Abstract
Objective
To investigate the effect of various frequencies of bolus use on the superficial dose of volumetric modulated arc therapy after modified radical mastectomy for breast cancer.
Methods
Based on the computed tomography images of a female anthropomorphic breast phantom, a 0.5 cm silicone-based 3D-printed bolus was created. Nine points evenly distributed on the breast skin were selected for assessing the skin dose, and a volume of subcutaneous lymphatic drainage of the breast (noted as ROI2-3) was delineated for assessing the chest wall dose. The treatment plans with and without bolus (plan_wb and plan_nb) were separately designed using the prescription of 50 Gy in 25 fractions following the standard dose constraints of the adjacent organ at risk. To characterize the accuracy of treatment planning system (TPS) dose calculations, the doses of the nine points were measured five times by thermoluminescence dosimeters (TLDs) and then were compared with the TPS calculated dose.
Results
Compared with Plan_nb (144.46 ± 10.32 cGy), the breast skin dose for plan_wb (208.75 ± 4.55 cGy) was significantly increased (t = −18.56, P < 0.001). The deviation of skin dose was smaller for Plan_wb, and the uniformity was significantly improved. The calculated value of TPS was in good agreement with the measured value of TLD, and the maximum deviation was within 5%. Skin and ROI2-3 doses were significantly increased with increasing frequencies of bolus applications. The mean dose of the breast skin and ROI2-3 for 15 and 23 times bolus applications were 45.33 Gy, 50.88 Gy and 50.36 Gy, 52.39 Gy, respectively.
Conclusion
3D printing bolus can improve the radiation dose and the accuracy of the planned dose. Setting Plan_wb to 15 times for T1-3N+ breast cancer patients and 23 times for T4N+ breast cancer patients can meet the clinical need. Quantitative analysis of the bolus application frequency for different tumor stages can provide a reference for clinical practice.
Keywords
Introduction
Tissue-equivalent bolus is usually used to increase the surface dose in patients with breast cancer undergoing post-mastectomy radiotherapy.1,2 The skin and chest wall dose are related to the frequency of bolus applications. Covering bolus throughout the entire radiotherapy planning process can lead to excessive surface dose and significant side effects. An appropriate bolus application frequency can keep the skin dose within the acceptable range while ensuring adequate chest wall dose to prevent a recurrence.
Traditional radical surgery will result in 5%-10% residual glandular tissue, and the abundant lymphatic vessel in subcutaneous tissue is an important pathway for tumor metastasis to the axilla or mammary lymph nodes. Therefore, subcutaneous lymphocytic plexus is an important target area for post-mastectomy chest wall radiotherapy.3,4 Apart from patients with T4b, T4c, and T4d breast cancer, skin is not a part of clinical target volume (CTV). The rates of immediate post-mastectomy reconstruction, primarily an implant/prosthesis-based reconstruction, as a surgical treatment for patients with early-stage breast cancer are rising.5,6 Skin sparing is crucial when it is not part of the CTV, in order to reduce treatment toxicity; nevertheless, skin is recognized as a complex organ. The skin's basal cell layer is located at a depth of roughly 70 µ and dermal vascularization is situated at a depth of 0.5-3 mm. Although skin is not generally part of the CTV, to reduce the possibility of a near-surface tumor recurrence, a radiotherapy chest wall treatment regimen should provide an adequate skin dose in selected patients.7,8 Due to the shape of the chest wall and the scar after breast cancer surgery, there is often an air gap between the conventional bolus and the chest wall, resulting in a deviation of the skin surface dose. 9 The 3D-printed bolus can reduce the air gap and improve dose uniformity in the target area, and the accuracy of the actual exposure dose. 10 However, even though some studies have investigated the thickness and materials of bolus for post-mastectomy radiotherapy,11–13 few studies have focused on the frequency of bolus use, and the optimal frequency remains unknown.
Thermoluminescent dosimeters (TLDs) are routinely used to measure the point dosimetry around brachytherapy sources. 14 It has many advantages such as small physical size, low energy dependence, good stability, high sensitivity, tissue equivalence, wide linear region, low cost, user-friendly, and simple annealing process. Therefore, TLDs can be applied to estimate radiation doses in radiotherapy for cancer and verify Monte Carlo (MC) simulation-based experiments. 15 It is a good choice for clinical dose distribution measurement in radiotherapy.
In this study, 3D printing technology was used to produce customized and highly conformal tissue-equivalent bolus and TLD was used to measure the breast skin dose. The impact of bolus application frequency on the dose to the breast skin and chest wall was analyzed, and the required frequency of bolus application for patients at different stages was quantified, providing a reference for clinical practice regarding the frequency of bolus use.
Material and Methods
Computed Tomography Simulation
The female anthropomorphic phantom (Alderson Rando, Chengdu, Sichuan) was positioned, and the metal markers were placed on the surface of the phantom based on laser crosslines while marking the upper, lower, left, and right boundaries of the bolus. A computed tomography (CT) simulator (Philips Brilliance CT Big Bore, Netherlands) was used for scanning, with a scanning range of from mid-neck to mid-abdomen, a tube voltage of 120 kV, a tube current of 350mAs, and a 0.3 cm hatch spacing and layer thickness. The above CT sets were used to create the virtual bolus in the treatment planning system (TPS) and to design the 3D-printed bolus.
Design and Production of 3D-printed Breast Bolus
After CT-guided localization, the images were imported into the Monaco TPS (Elekta, Inc. Sweden, version: 5.11.03). The CTV was delineated according to breast target volume delineation guidelines. 16 The ventral border, according to the ESTRO Guidelines, is 5 mm below the skin's surface, with the exception of T4b, T4c, and T4d breast cancer cases, when a bolus can be used to obtain a full radiation dose up to the skin. The planning target volume (PTV) was obtained by expanding the external margins of the CTV by 0.5 cm in the left-right and anterior-posterior and 1 cm in the cranial-caudal direction. A virtual bolus contour was defined based on the boundary of the positioning marker, with a relative electron density of 1.0 and a thickness of 0.5 cm. Export DICOM images for producing 3D-printed bolus. The structure set was imported into the Mimics17 software in the DICOM format and output in STL format for 3D printing. To create a more flexible and simulation-like bolus, a specific type of elastic printing material was chosen: thermoplastic polyurethane (TPU) filaments with a diameter of 1.75 mm, with a thickness of 5 mm, and a density of 1.10-1.14 g/cm3. However, the physical properties of TPU were not identical to water. Thus, the water-equivalent depth of TPU was simply calculated with the ratio of water and TPU physical densities. The bolus thicknesses of 0.5 cm for the TPU were reduced to 0.45 cm with 0.91 of the depth-scaling factors, respectively. Then, the 3D printer, which utilized fused deposition modeling technology, was used to print the bolus.
Treatment Planning
According to clinical practice standards, two plans were delivered with (plan_wb) or without bolus (plan_nb). The prescribed dose for the whole breast was 50 Gy in 25 fractions (2 Gy per fraction). All optimizations were performed using volumetric modulated arc therapy with a gantry angle range of 179° to 290°. A calculation grid size of 0.1 cm, a MC-based statistical uncertainty (per calculation mode) of 1%, a minimum subfield width of 0.5 cm, an increment of 20° for field segmentation, and a “low” fluence-smoothing level were set. The X-Ray Voxel Monte Carlo (XVMC) dose calculation algorithm was used. The treatment plans were created based on the frequency of bolus application, that is
Thermoluminescent Dosimeters Calibration
The skin dose of the breast was measured using lithium fluoride TLDs provided by China National Nuclear Corporation with a cross-section of 4.5 × 4.5 mm2 and thickness of 0.8 mm. The information of dose exposure was provided by a 2000B TLD annealing furnace and an RGO-3B TLD reader. This measurement system was calibrated annually by the Henan Institute of Metrology. TLD 100 chips were calibrated using a single-point calibration strategy as follows: with a source-to-surface distance of 100 cm, a field size of 10 cm × 10 cm, the TLD chips placed at a depth of 10 cm in a solid water phantom, and a dose of 100 MU (66.45 Gy) was delivered. This was repeated three times, and the average value was taken after excluding TLD chips with large dose deviations. Only those with stable doses were used to obtain the calibration factor for each TLD chip. The calibration factor was used to calculate the dose value of the measured TLD chip:
TLD Measurement of Breast Skin Dose
The anthropomorphic phantom was irradiated using the Elekta Axesse linear accelerator according to the treatment plan. The PTV center was aligned with the treatment isocenter based on the position of the Mark points. Nine measurement points were marked to represent different positions in the breast target volume. After CBCT scanning, calibrated TLD chips were placed at the corresponding marked points. To increase the accuracy and reproducibility of dose measurements, each plan was irradiated and measured five times. After completion of the irradiation, the TLD chips were collected, and the measurement and annealing were completed within 24 h. The mean value of breast skin TLD measurement was obtained according to the calibration factor and the formula (1).
TPS Calculation of Breast Skin Dose
The CBCT image acquired prior to treatment was registered with the planning CT in the planning system to determine the positions of the nine measurement points. The actual positions of the TLD measurements are shown in Figure 1. Points 3 and 5 were placed at the nipple and the field edge of the affected side, respectively, and were used as reference points for measurement. The other points were evenly distributed on the breast skin of the affected side. Treatment plans with different frequencies of bolus application were optimized using the TPS. Then, the dose values at the nine measurement points of interest were calculated based on the accumulated dose-volume histograms of each treatment plan.

Position of the nine measurement points on the thorax phantom.
TPS Calculation of Chest Wall Dose
Although the skin is not part of the CTV, except in patients with T4b, T4c and T4d breast cancer, the subcutaneous lymphatic plexus clearly is. The lymphatic plexus of the breast flows through the dermal plexus located within the subcutaneous tissue. In this study, the subcutaneous lymphatic drainage of the breast was used to evaluate the chest wall dose. The region of interest (noted as ROI2-3), which includes the subcutaneous tissue 2-3 mm below the skin surface, was delineated as the breast subcutaneous lymphatic drainage area, as shown in Figure 2. The dose distribution of ROI2-3 was statistically analyzed based on the accumulated dose-volume histograms with different frequencies of bolus application.
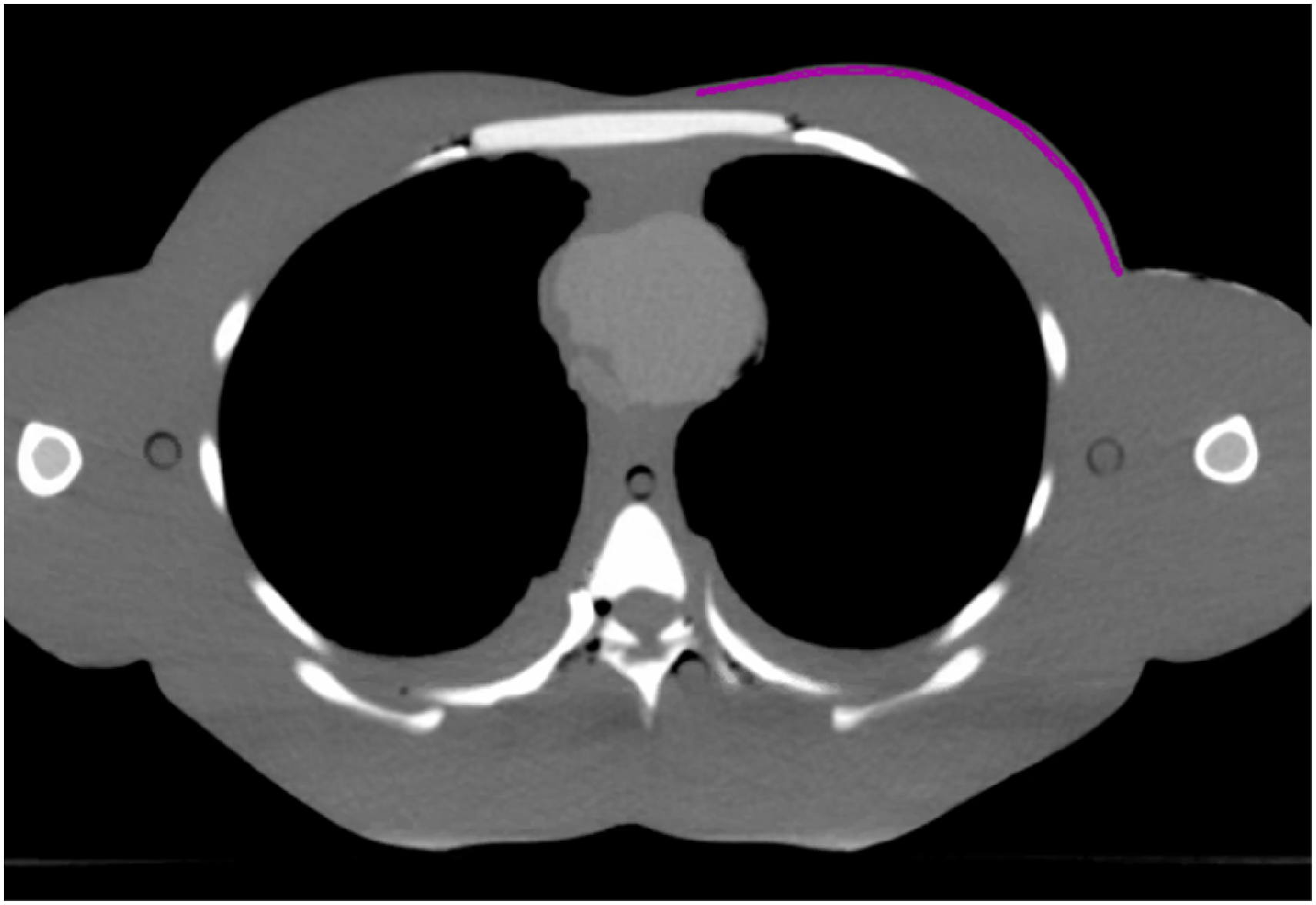
Breast subcutaneous lymphatic drainage area ROI2-3.
Statistical Analysis
All data were analyzed using Statistical Package for Social Sciences, version 16.0. P-values of 0.05 or less were considered statistically significant. Values are expressed as mean (range) ± standard deviation (SD).
The percentage difference (%diff) between the results of TPS calculations and TLD measurements was calculated as follows:
Results
Selection of TLD Chips
One hundred TLD chips were measured three times, and the standard deviation of each TLD chip measurement is shown in Figure 3. TLD chips with a dose standard deviation greater than 1 cGy were excluded, and 90 TLD chips with stable doses remained for skin dose measurements.

The standard deviation of the dose values of 100 TLD chips. TLD, thermoluminescence dosimeter
TLD Skin Dose Measurement
Plan_wb and plan_nb were performed five times, respectively, and the mean ± standard deviation of the TLD measurements of breast skin dose at different measurement points is shown in Figure 4. Compared with plan_nb, plan_wb showed a significant increase in the skin dose of the affected breast (t = −18.56, P < 0.001).

The mean ± standard deviation of the TLD measurements of breast skin dose at different measurement points. TLD, thermoluminescence dosimeter.
The Deviation Between TLD-measured Dose and TPS-calculated Dose
The three-view isodose distribution of the plan_nb and plan_wb in the plan system is shown in Figure 5. The result showed that the skin dose could be improved by bolus application. The relative deviation between the TPS-calculated values and TLD-measured values is shown in Table 1. The results showed that there was a significant deviation between the dose values of TPS and TLD at points 3 and 5, which were 3.18% and 4.44%, respectively. However, the TPS-calculated values and at other measurement points were in good agreement with the TLD-measured values, with a maximum deviation of less than 3%.

Three-view isodose distribution.
The Deviation Between TPS-calculated Values and TLD-measured Values.

TLD, thermoluminescence dosimeter; TPS, treatment planning system.
Breast Skin and ROI2-3 Dose Under the Different Frequencies of Bolus Applications
The changes in breast skin and ROI2-3 dose with treatment times of plan_wb are shown in Figure 6. As the number of plan_wb treatments increased, the breast skin and ROI2-3 dose also increased. When plan_wb was delivered 15 times of the total 25 fractions, the mean skin dose was 45.33 Gy, and the mean ROI2-3 dose was 50.88 Gy. When plan_wb was delivered 23 times of the total 25 fractions, the mean skin dose increased to 50.36 Gy, and the mean ROI2-3 dose increased to 52.39 Gy.
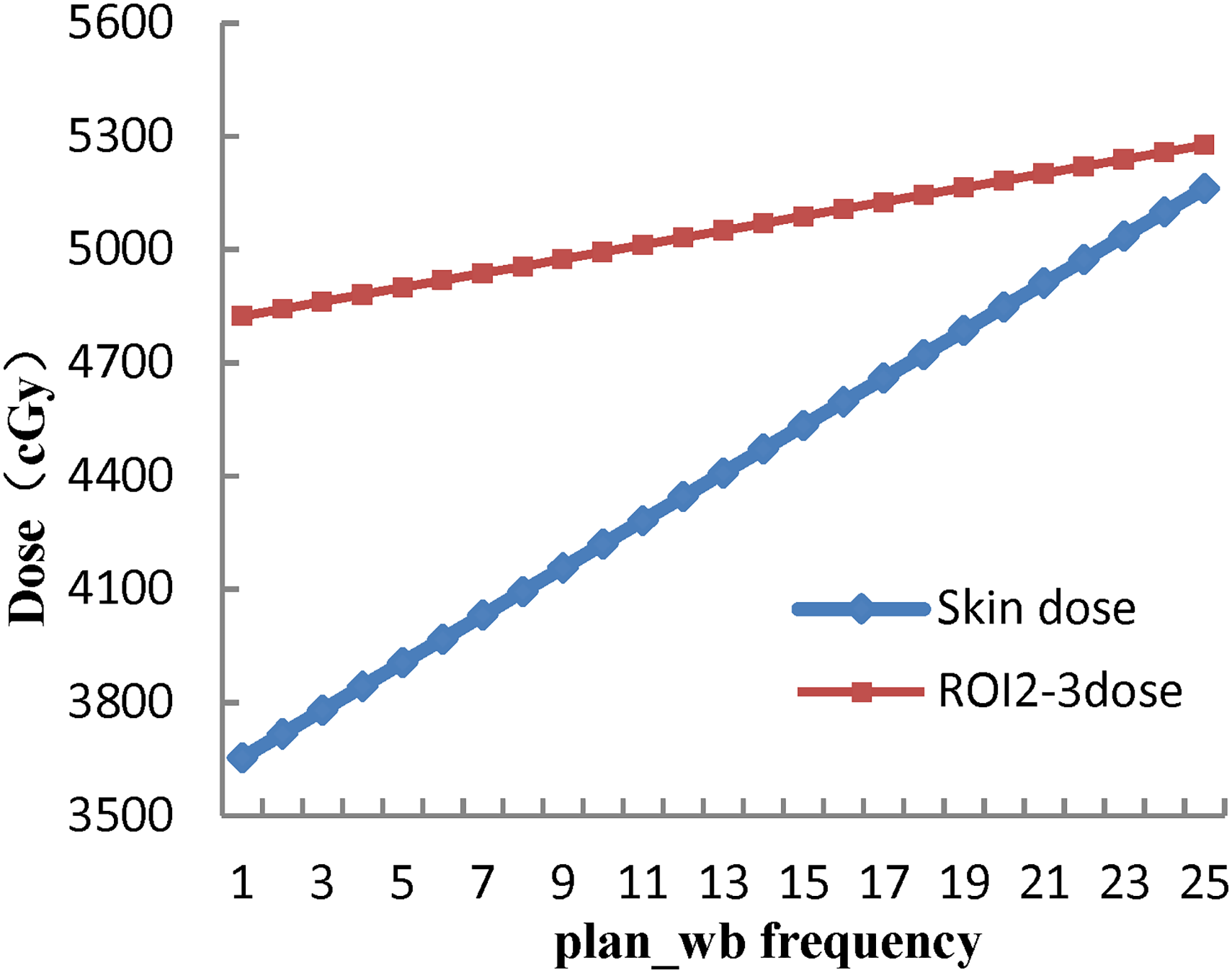
Breast skin and ROI2-3 dose under different frequencies of bolus applications.
Discussion
Breast cancer patients undergoing modified radical surgery need tissue compensators to increase the skin surface dose during radiotherapy. Due to the curvature of the chest wall and the appearance of postoperative scars, the most commonly used commercial flat boluses cannot perfect contact with the skin, resulting air gap that affects the second skin-sparing effect and reduces treatment accuracy. 17 Using 3D printing technology to produce personalized and highly conformal tissue compensators can reduce air gaps and improve dose homogeneity in the near-skin target area. 18 In this study, a 0.5 cm 3D printed bolus was used, and the results showed a 44.5% increase in mean breast skin dose, which means that the mean breast skin dose increased from 72.2% of the prescription dose (Plan_nb, 144.46 ± 10.32 cGy) to 104.4% of the prescription dose (Plan_wb, 208.75 ± 4.55 cGy). Compared to plan_nb, plan_wb showed a relatively small deviation in breast skin dose and significantly improved uniformity. The increase in breast skin dose was statistically significant (t = −18.56, P < 0.001).
The accuracy of radiation dose delivery is a critical requirement of modern precision radiation therapy.19,20 Calculation of skin doses is difficult in most TPSs due to their inability to account for all the factors that contribute to the surface dose. Doses calculated with different TPSs have been reported to underestimate or overestimate the measured skin doses.21,22 However, MC codes are now widely accessible and can be used to calculate extremely complicated radiation treatments, so this study adopted the MC TPSs to calculate skin doses.23–25 Skin dose is influenced by many factors such as delivery technique, field size, incident angle, air gap, bolus material, and skin measurement equipment.7,22 Capelle L et al used TLD/OSL measurements to assess the actual exposure dose to the patient's skin on the affected side. The results showed that there were relatively large deviations between the measured and calculated values at the upper and lower measurement points, while the other measurement points showed good agreement. 26 Elywa M et al used TLD measurements to evaluate the skin dose in patients undergoing cobalt-60 radiation therapy for breast cancer. The results showed that there was statistical significance between the TPS and TLD measurements at the points near the edge of the treatment field, while there was no correlation at other points. 27
It's important to note that TLD calibration should be performed regularly to ensure accurate dose measurements. In this study, one hundred TLD chips were calibrated using a single-point calibration method three times. TLD chips with a dose standard deviation greater than 1 were excluded, and 90 TLD chips with stable doses were obtained to ensure accurate dose measurements. Nine measurement points were selected to measure the skin dose of the breast. Among them, point 3 was located at the nipple position, with the small air gaps between the bolus and the irregular skin surface, resulting in a dose deviation of 3.18% between the TPS calculated value and the TLD-measured value. Point 5 showed a large discrepancy of 4.44%, which was attributed to its location at the edge of the treatment field, where the dose gradient was steep, and small positioning errors can cause large dose deviations. The TPS calculated values and TLD-measured values showed good consistency for the other measurement points, with a maximum deviation of less than 3%. There were four main reasons for the good consistency. First, surface markers were used for patient positioning, which improved the reproducibility of the treatment setup. Second, 3D printing technology was used to create personalized and highly conformal tissue compensators, which improved the accuracy of the skin dose. Third, the TLD calibration used a single-point calibration method, with 100 TLD chips measured three times to eliminate TLD chips with large dose deviations and improve the measurement accuracy. Finally, the TPS used in this study was Monaco 5.11.03, which employs the XVMC algorithm, a gold standard in radiation therapy dose calculation.28,29
The dose to the breast skin and chest wall is related to the frequency of bolus applications, and an appropriate use of bolus can ensure that the skin dose is within the tolerance range and the dose delivered to the chest wall can reduce the risk of treatment failure. Different radiotherapy institutions have different choices between improving skin dose and preventing skin adverse skin reactions,1,30–32 and different tumor stages require different doses for the skin and chest wall.3,4 For T1-3N+ breast cancer patients, the subcutaneous lymphatic drainage area, which is approximately 2-3 mm under the skin surface, needs to reach a prophylactic dose of 50 Gy. For T4N+ breast cancer patients, the skin is part of the CTV and needs to reach a prophylactic dose of 50 Gy. 4 In this study, the skin dose and ROI2-3 dose with different plan_wb treatment frequencies were analyzed. The bolus was produced by 3D printing technology, with a thickness of 0.5 cm. The required bolus application frequency for different stages of breast cancer was quantified, providing a reference for clinical practice. For T1-3N+ breast cancer patients, when the plan_wb treatment frequency was 15 times of the total 25 fractions, the breast skin and ROI2-3 irradiation doses were 45.33 Gy and 50.88 Gy, respectively. For T4N+ breast cancer patients, when the plan_wb treatment frequency was 23 times of the total 25 fractions, the breast skin and ROI2-3 irradiation doses were 50.36 Gy and 52.39 Gy, respectively, which met the clinical needs.
We have applied the above bolus regime in the afterward clinical practice, the patients were given radiotherapy once a day, 5 days a week, for a total of 5 weeks with 2 Gy each time. For T1-3N+ patients, the first 15 times of the radiation were covered with bolus. For T4N+ patients, the first 23 times of treatments were using bolus.
The data of the 3D-print bolus produced in this study comes from localization CT, which can be completed simultaneously with the formulation of radiotherapy plans without prolonging the patient's waiting time for radiotherapy. It is clinically feasible. A total of 87 patients were admitted to the Radiotherapy Department of Henan Cancer Hospital from February 2023 to August 2023. Among them, 83 patients were diagnosed with T1-3N+ breast cancer and 4 patients were with T4N+ breast cancer. Due to limited T4N+ group data, acute skin radiation injury statistics were conducted on 83 patients with T1-3N+ breast cancer. The radiation-induced acute skin injury grade was assessed according to the RTOG Acute Radiation Injury Grading; 33 83 cases were divided into the 15-times bolus-using group and the 25-times bolus-using group. Compared to adding bolus every time while receiving radiotherapy, the incidence rate of radiation dermatitis of grade III and grade IV in 15 times bolus-using group reduced from 19.0% (8/42) to 2.4% (1/41), (P = 0.038, supplementary table). These results showed that the incidence of radiation dermatitis from grade III to grade IV was significantly reduced.
Due to the limitations of the human phantom in this study, further long-term follow-up studies are needed to confirm the differences between skin and chest wall doses under different frequencies of bolus applications, as well as to assess the toxicity and clinical outcomes in patients treated with different plans.
Conclusions
The 3D printing bolus can help overcome the dosage disparity caused by undesired air gaps and a property bolus application frequency can keep the skin dose within the acceptable range while ensuring adequate chest wall dose to prevent recurrence.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241264848 - Supplemental material for Superficial Dosimetry Study of the Frequency of Bolus Using in Volumetric Modulated Arc Therapy after Modified Radical Mastectomy
Supplemental material, sj-docx-1-tct-10.1177_15330338241264848 for Superficial Dosimetry Study of the Frequency of Bolus Using in Volumetric Modulated Arc Therapy after Modified Radical Mastectomy by Lingling Tian, MS, Ronghu Mao, MM, Dingjie Li, MM, Wei Guo, MD, Bing Li, MD, Zhaoyang Lou, MD, and Leiming Guo, MM in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
Thanks to GenoSaber Biotech Co. Ltd. for providing writing assistance.
Conflict of Interest
The authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by A Study on the Open Teaching Method Combined with Scientific Research - Taking the Course of Radiotherapy Target Area Mapping as an Example [Wjlx2021332]; Immune Radiotherapy Research Fund Project of the Radiation Tumor Treatment Credit Committee of the Chinese Medical Association[Z-2017-24-2108].
Research Ethics and Patient Consent
This study did not involve any animal or human subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


