PFS was 10.15 and 2.09 years for patients aged 61–80 and >80 years, respectively. Similarly, median OS (SI Figure 1a) of the entire patient cohort examined across age categories differed significantly (log-rank p < 0.001). Again, younger patients exhibited the longest survival time. Accordingly, both PFS and OS were significantly shorted in patients aged below median (log-rank p < 0.001, SI Figure 1b). An overview of PFS and OS rates based on age categories is provided in SI Table 2.
In the Cox proportional hazards model (SI Table 3), age categories showed a significant effect for PFS (likelihood ratio test, χ2 = 23.37, p < 0.001) and for OS (likelihood ratio test, χ2 = 44.38, p < 0.001) for the entire patient population as well as for PFS (likelihood ratio test, χ2 = 24.77, p < 0.001) and OS (likelihood ratio test, χ2 = 44.4, p < 0.001) based on median age. Compared with patients <61 years, those aged >80 years had a 3.53 (95% CI: 2.12-5.88) times higher risk of progression, whereas the HR for 61–80 years was not statistically significant (HR = 1.44 (95% CI: 0.92-2.24), p = 0.104). Pairwise log-rank tests revealed significant differences in PFS between <61 years and >80 years (p < 0.001, Bonferroni-Holm adjusted), and 61–80 years and >80 years (p < 0.001, Bonferroni-Holm adjusted). A similar picture occurs for median split for PFS, where the patients older than median had a 2.46 (95% CI: 1.71-3.54) times higher risk of progression. For OS, patients older than 80 years had a 7.09 (95% CI: 3.91-12.86) times higher risk of death than <61 years, whereas the HR for 61–80 years was 2.50 (95% CI: 1.46-4.28) higher than younger patients. Pairwise log-rank tests revealed significant differences in OS between all age categories. Patients aged >80 years had significantly shorter OS than <61 years (p < 0.001, Bonferroni-Holm adjusted), and 61–80 years (p < 0.001, Bonferroni-Holm adjusted). Also, patients aged between 61–80 years had a significantly smaller OS than <61 years (p < 0.001, Bonferroni-Holm adjusted). Patients aged ≥ median had a 3.73 (95% CI: 2.47-5.64) fold higher risk of death compared with younger patients.
Median PFS of patients receiving an anthracycline-containing SOC therapy with curative intent was 10.15 years (95% CI: 7.29– Not estimable) and PFS rates at 1, 2, and 5 years were 78.45% (95% CI: 73.52-83.71), 73.16% (95% CI: 67.86-78.88), and 62.96% (95% CI: 57.18-69.33), respectively (SI Figure 1c). Among patients treated with curative SOC therapy, median OS was 11.91 years (95% CI: 10.17– Not estimable) with 80.24% (95% CI: 75.43-85.36) and 70.47% (95% CI: 64.95-76.46) of patients being alive at 2 years and 3 years, respectively. Kaplan-Meier curves depicting the PFS and OS probabilities of all treated patients and patients treated with curative intent SOC therapies are depicted in SI Figure 1c.
When stratifying outcomes for patients receiving SOC frontline therapy with curative intent based on IPI (SI Figure 2), median PFS was not reached for the low- (IPI=0-1) risk group compared to 11.1 (95% CI: 10.2–Not estimable) and 10.8 years (95% CI: 5.3– Not estimable) for the low-intermediate- (IPI=2) and high-intermediate- (IPI=3) risk groups, respectively. In contrast, median PFS rates in patients with high-risk IPI (IPI=4 or IPI=5) were 1.2 (95% CI: 0.7-5.7) years and 2.25 (95% CI: 0.4– Not estimable), respectively. SI Table 4 lists the PFS and OS outcomes of patients treated with SOC curative intent at the 1-, 2-, and 3-year landmarks according to the IPI risk group.
We also performed an analysis of frontline therapy outcomes based on the inclusion criteria of the phase 3 POLARIX and phase 3 ZUMA-23 trials to provide real-word reference values for patients fit for novel therapies but previously treated with prevailing SOC therapies (see SI Table 5 for the inclusion/exclusion criteria of the respective trials). Of all patients diagnosed within the study period, n = 151 (54.1%) and n = 32 (11.4%) met the main study inclusion criteria for either POLARIX or ZUMA-23 and were treated with curative SOC frontline therapy. The outcomes of patients treated with curative-intent SOC therapy meeting the POLARIX or ZUMA-23 inclusion criteria are shown in Figure 2a,b. Briefly, median PFS and OS were 11.9 and 12.8 years in the POLARIX-like population, whereas median PFS and OS were only 1.3 and 3.5 years in the ZUMA-23-like population, respectively (SI Table 6).
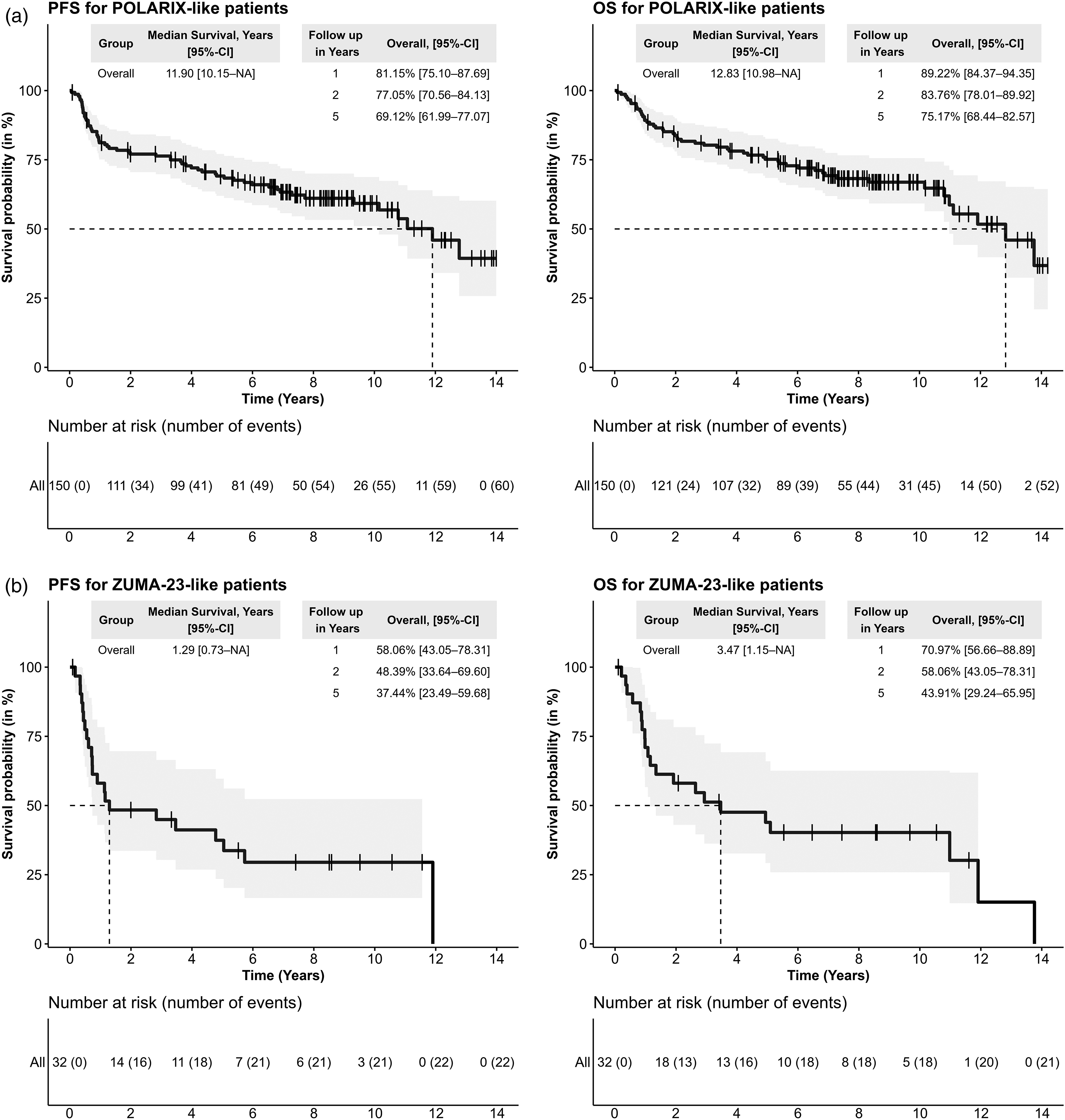
Kaplan-meier plots of POLARIX- and ZUMA-23-like patients treated with SOC Therapy. PFS and OS of newly diagnosed patients with DLBCL fulfilling POLARIX (2a - upper row) and ZUMA-23 (2b - lower row) clinical trial inclusion criteria. patients were treated with SOC curative-intent frontline therapy.
Second-Line Therapy and Outcomes of Patients Failing First-Line Therapy
Of the 278 patients treated with any systemic therapy in the first line, 59 patients (21.2%) also received second-line therapy. Of these, 56 patients (94.9%) received a SOC second-line therapy. Overall, intensive therapy with curative intent was delivered to 43 patients (72.9%), whereas 16 patients (27.1%) received only palliative/less intensive treatment.
Of the 59 patients receiving any second-line therapy, 12 (20.3%) were still alive at the time of data analysis, 43 (72.9%) had died and 4 (6.8%) had been lost to follow-up. All patients who were alive at the time of analysis were treated with potentially curative regimens.
A detailed summary of patient characteristics at the time of second-line therapy is shown in Table 3.
Patient Characteristics, Treatment Strategies, and Survival Outcomes in R/R Patients with DLBCL.
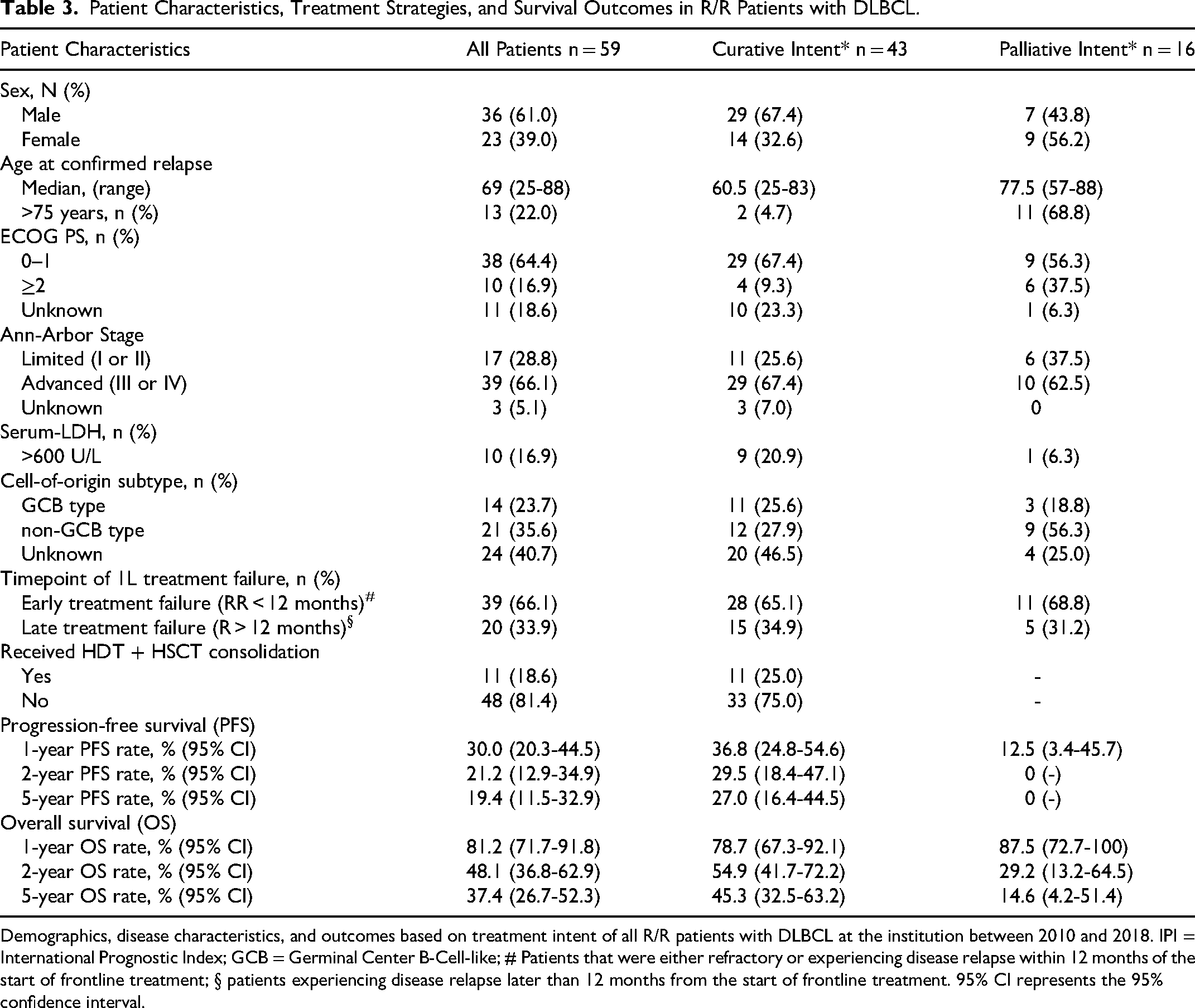
Demographics, disease characteristics, and outcomes based on treatment intent of all R/R patients with DLBCL at the institution between 2010 and 2018. IPI = International Prognostic Index; GCB = Germinal Center B-Cell-like; # Patients that were either refractory or experiencing disease relapse within 12 months of the start of frontline treatment; § patients experiencing disease relapse later than 12 months from the start of frontline treatment. 95% CI represents the 95% confidence interval.
The median age of patients at the start of second-line therapy was 69 (25-88) years, and 22% of patients were older than 75 years at the start of second-line treatment. Patients receiving more intensive therapies with curative intent were younger (median age 60.5 vs 77.5 years) and only a very small fraction of patients receiving curative-intent treatment were older than 75 years (4.7%). In contrast, 68.8% in the less intensive/palliative group were older than 75 years at the start of second-line therapy. Most patients receiving second-line therapy had advanced-stage disease (Ann-Arbor III or IV), 16.7% had LDH levels >600 U/L, and 35.6% had an unfavorable non-GCB type. Two-thirds (66.1%) of patients receiving any second-line therapy experienced early failure of frontline therapy (RR < 12 months), while one-third (33.9%) showed a late failure of frontline treatment (RR > 12 months). In total, 18.6% of patients received HDT followed by ASCT as part of their second-line treatment, and 25.0% of patients initially treated with curative intent were able to finally undergo ASCT consolidation. For the overall population receiving second-line therapy, the 1-, 2-, and 5-year PFS rates were 30.0% (95% CI: 20.3-44.5), 21.2% (95% CI: 12.9-34.9), and 19.4% (95% CI: 11.5-32.9), while the 2- and 5-year OS rates were 48.1% (95% CI: 36.8-62.9) and 37.4% (95% CI: 26.7-52.3), respectively.
Regarding the outcomes after second-line treatment based on treatment intention, the median PFS and OS differed between patients receiving curative therapy and those receiving palliative therapy (Figure 3). PFS only differed numerically between patients treated with palliative intent compared to curative therapy intent (log-rank p = 0.054) while Cox-regression revealed a 1.82 times higher risk for progression (p = 0.06). In contrast, OS was significantly inferior in patients receiving palliative treatment (log-rank p < 0.01) with a 2.38-fold increased risk for death compared to patients treated with curative intent (p < 0.01). In addition, the 1-, 2-, and 5-year PFS rates were different, with approximately one-third of patients treated with curative intent treatment being free from progression at the two- and five-year landmarks compared to zero patients in the palliative/less intensive group being progression-free at the two-year landmark. Correspondingly, also 1-, 2-, and 5-year OS rates were markedly different between the treatment groups, with 45.3% (95% CI: 32.5-63.2) of patients in the curative intent group being alive after five years compared to 14.6% (95% CI: 4.2-51.4) in the palliative/less intensive group.
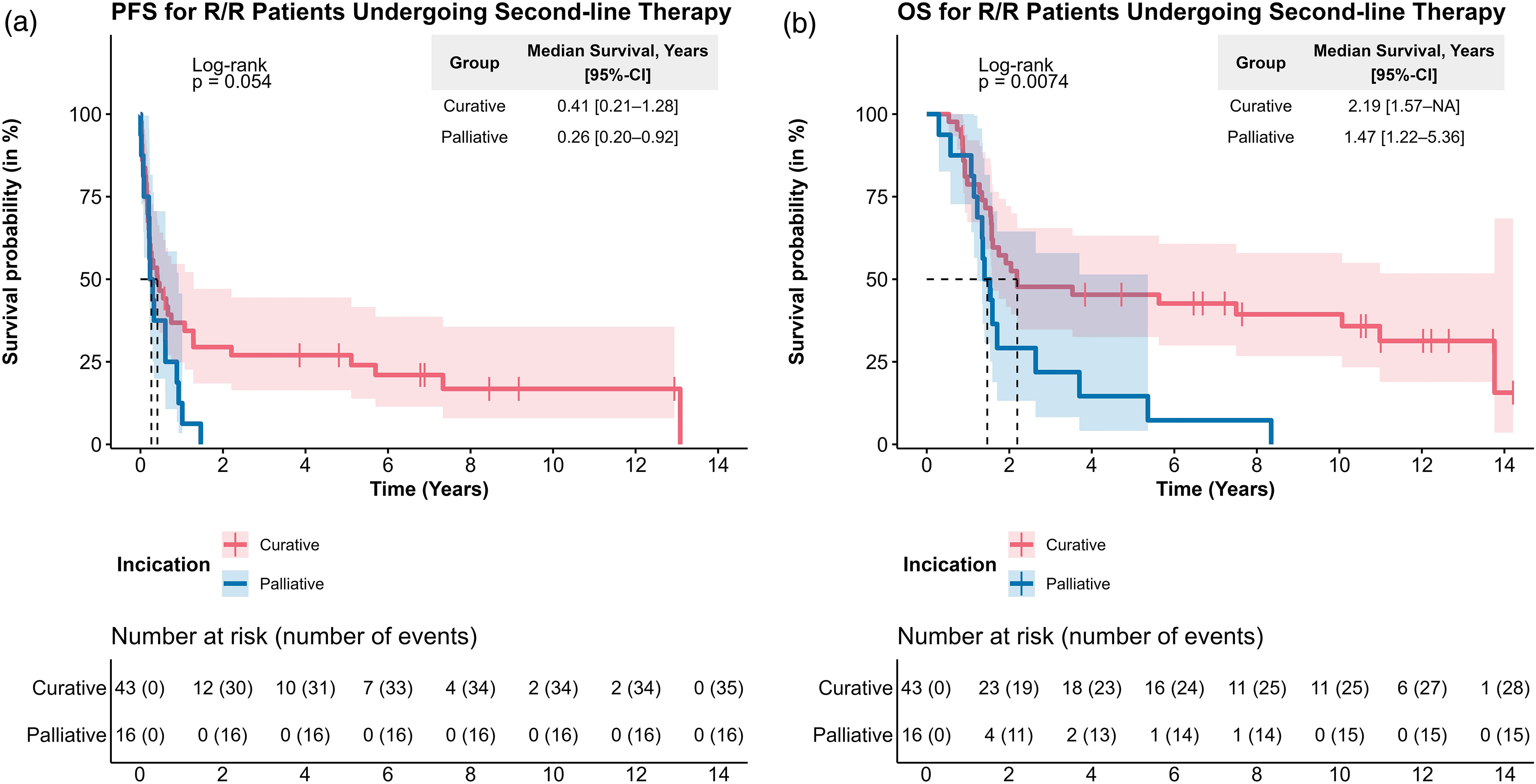
Outcomes of R/R patients with DLBCL treated with curative/less-intensive therapies kaplan-meier plots, including 95% CIs for PFS (a) and OS (b) in R/R patients with DLBCL treated with either curative (red) or non-curative (blue) intent.
Future Benchmark for Second-Line CAR T-Cell Therapy-Feasible Patients
Because CAR T-cell therapy has recently emerged as a novel SOC option for patients with early frontline (RR < 12 months) treatment failure in Austria, we aimed to provide a real-world benchmark for outcomes obtained with historic treatments (the post-rituximab but pre-CAR T era) in this patient segment within the Austrian healthcare environment as a reference for future analyses. For this, we examined CAR-T feasible patients as defined according to the institutional criteria (SI Table 7). While these criteria are broadly in line with both the EMA label and Onkopedia Guidelines, which provide no specific recommendations regarding age, comorbidities, and laboratory thresholds, an ECOG PS of 0–2 is assumed to adequately represent most CAR T-fit patients.
When looking at the OS rates of CAR T-cell therapy-fit patients from the beginning of frontline therapy, the median OS in these patients was less than 1 year (95% CI: 0.8-1.3), with 2- and 5-year OS rates of 26.6% (95% CI: 15.6-45.1) and 20.7% (95% CI: 11.0-39.0), respectively (SI Figure 3). Furthermore, 37% (n = 22/n = 59) of all patients receiving any second-line therapy and 56% (n = 22/n = 39) of all patients exhibiting early frontline treatment failure (RR < 12 months) were deemed fit enough to receive CAR T-cell therapy in a real-world setting.
Patients who would be theoretically eligible for second-line CAR T therapy in our institution and who had been treated with historical SOC exhibited a median PFS-2 of 0.19 years (95% CI: 0.15.5-0.31) and a median OS of 0.58 years (95% CI: 0.3–Not estimable) (Figure 4). The respective 1-, 2-/5-year PFS rates were 18.2% (95% CI: 7.5-44.1) and 13.6% (95% CI: 4.8-39.0), whereas the corresponding OS rates were 35.8% (95% CI: 20.3-63.1) and 25.6% (95% CI: 12.3-53.3), respectively.
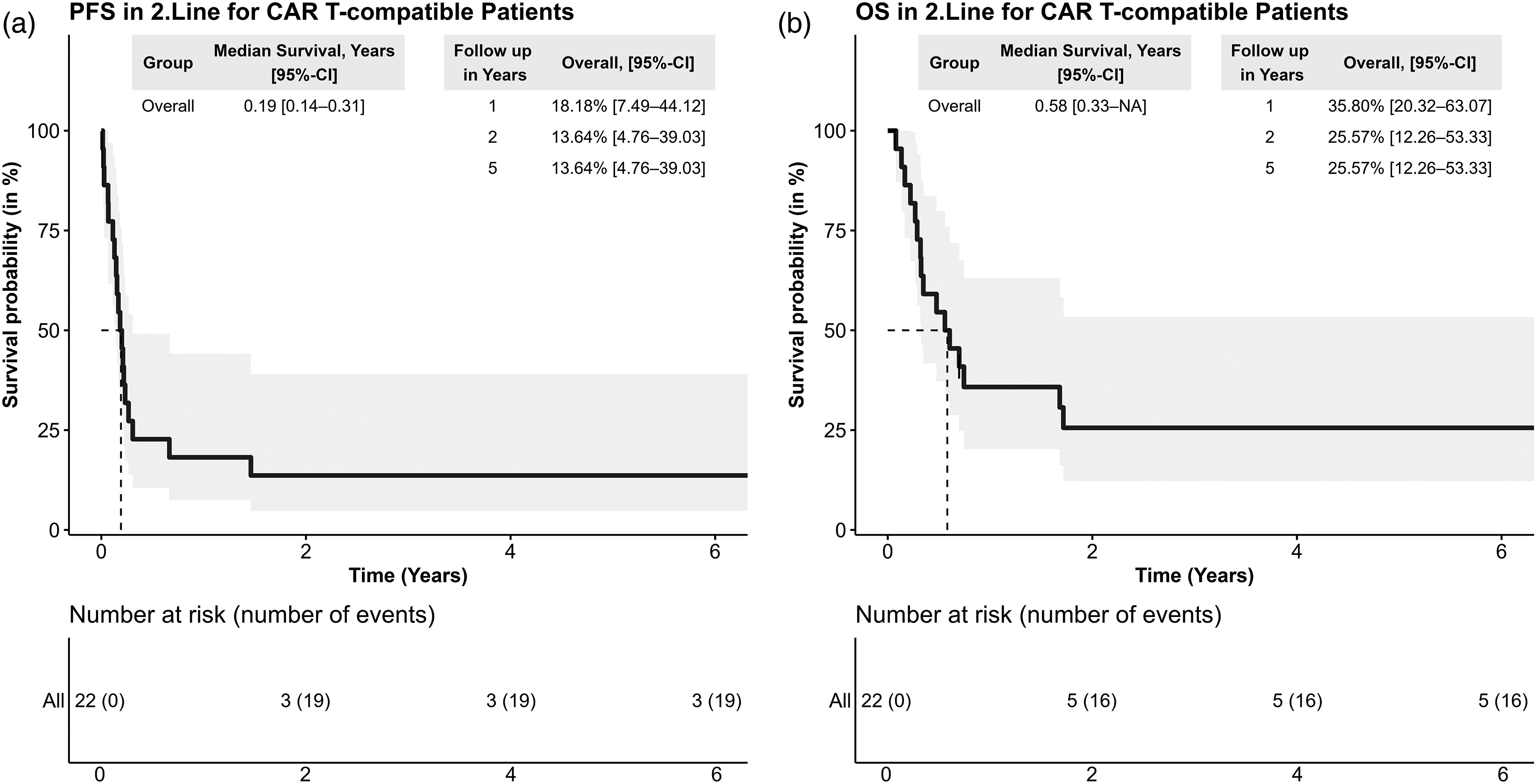
Outcomes of R/R patients with DLBCL meeting institutional CAR T-feasibility criteria Kaplan-Meier plots for PFS-2 (a) and OS (b) in R/R patients with DLBCL that were treated with SOC therapy and that would fulfill institutional real-world feasibility criteria for CAR T-cell therapy. feasibility criteria were defined as fulfilling the label (refractory or relapsed within 12 months) and other criteria like ECOG PS = 0–2.
Discussion
Given the tremendous progress made in recent years, treatment guidelines for DLBCL in Germany, Austria, and Switzerland have been adapted to include novel therapies, such as ADC-based and CAR T-cell therapies as well as other targeted therapies. 6 However, the ability to implement and realize these changes depends on the specific circumstances of the healthcare system in each country, such as the availability and accessibility of diagnostic techniques and therapeutics, preexisting recommendations, and experience, which may significantly impact the resulting clinical practice. 26 Accordingly, to provide optimal care for patients, national specifics should be considered, and treatment recommendations should ideally consider and reflect real-world experiences gained in a specific healthcare environment. For example, large national registries have been established to capture the access, efficacy, and safety of these novel regimens after their approval within the corresponding healthcare systems to evaluate real-world outcomes. 27
Both the typical DLBCL patient and the treatment regime applied in the first line in Austria in the years between 2010 and 2018 were largely comparable to that reported from real-world studies conducted in other countries.28,29 Accordingly, patients consistently had a median age of approximately 70 years at diagnosis, advanced Ann-Arbor disease stage of >50% at diagnosis, and slightly more than half of the patients presented with low/low-intermediate IPI scores. The typical front-line therapy in all countries is R-CHOP or an R-CHOP-like regimen applied in approximately 90% of the cases. Furthermore, outcomes based on known prognostic markers like patient age and IPI are also comparable with previous results. 28 In summary, our data is in line with reports from other large European and overseas registries and should provide a realistic estimation of the outcomes that can be obtained by SOC therapies in the post-rituximab and pre-CAR-T eras. While the outcomes in the low and low-intermediate groups in our cohort were good, there remains a need to improve treatment outcomes for patients presenting with high IPI scores, where PFS and OS estimates were markedly shorter than those in the other groups.
Based on positive results from the phase 3 POLARIX trial 5 and recent EMA approval, a new frontline treatment regimen that incorporates the CD79b-directed antibody-drug-conjugate polatuzumab (Pola) into the R-CHP regimen has emerged as a novel SOC. The R-Pola-CHP regimen seems especially effective in patients with high-risk features and an unfavorable ABC COO subtype. 30 While this combination has been widely adopted as a novel treatment option for patients with high IPI scores, a real-world analysis of its efficacy in a non-trial setting is lacking. To provide a real-world benchmark for future investigations, we analyzed patients receiving a standard R-anthracycline regimen within a large Austrian institution based on the inclusion/exclusion criteria of the POLARIX trial. Moreover, the current ZUMA-23 trial with anti-CD-19 is evaluating the efficacy of axicabtagene-ciloleucel (axi-cel) versus SOC in high-risk LBCL patients presenting with IPI4-5 and stage III/IV disease in a randomized phase 3 setting. 22 As this population usually suffers from suboptimal outcomes with standard therapies, we provided a real-world benchmark obtained with historic immuno-chemotherapies within this study population. This is especially relevant because the use of CAR T-cell therapy in the frontline setting might be associated with increased healthcare spending. Based on the inclusion criteria of both trials, the ZUMA-23-like cohort was enriched for ultra-high-risk patients (IPI 4-5, stage III/IV disease). Consequently, outcomes in the ZUMA-23-like cohort were significantly inferior compared to the POLARIX-like cohort, which included also patients with low-intermediate and intermediate IPI scores.
With respect to second-line treatment, we observed a similar proportion of patients receiving any treatment, with similar rates of primary refractory patients and patients experiencing early treatment failure compared to other real-world analyses.8,31-33 The proportion of patients receiving a HDT + ASCT consolidation as part of second-line treatment ranges between 17%–33%; thismight be because, at many institutions, a HDT therapy is only initiated if patients reach a PET/CT negative CMR after salvage immune-chemotherapy, as these patients seem to profit most from this procedure. 34 Our study is perfectly in line with previous results, showing that while a large proportion of patients are treated with curative intent, only 25% are finally able to receive HDT followed by ASCT. Nonetheless, global outcomes for R/R patients with DLBCL treated with standard therapies remain poor. This is especially true for patients who are unable to undergo HDT + ASCT consolidation, which represents a large proportion of all second-line patients, as confirmed by our and other recent real-world reports.29,35 Thus, novel therapeutic options are needed for patients with R/R DLBCL, especially for those who are not fit for intensive therapy and/or who are able to undergo HDT + ASCT. Indeed, novel options such as Pola-RB, Tafa-Len, and Lonca have been approved for the treatment of unfit R/R patients with DLBCL based on phase 2 trials,12-14 but the efficacy of these regimens has not been tested against the preferred SOC in fit R/R patients with DLBCL. Therefore, the most relevant change in the treatment paradigm for R/R patients with DLBCL has been the approval of anti-CD19 CAR T-cell therapy for fit patients experiencing early treatment failure based on two large, randomized, phase-3 trials and, consequently, the change in the respective treatment guidelines in Austria, Germany, and Switzerland. 6 Notably, axi-cel was the first treatment for 30 years to significantly improve OS compared to the historical SOC, while also providing a better quality of life. 36 Similarly, liso-cel provided a strong numerical OS advantage over SOC. 37 While both the ZUMA-7 and TRANSFORM trials compared the efficacy of anti-CD19 CAR cells in very fit patients intended for intensive chemotherapy and HDT + ASCT, both axi-cel and liso-cel have also been shown to be effective and safe in older and less-fit patients. Both the ALYCANTE 38 and PILOT 39 trials have enrolled patients who are not fit for intensive chemotherapy due to advanced age (>65 years) or significant comorbidities, and have shown that CAR T cells can be offered to a broad patient population. Based on these results, we recently presented a potential algorithm for treating patients with DLBCL outside of a clinical trial, emphasizing the relevance of CAR T-cell therapy for the treatment of relapsed/refractory patients. 40 While CAR T cells have demonstrated a similar long-term efficacy and safety profile in patients failing two or more systemic therapies in most European countries, including Austria,41-43 mature data confirming their efficacy in the second-line, high-risk setting are scarce, and only early and very limited reports on immature efficacy outcomes are available. For example, the median follow-up for efficacy in the recently presented DECAR T study of axi-cel as second-line therapy was only 3 months. 44 Long-term efficacy, feasibility, and safety data of CAR-T cells in a real-world setting with respect to relevant local, historic benchmarks in the second line are needed to confirm these data within a given healthcare system. We believe that our data provide a reasonable benchmark for future analyses of second-line CAR T-cell therapy in a real-world setting in Austria.
Based on recent analyses from the Swedish and Canadian registries,17,35 the estimated proportion of potentially eligible CAR T patients among all patients requiring second-line therapy ranged between 13%–17% when selecting patients stringently based on the pivotal phase 3 study inclusion/exclusion criteria (patients with early treatment failure that are intended to receive HDT + ASCT and meet patient eligibility criteria based on the ZUMA-7 protocol). However, this estimation may be too restrictive, as indicated in both reports. Thus, based on a more relaxed eligibility assumption, more patients with early frontline treatment failure might be eligible for treatment if CNS involvement or ECOG PS2 does not preclude them from CAR T therapy. This is especially relevant, as recent data on the use of CAR T therapy in the second-line setting have shown that patients who would not qualify for HDT + ASCT (based on age and/or comorbidities) display similar efficacy and safety outcomes when treated in routine clinical settings. 45 Furthermore, CAR T cell therapy in patients with primary or secondary CNS involvement has been shown to be well tolerated and effective in real-world settings. 46 Therefore, we believe that the selection criteria we used to estimate the proportion of CAR T cell-eligible patients (56% of all patients experiencing early frontline treatment failure) in the second-line setting and the efficacy benchmarks obtained with historic standard-of-care therapy within the CAR T-eligible population in the real-world setting are a better estimate, although numbers are small and based on retrospective evaluations. A recent analysis indicated that the total number of DLBCL patients eligible for second-line CAR T-cell therapy in Austria may be as high as 89 patients per year. 2 Although these data were obtained by theoretical calculations, our analysis seems to be in line with this observation.
When comparing the outcomes of potentially eligible CAR T patients obtained in our study and other real-world studies, we see that outcomes with SOC in terms of OS are dismal. This was different when looking at the SOC arms in ZUMA-7, where the median OS was 31.1 mo and 2-, 3-, and 4-year OS rates were 61%, 48%, and 46%, respectively. 47 These favorable outcomes might be mainly caused by the high rates of crossover; thus, a large proportion of patients readily received CAR T therapy in the third-line setting. In fact, for patients receiving subsequent therapies in ZUMA-7, the median PFS was 6.3 mo in SOC patients receiving third-line CAR Ts versus 1.9 months for patients receiving alternative therapies with a median OS of 16.3 versus 9.5 months, respectively. 48
Since the bsAB combination therapy of Glofi-GemOx has been approved for R/R DLBCL patients after at least one prior therapy, the recently published EHA Clinical Practice Guidelines recommended the use of this regimen for either refractory/early relapsing CAR T-unfit or late relapsing HDT-unfit patients. 49 While it is yet hard to define this patient cohort due to missing data on its real-world feasibility, we believe the most relevant benchmark cohort for this regimen, and other targeted treatments like ADC-based combinations, is represented by the outcomes of patients that received only less-intensive/palliative treatment in our cohort, demonstrating a 5-year OS probability of only 14.6%.
This study had several limitations. First, this was a retrospective study; in particular, the retroactive assessment of study inclusion or CAR T-eligibility criteria was potentially based on incomplete data. Furthermore, the number of events was generally sufficient to ensure statistical power. However, this was not the case for all analyses, such as the comparison between age categories as well as PFS analyses in relapsed and refractory patients receiving second-line therapy. These non-significant results were associated with a small number of events and wide confidence intervals, indicating that a clinical effect cannot be completely excluded. Therefore, the results must be interpreted as inconclusive rather than a missing effect. Moreover, the monocentric nature of the study might be seen as a limitation, as institutional guidelines and practices might not be generalizable to all Austrian centers, and the results might thus not be precisely representative. However, an indirect comparison with other monocenter and multicenter studies in Europe revealed comparable results across many large sites. Additionally, clinical practice and international guidelines regarding the standard-of-care treatment of patients with DLBCL, especially in second-line settings and for fit patients, did not change dramatically during the study period. Furthermore, as CAR T-cell therapy has been approved and reimbursed in Austria since 2020, OS outcomes in patients in the second-line setting might be conflated as an impressive outcome with CAR T in patients who have failed two or more lines of therapy in Austria have been reported. However, as already indicated, the overall number of patients treated with CAR T during the study period in Austria was low, with a potentially limited impact on the overall outcome in our cohort. Finally, we admit that data evaluating real-world feasibility of frontline CAR T therapy is lacking and data on Pola-R-CHP in a European context is scarce. However, recent real-world evidence studies suggest that Pola-R-CHP is feasible and safe for the majority of newly diagnosed DLBCL patients. 50 These patients would otherwise have also qualified for R-CHOP or R-COMP, which represents 91% of all newly diagnosed patients in our cohort and as such serve as a reasonable benchmark for this patient population. The strengths of this study include the comprehensive and long-term follow-up of the patients included in our registry. Furthermore, this is the first study of Austrian patients with DLBCL to analyze the detailed patient trajectory from diagnosis to second-line treatment by including all patients diagnosed within the given observation period.
Conclusion
In summary, our study provides the first insights into the real-world trajectories and outcomes of patients with DLBCL treated at a large Austrian site. Overall, the outcomes were consistent with those of previous analyses, highlighting the high unmet need for novel treatment approaches, especially in patients with high-risk features at diagnosis and in patients with R/R disease. This is especially true for patients eligible for CAR T-cell therapy. Additionally, as new therapies such as CAR T-cell therapies have become novel SOC, we provide an outcome benchmark for patient populations now receiving such treatment for future comparative analyses.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251410356 - Supplemental material for Historic Real-World Outcomes and Future Benchmarks for Patients with Diffuse Large B-Cell Lymphoma Receiving First- and Second-Line Therapy in Austria – a Large Single-Center Experience
Supplemental material, sj-docx-1-tct-10.1177_15330338251410356 for Historic Real-World Outcomes and Future Benchmarks for Patients with Diffuse Large B-Cell Lymphoma Receiving First- and Second-Line Therapy in Austria – a Large Single-Center Experience by Michael Leisch, Dominik Kiem, Christoph Grabmer, Anton Kugler, Gianfranco Pocobelli, Mayer Marie-Christina, Bernd Schöpf and Alexander Egle, Richard Greil, Thomas Melchardt in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
We thank Michaela Schachner for support with data entry, database curation, and administration.
Ethics Statement
This study was evaluated by the regional ethics committee “Ethikkommission für das Bundesland Salzburg” via proposal EK Nr: 415-EP/73/127-2012 and has been approved via ethics committee vote EK Nr: 1046/2022 on 18 May 2022. The study has been conducted in accordance with the Declaration of Helsinki. All the patients included in this analysis provided written informed consent.
Author Contributions
TM, BS, DK, and ML conceptualized and designed the study. All authors, except BS, were involved in data curation and verification. TM and GP performed formal data analyses. TM, ML, BS, DK, and GP visualized the data and created the tables and figures. TM supervised the study. All authors contributed to data collection, interpreted the data, participated in writing the manuscript, provided feedback throughout the development process, and approved the final version of the manuscript submitted for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Kite, a Gilead Company. Medical writing assistance was provided by Medical-Writing Gerhard Krumschnabel (![]() ) and funded by Kite, a Gilead Company
) and funded by Kite, a Gilead Company
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Honoraria and research support from BMS, Incyte, Kite/Gilead and Roche. Medical writing assistance was provided by Medical-Writing Gerhard Krumschnabel.
TM: Honoraria from KITE, Roche and Incyte
CG: Honoraria from KITE
ML: Honoraria from BMS, Kite
AE: Honoraria from KITE, Roche
BS: employment with Kite Pharma, a Gilead company and Gilead stock ownership
RG: Honoraria and research support from KITE, Roche
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
