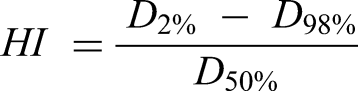Abstract
Introduction
Left-sided breast cancer radiotherapy requires precise dose modulation to balance target coverage and organ-at-risk (OAR) sparing. This study evaluates a novel Organ and Arc-based Directional Block (OABD Block) in helical tomotherapy planning to address this challenge.
Methods
In this single-institutional retrospective study, 10 post-mastectomy patients with left-sided breast cancer receiving adjuvant radiotherapy were studied. Target volumes included chest wall, internal mammary, axillary, and supraclavicular lymph nodes, with a dose of 50 Gy over 25 fractions. Using a tomotherapy planning system, an OABD Block was configured to incorporate arc structures and protect organs-at-risk. For each patient, helical tomotherapy plans were prepared with and without the OABD Block, keeping field width, pitch, and modulation factors identical. Additionally, static intensity-modulated radiotherapy (IMRT) plans were created on a United Imaging system. Treatment plans were evaluated by dose-volume parameters, conformity and homogeneity indices, and mean doses to targets and normal tissues.
Results
Helical tomotherapy with the OABD Block provided a mean conformity Index of 0.79 for the Planning Target Volume, higher than plans without the block (0.73) but below IMRT plans (0.88). The homogeneity Index averaged 0.14 with the block, 0.18 without, and 0.11 in IMRT. For the internal mammary lymph node region, D95% reached 5007.7 cGy with the block, compared to 5001.1 cGy without and 4897.9 cGy in IMRT. The OABD Block reduced the mean heart dose to 478.7 cGy, compared to 533.5 cGy without and 638.9 cGy in IMRT. Left lung V5 was 48.0% with the block, 52.7% without, and 53.2% in IMRT; V20 was also lowest with the block (17.5%) versus without (20.3%) and IMRT (24.3%).
Conclusion
Adding the OABD Block to helical tomotherapy improved internal mammary lymph node dose coverage and reduced exposure to organs at risk.
Introduction
Radiotherapy plays an indispensable role in the comprehensive management of breast cancer. In a 15-year study, Poortmans et al demonstrated that irradiating the internal mammary and medial supraclavicular lymph nodes significantly lowers mortality and recurrence rates in patients with stages I-III breast cancer. 1 For early-stage breast cancer patients with positive nodal involvement, irradiating the internal mammary nodes reduces the risks of distant recurrence and death, thus improving long-term survival. 2 However, in left-sided breast cancer, this irradiation may inadvertently increase the likelihood of cardiac toxicity, coronary atherosclerosis, pneumonia, and pulmonary fibrosis. 3 Therefore, minimizing radiation exposure to the heart and lungs while maintaining adequate dosing to the internal mammary lymph nodes is crucial for both clinical care and research advancement.
Traditional three-dimensional conformal radiotherapy (3DCRT) in breast cancer treatment is linked to adverse effects, including dermatitis, pneumonitis, and cardiac injury. These toxicities—both acute and late—occur because 3DCRT plans often lack uniform target coverage, creating hotspots that expose lung and cardiac tissues to excessive radiation. 4 Intensity-modulated radiation therapy (IMRT) addresses these limitations by using more beam angles and greater modulation, improving uniformity across the target volume and limiting radiation doses to surrounding healthy tissues, like the heart and lungs.4-6
Helical tomotherapy (TOMO) is an innovative technique that offers advantages over 3DCRT and IMRT. It uses a CT-scanner-like modality to deliver focused, helical, and volumetric irradiation from a 360-degree perspective. This method ensures superior target conformity and dose distribution uniformity, thereby maximizing the sparing of normal tissues and organs. Additionally, tomotherapy incorporates megavolt computed tomography for precise image-guided radiation therapy.7,8 It has been widely applied across various tumor types, showing notable benefits in complex cases such as head and neck cancers, whole brain and spinal irradiation, and multiple tumor scenarios. 9 In breast cancer radiotherapy, tomotherapy demonstrates superior long-term efficacy, reduced toxicity, and a lower tumor recurrence rate compared to traditional methods. 10 This technique is particularly beneficial for patients with complex breast cancer anatomy, bilateral breast cancer, the need for internal mammary lymph node irradiation, and pre-existing cardiac or pulmonary conditions from chemotherapy, offering exceptional protection for organs at risk while maintaining excellent target volume coverage. 11
For left-sided breast cancer with regional lymph node involvement, tomotherapy offers superior dose conformity and uniformity compared to 3DCRT, potentially sparing the heart and left lung from high doses associated with complications, though it may increase exposure to the contralateral lung and breast. 11 While IMRT uses controlled beam angles to minimize radiation exposure to organs at risk, tomotherapy relies on block configurations, including Exit-Only and Never modes, to shape the radiation field. In Exit-Only mode, radiation passes through the target before reaching an organ; in Never mode, radiation is fully blocked from the organ.
One study 12 assessed block configurations for tomotherapy in left-sided breast cancer, finding that a full block with partial transparency provided the best results but was complex to implement. While this setup reduces the V5 and V20 lung volumes, it unexpectedly raises the heart's mean dose.
This study presents an innovative approach using the OABD Block (Organ and Arc-based Directional Block), which integrates organs at risk with an arc-shaped structure based on the target volume's expansion. This strategy ensures adequate dose coverage while minimizing radiation exposure to critical organs, thereby enhancing the effectiveness of helical tomotherapy treatment plans.
Methods and Materials
Patient Selection and Delineation of Regions of Interest
The research process of this study is depicted in Figure 1. This retrospective study examined ten patients with left-sided breast cancer who underwent modified radical mastectomy followed by radiotherapy at a tertiary hospital from February 2021 to January 2022. The study was approved by the Institutional Review Board of the participating hospital. We collected comprehensive data, including radiotherapy prescriptions, positioning CT scans, and contours of target volumes and organs at risk, ensuring all procedures adhered to the hospital's ethical standards. Written informed consent was obtained from all participants for the use of their clinical data in this study.
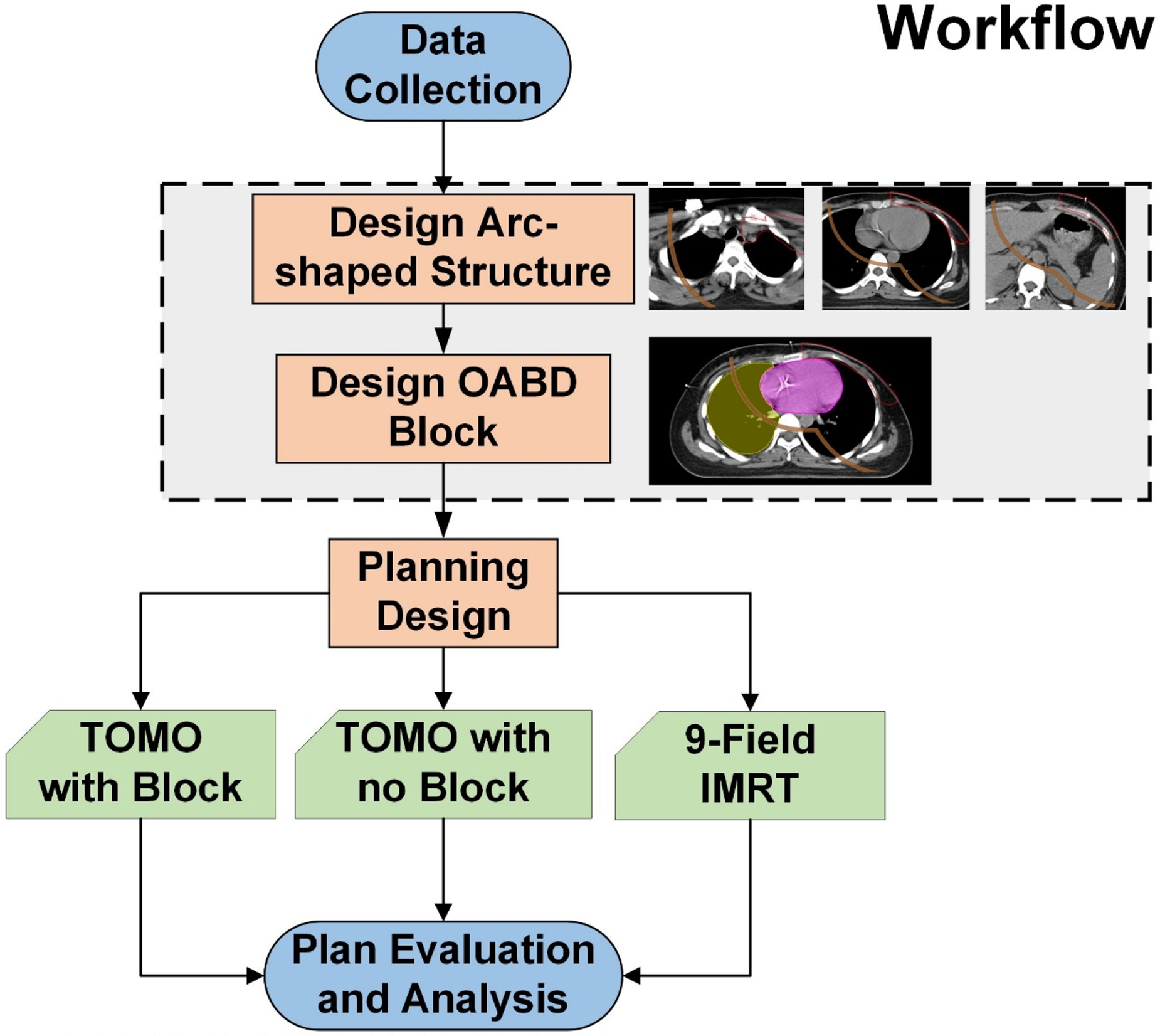
Research Workflow.
The patient positioning CT scans were acquired using a Siemens SOMATOM Definition AS scanner with a slice thickness of 5 mm. These scans were then imported into the United Imaging treatment planning system for delineation by radiation oncologists. Target volumes targeted for radiation therapy included the ipsilateral chest wall, supraclavicular and infraclavicular lymph nodes, high-risk axillary lymph nodes, and internal mammary lymph nodes. To ensure effective dose delivery to areas adjacent to the skin, we designed a 3 mm-thick virtual bolus with a density comparable to water (1 g/cm³), placed at the interface between the body surface and air. This virtual bolus was included in all treatment plans. The organs at risk, as contoured by the radiation oncologists, consisted of the left and right lungs, heart, contralateral breast, spinal cord, left humeral head, thyroid gland, trachea, and esophagus. Figure 2 provides a representative illustration of the delineated target volumes and organs at risk for one patient.
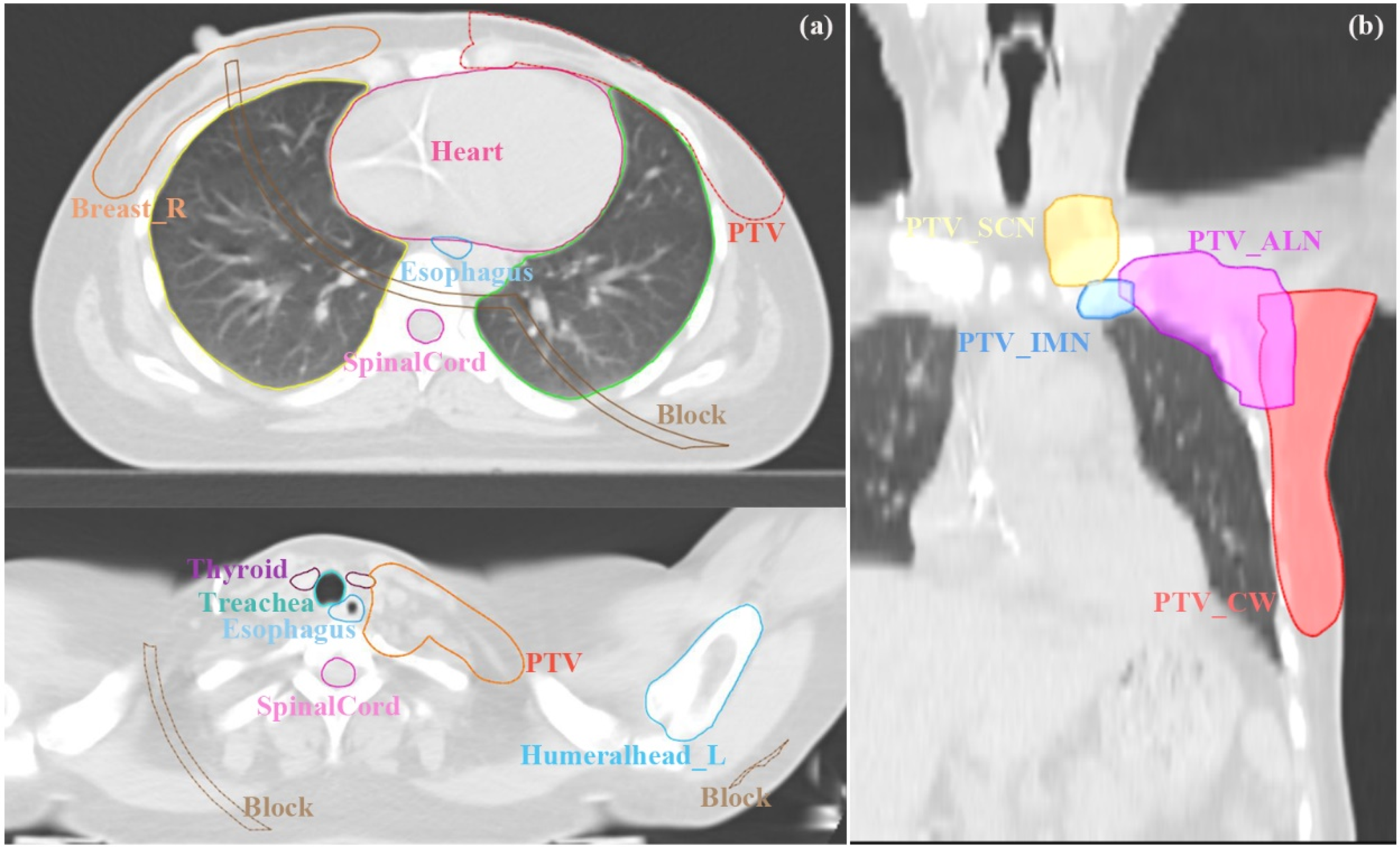
Graphical Description of the Organs at Risk (a) and Four Separated Planning Target Volumes (b) for Left-Sided Breast Cancer Patients Post-Modified Radical Mastectomy by Oncologist.
Block Design and Planning Optimization Strategy
In helical tomotherapy planning, we developed an arc-shaped structure by expanding the target volume to control beam incidence angles. This was done through the following steps:
Step 1: Enlarge the target volume by 9 cm to form a 5 mm-wide ring.
Step 2: Intersect this ring with the body contour, reduced inward by 1 cm, to produce the final arc shape (illustrated by the brown contour in Figures 1 and 2).
For planning design, we incorporated this arc shape, along with the heart and contralateral lung, into a composite virtual block. This was done as follows:
Step 3: Combine the arc-shaped structure with the heart and contralateral lung to form a composite virtual block.
Step 4: Within the planning system, configure this block as Exit-Only to manage beam angles effectively in the tomotherapy plan. This block is termed the OABD Block (Organ and Arc-Based Directional Block).
In this study involving ten patients, TOMO plans were developed using the Precision treatment planning system v1.1.1.1[11] (Accuray, Inc., Madison, Wisconsin, USA), with the GPU-based VOLO optimizer and the Collapsed Cone Convolution Superposition dose calculation algorithm. We developed two types of plans: one that included the Organ and Arc-Based Directional Block (OABD Block, referred to as TOMO_Block) and another that excluded it (denoted as TOMO_no_Block). Both plans were configured with Dynamic Jaw Mode, a field width of 2.5 cm, a pitch of 0.287, a modulation factor of 3.8, and a fine dose calculation grid (1.17 mm × 1.17 mm × 5 mm). The optimization parameters and dose constraint functions remained identical for both plans, except for the application of the OABD Block.
The prescribed dose to the Planning Target Volume (PTV) was 50 Gy in 25 fractions. In the optimization process, the dose constraints, established based on our institutional clinical experience and international guidelines, 13 are as follows: At least 95% of the PTV must receive 50 Gy, with the maximum PTV dose kept below 60 Gy (120% of the prescribed dose); the mean heart dose must be less than 7 Gy; for the ipsilateral lung, V20Gy must be ≤30% and V5Gy ≤65%; the maximum spinal cord dose must be less than 45 Gy; the esophagus Dmean ≤25 Gy; the thyroid Dmean ≤25 Gy; the humeral head Dmean ≤25 Gy; the contralateral breast V5Gy ≤10%; and the contralateral lung V5Gy ≤10%.
During the TOMO plan optimization, we progressively increased the penalty weights for organs at risk to achieve minimal doses while meeting target constraints. The optimization concluded when no further significant improvements were evident, leading to the final dose calculations.
Additionally, for these ten patients, we constructed IMRT plans on the United Imaging treatment planning system, using one 0-degree field and eight tangential fields with gantry angles of 295°, 305°, 325°, 340°, 0°, 95°, 110°, 130°, and 150°. The prescription and dose constraints for the IMRT plans were consistent with those of the TOMO plans. During optimization, meeting the PTV objective took the highest priority, followed by satisfying the dose constraints for the heart, ipsilateral lung, and contralateral breast. For all cases, the dose was computed using the Collapsed Cone Convolution algorithm, with the dose calculation grid set to 2.5 mm for the IMRT plan on the United Imaging treatment planning system vR001 (United Imaging Healthcare, UIH, Shanghai, China).
Evaluation Metrics
We evaluated the treatment plans using the Homogeneity Index (HI) and Conformity Index (CI) to assess dose uniformity and conformity within the target volume.
14
The HI formula is:
The CI formula is:
To measure OAR protection, we analyzed V5, V20, and mean doses. To quantify improvements from the OABD Block in TOMO plans, we used
Since a higher CI signifies a superior plan for the target volume, while lower values indicate better OAR protection, we adjusted the improvement formula for the target volume CI to
Results
Table 1 provides a comparative analysis of dosimetric parameters for the three treatment plans. The Conformity Index (CI) for the PTV was 0.79 for the plan with the OABD Block, 0.73 without it, and 0.88 for the IMRT plan. Homogeneity Index (HI) values were 0.14, 0.18, and 0.11, and the D95% for the internal mammary lymph nodes was 5007.7 cGy, 5001.1 cGy, and 4897.9 cGy, respectively. The maximum PTV dose was 5785 cGy with the Block versus 5971.1 cGy without it (p = 0.005), with no significant difference from the IMRT plan's 5741.4 cGy (p = 0.386).
Comparative Dosimetric Parameters of TOMO_Block, TOMO_no_Block, and IMRT Plans.

Abbreviations: TB, TOMO plan with Block; TnB, TOMO plan with no Block; CI, Conformity index; HI, Homogeneity index; PTV IMN, the PTV of Internal Mammary Nodes.
The mean heart dose, a critical metric, was 478.7 cGy with the OABD Block, 533.5 cGy without, and 638.9 cGy for IMRT. For the ipsilateral lung, the V5 values were 48.0%, 52.7%, and 53.2%, respectively, while the V20 values were 17.5%, 20.3%, and 24.3%, respectively. The contralateral lung exhibited V5 values of 8.4%, 21.3%, and 1.5%, and the contralateral breast showed V5 values of 9.1%, 25.3%, and 8.7%, respectively. The thyroid received mean doses of 1504.2 cGy, 1574.4 cGy, and 2085.4 cGy, and the left humeral head, 1247.1 cGy, 1329.8 cGy, and 1962.1 cGy. Other organ doses were 1000.6 cGy, 1209.7 cGy, and 1189.3 cGy for the trachea and 946.4 cGy, 1123.1 cGy, and 693.5 cGy for the esophagus. Spinal cord maximum doses were 2798.5 cGy, 3235.7 cGy, and 902.7 cGy.
Figure 3 illustrates the improvements in critical metrics for TOMO plans following the integration of the OABD Block. The incorporation of the OABD Block led to a 21.2% increase in the Homogeneity Index (HI) and a 7.2% improvement in the Conformity Index (CI) for the Planning Target Volume (PTV). There was a corresponding reduction of 8.9% in the V5 and 13.6% in the V20 for the ipsilateral lung. Furthermore, the mean dose to the heart decreased by 11.1%, while the V5 for the contralateral lung and contralateral breast decreased by 58.2% and 61.3%, respectively. However, these enhancements in dosimetric outcomes were accompanied by a 36.7% increase in the average time required for treatment using the TOMO plans.

the Degree of Improvement in key Metrics of TOMO Plans Using the OABD Block. the Lower Boxplot Displays the Mean and Interquartile Ranges, While the Upper Portion Shows a Scatter Plot. Since a Higher PTV CI Indicates a Better Plan, to More Intuitively Demonstrate the Improvement Effect on PTV CI, we Have Adjusted the Calculation Formula for the Improvement Degree of PTV CI to
In Figure 4b, the TOMO plan without the OABD Block displayed increased radiation doses in areas adjacent to the target volume, particularly in the right breast, ipsilateral lung, and lower ribs, compared to the plan that utilized the OABD Block. Furthermore, Figure 4c illustrates that the IMRT plan provided inadequate dose coverage of the PTV IMN relative to the TOMO plan with the OABD Block and resulted in higher radiation doses to the ipsilateral lung near the target volume. The dose-volume histograms in Figure 4d further corroborate these findings. The detailed views of the sagittal and coronal dose distributions are presented in Supplementary Figure 1.

Comparative Dose Distributions on the Same Transverse Slice for the Three Plans (a, b, c) and Dose-volume Histogram Comparisons (d) for the Same Patient.
In Figure 5, we explored the correlation between the dose variations resulting from the implementation of virtual blocks in TOMO therapy and the volumes of the regions of interest. The detailed R-squared values are provided in Supplemental Table 1. The analysis indicates a significant negative correlation between the right lung's V5, the mean dose to the left lung, and the left lung's V20 with the volume of the left lung. Likewise, the right lung's V5 and the left lung's V20 demonstrate a significant negative correlation with the volume of the right lung.
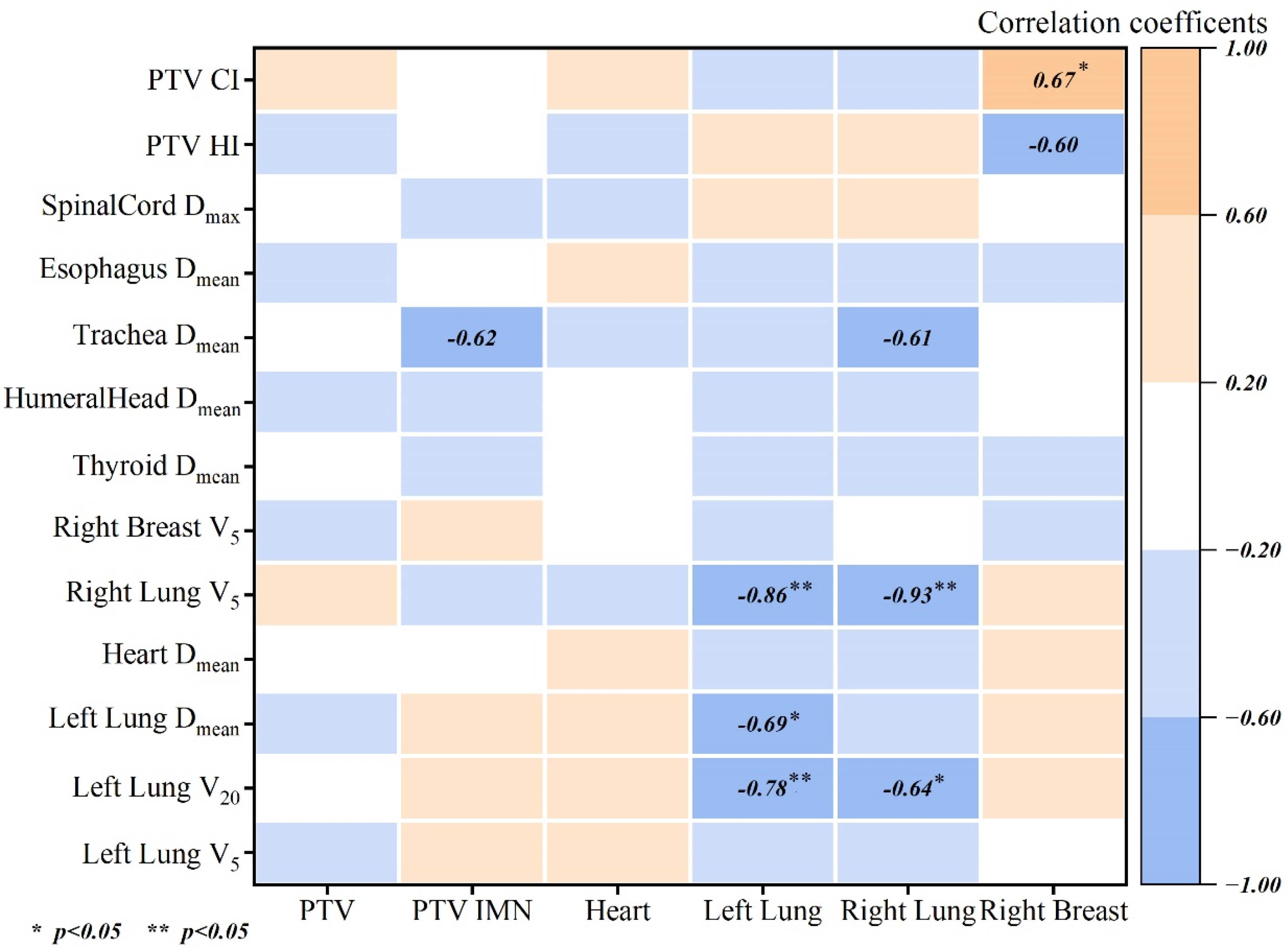
Pearson Correlation Coefficients Between the Differences in Dose Parameters Induced by the use of Virtual Blocks in TOMO Plans and the Volumes of the Regions of Interest.
Discussion
The TOMO plans incorporating the OABD Block demonstrated improved target volume coverage and reduced radiation exposure to organs at risk, outperforming both the IMRT plans and TOMO plans without the block.
In the PTV evaluation, the TOMO plan with the OABD Block outperformed the plan without the block in maximum dose, mean dose, homogeneity, and conformity. It also surpassed IMRT in PTV D95% and Dmean, with comparable maximum doses. Regarding PTV IMN, the IMRT plan's D95% of 4897.9 cGy was subthreshold and significantly lower than that of the TOMO plan with the OABD Block.
For the ipsilateral lung, the TOMO_Block plan showed significantly reduced V5 (48.0% vs 52.7%/53.2%), V20 (17.5% vs 20.3%/24.3%) when compared to both TOMO_no_Block and IMRT, and the increased lung V20 was associated with an increased risk of ≥ grade 3 pneumonitis. 15 TOMO plans often have difficulty minimizing low-dose areas in the contralateral lung and breast. However, by incorporating the OABD Block, the TOMO plan can effectively restrict these low-dose regions, meeting the clinical criterion of V5 less than 10%. In contrast, the non-blocked TOMO plan fails to satisfy this important criterion.
The TOMO_Block plan significantly reduced heart mean dose by 54.8 cGy compared to TOMO_no_Block and by 160.2 cGy compared to IMRT, highlighting its superior sparing of heart tissue. Van den Bogaard et al demonstrated that within 9 years of radiotherapy, each 100 cGy increase in mean heart dose raises the cumulative incidence of acute coronary events by 16.5%. 16 By reducing cardiac dose, the OABD Block may improve patients’ long-term survival and quality of life.
Quantitative assessment of the OABD Block's effects on high-dose regions of critical organs and body low-dose exposure is summarized in Table 2. Cardiac high-dose parameters remained stable, while left lung Dmax decreased by 3.4% (5536.3 vs 5725.4 cGy, p < 0.001), demonstrating ipsilateral organ protection. The right lung showed dramatic dose reshaping: 16.2% lower Dmax (2808 vs 3350 cGy, p = 0.002) and 37.2% reduced D2% (845.8 vs 1347.1 cGy, p < 0.001), attributable to beam angle optimization minimizing contralateral scatter. Paradoxically, right breast Dmax increased (2875.8 vs 2579.7 cGy, p = 0.003) but D2% decreased (1402.3 vs 1666.5 cGy, p = 0.007), suggesting spatial redistribution toward target-proximal regions. Whole-body V5 decreased by 23.4% (32.1% vs 41.9%, p < 0.001), significantly reducing low-dose exposure.
Comparison of maximum Dose to Critical Organs at Risk and low Dose to Body Between TOMO Plans with and Without Block.

Abbreviations: TB, TOMO plan with Block; TnB, TOMO plan with no Block.
The OABD Block enhances dose protection for smaller lungs and improves PTV conformity for larger right breasts. No significant correlations were found for other dosimetric parameters and organ volumes, as illustrated in Figure 5. This, along with Table 1, suggests that the OABD Block in TOMO plans optimizes dose distribution to targets and at-risk organs, independent of organ size.
Two studies have investigated the use of blocks in TOMO breast radiotherapy planning. One study 17 employed a complete block, positioned parallel to the target, which effectively protected organs at risk but increased treatment time by approximately 22 min, thus limiting its clinical applicability. The other study 12 utilized a complete block with partial transparency. While this approach improved dose coverage for the internal mammary region by setting a small area of the block near the inner breast target to exit-only mode, it involved a complex hole-punching process, extended treatment time, and resulted in a significant increase in cardiac dose. The OABD Block's arc shape in our study is based on three hypotheses: (1) A 9 cm target expansion controls radiation angles, preventing under-coverage from proximity and angle restriction loss from excessive distance. (2) A 5 mm ring intersects at-risk organs, optimizing beam angle limitation without affecting dose constraints for lower-priority organs. (3) A 1 cm setback from the body contour enhances tangential beam incidence, particularly for deep targets like internal mammary lymph nodes, improving target dose coverage. This design, integrated into TOMO Helical plan optimization, restricts beam angles, maximizes TOMO's modulation, and minimizes critical organ exposure.
This study also has some limitations. The TOMO plan's delivery time increased by 40% with the OABD Block, potentially reducing machine efficiency and extending patient treatment duration. This highlights the need for improved patient immobilization in TOMO plans with virtual blocks and careful consideration of the patient's physical condition when deciding to use the OABD Block. Further research is needed to explore how to reduce TOMO delivery time while maintaining the dosimetric advantages of using Block.
Furthermore, the deep-inspirational breath-hold (DIBH) technique, 18 which is commonly used in radiotherapy for left-sided breast cancer and can reduce the dose to the heart and lungs, was not included in the study framework. A study 19 found that using DIBH with Helical Tomotherapy for left-sided breast cancer reduced mean heart dose from 5.76 Gy to 3.8 Gy and left lung V5 from 59.3% to 56.7%, while left lung V20 slightly increased from 24.8% to 25%. If the OABD Block were combined with the DIBH technique, there is potential to further enhance the effectiveness of TOMO plans. However, the combination of these two techniques would also likely extend treatment time, which warrants further exploration in subsequent studies.
Conclusion
In patients with left-sided breast cancer who have undergone radical surgery, employing the Organ and Arc-Based Directional Block method during the formulation of helical tomotherapy treatment plans enables the preservation of the stipulated doses to the internal mammary lymph node target regions, concurrently leading to a marked reduction in the irradiated dose to organs at risk.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251363288 - Supplemental material for Optimizing Helical Tomotherapy for Left-Sided Breast Cancer: A Retrospective Dosimetric Study of a Novel Virtual Organ-Arc Block
Supplemental material, sj-docx-1-tct-10.1177_15330338251363288 for Optimizing Helical Tomotherapy for Left-Sided Breast Cancer: A Retrospective Dosimetric Study of a Novel Virtual Organ-Arc Block by Yingtao Fang, Wenliang Yu, Jian Qiao, Yanju Yang, Jing Mi, Lei Yu, Ying Guo, Jiazhou Wang and Weigang Hu in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
The authors have no individuals or institutions to acknowledge.
Ethical Approval and Informed Consent Statements
The ethical approval for this retrospective study is covered under institutional approval number 1612167-18, which pertains to our institution's informed consent form in place since 2017. Under this form, all patients provided explicit consent for the use of their treatment-related data, including clinical history, for scientific research, with strict assurance of confidentiality. The present study involved no interventions or active patient participation; it solely involved the reanalysis of existing clinical data, such as previously acquired CT scans and contouring and the redesign of radiotherapy plans to compare outcomes across different accelerator platforms. As no new data were collected and all patients had previously consented to the use of their data for research, this study is fully compliant with institutional and ethical standards governing retrospective research.
Author Contributions
Yingtao Fang, Wenliang Yu, and Jian Qiao contributed equally to this work. Study conception and design: Yingtao Fang, Wenliang Yu, Jian Qiao, Weigang Hu. Data collection: Wenliang Yu, Yanju Yang, Jing Mi. Analysis and interpretation of results: Yingtao Fang, Wenliang Yu, Lei Yu. Draft manuscript preparation: Yingtao Fang, Wenliang Yu, Jian Qiao. Critical review of manuscript: Jian Qiao, Jiazhou Wang, Weigang Hu. All authors reviewed the results and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting this study are not publicly available.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.