Abstract
Background
Lymphovascular invasion (LVI) is a critical factor in breast cancer (BC) prognosis and treatment planning, yet preoperative non-invasive assessment remains challenging. This research proposes the design and validation of a comprehensive artificial intelligence (AI) system that combines intratumoral and peritumoral radiomic analysis, deep learning (DL)-derived features, and clinical risk indicators extracted from dynamic contrast-enhanced MRI (DCE-MRI), with the goal of predicting LVI status in patients with BC.
Methods
This multi-institutional retrospective study included 496 IBC patients (training cohort: n = 344; validation cohort: n = 152). DCE-MRI scans were acquired preoperatively, and intratumoral/peritumoral (0-1, 1-3, 3-5 mm) radiomics features were extracted. A ResNet-50-based DL model was applied to 2.5D tumor slices, and clinical risk factors were identified via logistic regression. The least absolute shrinkage and selection operator (LASSO) method was employed to identify the most relevant features. The ensemble model was created by combining the Intra- Peri Fusion model with the clinically independent risk factors. Model performance was evaluated by sensitivity, specificity, AUC, and decision curve analysis (DCA).
Results
LVI was present in 33.8% and 32.7% of the training and validation cohorts. The SVM (Support Vector Machine) Intra-Peri Fusion model reached AUCs of 0.921 and 0.906, showing enhanced discriminative performance over single-region approaches. The ensemble model, derived from integrating a fusion model with clinical risk factors, demonstrated superior performance with AUCs of 0.951 (training) and 0.929 (validation) and high net benefit in DCA. Calibration curves confirmed excellent agreement between predicted and observed outcomes.
Conclusion
The AI-driven ensemble model combining radiomics, DL, and clinical features enables accurate preoperative prediction of LVI in IBC, which holds potential for optimizing surgical planning and adjuvant therapy strategies.
Introduction
Breast cancer (BC) remains one of the most prevalent cancers affecting individuals worldwide, with a cancer mortality rate of approximately 6.9%, posing a significant threat to women's health. 1 The majority of BC-related deaths result from primary tumor proliferation or distant metastasis. Lymphovascular invasion (LVI), defined as the presence of malignant cells within lymphatic and/or blood vessels adjacent to the tumor, plays a pivotal role in the processes of tumor infiltration and metastatic spread. 2 LVI positivity is closely associated with an increased risk of local recurrence and distant metastasis in BC patients, making it an important factor influencing treatment choices and long-term prognosis.3,4 In the context of early-stage BC with LVI, a comprehensive treatment plan incorporating mastectomy and systemic therapy should be considered, notably in patients needing additional surgery. 5 Consequently, preoperative identification of LVI status is essential to guide clinical decision-making and enhance treatment outcomes for BC patients.
Accurate preoperative assessment of LVI remains a challenge. The limitations of needle biopsy prevent the specimen from providing information about the lymphovascular status surrounding the tumor, leading to lower accuracy in preoperative LVI assessment. 6 Postoperative pathology is the only method for confirming LVI; however, it may result in diagnostic delays and increase the psychological burden on patients. Therefore, it is crucial to explore a non-invasive and accurate method for preoperative LVI evaluation. Dynamic Contrast-Enhanced MRI (DCE-MRI) offers data on tumor vascular system volume, permeability, and angiogenesis. Accumulating studies suggest that specific features extracted from DCE-MRI are significantly associated with LVI occurrence in BC cases.7,8 However, routine image analysis relies on the expertise of the physician, and inexperienced radiologists may lead to unnecessary or delayed treatments.9,10
Advancements in AI algorithms, particularly deep learning (DL) and radiomics, have become invaluable tools in BC research. Radiomics enables the extraction of numerous quantitative features from medical images in a non-invasive manner. In contrast, DL techniques exploit multi-level neural architectures to detect nuanced differences in image patterns, producing high-dimensional feature maps. 11 Prior researches have shown that the peritumoral region contains critical information, such as blood vessels, lymphatic vessels, immune infiltration, and stromal reactions, which are closely associated with tumor biology and patient prognosis. 12 While prior studies have explored radiomics or deep learning to predict LVI in breast cancer, most rely solely on intratumoral features, 13 use single peritumoral distances, 12 or lack integration with clinical factors.13,14 Many also suffer from small sample sizes and lack external validation. 14 In contrast, our study integrates multi-scale peritumoral and intratumoral radiomics, ResNet-based deep features, and clinical risk factors in a unified SVM model, validated on a multi-center cohort.
The purpose of this work is to assess the potential of DL and radiomics models, derived from intra- and peritumoral DCE-MRI data, for accurate preoperative prediction of LVI in BC patients.
Methods
Study Population
In this multi-institutional study, we recruited patients from three centers. The enrollment period ranged from July 2020 to June 2023, including women who underwent breast MRI scans. The inclusion criteria were: (1) histologically confirmed BC; (2) breast MRI performed within two weeks prior to surgery; (3) Availability of both complete clinical profiles and corresponding pathological data. The exclusion criteria were: (1) Preoperative interventions such as immunotherapy, chemotherapy, or radiation therapy; (2) concurrent malignancies; (3) incomplete MRI protocol or suboptimal image quality. The procedure for patient selection is depicted in Figure 1. A total of 496 BC patients were ultimately enrolled in this study. Participants from Center 1 and Center 2 (n = 344) were allocated to the training cohort, while those from Center 3 (n = 152) constituted the validation cohort. LVI was defined by histopathological evidence of tumor cell infiltration into adjacent lymphatic or blood vessels. 15 Based on LVI status, patients were categorized into two cohorts: those with (LVI-positive) and those without (LVI-negative). The study was designed and executed in accordance with the Medical Imaging Artificial Intelligence Checklist guidelines. 16 The study received approval from the Ethics Committee, and informed consent was waived. Ethical approval number: YX-Z-2023-063-01.

Flow Chart for Patient Recruitment.
Clinical Data Collection and Clinical Model Construction
Figure 2 shows the workflow of our study. Patient clinical information was recorded, including Age, Size, Menstruation, Location, LymphNode_Metastasis, Quadrant, Skin_Edema, Nipple_Retraction, Morphology, Margin, Enhancement_type, Enhancement_Curve, Vascular_shadow, BI-RADS. Univariate analysis was conducted on all clinically relevant variables. Predictive variables identified through univariate analysis (P < 0.05) were subjected to multivariate logistic regression to isolate independent LVI-related factors, which were then used to build the clinical model.

The Schematic Workflow of Model Development and Validation.
Image Segmentation and Preprocessing
A high-pressure injector delivers gadopentetate dimeglumine into the antecubital vein at a concentration of 0.2 mmol/kg and a flow rate of 2.0 ml/s during DCE-MRI acquisition. Following this, 15 ml of saline is injected at the same flow rate. The scan continuously acquires data across 8 phases without intervals, with each phase having a scanning duration of approximately 68 s. Each individual scan phase consists of 160 slices. Detailed MR imaging parameters are provided in Supplementary Table S1. MRI images were preprocessed using voxel resampling, grayscale discretization, and grayscale normalization. First, linear interpolation was applied to resample the images to a resolution of 1 mm × 1 mm × 1 mm. A bin width of 25 discrete grayscale levels was chosen to minimize the impact of noise heterogeneity in the images. Grayscale values were normalized to the range of [0, 1] to ensure data comparability. Manual 3D ROI segmentation was conducted on DCE-MRI slices using ITK-SNAP (v3.8.0). Both solid and cystic tumor components were included, with the largest lesion selected in multifocal cases. Two radiologists (10 years of experience) performed the delineation independently, and disagreements were resolved by a senior radiologist with 25 years of chest MRI experience. Clinical and pathological data were blinded during segmentation.
Tumor ROIs were dilated by 1 mm, 3 mm, and 5 mm beyond their original boundaries using the “SimpleITK” in Python (version 3.6). These distances were selected based on previous studies indicating that most tumor-associated stromal and microvascular changes occur within the 5 mm zone around the tumor boundary.12,17,18 To minimize the inclusion of non-tumor structures such as adipose tissue or normal glands in the peritumoral regions, all expanded masks were visually inspected by two board-certified radiologists with 10 and 12 years of experience in breast MRI. Manual adjustments were applied when the peritumoral zone extended beyond the anatomical boundaries of the breast parenchyma. In cases of disagreement, a third senior radiologist (20 years of experience) provided the final decision to ensure consistency and minimize inter-observer variability. Radiomic feature extraction was performed using PyRadiomics, 11 yielding 1834 features were calculated, including shape features, first-order statistical features, and texture features derived from gray level co-occurrence matrix (GLCM), gray level run length matrix (GLRLM), gray level size zone matrix (GLSZM), gray level dependence matrix (GLDM), and neighborhood gray-tone difference matrix (NGTDM). Features were extracted not only from the original image but also from a set of filtered images to enhance diversity and capture multi-scale signal patterns. The applied filters included wavelet (8 decompositions), exponential, square, square root, logarithm, and gradient. All images were normalized (z-score) before feature extraction.
Deep Learning Feature Extraction
In this study, a DL model was constructed based on the ResNet-50 architecture. The input comprised 2.5D representations, generated by selecting the largest slices in the axial, coronal, and sagittal planes. These images were resized to 224 × 224 voxels to match the input dimension requirements. To mitigate the limitations posed by a relatively small dataset, transfer learning was employed. During model training, real-time data augmentation techniques—such as adjustments in brightness and contrast, random rotations, and horizontal or vertical flips—were applied. Additionally, on-the-fly augmentation with random cropping and flipping further enhanced variability. Model optimization was performed using the Adam optimizer, configured with batch size of 64, weight decay of 0.01, and trained over 50 epochs. To handle class imbalance, the focal loss function was adopted. High-level features extracted from the convolutional layers were subsequently compressed using Principal Component Analysis (PCA), reducing feature dimensionality to 128 components to improve generalization and reduce overfitting.
Feature Selection and Model Construction
For each feature type (radiomics features, ResNet-derived deep features, and their combinations with clinical variables), we evaluated three commonly used classifiers: support vector machine (SVM), logistic regression (LR), and random forest (RF). Classifier performance was compared using five-fold cross-validation on the training set, and the best-performing classifier (SVM) was selected and used for all final models. To prevent overfitting, dimensionality reduction was performed using PCA (for deep features), and feature selection was conducted using mRMR and LASSO. SMOTE was applied exclusively to the training set to address class imbalance. Final model performance was evaluated both on cross-validation and on an independent external test cohort.
Specifically, features extracted from the intratumoral region were used to develop the Intra-Rad model, while DL features derived from the same region constituted the Intra-DL model. For the peritumoral areas (0-1 mm, 1-3 mm, and 3-5 mm from the tumor boundary), separate models were established, denoted as Peri-1, Peri-3, and Peri-5, respectively. A Peri-Fusion model was subsequently created by integrating features across all three peritumoral distances. Finally, an Intra-Peri Fusion model was generated by combining intratumoral radiomic features, peritumoral radiomic features, and DL-derived representations. To enhance predictive performance, we developed an ensemble model, which integrates radiomic features, deep learning-derived features, and clinical risk factors.
Model Training and Classifier Comparison
For each patient, a total of 1834 × 3 peritumoral radiomic features, 1304 intratumoral radiomic features, and 128 DL features were extracted from the MR images. After dimensionality reduction and feature selection, 23 peritumoral radiomic features, 24 intratumoral radiomic features, and 24 intratumoral DL features were retained for model construction. Based on these features, three machine learning algorithms—SVM, LR, and random forest—were used to construct the Peri-fusion model, Intra-Rad model, and DL model. The implementation scripts are available in Supplementary Code S1.
Statistical Analysis
All statistical evaluations were performed in SPSS 26.0 and R 4.1.2. Parametric data were presented as mean ± SD and compared using t-tests. Categorical variables were analyzed via Chi-square tests, and ordinal variables via rank-sum tests. ROC analysis provided AUC, sensitivity, specificity, and accuracy for model evaluation. Delong tests were used to compare AUCs, while calibration curves and DCA were applied to assess model reliability and clinical benefit. Significance was defined as P < 0.05.
Result
Patients
A total of 496 BC patients were included in this study, comprising 344 cases in the training cohort and 152 cases in the validation cohort. Postoperative pathological examination confirmed the presence of LVI in 116 cases (33.8%) from the training cohort and 50 cases (32.7%) from the validation cohort (Table 1).
Baseline Clinical Characteristics of the Tumour Budding Low, Medium and High Grade Cohorts.

Clinical Model Development and Evaluation
We evaluated the relationship between clinical data and LVI in patients with BC in the training cohort. Univariate and multivariate logistic regression analysis revealed that lymph node metastasis, tumor size, and BI-RADS staging were independent risk factors (Table 2). Based on these factors, a clinical model was developed to predict LVI in IBC patients, incorporating lymph node metastasis, tumor size, and BI-RADS staging. The AUC of the model in the training cohort was 0.657, and in the validation cohort, it was 0.611 (Table 3).
Independent Risk Factors After Univariate and Multivariate Logistic Regression Analysis.

Diagnostic Performance of Different Model.
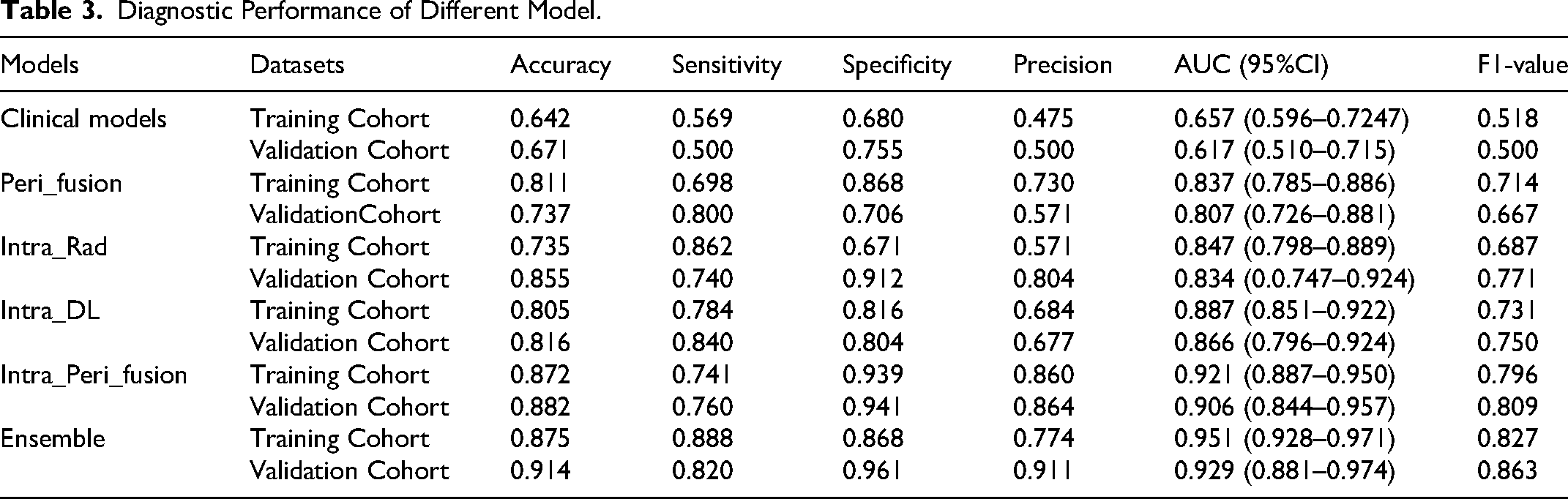
Image Model Construction and Evaluation
As indicated in Supplementary Table S2, for the machine learning model based on peritumoral radiomic features, SVM showed the optimal diagnostic performance (AUC = 0.807 [95% CI: 0.726-0.881]), with a sensitivity of 0.800 and specificity of 0.706 in the validation cohort. As shown in Supplementary Table S3, for the machine learning model based on intratumoral radiomic features, SVM maintained a leading position in the validation cohort (AUC = 0.834 [95% CI: 0.0.747-0.924]). Compared to the LR and random forest models, the SVM model exhibited higher sensitivity and specificity. As shown in Supplementary Table S4, for the model based on intratumoral DL features, SVM also demonstrated the optimal diagnostic performance (AUC = 0.866 [95% CI: 0.796-0.924]), with a sensitivity of 0.840 and specificity of 0.804. Building on this, the study further integrated peritumoral and intratumoral radiomic features, as well as intratumoral DL features, to construct the Intra Peri Fusion model. As shown in the Table 3 and supplement table S5, SVM again demonstrated the optimal diagnostic performance (AUC = 0.906 [95% CI: 0.844-0.957]), with a sensitivity of 0.760 and specificity of 0.941. Overall, the three models demonstrated their effectiveness in predicting LVI in BC patients, with the fusion model significantly outperforming the individual models (Table 3). Given the superior performance of the fusion model, the Intra Peri Fusion model was selected to participate in the construction of the ensemble model.
Ensemble Model Construction and Evaluation
To enhance the prediction of LVI in BC, this study integrated key independent clinical risk factors—including lymph node involvement, tumor diameter, and BI-RADS classification—with the Intra-Peri Fusion features to construct an ensemble model. This integrated approach demonstrated superior predictive performance when compared to models based solely on feature fusion or clinical variables, achieving AUC values of 0.951 and 0.929 in the training and validation cohorts, respectively.
DCA revealed that within a threshold probability range of 0 to 0.9, the ensemble model yielded the highest net clinical benefit in the training cohort, indicating strong clinical utility (Figure 3). Model calibration was further supported by the Hosmer–Lemeshow goodness-of-fit test, which showed no statistically significant deviation in either the training (P = 0.192) or validation cohort (P = 0.845), suggesting satisfactory model fit. Additionally, calibration plots (Figure 3) illustrated close agreement between predicted probabilities and actual outcomes.

ROC Curves, Decision Curve Analysis, and Calibration Curves of Different DL Models in Training Cohort (a-c) and Validationt Cohort (d-f).
Discussion
In this study, we developed the Peri-fusion model, Intra-Rad model, Intra-DL model, and Intra-Peri fusion model based on radiomic features from the peritumoral (0-1, 1-3, 3-5 mm) and intratumoral regions, as well as DL features, to explore their value in predicting LVI status in IBC patients. Among the models constructed, the Intra-Peri fusion model, which integrated peritumoral radiomic features, intratumoral radiomic features, and DL features, demonstrated the best diagnostic performance. Furthermore, an ensemble model was constructed by combining the Intra-Peri fusion model with independent risk factors, such as lymph node status, tumor size, and Bi-Rads category. The results showed that the ensemble model achieved the highest AUC values of 0.951 in the training cohort and 0.929 in the validation cohort, outperforming all other models. Additionally, calibration curve and DCA results indicated that it had good stability and clinical applicability. LVI is a critical process in BC metastasis, and accurately determining the LVI status before surgery plays a vital role in guiding physicians in understanding the patient's condition and planning subsequent treatments. 19 Currently, the gold standard for confirming LVI remains postoperative pathological results, as preoperative assessment remains challenging. MRI is an essential imaging technique for preoperative evaluation of BC. We identified lymph node status, tumor size, and Bi-RADS as independent clinical risk factors for LVI. Tumor size has been widely established as one of the important indicators influencing the prognosis of BC patients. 20 We found that larger tumors were associated with a higher risk of LVI, which is consistent with the findings of NIJIATI et al, 21 suggesting that LVI-positive BCs are more malignant and aggressive, often exhibiting a rapid growth pattern.
As a precursor to axillary lymph node metastasis, LVI is considered not only an independent predictor of axillary lymph node metastasis but also its initial stage.22,23 In clinical settings, MRI assessment of axillary lymph node involvement serves as an important predictor of metastasis in BC and demonstrates consistent diagnostic accuracy. 24 Consistent with previous studies, our findings further emphasize the correlation between MRI-based axillary lymph node status and LVI in BC. Bi-RADS, as the standardized grading system for breast imaging, reflects the malignancy of tumors by assessing their imaging features.4,8 We also found a significant association between higher Bi-RADS scores and LVI, likely due to the fact that higher-grade tumors exhibit irregular borders, neovascularization, and enhanced blood flow signals, which suggest greater invasiveness and may indicate local dissemination, thus increasing the risk of LVI. After constructing a model based on these three clinical independent risk factors, we found that the predictive performance for invasive LVI in BC was moderate. While specificity was high (75.5%), sensitivity was suboptimal (50.0%). Therefore, relying solely on clinical factors may not provide an accurate assessment of LVI status in BC.
Artificial intelligence, including radiomics and DL, has provided new opportunities for tumor research, particularly in the context of BC. 25 Zhang et al constructed a model combining radiomics features extracted from T2WI, ADC, and DCE-MRI images, achieving an AUC of 0.919, sensitivity of 0.902, and specificity of 0.829 in the training cohort, and an AUC of 0.863, sensitivity of 0.778, and specificity of 0.818 in the validation cohort. 14 Although this model greatly improved predictive ability compared to individual sequences, it is complicated to use in clinical practice due to the combination of multiple sequences, which reduces its clinical applicability. Yang et al developed a Joint model combining radiomics and DL features based on DCE-MRI, achieving an AUC of 0.827, accuracy of 0.756, sensitivity of 0.775, and F1-score of 0.755 in the validation cohort. 13 However, their study had a small sample size and only analyzed tumor internal features. The peritumoral region represents the tumor microenvironment, which contains significant biological information such as immune cells, extracellular matrix, and neovascularization. 26 Studies have shown that BC invades the surrounding microenvironment during progression, leading to tissue remodeling around the tumor.27,28 Therefore, extracting radiomics features from the peritumoral region may help quantify the invasiveness of the tumor. However, there is still controversy regarding the optimal definition of the peritumoral region in BC. To date, there is no universally accepted gold standard for peritumoral region definition. Our use of multi-scale fixed-distance zones was based on literature evidence and intended to provide a balance between biological relevance and practical feasibility. Wang et al developed radiomics models targeting peritumoral regions expanded by 1 mm, 3 mm, and 5 mm, and reported that the 3 mm model exhibited superior performance in differentiating benign from malignant breast lesions, achieving an AUC of 0.930, accuracy of 0.872, sensitivity of 0.900, and F1-score of 0.878 in the validation cohort. 12 Li et al provided evidence that radiomic characteristics derived from the 4 mm peritumoral area are predictive of HER-2 and Ki-67 expression in BC patients. 17 Additionally, Xie et al found that texture features in the 5 mm peritumoral region can predict the ability to achieve pathological complete response after neoadjuvant therapy in BC. 18 A comparative summary of the performance metrics reported in these studies is presented in Supplementary Table S6.
In light of these findings, we extracted radiomics features from peritumoral regions of 1 mm, 3 mm, and 5 mm to predict the LVI load in BC. The results showed that a model combining features from peritumoral regions of 0–1 mm, 1–3 mm, and 3–5 mm outperformed models using individual regions, with the 1 mm peritumoral region having the highest weight coefficient. This emphasizes the importance of the 1 cm peritumoral features, consistent with previous studies showing the key role of the tumor microenvironment in metastasis and prognosis. 29 Most microvascular invasions occur within a 1 cm region.
To enhance model diagnostic efficacy, this study also used DL to explore tumor internal features. The results indicated that the ResNet50-based DL model outperformed the radiomics model slightly in predicting LVI in IBC patients. This may be due to the strong deep structure and residual connection advantages of ResNet50, which can capture low-level details such as edges and textures, while also identifying high-level semantic information, thereby improving diagnostic efficacy by extracting complex information from DCE-MRI images. Furthermore, after feature selection in the ensemble model, tumor internal radiomics features were excluded, likely becauseDL features can capture more complex, non-linear patterns, showing stronger feature representation abilities. There was high redundancy or correlation between radiomics and DL features, so radiomics features were compressed, leaving only DL features.
We innovatively integrated clinical features (such as tumor size and staging), peritumoral and tumor internal radiomics features, and DL features using an SVM model, significantly improving prediction accuracy. This integration takes advantage of the complementary nature of the three types of features: clinical features capture macroscopic characteristics, radiomics features reveal microscopic details, and DL features uncover complex high-order non-linear patterns. The SVM model, with its kernel function, can capture complex interactions between different features, mapping data to high-dimensional space, thus improving the model's robustness and generalization ability. The superior performance of the ensemble model in predicting LVI, especially when compared with existing studies, demonstrates its potential in complex tasks. It is important to note that no single imaging sequence can consistently deliver excellent results across all validation cohorts, highlighting the high heterogeneity of IBC tumors. Therefore, an ensemble model integrating multiple data sources is better suited to reflect the tumor's complex characteristics, improving the accuracy of LVI prediction.
Although we did not implement an end-to-end slice-based CNN classifier, we extracted deep semantic features from ResNet and integrated them with radiomic and clinical features using a unified SVM model to enhance robustness and interpretability. To further improve model transparency, we applied SHAP exPlanations (Figure S1) to the SVM-based models, which revealed that key features such as peritumoral entropy, intratumoral homogeneity, and tumor size contributed strongly to the predictions—consistent with known imaging correlates of LVI. Additionally, Grad-CAM was used on the 2.5D ResNet inputs (ie, the largest tumor slice and its adjacent layers) to visualize spatial attention. The resulting heatmaps demonstrated that the model primarily focused on tumor borders and surrounding regions. These areas are often associated with LVI and may reflect relevant morphological or perfusion patterns, supporting the biological plausibility and clinical relevance of the model's predictions. (Figure S2)
Despite the good predictive ability shown in this study for LVI status in BC, some limitations remain: (1) Although our model shows promising performance, it remains a preliminary tool. Future prospective studies with larger, more diverse cohorts are necessary to validate its clinical utility. (2) Manual 3D segmentation is time-consuming. Although reviewed by experienced radiologists using a standardized process, manual correction of peritumoral masks may still introduce subjectivity. Automated approaches could improve consistency in future work. (3) As our deep learning model used a 2.5D input composed of the largest tumor slice and its adjacent layers, some spatial information and volumetric heterogeneity may have been lost. While this strategy improves clinical feasibility and reduces computational burden, we acknowledge its limitations and suggest that future studies should explore full 3D convolutional networks to better capture tumor morphology and peritumoral context.
In conclusion, the radiomics and DL features based on DCE-MRI in the tumor internal and peritumoral regions have good predictive value for LVI status in IBC patients. The ensemble model combining clinical risk factors further improves performance and may play an important role in clinical decision-making, avoiding unnecessary invasive examinations.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251374945 - Supplemental material for Integrating Peritumoral and Intratumoral Radiomics with Deep Learning for Preoperative Prediction of Lymphovascular Invasion in Invasive Breast Cancer Using DCE-MRI
Supplemental material, sj-docx-1-tct-10.1177_15330338251374945 for Integrating Peritumoral and Intratumoral Radiomics with Deep Learning for Preoperative Prediction of Lymphovascular Invasion in Invasive Breast Cancer Using DCE-MRI by Qiaomei Zhao, Hui Zhang and Wei Xing in Technology in Cancer Research & Treatment
Footnotes
Ethics Approval and Consent to Participate
This retrospective study received ethical approval from the Ethics Committee of the Third Affiliated Hospital of Soochow University (approval code: 2023017), the Huai’an Maternity and Child Clinical College of Xuzhou Medical University (approval code: 2025028), and the Affiliated Huaian No.1 People's Hospital of Nanjing Medical University (approval code: YX-Z-2023-063-01), and the requirement for informed consent was waived because the study used only de-identified patient data.
Author Contributions
Qiaomei Zhao and Wei Xing proposed the initial study concept, while Hui Zhang was responsible for study design. Data collection was carried out by Qiaomei Zhao. Wei Xing and Hui Zhang jointly conducted data analysis and interpretation. Statistical analysis was performed by Hui Zhang. The manuscript was drafted by Qiaomei Zhao and Wei Xing, who also contributed to its critical revision for intellectual content. All authors reviewed and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangsu Province Huai'an Basic Research Program (Joint Special Project) Health and Medical Research Project, (grant number HABL2023061).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the present study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
