Abstract
Keywords
Introduction
Ductal carcinoma in situ (DCIS) is a commonly diagnosed disease worldwide, accounting for an estimated 48 100 cases in 2019 in the United States. 1 The standard therapeutic regimen entails breast-conserving surgery (BCS), followed by external beam radiotherapy (RT). Adjuvant RT halves the risk of local recurrence of DCIS and the resurgence of breast cancer in the treated breast. 2
Recent data have shown that patients with DCIS face a risk of 3-fold higher mortality after breast cancer surgery. 3 Furthermore, the standardized mortality risk was lower in patients who underwent lumpectomy and RT for DCIS than in those who underwent surgery alone. 3 In another retrospective study, 138 DCIS patients with BCS without adjuvant RT were followed up for 9 years in mean. Recurrence rate was 19% with an annualized recurrence rate of 2%. 4
A recent study investigated whether all patients diagnosed with DCIS may require adjuvant treatment. 5 Among all patients (n = 1286) with a median age of 60 years, who did not undergo loco-regional therapy, the 10-year cumulative incidences of ipsilateral invasive cancer and all-cause mortality were 11% and 24.%, respectively. 5 Li et al showed that women who had previously received RT for DCIS had higher mortality rates after developing invasive second breast cancer than those who did not receive RT. 6 Chien et al investigated the value of mastectomy compared to BCS with adjuvant RT in young women with DCIS and reported a lower incidence of DCIS and invasive breast cancer in the mastectomy group. 7 Xia et al analyzed 5432 patients with DCIS with microinvasion from the Surveillance, Epidemiology and End Results (SEER) database and found a significant increase in the overall survival (OS) and breast cancer-specific survival after BCS and adjuvant RT compared to mastectomy. 8 Furthermore, a boost application (16 Gy in 8 fractions) of RT to the DCIS tumor bed in addition to postoperative whole-breast irradiation (50.4 Gy, normo-fractionated in 25 fractions or 42.5 Gy, hypo-fractionated in 16 fractions) significantly improved local control rates. 9
An analysis of secondary cancer risk after BCS reported that the cumulative lung cancer risk was 8.2% at 25 years after RT versus 4.3% without RT. 10 Interestingly, recent studies on cardiac morbidity after postoperative RT for DCIS have not shown an increased risk of ischemic heart disease,11,12 in contrast to many other studies on breast cancer RT.13,14
Currently, 3-dimensional conformal radiotherapy (3D-CRT) is the standard irradiation technique after BCS. In some cases, standard 3D-CRT resulted in a presumably unfavorable dose distribution with regard to the target volume or to organ-at-risk (OAR). 15 Common reasons are complicated anatomical situations, such as unfavorable positioning of the heart or parts of the lungs, or if the chest wall had a complex shape, for example, funnel chest/pectus excavatum. A tall heart or a large breast gland could also be a reason.
Further developments in RT techniques include intensity-modulated RT (IMRT) in combination with image-guided RT. Step-and-shoot IMRT employs 5 to 11 intensity-modulated beams in different directions at discrete angles. IMRT allows adequate target-dose coverage with simultaneous reduction in the mean dose exposure to the OARs. 16
Newer technical advances in IMRT include rotational IMRT techniques, such as volumetric intensity-modulated arc therapy (VMAT) and helical tomotherapy (hT).17,18 However, IMRT increases the integral low-dose exposure to the OARs. In the case of adjuvant RT of the breast after BCS, the contralateral breast, lungs, and heart are the focus of dose exposure reduction. To address the special anatomic milieu in adjuvant RT of the breast, special technical adaptations help reduce dose exposure in OAR. More tangential beam directions led to the advent of tangential beam IMRT and VMAT.19,20
hT is a form of percutaneous photon beam combined with IMRT. Radiation is delivered using a rotating fan-beam, and the photon beam forms a helix when the patient moves through the gantry bore. 21 The beam is modulated by a pneumatically driven, binary multileaf collimator (MLC). The treatment plan quality of hT is superior to other forms of IMRT, since it achieves favorable OAR sparing in most cases, 22 in addition to improved target-dose coverage and dose homogeneity. 23
TomoDirect is a special form of treatment with a tomotherapy accelerator, which is similar to 3D-CRT using 2 to 4 intensity-modulated, tangential fixed beams. Similar to hT, the table moves slowly through the gantry during RT but without movement of the linac. TomoDirect is in use regularly in breast RT, particularly when the patient's anatomy is not complex. TomoDirect yields extremely low exposure of middle and low doses to the whole-body volume compared to other IMRT techniques, similar to 3D-CRT.24,25 Takano et al reported in his RT-plan comparison of left-sided breast cancer following values for V5 Gy (%) of the ipsilateral lung, the contralateral lung, and the contralateral breast: 53%, 0.4%, and 2.9% (TomoDirect) versus 55%, 0.3%, and 5.6% (3D-CRT) versus 99%, 91%, and 51% (hT). 25
Generally, IMRT techniques result in low scattered doses to a larger part of the whole-body volume, for example, to the ipsilateral and contralateral lungs, compared to traditional 3D-CRT. hT achieves the steepest dose gradient (conformity index [CI] approximating 1), has excellent dose homogeneity within the target volume, and has the lowest maximum dose values compared to other IMRT and rotational techniques. In addition, the middle-dose exposure for normal tissue is lower in hT.23,26 Among the different IMRT techniques, hT shows relatively low scattered doses in the vertical direction compared to other IMRT techniques. For larger planning target volume (PTV), hT showed slightly increased horizontal peripheral scattered doses (low dose). 27 In breast cancer patients with a funnel chest, significantly better uniformity and conformity combined with a significant reduction of high-dose exposure to the OARs can be reached using hT in comparison with 3D-CRT. 15
Therefore, this study aimed to investigate the long-term outcomes and adverse events (AEs) in a homogenous, single-center cohort of patients treated with hT, a modern irradiation technique, after BCS for DCIS. The objective was to use hT in cases where standard 3D-CRT resulted in presumably unfavorable dose distribution.
Methods
Patients’ Characteristics
This study enrolled 28 consecutive patients (median age, 58 years; range, 41-81 years) who were diagnosed with DCIS and treated with BCS and postoperative hT as the standard of care between 2011 and 2020. This retrospective study conformed to the STROBE guidelines. 28 DCIS was located in the left breast in 54% and in the right breast in 46% of patients. Re-excision due to insufficient resection margins was performed in 39% of all patients. Finally, the median resection margin was 3 mm. In 75% of cases, DCIS was positive for hormonal receptors. The mean time span between BCS and RT was 5 weeks.
Prior to hT, all patients were counseled about RT and its alternatives, such as mastectomy. Generally, adjuvant RT is indicated after BCS. In our institution, the contraindications for adjuvant RT are life expectancy less than 5 years and breast ablation. hT was indicated when 3D-CRT was insufficient to cover the PTV or when the radiation doses delivered to the OARs were not within the tolerable range (eg, complicated anatomical situations, such as unfavorable positioning of the heart or parts of the lungs, or if the chest wall had a complex shape). The patients’ characteristics are presented in Table 1.
Patients’ Characteristics.

Abbreviation: BCS, breast-conserving surgery.
Radiation Treatment
The PTV of the treated breast was defined according to the institutional standards and included the entire mammary gland and chest wall behind it with safe lateral, cranial, and caudal margins of 2 cm and a safe medial margin of 1 cm. The ipsilateral and contralateral lungs, the entire heart, the left ventricle, and the contralateral breast were delineated on the computed tomography (CT) images as OAR.
hT was performed when standard 3D-CRT didn’t appear promising and resulted in a presumably unfavorable dose distribution with regard to the target volume or to OAR.
The prescribed radiation total (fraction) dose was 50.4 Gy (5 × 1.8 Gy/week) on the PTV of the whole breast in all cases. The actual median dose delivered to the PTV was 50.2 Gy (range, 49.9-50.5 Gy). None of the patients received hypofractionated RT. Three patients received additional boost RT (16 Gy, 5 × 2 Gy/week) to the initial tumor bed since the resection margins were less than 1 mm (Table 1).
Treatment was performed using the TomoTherapy Hi-Art System (Accuray, Sunnyvale, CA, USA). This helical IMRT system is equipped with a 6-MV photon beam. A beam field width of 2.5 or 5 cm was used for all plans, and calculations were performed using a fine dose grid. Radiation was delivered using a rotating fan-beam and the photon beam forms a helix as the patient moves through the gantry bore. 21 The beam is modulated by a pneumatically driven, binary MLC. During inverse treatment planning in the tomotherapy planning system, MLC conformation is optimized to obtain highly conformal radiation doses at the target. 29 The applied dose-volume constraints for the OARs used in the hT planning system were adapted from the National Radiation Gynecology (NRG) Protocol Radiation Therapy Template (whole-breast photon and proton therapy) for normo-fractionated RT (cumulative dose, 50.4 Gy; single dose, 1.8 Gy). Generally, in the first instance, dose coverage of the PTV was optimized by increasing importance of PTV and penalties of maximal and minimal PTV doses step-by-step, until aimed dose coverage was reached (95%-107% of the described dose of 50.4 Gy). That followed, penalties of OAR were risen successively as far as possible to obtain sought doses without collapsing dose coverage of the PTV. Further details were as follows: ≤15% of the ipsilateral lung should receive ≥18 Gy, ≤20% of the ipsilateral lung should receive ≥10 Gy, ≤30% of the ipsilateral lung should receive ≥5 Gy, and ≤15% of the contralateral lung should receive ≥5 Gy. For left-sided breast cancers, ≤10% of the whole heart should receive ≥19 Gy and the mean heart dose should be ≤5 Gy.30,31 At the beginning of the calculation, the initial dose constraints for OAR in the planning system were as follows: ipsilateral lung: maximum dose, 49 Gy and 20%/10 Gy; contralateral lung: maximum dose, 40 Gy and 10%/5 Gy; heart: maximum dose, 40 Gy and 10%/10 Gy; and contralateral breast: maximum dose 25 Gy and 15%/5 Gy. See Figure 1 (initial constraints for RT of right breast) and Figure 2 (initial constraints for RT of left breast).

Initial dose-volume constraints for PTV (right-sided breast gland)/OAR and dose objectives are presented in the treatment planning system of helical tomotherapy at the beginning of optimization.

Initial dose-volume constraints for PTV (left-sided breast gland)/OAR and dose objectives are presented in the treatment planning system of helical tomotherapy at the beginning of optimization.
To objectify the advantage of hT, a retrospective plan comparison of 3D-CRT with hT was carried out in 10 representative cases of the cohort (right and left breast each n = 5). 3D-CRT plans were created with the Eclipse planning system within the Varian ARIA oncology information system (Varian Medical Systems, Inc., Palo Alto, CA, US). Therefore, 2 tangential 6-mega-electron-volt (MeV) photon beams of a linear accelerator (Varian Medical System) and dynamic wedge filter were used. Please see Figure 3 (right breast) and Figure 4 (left breast).

Dose distribution in a transversal CT scan of female patient with right-sided DCIS performed with 2 different radiation techniques: 3D-CRT and hT. Colored isodoses are shown relative (%) to the described dose (50.4 Gy) for planning target volume (red line). The maximal dose point is marked in red.
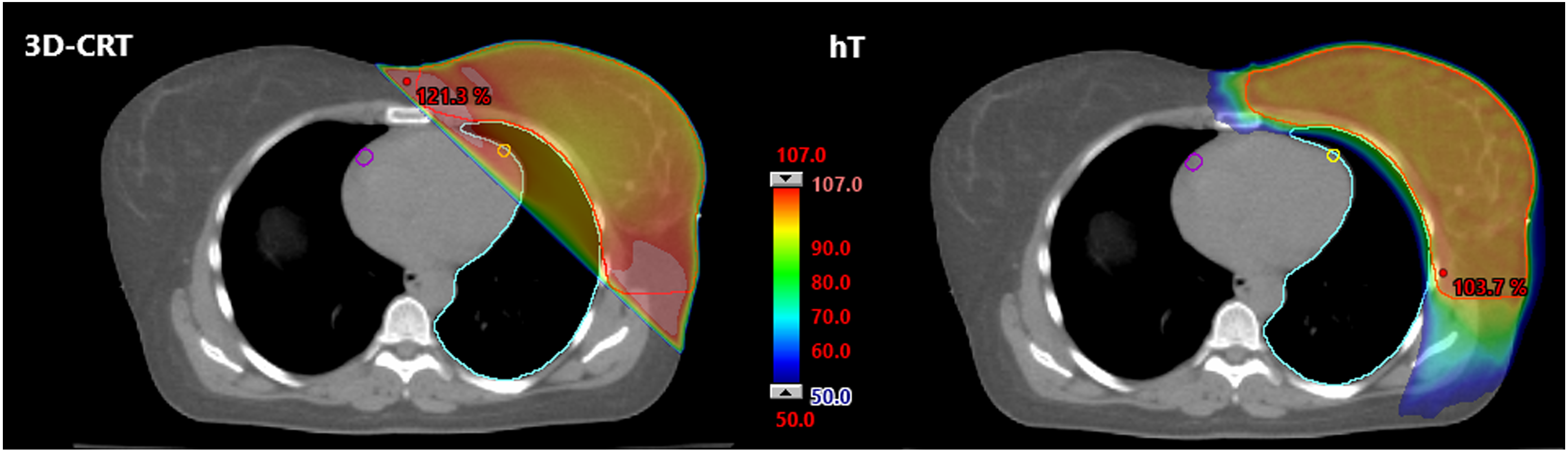
Dose distribution in a transversal CT scan of female patient with left-sided DCIS performed with 2 different radiation techniques: 3D-CRT and hT. Colored isodoses are shown relative (%) to the described dose (50.4 Gy) for planning target volume (red line). The maximal dose point is marked in red.
With regard to the plan comparison (3D vs hT), left coronary (left main coronary artery and left anterior descending coronary artery) and right coronary were contoured additionally and analyzed retrospectively. Evaluation included doses of the lung, the heart, and the ventricle. Maximal dose in PTV (PTV Dmax), minimal dose in PTV (PTV Dmin), and the PTV that achieved 95% respectively 107% of the described dose or more (PTV V95% respectively PTV V107%) were determined.
Treatment Plan Evaluation
The dose-volume-histograms of the treatment plans were reviewed to extract the heart dose (mean dose, maximal dose, volume that received 25 Gy and more [V25 Gy]), the left ventricle dose (mean and maximal dose), the ipsilateral and contralateral lung doses (mean dose, V5 Gy, V20 Gy), the contralateral breast dose (mean dose and dose-covering 5% of the volume [D5%]), and the PTV doses (dose covering 98% of the volume [D98%], dose covering 2% of the volume [D2%], and dose covering 50% of the volume [D50%]). Furthermore, we calculated the homogeneity index (HI) and CI to evaluate the plan quality. The CI is an objective measure of how well the distribution of the radiation dose conforms to the PTV. The CI was calculated using the following formula:

Statistical Design and Classifications
The primary endpoint of this retrospective analysis was the local recurrence-free survival (LRFS) and the secondary endpoints were OS and the incidence of secondary cancers. The time estimates for OS and follow-up pertain to the initial diagnosis and the end of RT, respectively. LRFS was defined as the absence of local tumor recurrence. Secondary cancer-free survival (SCFS) was defined as the lack of secondary cancer after RT. Statistical analyses were conducted using Microsoft Excel and SPSS 16 (IBM). The statistical approach included event analyses of local recurrence, death, and secondary cancers with respect to the time frame. Kaplan–Meier analysis was not performed because of the absence of events. AEs were classified according to the Common Toxicity Criteria for Adverse Events (CTCAE), version 5. The first clinical follow-up examination by the radiation oncologist was performed 6 to 8 weeks after the completion of RT and on a regular basis thereafter, including regular mammography and/or breast ultrasonography. Acute and late AEs were analyzed by determining the absolute and relative mean values and the ranges. We confirm that fellow researchers can reproduce our methodology based on the descriptions provided in this section.
Ethics Approval and Consent to Participate
The relevant ethics committee (Landesärztekammer Baden-Württemberg, Stuttgart, Germany; AZ: F-2021-082) approved the study and waived the need for informed consent for participation, since this study entailed an anonymized retrospective analysis. There were minimal risks to the patients in the non-interventional analysis. This study was conducted in accordance with the principles of the Declaration of Helsinki. All patient details have been de-identified.
Results
Plan Comparison (3D-CRT vs hT)
Relating to PTV parameters (Dmax, Dmin, V95%, V107%), hT indicated significant better results than 3D-CRT. The middle- and high-dose exposure of the ipsilateral lung was very much lower in hT. In case of the left breast gland RT, hT also showed distinct dose reduction in the volume of the heart, the ventricle, and the left coronary. In treatment of the right breast gland, hT demonstrated significant lower-dose exposure of the right coronary. On the other hand, increased low-dose exposure of the contralateral lung and breast gland opposed to these clinical relevant advantages of hT. Data values and significances are shown in Table 2 (DCIS right breast) and Table 3 (DCIS left breast).
Data of Plan Comparison of Patients With Right-Sided DCIS: 3D-CRT Versus hT.

Presumed clinical relevant differences were marked green (advantageous) or red.
Abbreviations: DCIS, ductal carcinoma in situ; 3D-CRT, three-dimensional conformal radiotherapy; hT, helical tomotherapy; PTV, planning target volume; OARs, organs-at-risk; Dmax, maximal dose; Dmin, minimal dose; Vx%, PTV that achieved x% of the described dose (50.4 Gy) or more; StDev, standard deviation.
Significant difference: P < .05.
Data of Plan Comparison of Patients With Left-Sided DCIS: 3D-CRT Versus hT.

Presumed clinical relevant differences were marked green (advantageous) or red.
Abbreviations: DCIS, ductal carcinoma in situ; 3D-CRT, three-dimensional conformal radiotherapy; hT, helical tomotherapy; PTV, planning target volume; OARs, organs-at-risk; Dmax, maximal dose; Dmin, minimal dose; Vx%, PTV that achieved x% of the described dose (50.4 Gy) or more; StDev, standard deviation.
Significant difference: P < .05.
Patients’ Characteristics
The median age of the female patients was 58 years at the time of radiation therapy. DCIS was located in the right breast in 46% of patients and in the left breast in 54% of patients. Hormonal receptors were expressed in 75% of patients. Re-excision was performed in 39% of all patients, leading to a final median resection margin of 3 mm (Table 1).
Treatment Plan Parameters
The analysis of the plan data showed high conformity and homogeneity of dose application in hT. The CI value approximated 1 with a mean value of 0.9 (range, 0.86-0.96). Correspondingly, the mean HI value was close to 0 (0.056), with a range of 0.050 to 0.064. The mean doses to the OARs were as follows: contralateral and ipsilateral lung: 10.9 and 3.9 Gy, respectively; and heart: 5.8 Gy (range, 0.7-8.5 Gy). Additional results on the radiation doses to the OARs and the plan parameters are shown in Table 4 and Figure 5.

Box and whisker plots of organ at risk doses for all 28 patients treated with helical tomotherapy (hT). Y-axis: numerical quantity value depending on value of different box and whisker plots (Gy or volume-%). Box and whisker plot explanation: lower whisker (1.5-fold of the interquartile range)—box (lower quartile–median–upper quartile)—upper whisker (1.5-fold of the interquartile range), ° = outlier, * = extreme value. Y-axis from left to right: (1) Mean heart dose (Gy), (2) maximal left ventricle dose (Gy), (3) mean left ventricle dose (Gy), (4) heart V25%, (5) mean ipsilateral lung dose (Gy), (6) V20% ipsilateral lung, (7) V5% ipsilateral lung, (8) mean contralateral lung dose (Gy), (9) V20% contralateral lung, (10) V5% contralateral lung, (11) mean contralateral breast dose (Gy).
Plan Parameters of the Dose-Volume-Histograms of all Patients.

Abbreviations: VXGy, volume that receives X Gy or more; D0.03cc, an approximate maximum dose for organ at risk.
Survival and Tumor Control
At the time of data analysis, all patients (n = 28) were alive and none was diagnosed with in-breast recurrence or ipsilateral breast cancer. The median follow-up duration was 61 months (range, 3-123 months). The 1-, 3-, and 5-year OS rates were 100% each (Figure 6). Furthermore, the 1-, 3-, and 5-year LRFS rates were 100% each. Up to 10 years, no secondary tumor occurred in the cohort. SCFS was 100%.

Kaplan–Meier curve for OS regarding to all DCIS-patients (n = 28).
Acute and Late AEs
No relevant interruptions in treatment were documented. The most common acute AE was mild dermatitis at the ipsilateral breast. CTCAE grade 1 to 2 dermatitis occurred in 26 of 28 patients and CTCAE grade 3 dermatitis in 1 of 28 patients. Four patients experienced fatigue, 4 developed thrombopenia, and hyperpigmentation occurred at the ipsilateral breast in 3 patients (Table 5). The most common late AEs were induration at the surgical scar (n = 7), hyperpigmentation (n = 5), and dry skin (n = 2) (Table 6). One 79-year-old patient with left-sided DCIS was diagnosed with coronary artery disease (CAD)/myocardial infarction (grade 3/severe AE) and underwent stenting 1 year after RT. The patient recovered and she is still alive. The initial treatment planning CT scan revealed arteriosclerosis of the left cardiac arteries. The applied mean heart dose was 8.1 Gy. None of the patients experienced acute or late AEs > grade 3. Other acute and late AEs as well as more details are provided in Tables 5 and 6.
Acute Adverse Events in 28 Patients Treated With Breast Conserving Surgery and Postoperative Helical Tomotherapy.

Abbreviation: CTCAE, Common Toxicity Criteria for Adverse Events.
Late Adverse Events in 28 Patients Treated With Breast Conserving Surgery and Postoperative Helical Tomotherapy.

Abbreviation: CTCAE, Common Toxicity Criteria for Adverse Events.
Discussion
Short Resume
We retrospectively assessed the long-term treatment outcomes and AEs in a cohort of 28 patients with DCIS who were treated uniformly with hT between 2011 and 2020 owing to presumably unfavorable dose distribution with standard 3D-CRT. The reason for this purpose was a complex shape of the chest wall (eg, funnel chest/pectus excavatum), a large breast gland, or a tall heart respectively adverse position of the heart. With regard to oncological control and secondary tumor occurrence, hT shows excellent results in the investigated cohort without any event. At once RT with hT was compatible well with mainly minor side effects. All patients with DCIS treated with hT were included in the analysis and no patient was excluded. Furthermore, our patients represent a real-life cohort in routine clinical practice.
Considerations for Radiation Technique and Dosimetry
Advantages in dose distribution of hT in comparison with the 3D-CRT are shown in Figures 1 and 2. In case of pectus excavatum, the advantage of hT was shown by Uhl et al within a dosimetry planning study. 15 Within this study, the uniformity and conformity of the target dose distribution and the median/average dose of the PTV were inferior in 3D-CRT compared to hT. By using hT, the volume of the lungs, the contralateral breast, and the heart, which received high-dose exposure, could be significantly reduced. hT led to a significantly higher low-dose exposure to the lungs and contralateral breast. 15 Evaluation of the plan comparison inside this cohort showed corresponding results (Tables 2 and 3).
Recently Palumbo et al have published a planning study, where 3D-CRT was compared to hT in 20 left-sided breast cancer patients with common standard anatomy. 32 hT provided the best target coverage and significantly reduced high and mean dose to the left anterior descending artery, the ventricle, and to the heart. As expected, due to the rotational delivery, the low dose to the right coronary arteries, the contralateral breast, and the lungs was higher with hT. 32 Authors mentioned that hT could have advantages in RT of patients with left-sided breast cancer and heart diseases. They noted that further studies are needed to correlate dosimetric findings with in-depth cardiac monitoring. 32
In principle, increased target coverage or reduced high and mean dose to heart, ipsilateral lung, or coronary artery in case of difficult anatomic situations or patients with heart disease should be also archived by alternative radiation techniques with rotational dose delivery, for example, VMAT.
Local Control and Survival
Several studies have reported the benefits of postoperative RT for DCIS. Wapnir et al reported on 2 trials of patients treated with lumpectomy ± RT and lumpectomy + RT ± tamoxifen and showed that postoperative RT reduced the ipsilateral in-breast tumor recurrence risk by 52% and the long-term prognosis remained excellent in these cohorts. 33 McCormick et al recently conducted a trial on good-risk DCIS treated with lumpectomy and postoperative RT and reported a significant reduction in all (7.1% vs 15.1%) and invasive (5.4% vs 9.5%) in-breast recurrences at 15 years. 2 Narod et al investigated breast cancer mortality after DCIS in 108 196 patients with DCIS registered in the SEER database and found that the risk of death increased after recurrent breast cancer; however, the prevention of invasive breast cancer recurrence by RT did not significantly reduce breast cancer mortality after 10 years (hazard ratio 0.8% vs 0.9%). 34 In our cohort, no recurrences were documented during follow-up, resulting in an excellent LRFS rate after surgery and postoperative hT (Figure 2). The distinct dose distribution and conformity of the hT technique compared to other radiation techniques (documented by high-level HI and CI values) averted underdoses in the PTV and could have been responsible for the absence of recurrence events in the current study. Events of death, secondary breast cancer, or death due to secondary breast cancer was not observed in this study.
Side Effects
In RTOG 9804, McCormick et al performed whole breast RT by administering 42.5 to 50.4 Gy in 16 to 28 fractions and reported grade 1 to 2 AEs in 76% of patients as well as ≥grade 3 acute AEs in 4% of patients. The late AEs were grade 1 in 30%, grade 2 in 4.6%, and grade 3 in 0.7% of patients. 2 Joseph et al conducted a phase III trial in patients with early breast cancer using hT or forward field-in-field (FiF) with IMRT. Patients treated with hT had significantly fewer AEs compared to those treated with FiF IMRT. Acute dermatitis of CTCAE grade 0 to 1 occurred in 63% of patients undergoing hT and grade 2 to 4 in 37%. 35 Acute erythema was reported as faint in 61%, bright in 34%, and acute moist desquamation in 11% of patients. The late AEs were skin induration/fibrosis in 67% and hyperpigmentation in 63% of cases. 35 Favorable rates of acute and late AE related to the skin and breast were documented in our cohort. None of the patients experienced acute or late AEs > grade 3 (Tables 3 and 4). The dose distribution in hT prevents dose values >107% of the described doses in the PTV. 26 This factor seems to be important in preventing severe acute or late toxicity events in the treated breast tissue.
Vasiljevic et al performed a prospective study on pneumonitis after RT for breast cancer and reported a pneumonitis incidence of 13%, for which smoking history and chronic obstructive pulmonary disease served as positive predictors. 36 A prospective cohort study by Karlsen et al reported that the rates of radiological and clinical radiation pneumonitis after adjuvant tangential 3D-CRT were 78% and 19%, respectively. 37 In our analysis, 1 patient each (4%) developed localized acute pneumonitis (CTCAE grade 2) and localized late pulmonary fibrosis (CTCAE grade 1). Generally, dose distribution in hT prevents high values of maximum doses in the PTV and the surrounding areas with reduced middle-dose exposure.13,14 This could be the principal factor that averts severe acute or late toxicity events. In the present study, none of the patients experienced acute (or late) AEs > grade 3 (Tables 5 and 6).
Skyttä et al performed a prospective trial on cardiac changes and morbidities during a 3-year period after postoperative breast RT for DCIS. They reported negative subclinical changes in cardiac biomarkers as well as left ventricular function, which were more distinct in left-sided breast cancer. 38 In contrast, Haque et al did not report any increase in the cardiac mortality risk after left-sided RT using modern RT techniques and maintaining low cardiac doses. 12 However, in their cohort, the cardiac mortality risk was higher in patients who received RT before 1983. 12 Wadsten et al investigated patients registered in the Breast Cancer Data Base Sweden between 1992 and 2012 (median follow-up: 8.8 years) and found that the risk for ischemic heart disease did not increase after surgery with postoperative RT versus surgery without RT (hazard ratio 0.77). 11 However, they mentioned that the effect might be biased by patient selection, but the laterality of RT was not associated with an elevated risk (hazard ratio: 0.85). 11 Takx et al investigated breast cancer survivors and quantified coronary artery calcium on CT imaging. The median time between RT and CT was 2 years and no differences were found between irradiated patients and the control group. 39 They concluded that their results do not provide evidence of radiation-induced accelerated CAD in patients with breast cancer. 39 Therefore, we assumed that the diagnosis of CAD on planning CT and subsequent stent placement after 1 year in one of our patients was unlikely a result of radiation treatment.
Second Primary Tumor Risk
Pignol et al analyzed the second primary tumor risk after BCS and discovered that the lung cancer risk was 8.2% 25 years after RT versus 4.3% without RT. 10 They concluded that a reduction in the mean lung dose in RT prevented secondary lung cancer. 10 Another study on second primary cancers after RT for DCIS reported an estimated cumulative excessive risk of 0.8% by 15 years after the diagnosis of DCIS. 40 In our trial, none of the patients was diagnosed with secondary cancer during follow-up, corresponding to a SCFS rate of 100%. This finding also reinforces evidence that IMRT techniques with their higher low-dose exposure to normal tissue volumes do not increase the second primary cancer risk compared to the classic 3D-CRT.
Limitations
The limitations of this study include its retrospective design and relatively small sample size. Power calculation was not done for estimation of sample size selected for the study, because it was a retrospective analysis. Furthermore, a multivariate analysis of the prognostic factors was not possible due to the lack of events pertaining to OS, LRFS, and secondary cancers. However, the homogeneity of our patient cohort represents real-life outcomes in routine practice and provides rare data on the long-term outcomes of DCIS exclusively treated with hT as adjuvant RT.
Conclusion
hT is feasible and safe in the long term, after BCS for patients with DCIS, which exhibit a complex shape of the chest, a large breast gland, or a tall heart respectively adverse position of the heart. In this situation, hT has advantages in dose distribution in PTV and OARs compared to 3D-CRT. In the investigated collective, there were no recurrences, invasive breast cancer diagnoses, or secondary cancers during follow-up. Main AEs were acute mild dermatitis, fatigue, thrombopenia, late induration at the surgical scar, and skin hyperpigmentation. The results of this study contribute to knowledge about adjuvant RT for DCIS. The findings of the current study indicate that a wider implementation of hT as adjuvant RT after BCS can be beneficial to patients with DCIS with a complex thoracic anatomy.
Footnotes
Abbreviations
Author Contributions
F.Z. initiated and supervised the project. F.Z. collected data. H.H., M.S., P.L., and S.H. carried out the data analysis. F.Z., P.E.H., J.D., and H.H. interpreted the data. H.H. and F.Z. wrote the manuscript. H.H., P.E.H., J.D., and F.Z. were involved in article revision.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jürgen Debus reports grants from Viewray Inc, CRI The Clinical Research Institute GmbH, Accuracy International Sarl, RaySearch Laboratories AB, Vision RT Limited, Merck Serono GmbH, Astellas Pharma GmbH, Astra Zeneca GmbH, Siemens Healthcare GmbH, Solution Akademie GmbH, Ergomed PLC Surrey Research Park, Quintiles GmbH, Pharmaceutical Research Associates GmbH, Boehringer Ingelheim Pharma GmbH&CoKG, PTW Freiburg Dr Pychlau GmbH, and Nanobiotix S.A., outside the submitted work. All other authors declare that they have no competing interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
The accountable ethics committee approved the analysis on July 26, 2021 (Medical Board Baden-Wuerttemberg, Stuttgart, Germany, protocol number AZ: F-2021-082). In Germany, for non-university medical centers, the ethics committee is at the Medical Board of the according federated state, in this case in Stuttgart. The ethics committee at the Medical Board waved the need for an informed consent for inclusion in the anonymized retrospective analysis. There were no more than minimal risks for the patients in the non-interventional analysis. This analysis was conducted in accordance with the Declaration of Helsinki.
