Abstract
Background:
A 2-level factorial pilot study was conducted in 2007 just before starting a randomized clinical trial comparing tomotherapy and conventional radiotherapy (CR) to reduce cardiac and pulmonary adverse effects in breast cancer, considering tumor laterality (left/right), target volume (with/without nodal irradiation), surgery (tumorectomy/mastectomy), and patient position (prone/supine). The study was revisited using a penalty score based on the recently developed mean absolute dose deviation (MADD).
Methods:
Eight patients with a unique combination of laterality, nodal coverage, and surgery underwent dual tomotherapy and CR treatment planning in both prone and supine positions, providing 32 distinct combinations. The penalty score was applied using the weighted sum of the MADDs. The Lenth method for unreplicated 2-level factorial design was used in the analysis.
Results:
The Lenth analysis identified nodal irradiation as the active main effect penalizing the dosimetry by 1.14 Gy (P = 0.001). Other significant effects were left laterality (0.94 Gy), mastectomy (0.61 Gy), and interactions between left mastectomy (0.89 Gy) and prone mastectomy (0.71 Gy), with P-values between 0.005 and 0.05. Tomotherapy provided a small reduction in penalty (reduction of 0.54 Gy) through interaction with nodal irradiation (P = 0.080). Some effects approached significance with P-values > 0.05 and ≤ 0.10 for interactions of prone × mastectomy × left (0.60 Gy), nodal irradiation × mastectomy (0.59 Gy), and prone × left (0.55 Gy) and the main effect prone (0.52 Gy).
Conclusions:
The historical dosimetric analysis previously revealed the feasibility of tomotherapy, but a conclusion could not be made. The MADD-based score is promising, and a new analysis highlights the impact of factors and hierarchy of priorities that need to be addressed if major gains are to be attained.
Keywords
Introduction
Meta-analyses on breast cancer have shown that local radiotherapy (RT) is associated with two-thirds reduction of local recurrence, regardless of the type of surgery and axillary nodal status. 1 A definite survival advantage is observed with long follow-up duration. 2 However, the survival advantage can be offset by non-negligible toxicity notably on the heart and lungs. 3,4 Advanced techniques hold promise to improve the benefit ratio of tumor control vs. toxicity, such as inverse-planned intensity-modulated RT (IP-IMRT) and volumetric image-guided RT (V-IGRT), although there are trade-offs. 5 Despite the advances, conventional RT without IP-IMRT in breast cancer remains the standard treatment, and complex techniques are restricted to highly selected groups. 6 A US practice survey found that volumetric imaging was used in all sites, except the breast. 7 Breast tomotherapy was deemed inferior to conventional RT in a dosimetric comparison of tomotherapy versus tangential fields with tissue compensation. 8 Despite the contradicting observation, 9 complex IMRT is still considered inappropriate, and < 20% of patients undergoing lumpectomy received IMRT based on surveys of Medicare beneficiaries. 10,11 In Australia and New Zealand, where patients with breast cancer represent the largest population receiving curative RT, IGRT has been seldom applied; 2-dimensional portal imaging was the predominant imaging mode for breast cancer (82% and 86%, respectively). 12,13 To further compound the disease burden, reimbursement of IP-IMRT/V-IGRT is denied to most patients with breast cancer in several western countries. In the USA, several Medicare carriers discontinued coverage of IMRT for breast cancer, or changed local coverage decisions to only allow IMRT in specific situations around 2008-2009. 10 In France and Belgium, breast IMRT using dedicated V-IGRT is excluded from the list of admissible radiation billing (Assurance Maladie, website ameli.fr, accessed February 19, 2020). Presently, despite the breast radiation breakthrough in the early 2000s, advanced techniques in breast cancer face an overwhelming disincentive.
Against that backdrop, TomoBreast is one of the extremely rare breast cancer randomized clinical trials and posits that RT outcomes can be improved with the use of advanced radiation techniques. 14,15 Before the trial, the original investigators conducted a preliminary evaluation of factors most likely to affect the feasibility of tomotherapy (T) (throughout the text, we will use “tomotherapy” as an instantiation of IP-IMRT/V-IGRT acknowledging there are now several alternatives) compared with conventional RT, according to left (L) versus right breast laterality, mastectomy (M) versus tumorectomy, irradiation of regional lymph nodes (N) versus no lymph node irradiation, and prone (P) versus supine position. 16 A formal analysis of the pre-TomoBreast study has not been conducted because of the lack of appropriate tools. However, the situation has changed with the recently developed metric, the mean absolute dose deviation (MADD). 17 The MADD allows combining dose-volume histograms into a single dosimetric outcome that makes it feasible to have another perspective in the pre-TomoBreast study. Information thereof might be valuable only for identifying strengths and caveats for future applications. This study was not hypothesis driven; the report explored the factors and aimed to evaluate the utility of the MADD-based score as applied to the pre-TomoBreast 2 5 data.
Materials and Methods
Source of Data
The accrual of patients and the treatment plans were performed in 2006–2007 at the Radiotherapy Department of the Universitair Ziekenhuis Brussel (UZ Brussel). The data was provided courtesy of the original investigators. 16,18 Eight adult women presented with a primary unilateral breast cancer, each with a distinct combination of left or right breast laterality, presence or absence of nodal irradiation, and tumorectomy or mastectomy, who were referred for postoperative RT, provided informed consent, and underwent double simulation in supine and prone positions (Supplemental Material, Protocol). Conventional RT used tangential breast/chest wall and anterior axillary supraclavicular fields. 19,20 Additional multileaf collimator segments were allowed to optimize the planning, using the Pinnacle3 Station, collapsed cone convolution algorithm. For tomotherapy, we used the TomoTherapy treatment planning system (Hi-Art, Accuray Inc., Sunnyvale, CA) for inverse planning (dynamically penalized likelihood method-cost function). The following structures were delineated for the study: clinical target volume (CTV), planning target volume (PTV; PTVbw for either the breast after breast-conserving surgery or the chest wall after mastectomy, PTVn for nodes), ipsilateral and contralateral lung, heart, and contralateral breast. The CTV, PTV, and organs at risk (OARs) were outlined on all CT slices. The planning dose prescription was 42 Gy in 15 fractions of 2.8 Gy daily. The constraints and planning procedures are detailed in Supplementary Materials (Part B and Supplementary Table ST-1).
Penalty Score
For a definitive evaluation of the treatment plans, we defined a penalty score using MADDs. The MADD is a measure of dispersion that quantifies how much a DVH deviates from a given dose specification. 17 Using the notation where x represents the dose and y the corresponding volume values on a cumulative dose-volume histogram of a structure with volume V0 and for which a reference dose A has been specified, the MADD M(A) of the structure can be defined as follows:
which trivially represents the area between curves, between the DVH of a structure and the reference dose A. In the case of an OAR where the ideal reference dose is 0, it is also the area under the cumulative DVH, which equates the mean dose to the organ scaled to the volume of the organ. In the case of a target volume where the prescribed reference dose is not 0, the area between the DVH and prescribed dose equates the sum of under- and overdosage to the target. A MADD value of 0 indicates a perfect organ or target DVH, where the dose to the organ at risk is 0 and the dose to the target structure perfectly matches the prescription to the target. The definition of the MADD is the same regardless of the structure being labeled as an OAR or target. The MADD for all structures is expressed in the same dose unit as per planning. A larger MADD value indicates a larger deviation from the ideal DVH.
In the present study, the MADDs were computed for all structures for every planning combination. The penalty score pertaining to a planning combination was constructed using the weighted sum of the MADDs:
where
Mi
is the MADD of the structure i, where i = 1 designates PTVbw, 2 PTVn, 3 ipsilateral lung, 4 heart, and 5 contralateral breast.
Ai
is the reference dose to the structure i, 42 Gy for PTVbw and PTVn, and 0 Gy for the ipsilateral lung, heart, and contralateral breast.
wi
is the weight assigned to the structure i with
The constraint that weights sum to 1 allows expressing the penalty score at the same dose scale as the planned treatment dose and reading the score as a weighted average of excess dose deviations. We assigned the same weight to all structures, wi = 0.20, for i = 1…5. The theoretical best possible penalty score is 0 when all MADDs are 0. Increasing values of the score indicate increasingly poorer dosimetric outcomes.
The OAR mean doses were previously recorded, and these required no new calculation. The MADD of the PTVbw and PTVn were computed on the DVHs retrieved by digitizing the respective curves from the patients’ DVH graphs using DigitizeIt version 2.3.3 (I. Bormann, digitizeit.de, 38108 Braunschweig, Germany). For PTVn, not contoured in node-negative patients, 0 was assigned to the corresponding MADD.
Lenth Analysis
The study design is an unreplicated factorial experiment, indicating that there is one and only one observation for any combination of factors. Traditional regression-based inferences cannot be made because there is no estimate of residual error. 21 We applied the Lenth statistical method for unreplicated factorial designs to identify factors and combinations of factors most significantly associated with penalty scores. 22 The Lenth method computes a pseudo standard error (PSE) from the median scores. The main assumptions are equal variance and sparsity of the effects, indicating that the majority of the effects are expected to be inactive and only a handful are active. The margin of error (ME) is calculated from the PSE to identify the threshold value of the outcomes that reach significance. Furthermore, because several inferences are made, simultaneously increasing the risk of false discovery, the method computes a simultaneous ME, which provides a higher significance threshold adjusting for multiple comparisons.
The analysis was applied to full data (all 5 factors, 25 = 32 runs) and data excluding the prone plans (4 factors, 24 = 16 runs), considering that supine position is more commonly used in practice.
Data processing, tabulation, factorial analysis, and significance testing were performed using the statistical software R, version 3.6.3. 23 The Lenth method was implemented using the R package unrepx.
Additional Notes
The supplemental material includes the study protocol, dosimetric procedures, and all patients’ DVH printouts (digitized in Supplementary Figure SF-1, thumbnails of originals in Supplementary Figure SF-2). Although not detailed in the study protocol, all treatment plans were prospectively performed within the department’s workflow, received final approval from the supervising radiation oncologist, and underwent staff review. We did not conduct after-the-fact dosimetry; all present analyses used the original investigators’ approved plans.
The presentation of results will be divided into 2 parts: listwise structure-by-structure review, and new factorial analysis.
Results
Patient characteristics are summarized in Table 1. As mentioned in the Methods section, each patient presented with a distinct combination of left or right breast laterality, presence or absence of nodal irradiation, and tumorectomy or mastectomy, placed in prone and supine position, and received conventional and tomotherapy plan, resulting in a total of 32 runs (Table 2).
Patient Characteristics.
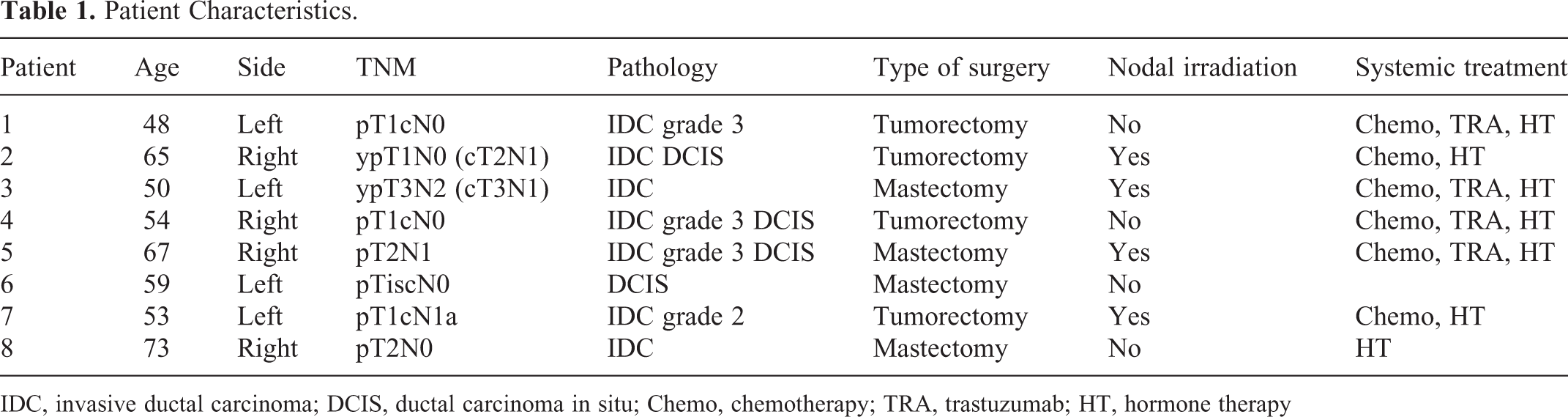
IDC, invasive ductal carcinoma; DCIS, ductal carcinoma in situ; Chemo, chemotherapy; TRA, trastuzumab; HT, hormone therapy
MADDs in all Factor Combinations.

RT, radiotherapy; NA, not available; assigned 0 for score calculation.
Listwise Outcomes by Structures
PTV coverage of the breast/chest wall was generally good, with small values of the breast/chest wall MADDs, indicating a homogeneous dose (Supplementary Table ST-2). PTVbw for the breast post-conservative surgery was between 95% and 110% of the prescribed dose in 15/16 plans. Among the plans that reached dose constraints, the highest V95% was obtained with supine tomotherapy in 3/4 patients. PTVbw for chest wall post-mastectomy was between 95% and 110% of the prescribed dose in 14/16 plans. Among the plans within dose constraints, the highest value of V95% was with tomotherapy planning in 3/4 patients.
The coverage of the nodal PTVs was poor by conventional therapy, with an average MADD of 4.0 Gy, compared with an average MADD of 1.8 Gy by tomotherapy. Only 2 plans achieved delivery between 95% and 110% of the prescribed dose to the nodal PTVs, with supine tomotherapy. Prone position was unfavorable for PTVbw in cases of lymph node irradiation, with either tumorectomy or mastectomy.
In the ipsilateral lung, dose constraints were respected in 21 plans (11 without supraclavicular irradiation), of which 13 with tomotherapy, and 12 in prone position. By patients, the lowest value of V20 Gy (range from 0.46% to 9.8%) was achieved in 6/8 patients with prone tomotherapy plans (patients #1, 3, 4, 5, 7, and 8), and in 1/8 with prone conventional plan (patient #2). V20 Gy dose constraints have always been respected with prone tomotherapy except for patient #6 (V20 Gy 8.72%, which exceeded 8% lung constraint for node-negative). In 6/8 cases (patients #1, 2, 3, 4, 5, and 7), V20 Gy was halved with prone tomotherapy compared with supine conventional planning. In 1 case (patient #1, left tumorectomy, N0), this value was divided by a factor of 14 (0.46% vs. 6.5%).
Regarding the heart with right-sided tumors, all plans reached the dose constraints for V20 Gy, except for one prone conventional planning in patient #4 (tumorectomy, N0) with a value of 5.4%. This was explained by cardiomegaly in this patient. With left-sided tumors, all plans respected the dose constraints except for 3 of them (in prone position). The lowest V20 Gy was achieved in 3/4 cases (patients #3, 6, and 7) with supine tomotherapy (values of 3.24%, 0.03%, and 0%, respectively).
Regarding the contralateral breast, only 6 plans respected Dmax dose constraint, all 6 with conventional supine planning. With conventional techniques, only 2 fields (tangential) are used; with tomotherapy, many can be used. However, tomotherapy planning did not always significantly increase the mean dose to the contralateral breast. With tomotherapy, in the supine or prone position, the mean dose was inferior to 4 Gy in 8 and 5 cases, respectively. In patients #7 and 8, the mean dose to the contralateral breast for all plans was inferior to 1 Gy. The 3 patients with an increased (> 4 Gy) mean dose were patients #3, 4, and 8 (1 left mastectomy, N+, and 2 tumorectomy N0) with values of 11.21 Gy, 7.25 Gy, and 8.58 Gy, respectively.
MADD-Based Penalty Scores Factorial Analysis
The MADDs pertaining to each structure (PTV breast/chest wall, PTV nodes, and OAR lung, heart, and contralateral breast) and the penalty score in each of the 32 patient-treatment combinations are shown in Table 2.
The Lenth analysis of Table 2’s scores is shown in Table 3. Figure 1 presents the barplot representation of Table 3. The analysis identified nodal irradiation (N) as the outstanding statistically significant main effect (Figure 1, N, unadjusted P = 0.001). Nodal irradiation contributes an estimated main effect of 1.14 Gy to the penalty score (Table 3). Left laterality (L, 0.94 Gy), mastectomy (M, 0.61 Gy), the interactions of mastectomy × left (ML, 0.89 Gy) and prone × mastectomy (PM, 0.71 Gy) were of intermediary significance with unadjusted P-values between 0.005 (left laterality) and 0.049 (mastectomy).
Estimated Factorial Penalty Score Effects.
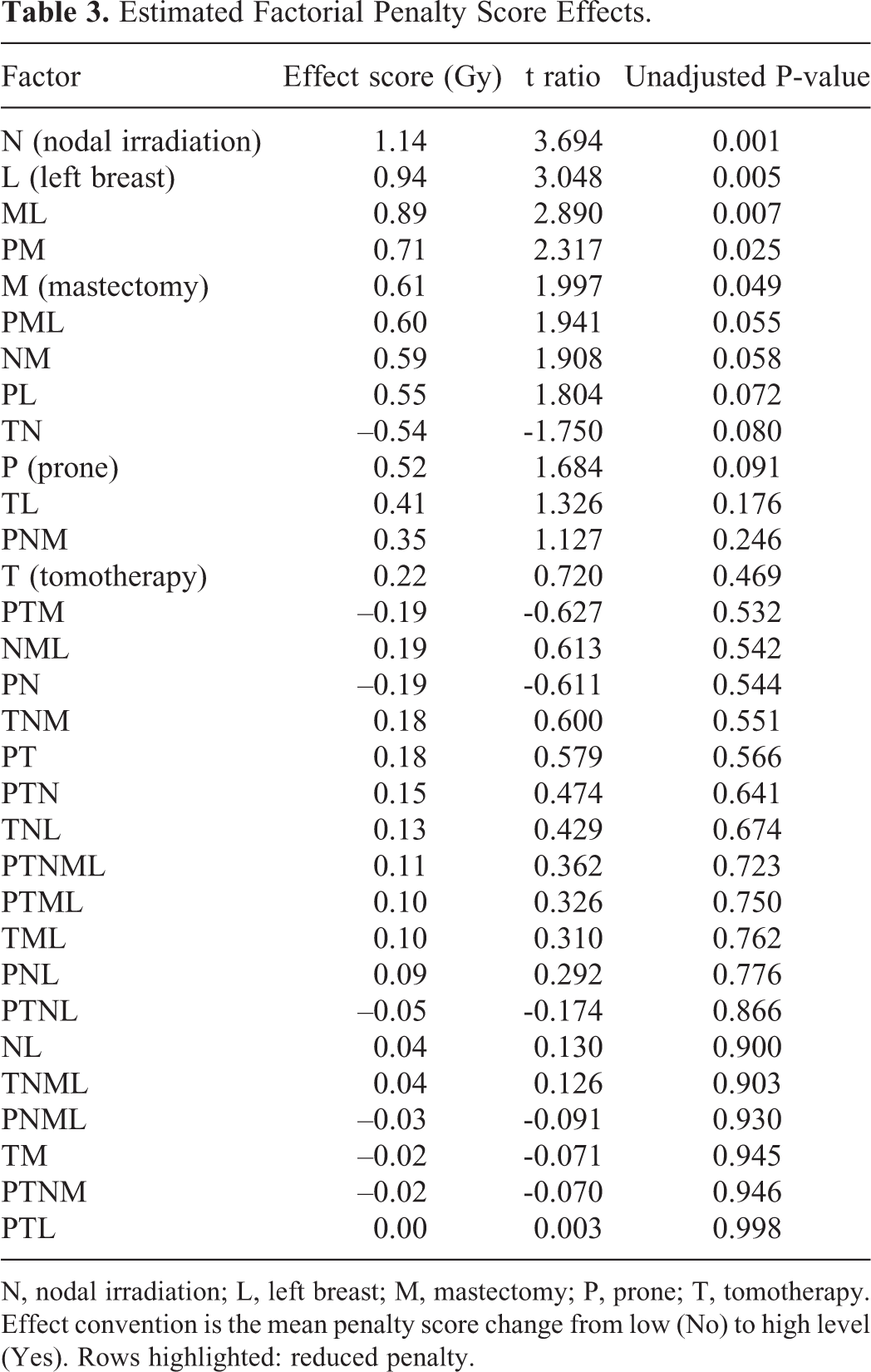
N, nodal irradiation; L, left breast; M, mastectomy; P, prone; T, tomotherapy. Effect convention is the mean penalty score change from low (No) to high level (Yes). Rows highlighted: reduced penalty.

Barplot 25 factorial penalty score effects. N, nodal irradiation; L, left breast; M, mastectomy; P, prone; T, tomotherapy; ME, margin of error; SME, simultaneous margin of error. Bars beyond ME indicate an absolute effect significant at P ≤ 0.05, beyond SME at P < 0.002. Blue codes, positive effects; orange codes, negative effects.
A few interactions approached significance, with P-values >0.05 and ≤ 0.10 for prone × mastectomy × left (PML, effect = 0.60 Gy, P = 0.055), nodal irradiation × mastectomy (NM, effect = 0.59 Gy, P = 0.058), prone × left (PL, effect = 0.55 Gy, P = 0.071), tomotherapy × nodal irradiation (TN, effect = –0.54 Gy, i.e. score reduction, P = 0.079), and prone (P, effect = 0.52 Gy, P = 0.089) (Table 3). The other factors and interactions shown in Figure 1 and Table 3 were far from significant. Overall, most factors and interactions were associated with an increased penalty (blue bars), whereas 7 interactions were associated with a reduced penalty, of which 5 were linked with tomotherapy (Figure 1, orange bars, TN, PTM, PTNL, TM, and PTNM, also highlighted in Table 3).
In an analysis excluding prone data, which corresponds to removing all prone rows from Table 2, reducing the experiment from 5 to 4 factors, only 2 factors were found significant (Figure 2): nodal irradiation (N, effect = 1.32 Gy, P = 0.0003) and the interaction tomotherapy × nodal irradiation (TN, effect = –0.68 Gy, i.e. score reduction, P = 0.026). The tomotherapy main effect was small (T, effect = 0.04 Gy, P = 0.886). The other estimated effects shown in Table 4 were not significant.

Barplot 24 penalty score effects, supine position only. N, nodal irradiation; L, left breast; M, mastectomy; T, tomotherapy; ME, margin of error at 0.05 significance level; SME, simultaneous margin of error. Blue codes, positive effects; orange codes, negative effects.
Estimated Factorial Effects, All Supine Positions Only.

N, nodal irradiation; L, left breast; M, mastectomy; T, tomotherapy. Effect convention is the mean penalty score change from low (No) to high level (Yes). Rows highlighted: reduced penalty.
Discussion
Listwise Analysis
Not much can be said of the historical analysis which applied the listwise evaluation of structures’ dosimetric outcomes. The conclusion drawn at the time and unchanged today was the feasibility of tomotherapy. The burden of the proof of tomotherapy superiority relies on the subsequent TomoBreast randomized clinical trial (ClinicalTrials.gov NCT00459628) that started immediately after. The experience served its purpose as a precursor for the trial. However, before moving on to the discussion of the factorial analysis, the experiment deserves a few comments on the time validity. We also discuss the descriptive utility of the penalty score.
Dataset Validity Over Time
Treatment planning outcomes are operator dependent. 24,25 Efficiency increases with experience. Doses to OAR decrease over time. 26 “Conventional” RT at the UZ Brussel implemented the earliest inception of breast IMRT as later described by others. 27,28 Tomotherapy was barely starting, and few had much or any experience with its use in breast cancer as shown in a correspondence that highlighted and corrected dosimetric shortcomings. 9 Both breast “conventional” RT and IP-IMRT performed in the pre-TomoBreast experiment were in their infancy; therefore, one should be cautious against transposing today. However, a study in 2016 compared tangential IMRT and tomotherapy, with and without breath-hold, in a group of women without nodal irradiation. 29 The old lung and heart doses reported in the present study are comparable to those in that 2016 study. With regard to nodal irradiation, the present study lung doses are lower than reported in the breath-hold literature, 20,30 demonstrating that the pre-TomoBreast dosimetric comparison remains actual.
Qualitative or Quantitative Scoring
Qualitative scoring has been used to summarize dosimetric outcomes. 18 According to the historical analysis, qualitative scoring indicated an overall advantage with supine tomotherapy, as compared with the other 3 combinations (Supplementary Table ST-3). However, the penalty score computed now would conclude that supine conventional RT provides the best dosimetric outcome with a penalty score (weighted mean excess dose deviation) of 2.16 Gy compared with the supine tomotherapy penalty score of 2.21 Gy. Note from the same Supplementary Table ST-3 that the qualitative scoring shows an advantage of tomotherapy on the breast/chest wall PTV. However, the MADDs are almost identical. Between the highest and lowest breast chest/wall PTV MADDs, a difference of 0.34 (=1.28 – 0.94) Gy for a prescribed dose of 42 Gy can hardly be considered as the sole determinant in selecting a technique. The exercise shows that the MADD and penalty score provide objective and physically meaningful quantitative summaries that can boost data interpretation.
Byproduct: DVH Visual Quality Relationship
To further explore the descriptive quantitative utility of the penalty score, we transcribed all 32 scores from Table 2 onto the full set of DVH figures (Supplementary Figure SF-1). The scores match quite well with the visual quality of the DVH graphs. The pairs of DVH graph-scores suggest that scores < 1.50 for a dose prescription of 42 Gy indicate reasonably acceptable plans. Higher scores indicate increasingly poor plans. This might lead to a prospective evaluation of the visual-score relationship.
Factorial Analysis
Most factors considered are well known. It is trivial that nodal irradiation involves more lung areas, that the left breast might increase heart dose, and that post-mastectomy RT is prescribed when the disease is locally/regionally advanced, which increases the radiation volume, all of which contribute to poorer dosimetric outcomes. However, the interplay of these factors with different treatments is unknown.
Hierarchy of Priorities
The factorial analysis provides a sensible hierarchy of priorities at a glance. Figure 1 groups factors by effect size (the effects are shown in the original units of measurements). 22 Despite the daunting number of factor combinations, the experimenter can identify which set of factors are most likely to provide a substantial gain if they could be addressed. As an example, we noted that the factor N, nodal irradiation, is the foremost effect. Notwithstanding other factors, as shown in Table 3, nodal irradiation with conventional RT would incur an estimated mean increase of penalty score of 1.14 Gy relative to N0 (=factor N alone). Nodal irradiation with tomotherapy would instead incur a mean increase of penalty score of 0.82 Gy relative to N0 = sum of the effect of N (1.14 Gy) + effect of T (0.22 Gy) + effect of interaction TN (–0.54 Gy). The example is shown in Figure 3 using Table 2’s raw penalty scores. This shows that tomotherapy increased the penalty score in node-negative cases but decreased the penalty score in nodal irradiation. Incidentally, if confirmed, the observation would raise the uncomfortable question that health policy makers restrict tomotherapy reimbursement to lowest-risk tumors for which there is little gain, yet deny it in the higher-risk cases more likely to have greater gain.

Interaction plot. Effect of “IGRT” (tomotherapy) compared with “IMRT” (conventional), according to presence or absence of nodal irradiation.
Power of the 2 k Factorial
The 2-level factorial is a powerful efficient design. 21,31 However, its use in clinical medicine is exceedingly rare. 32,33 A PubMed search on April 4, 2020, found that, among 349,364 “breast cancer” communications, 107 human non-laboratory studies could qualify as factorial experiments. These included 78 papers with 2 factors (22, commonly noted 2 × 2), 8 with 3 factors (23), 2 with 4 factors (24), one of which emulated factorial through regression, 34 and 1 that was truly full factorial, 35 1 with 5 factors (25), 36 and 1 with 7 factors (27). 37 That is, only three 2k breast studies were performed with k > 3, 0.0009%. A possible explanation is the complexity even with textbook guidance and tutorials. 21,31,38 Implementation might also have been hindered by the perception that the 2-level factorial design requires large amounts of data. Indeed, for each factor added, the size of the unreplicated experiment doubles. However, the power yield per run is considerable. Daniel’s rule of thumb (page 76) 39 notes that 4 real effects are averaged for a 24, and 7 real effects for a 25 unreplicated experiment. We can find further discussion in Ryan (page 209), 40 considering a model with main effects only and 2-factor interactions: for an unreplicated 24 design (the present study size, halved to 16 runs by excluding prone position), the power was 0.887 for a 2σ effect and 0.368 for a 1σ effect. For an unreplicated 25 design (total size of the present study, 32 runs), the power was 0.999 for a 2σ effect and .757 for a 1σ effect. The 2 active effects found in this study with the reduced 4-factor set and 5 with the full data are in good agreement with the power considerations.
Caveats
We have discussed some limitations of the study, notably the time delay. However, the major limitation lies elsewhere. Even though the original experiment’s layout is a full factorial, the runs were not randomized, the measurements were nested by patients, and the observations were not independent. Without replication, we cannot test the assumption of variance equality. Future implementation would have to strive to assign one different patient per run, as random as possible. Replication would be advisable.
Note that part of the present study overlaps with a subsequent investigation that added TOMODirect. 18 TOMODirect supine (6 beams) is optimal for a left-sided tumor, but the data were not released for the present analysis.
Altered Fractionation
Hypofractionation in RT for breast cancer has received increasing acceptance. 41 Although fractionation is not a topic of the present study, the TomoBreast randomized clinical trial implemented 42 Gy in 15 fractions to the breast/chest wall and implemented a 51-Gy integrated boost in 15 fractions to the tumor bed after conservative surgery. The choice was based on a comparison of published protocols. 42 -49 The best evidence available at the time was the Canadian trial that compared 42.5 Gy in 16 fractions to conventional RT, without boost. 43 The original investigators learned informally from Canadian colleagues that 10-Gy boost in 4 fractions were given in clinical practice. They found (1) a schedule similar to the Canadian trial but requiring < 16 fractions, and (2) that would be close to standard fractionation doses of 50 Gy to the breast/chest wall and 66 Gy to the tumor bed with regard to ranges of alpha/beta ratio from 3 to 10. 50 As shown in Table 5, 42-Gy to breast/chest wall in 15 fractions are comparable to the Canadian doses, while 51-Gy simultaneous integrated boost in 15 fractions provides a biologically effective dose (BED) and equivalent dose in 2 Gy fractions (EQD2) close to standard fractionation; furthermore, 42 and 51 Gy/15 fractions are arithmetically exact to 1 decimal to compute fraction dose.
Selected Breast Hypofractionation Schedules.

Biological effective dose, BED = D·(1+d/α/β). Equivalent dose in 2 Gy fractions, EQD2 = D·(α/β+d)/(α/β+2). 50
Clinical outcome data was not released for the 8 patients in the present study. However, TomoBreast has randomized 123 women presenting with stage I–II breast cancer to hypofractionated tomotherapy 42/51 Gy versus normofractionated conventional RT (50/66 Gy). In a congress presentation at a median follow-up duration of 10 years, there was no difference in overall or disease-free survival, but patients randomized to tomotherapy had a significantly better survival free from deterioration in any of dyspnea, fatigue, physical functioning, or pain self-reported measures. 51 The present study provides the rationale to propose subgroup analyses according to breast laterality, mastectomy, and nodal status.
Further Studies
The MADD-based penalty score is a new tool. Validation requires prospective evaluation. Experience needs to be accumulated to define the best domain of application, which will depend on the population of patients treated in different centers and questions considered. We have started implementing the MADD in studies that we feel are most urgently needed in our respective centers.
In a study originating from Geneva, HUGProne (ClinicalTrials.gov, NCT02237469), prone breast RT is compared with supine breast RT, with or without breath-hold. HUGProne aimed to evaluate the dosimetric gain associated with the prone or supine dosimetric gain, which is the lowest radiation dose to non-target organs (heart, lungs, contralateral breast), while giving the prescribed dose to the tumor bed and ipsilateral breast. The penalty score used in the present study will be highly appropriate as a measure of dosimetric gain.
Another study is being conducted in Martinique, where the major disease burden is prostate cancer. The issue in RT of prostate cancer is how to balance between a high dose to the prostate and sparing the immediately adjacent OARs, the bladder and rectum. Currently, patients in Martinique’s study are treated with either Tomotherapy or TrueBeam volumetric modulated arc therapy, with allocation depending only on time slot availability. The MADD will be applied to compare the 2 treatments to identify which patients would best benefit from one technique.
We cannot advise how MADD should be used. If the focus is on the lowest possible dose to OAR or only on the highest possible coverage dose to PTV, many indices are available 52 : there is no need for the MADD. However, when there is a need to balance the radiation doses to OARs and PTVs, as suggested in the ongoing Geneva breast and Martinique prostate studies, or in situations such as stereotactic RT that delivers ablative doses to spinal malignancies while sparing the spinal cord, 53 the contribution of the MADD and penalty score to evaluate the dosimetric gain between techniques can be important.
Another domain that we discovered is the analysis of recurrences. A recent French study found no relationship between the doses delivered to the sites of nodal recurrences in head and neck cancer. 54 Head and neck RT is complex with many OARs and PTVs. Beyond standard metrics, MADD has the potential to provide a new look.
Conclusions
The key to the study is the development of the MADD that scores the OAR and PTV on the same physical dose scale as the DVHs. The overall penalty score follows naturally and provides a summary of dosimetric outcomes. It integrates seamlessly in a 2-level factorial analysis that shows that nodal irradiation, left laterality, and mastectomy significantly penalize dosimetric outcomes. The interaction between tomotherapy and nodal irradiation improved the outcome, although it did not reach significance.
The MADD-based score appears to be a promising tool to summarize radiation treatment plans and highlight relationships, encouraging further evaluation in breast and other cancers.
Supplemental Material
Supplemental Material, maddapplied_suppl_6 - Two-Level Factorial Pre-TomoBreast Pilot Study of Tomotherapy and Conventional Radiotherapy in Breast Cancer: Post Hoc Utility of a Mean Absolute Dose Deviation Penalty Score
Supplemental Material, maddapplied_suppl_6 for Two-Level Factorial Pre-TomoBreast Pilot Study of Tomotherapy and Conventional Radiotherapy in Breast Cancer: Post Hoc Utility of a Mean Absolute Dose Deviation Penalty Score by Steve Heymann, Giovanna Dipasquale, Nam P. Nguyen, Meymey San, Olena Gorobets, Nicolas Leduc, Dirk Verellen, Guy Storme, Hilde Van Parijs, Mark De Ridder and Vincent Vinh-Hung in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
Data Sharing: Manuscript data is available on Mendeley. Reserved DOI: 10.17632/8w3ckgmt84.1. Shared link: ![]() . Ethics statement: Not applicable. The study uses previously published data without involving actual patients. According to local institutional practice all patients gave oral informed consent when they underwent CT-simulation procedures in 2006-2007. The procedures were part of the UZ Brussel Radiation Oncology department’s standard quality management for which ethics committee approval was not mandated.
. Ethics statement: Not applicable. The study uses previously published data without involving actual patients. According to local institutional practice all patients gave oral informed consent when they underwent CT-simulation procedures in 2006-2007. The procedures were part of the UZ Brussel Radiation Oncology department’s standard quality management for which ethics committee approval was not mandated.
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The Radiation Oncology Department, Universitair Ziekenhuis Brussel, Vrije Universiteit Brussel (VUB), Laarbeeklaan 101, 1090 Brussels, Belgium, had a research agreement unrelated to patient’s management with Accuray, Sunnyvale, CA. Vincent Vinh-Hung and Nam P. Nguyen have patents pending for the MADD, USPTO 62/608,751, WO/2019/014384.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


