Abstract
Background
Cancer remains a leading cause of death worldwide, necessitating the development of affordable and innovative therapies to reduce its human and economic burden.
Objectives
In this study, we aimed to develop a synergistic anticancer formula encapsulated in nanoliposomes to enhance efficacy and minimize side effects. Additionally, we explored the effect of aptamer conjugation on the efficacy and stability of the formula.
Methods
The Etoricoxib-β-cyclodextrin complex was prepared using the kneading method, and nanoliposomes were developed via thin film hydration. The AS1411 aptamer was conjugated to the nanoliposomes to target nucleolin, a protein overexpressed in cancer cells. The etoricoxib-β-cyclodextrin complex was characterized using proton nuclear magnetic resonance, and various liposome properties, including size, encapsulation efficiency, and stability, were optimized. The release profiles of the active compounds were evaluated using high-performance liquid chromatography, and their cytotoxicity was assessed in human cancer cell lines.
Results
The nanoliposomes co-loaded with the three agents and their aptamer-conjugated counterpart showed optimal characteristics, with particle sizes of 133.3 ± 1.45 nm and 174.8 ± 4.78 nm, and zeta potentials of −15.26 ± 1.80 mV and −15.66 ± 2.57 mV, respectively. The encapsulation efficiencies were 88.63% (raloxifene), 41.73% (etoricoxib), and 39.26% (naringin) without the aptamer, and 81.99%, 36.66%, and 38.33%, respectively, with the aptamer. The IC50 of the formula for the three co-loaded agents was 167.4 µg/mL for A549 cells and 2.6 µg/mL for MCF-7 cells. Cytotoxicity was further enhanced using their aptamer conjugate, particularly against the MDA-MB-231 cell line.
Conclusion
The novel triple-drug-loaded, aptamer-conjugated nanoliposome formula may be a future cancer treatment strategy.
This is a visual representation of the abstract.
Introduction
In 2022, cancer caused approximately 9.6 million deaths worldwide, making it the second leading cause of death. 1 Conventional treatments such as chemotherapy and radiation remain common; however, they are limited by severe side effects, non-specificity, and the risk of drug resistance, emphasizing the need for more targeted and effective therapies.2,3 Moreover, the limitations of chemotherapy emphasize the need for targeted drug delivery systems to improve treatment precision. These systems aim to enhance drug efficacy while minimizing damage to healthy tissues. 4 Developing new therapeutic strategies and effective treatment formulas is crucial to addressing the tremendous global human and economic tolls of cancer.5,6 Repurposing existing medications with approved cytotoxic and chemo-preventive activities and creating additional synergistic medical effects can help reduce costs and accelerate drug development timelines.7,8
Raloxifene, a selective estrogen receptor modulator and non-steroidal benzothiophene derivative, was initially used to prevent and treat osteoporosis in postmenopausal females because of its estrogenic activity in bone tissue. 6 Lately, it has demonstrated anticancer activity by exerting antiestrogenic effects in the breast tissue, leading to its approval by the Food and Drug Administration (FDA) as a preventive medication for reducing the risk of invasive breast cancer in postmenopausal women. The mechanism of Raloxifene's anticancer activity has been reported to involve a reduction in tumorigenicity and migration of breast cancer cells.9,10 However, its clinical application is hindered by poor physicochemical properties, low aqueous solubility, and poor bioavailability. 11
Naringin (4′,5,7-trihydroxyflavanone-7-rhamnoglucoside) is a natural flavonoid found predominantly in citrus fruits. 12 Naringin exhibits pleiotropic effects, influences various cellular signaling pathways, suppresses cytokine and growth factor production, and arrests the cell cycle. Furthermore, it has potential antioxidant, anti-androgenic, anti-inflammatory, antiulcer, anti-allergic, blood lipid-lowering, and anticancer activities.13,14 Additionally, studies have demonstrated that Naringin synergistically enhances the anticancer activities of antitumor drugs in combination therapies. 12 Naringin has been shown to enhance the efficacy of several chemotherapeutic agents through various mechanisms. For example, Naringin enhances the cytotoxicity of doxorubicin by inhibiting efflux pumps such as P-glycoprotein, thereby increasing intracellular drug accumulation. 15 Additionally, it synergizes with paclitaxel by modulating inflammatory and apoptotic pathways, 16 and it also shows enhanced anticancer effects when combined with natural compounds like curcumin, amplifying their pro-apoptotic and antioxidant activities. 17 Thus, naringine has emerged as a promising candidate for cancer prevention and treatment, providing substantial support for clinical applications in the future. 11 However, challenges such as low water solubility, insufficient permeability, and poor bioavailability limit its use.18,19
Selective cyclooxygenase-2 (COX-2) inhibitors have contributed to significant regression of existing cancer cells, as observed in clinical studies. 13 Therefore, targeting inflammation may play a crucial role in cancer management. The proposed mechanisms of action for these drugs include immune suppression, carcinogenesis, neoangiogenesis, and apoptosis inhibition in multiple cancer types. However, their precise mechanism remains unknown.20,21
Etoricoxib, a second-generation selective COX-2 inhibitor, in addition to its anti-inflammatory properties, reduces cellular proliferation and induces apoptosis in angiogenic endothelial cells. 22 It is deemed a suitable candidate for long-term chemo-preventive use owing to its minimal side effects. 23 Nevertheless, its low water solubility and consequently, poor oral bioavailability also limit its clinical utility. 20
Enhancing the delivery of anticancer agents to malignancy sites can overcome physiochemical challenges and poor bioavailability and minimize undesirable effects. 24 The nanoscale formulation of therapeutic agents facilitates the development of intelligent drug delivery carriers tailored to specific illnesses.25,26 PEGylated nanoliposomes were selected as the delivery system in this study due to their proven ability to encapsulate drugs with varying polarities, their excellent biocompatibility, controlled release properties, and clinical scalability. Compared to other nanocarriers such as transferosomes, which are more suited for transdermal delivery, and polymerosomes, which are more complex and less biocompatible, nanolipoomes offer a simpler yet highly effective platform. Additionally, their surface can be easily functionalized—such as with aptamers—for targeted delivery, making them particularly suitable for our multi-drug, multi-targeted anticancer strategy.27–29
Aptamers, single-stranded DNA or RNA molecules, have emerged as promising tools for cancer prevention and treatment. 30 These short synthetic nucleic acid sequences possess unique three-dimensional structures that enable them to bind selectively to specific target molecules, including cancer-related proteins.31,32 Their specificity and high binding affinity make them valuable candidates for application in various cancer research. 33 Aptamers hold the potential for cancer prevention through their ability to recognize and bind to biomarkers associated with early-stage cancer development. Early detection is crucial for improving cancer prognosis, and aptamers can contribute to the development of sensitive and specific diagnostic assays. Additionally, aptamers can be engineered to inhibit the activity of oncogenic proteins or pathways, thereby impeding cancer progression. 34 In the context of cancer treatment, aptamers can be designed to directly target cancer cells or deliver therapeutic agents with precision. 35 Functionalized aptamers can be conjugated to drugs, nanolipoomes, or other therapeutic payloads to facilitate targeted drug delivery to cancer cells, which enhances therapeutic efficacy while minimizing damage to healthy tissues. 36 Several cancer target-specific aptamers have been identified and studied, including AS1411, which targets nucleolin, a protein overexpressed in cancer cells. 37 Aptamers have shown potential in preclinical investigations and early-stage clinical trials for cancer treatment. However, the availability of FDA-approved aptamer-based drugs for cancer therapy remains limited. 35
In this study, our primary aim was to develop and characterize an aptamer-conjugated PEGylated nanoliposomes encapsulating Raloxifene and Naringin, along with a β-Cyclodextrin-Etoricoxib complex.38,39 This study delivers the first liposome that packs three complementary anticancer drugs and actively seeks tumour cells
The rationale behind the selected triple combination—Raloxifene, Naringin, and the β-Cyclodextrin–Etoricoxib complex—is based on their complementary mechanisms of action, including estrogen receptor modulation, 40 anti-inflammatory effects, and inhibition of drug efflux pumps, 41 which collectively aim to enhance anticancer efficacy while reducing systemic toxicity. 42 By co-encapsulating these agents within PEGylated nanoliposomes and functionalizing the surface with the AS1411 aptamer to target nucleolin-overexpressing cancer cells, 43 we hypothesize that the formulation will offer improved drug stability, targeted delivery, and superior cytotoxicity, representing a promising multi-targeted therapeutic strategy for cancer treatment.
To evaluate the broad-spectrum potential of the proposed formulation, we selected breast (MCF-7), lung (A549), and colon (HT-29) cancer cell lines—representing malignancies with distinct etiologies—all of which exhibit nucleolin overexpression, the molecular target of the AS1411 aptamer. This strategic selection supports the rationale for aptamer-mediated targeting and allows for assessing the formulation's efficacy across multiple nucleolin-positive cancer types. 44
The result is a monodisperse nanoliposome that stays stable, releases its cargo in a controlled fashion, and shows markedly higher cytotoxicity in MCF-7, HT-29 and A549 cancer cells than any single or dual treatment. By melding multi-pathway pharmacology with precise aptamer targeting, we offer a new, more potent way to attack tumours while sparing healthy tissue.43,45
This study presents a novel triple-drug-loaded, aptamer-conjugated nanoliposome formula targeting cancer cells via nucleolin. The approach enhances drug efficacy and reduces side effects, with improved cytotoxicity observed in various cancer cell lines. These findings suggest a promising future for this formula in cancer therapy.
Materials and Methods
Cells and Materials
Methods
Etoricoxib Complexation with Hydroxypropyl-β-Cyclodextrin
Etoricoxib was complexed with hydroxypropyl-β-cyclodextrin (HPβCD) using the kneading method. Equal weights of etoricoxib and HPβCD were mixed in a mortar. Aqueous ethanol (50%) was gradually added as a kneading vehicle until a paste was formed. Subsequently, the paste was manually triturated in one direction for 1 h and dried at 38 °C for 24 h. The dried material was scraped from the mortar, sieved, and stored until needed. This technique was selected for its simplicity, low solvent requirement, and proven ability to enhance complexation efficiency without the need for specialized equipment, making it suitable for laboratory-scale formulation development. 46
Nuclear Magnetic Resonance Spectroscopy Investigation of Complexation
Following Etoricoxib complexation with HPβCD, nuclear magnetic resonance (NMR) spectroscopy was performed in d6-DMSO (99.0% atom, Across Organics, USA) using a Bruker Avance III 500 mHz instrument (Bruker BioSpin, Switzerland) with a broadband observer (BBO) probe equipped with a z-gradient. The instrument temperature was controlled using a variable temperature unit (VTU) and held constant at 300 K. Chemical shifts in parts per million (ppm) were referenced to tetramethylsilane (TMS) as an internal reference. Data processing and analysis were performed using the Topspin framework (Bruker Biospin GmbH, Version 3.6.0) (Supplementary 1, Appendix 1).
Nanoliposome Formulation (Thin-Film Hydration Method)
Next, we prepared the nanoliposome formulation using the thin-film hydration method following published specifications. 47 The formula consisted of DSPC: Cholesterol: DSPE-PEG (2000) Amine in a ratio of 65:30:5, with a fixed loading of Raloxifene and Naringin (2 mg/mL each) and β-cyclodextrin-etoricoxib (4 mg/mL) at a total lipid concentration of 0.51 mmol (Figure 1). 48 This formulation was constructed to investigate the combined impact of Raloxifene, β-cyclodextrin-etoricoxib, and Naringin on nanoparticle characterization.

Schematic Representation of the Thin Film Hydration- Extrusion Method.
Nanoliposome Formulation (NCL Aptamer Post-Insertion)
The nanolipoomes were decorated with 3′-cholesterol tagged-anti-nucleolin AS1411 aptamer (NCL) using a post-insertion approach, utilizing the cholesterol as a wedge to incorporate aptamer into the surface of the liposome bilayer. For post-insertion, the NCL aptamer was developed by incubating nanolipoomes lipids (1 mg) with 3′-cholesterol tagged NCL (2.0 nmol) for 1 h at 60 °C with continuous stirring, allowing the hydrophobic cholesterol moieties to anchor within the liposome's bilayer. Following aptamer post-insertion, the unconjugated free aptamers (MW of 13.7 kD) were removed via ultrafiltration at 13 500 rpm using 100 KD Amicon filters, Merck Millipore® Ltd (Ireland). Subsequently, the NCL aptamer was folded into its G-quadruplex structure using a binding buffer containing 5 mM PBS, 2.5 mM MgCl2, and 140 mM KCl. 43
Characterization of Nanoliposomes
Encapsulation Efficiency, Size, Polydispersity index, and zeta Potential
The nanoliposomes were then characterized to evaluate their physicochemical properties. The average particle size, zeta potential (charge), and polydispersity index (PDI) of the nanoliposomes loaded with Raloxifene, Naringin, etoricoxib-formula-1 (RNE-f1), and the aptamer-RNE-formula 2 (Apt-RNE-f2), were measured using dynamic light scattering (DLS) on a Zetasizer (Malvern Instruments Ltd, Malvern, UK). The samples were diluted (1:20) with distilled water before measurement. 18
Structure and Morphology of Nanoliposomes
The structure and morphology of the aptamer-functionalized RNE-loaded nanoliposomes were analyzed using transmission electron microscopy (TEM). TEM analysis of the liposome samples was conducted using the negative staining method following previously published specifications. 47 First, 200 mesh formvar copper grids (SPI Supplies, USA) were coated with carbon using a low-vacuum Leica EM ACE200 glow discharge coating system (Leica, Austria). The carbon-coated grids were then coated with a 1.5% vinyl K solution in chloroform. A drop of deionized water-diluted liposome suspension was placed into the 200 mesh formvar copper grid and air-dried. The loaded grids were then stained with 3% (v/v) aqueous solution of uranyl acetate and incubated for 20 min at room temperature. After incubation, the grids were washed with distilled water and dried at room temperature before imaging using a Versa 3D TEM operating system (FEI, Netherlands) at an acceleration voltage of 30 kV. 39
Encapsulation Efficiency
Encapsulation efficiencies of Naringin, Etoricoxib, and Raloxifene were determined using high-performance liquid chromatography with UV detection (HPLC-UV), based on a method adapted from Ishii et al (J Chromatogr B Biomed Sci Appl. 1997;704(1-2):299-305). Compound-specific modifications were applied to optimize detection parameters: Naringin was quantified using the original method settings, while Etoricoxib and Raloxifene were analyzed at their respective optimized wavelengths and mobile phase compositions. A unified sample preparation protocol was employed for all analytes. The method was validated for each drug, demonstrating linearity (R² > 0.99) and precision (RSD < 5%), ensuring reliable quantification of encapsulated compounds. 49
Stability Investigation of Nanoliposomes Loaded with RNE (F1) and Apt-RNE (F2)
Stability After Lyophilization
Liposomal formulations were lyophilized to enhance their long-term stability. Prior to lyophilization, a cryoprotectant—sucrose—was added at a final concentration of 10% w/v to protect the liposomal structure during the freezing and drying processes. The nanolipoomes–sucrose mixture was first frozen at −80 °C for 12 h, followed by primary drying at −40 °C under vacuum (≤0.1 mbar) for 24 h, and secondary drying at 20 °C for 6 h to remove residual moisture. This protocol helped maintain the physicochemical characteristics and integrity of the nanolipoomess after reconstitution. The inclusion of sucrose effectively preserved particle size and encapsulation efficiency post-lyophilization. 50
The stability of the encapsulated nanoliposome after and under storage conditions was also investigated. Nanoliposome suspensions loaded with RNE-f1 and Apt-RNE-f2 were prepared using the Thin-Film Hydration under optimized conditions according to the specifications of the previously published reports.11,47,48
Stability Under Storage Conditions at 2 °C
Stability under storage conditions at 2 °C was assessed for nanoliposome suspensions loaded with RNE-f1 and Apt-RNE-f2. The nanoliposome suspensions loaded with RNE-f1 and Apt-RNE-f2 were stored at 2 °C and the size, charge, and PDI were examined at different time intervals (24, 48, 72, 96, 120, 144, and 168 h) to assess the stability of the nanoliposome suspensions under storage conditions at 2 °C. The measurements were compared with those of freshly prepared samples.47,48
Thermogravimetric Analysis (TGA)
Additionally, the thermal properties of the prepared nanolipoomes were determined using thermogravimetric (TGA) analysis (Netzsch-Proteus, 209F1_Iris, Selb, Germany). Lyophilized samples (5 mg each) were placed in open aluminum pans and heated at an increased rate of 10 °C/min to reach the temperature range of 40-500 °C under N2 flow (60 mL/min). 51
In Vitro Release
The
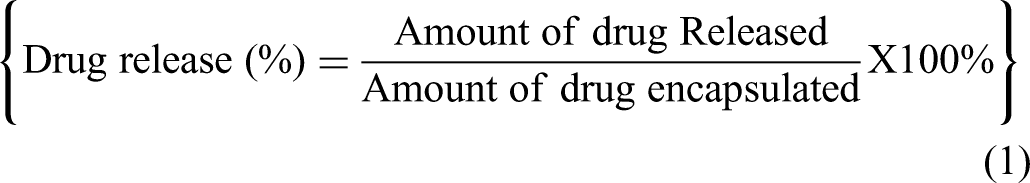
Cell Culture
For cell cultures, MDA-MB-231 (triple-negative breast cancer cell line), A549 (human non-small lung cancer cell line), Immortalized Human Vascular Endothelial Cells(EA.hy926,) and HT-29 (human colorectal adenocarcinoma cell line) cells were cultured in Dulbecco's modified eagle medium (DMEM; Gibco). MCF-7 (ER + breast cancer cell line) cells were cultured in RPMI 1640. All cultured media were supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBS) (EuroClone, Italy), 1% penicillin-streptomycin (EuroClone, Italy), and 2 mM L-glutamine. The cell lines were incubated at 37 °C under a 5% CO2 atmosphere and 90% relative humidity.47,48
Viability Assays
Cell toxicity assessment was conducted on a group of substances comprising etoricoxib, Naringin, etoricoxib-cyclodextrin, Raloxifene, and RNE co-loaded. Cytotoxicity was evaluated by measuring the vitality of cells in the culture environment. Cells were initially distributed into 96-well plates at a density of 1 × 104 cells per well and were incubated for 24 h at 37 °C in DMEM. Subsequently, these cells were exposed to varying concentrations at 1.5, 3, 6,12, 25, 50, 100, and 200 µg/mL of Etoricoxib, Naringin, Etoricoxib-β-cyclodextrin, Raloxifene, and RNE co-loaded for an additional 48 h. Following this exposure, an MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide)assay was carried out in the following manner: the culture medium was withdrawn, and the cells in each well were treated with a 20 µL solution of MTT (5 mg/mL) for 4 h at 37 °C. The MTT solution was then removed and replaced with 200 µL of dimethyl sulfoxide (DMSO) to dissolve any insoluble formazan crystals. The optical densities were measured at two distinct wavelengths: 570 and 630 nm. The data were collected from three replicates. Selective cytotoxicity was assessed using human endothelial cells as the primary cell culture to determine the lowest IC50 value for antiproliferative activity. All assays were performed in triplicate, and the derived IC50 values for antiproliferative activity were reported as mean ± standard deviations (n = 3).
Combination Index (CI) Determination
The interaction between Etoricoxib, Naringin, and Raloxifene in the co-loaded nanoliposome (RNE) formulation was assessed using the Chou-Talalay method based on IC₅₀ values from MTT assays. 53 Cancer cells (MCF-7, A549, HT29) were treated with individual drugs and the combination across a concentration range (1.5-200 µg/mL) for 48 h. IC₅₀ values were calculated, and the CI was determined using Equation 2:

Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows version 22. Descriptive statistics, including mean and standard deviation, were used to summarize the central tendency and variability of the dataset. Various statistical tests such as two-sample t-tests, paired t-tests, ANOVA, and repeated-measures ANOVA were employed for comparative analyses. Furthermore, graphical representations were created using GraphPad Prism to illustrate the key trends and patterns in the data. The combined use of SPSS and GraphPad Prism enabled comprehensive analysis and offered valuable insights into the outcomes of our study.
Results
NMR Analysis of Etoricoxib-β-Cyclodextrin complex Formation
The 1H-NMR spectrum of Etoricoxib in the absence of HPβCD exhibited a pair of doublets, each integrating for two protons and showing an A2B2 pattern at δ = 7.54 and 7.52 ppm, which were assigned to H-3/5 and H-2/6, respectively, of the methyl sulfonyl-substituted aromatic ring. The signal for the methyl group of methyl sulfonyl-substituted phenyl ring appeared as a singlet at δ = 3.25 ppm, while the singlet at δ = 2.33 ppm for the three protons was because of the methyl group of the pyridine ring (H-6′). Three doublets that appeared at 7.18, 8.30, and 8.80 ppm were assigned to the H-5,′ H-4,′ and H-2′ protons of the methyl-substituted pyridine ring, respectively. Additionally, two doublets appeared at 7.90 and 8.08 ppm, each integrating for one proton, and were attributed to the chloro-substituted pyridine ring protons (H-6ʺ and H-4ʺ). Significant changes in the nature and position of signals for the protons of Etoricoxib were observed in the presence of HPβCD products. The signals for H-3/5 of the methyl sulfonyl-substituted aromatic ring exhibited an upfield shift in both binary systems of Etoricoxib with HPβCD, whereas those for H-2/6 protons exhibited a significant downfield shift. A significant upfield shift was observed in the H-2/6 signals of the products. However, the signals for H-2/6 in the products were shifted downfield. The peaks corresponding to the protons of the methyl group of the sulfonyl group shifted downfield. For the two pyridine rings, the signals for the H-5,ʹ H-4,ʹ H-2′, and H-6ʺ protons exhibited a downfield shift in the presence of HPβCD. In contrast, upfield shifts were observed for the CH3 and H-4ʺ protons of the methyl group of pyridine- and chloro-substituted pyridine ring, respectively, in the products. Notably, the signals for all protons, except the methyl group of the Etoricoxib pyridines, exhibited a significant downfield shift in the product. In all spectra of systems of Etoricoxib with HPβCD, the signals for protons of HPβCD, situated inside the HPβCD cavity, exhibited upfield shifts compared with the pure HPβCD. Figure 2 shows the Etoricoxib-β-Cyclodextrin complex.
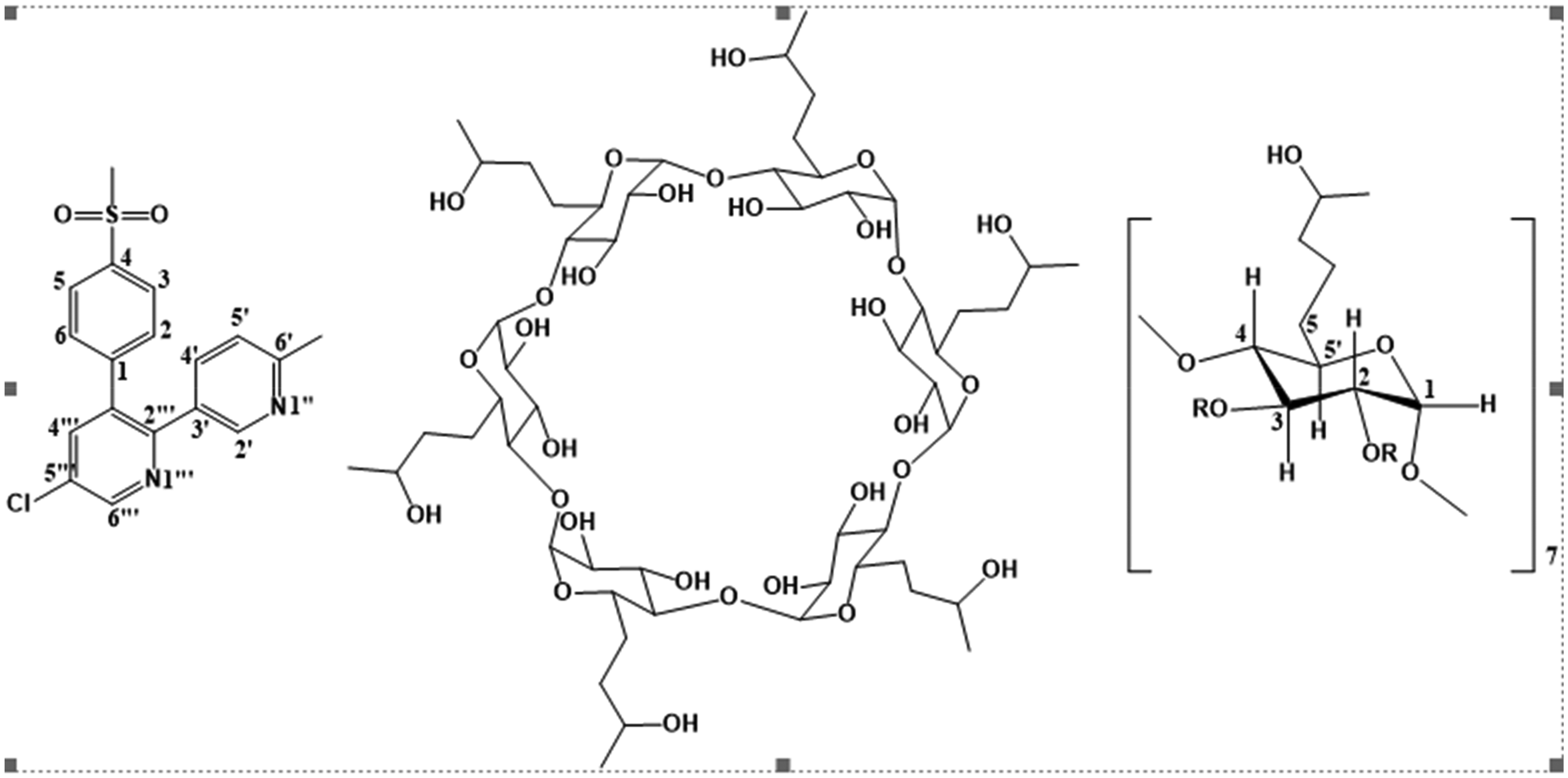
Etoricoxib-β-Cyclodextrin Complex.
Nanoparticle Characterization: Size, Charge, PDI, Encapsulation Efficiency, and Structure Morphology
Investigation of the Impact of Aptamer Conjugation on Size, Charge, and PDI Characterization
In this study, we compared the size (nm), PDI, and charge (mV) of RNE-loaded nanoliposomes and Apt-RNE-loaded nanoliposomes. All data fell within the optimal range for nanoliposome characterization, with sizes below 300 nm, PDIs less than 0.4, and charges within the accepted range of −20 to −20 mV. However, our measured zeta potential values were around −15 mV. This negative surface charge contributes to sufficient electrostatic repulsion, helping maintain the physical stability of the PEGylated nanoliposomes. Moreover, the presence of PEG chains on the liposome surface provides steric stabilization, which further enhances colloidal stability by preventing aggregation even at relatively low zeta potential values.
54
Two-sample t-test results indicated significant differences in size (

Comparison of Size (nm), Polydispersity index (PDI), and Charge (mV) Between Nanoliposomes Loaded with RNE (F1) and Those Loaded with Apt-RNE (F2).
Investigation of the Impact of Aptamer Conjugation on EE
A Two-sample test was also performed to determine significant differences in EE of each active constituent in the RNE formula before and after aptamer conjugation. Naringin and etoricoxib showed no significant differences in EE (
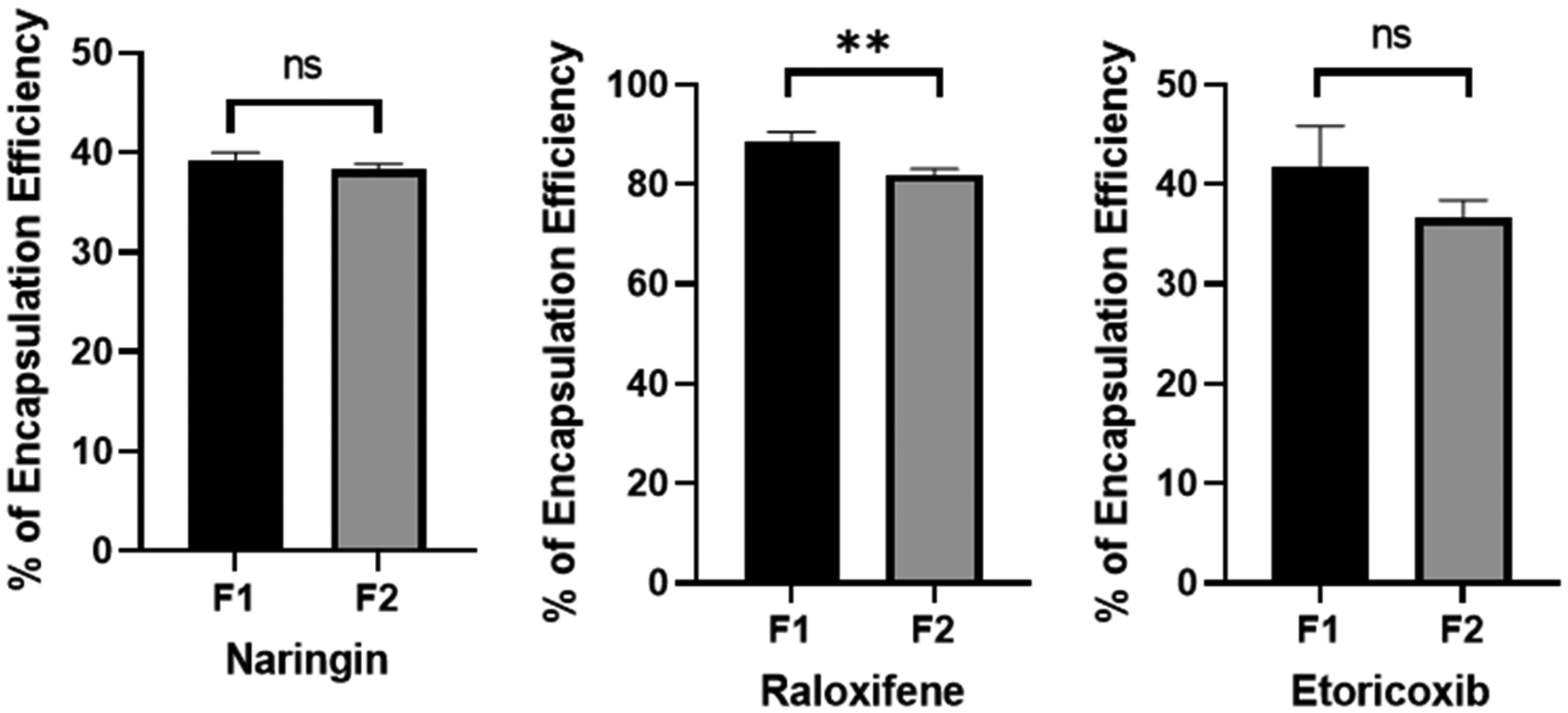
Impact of Aptamer Conjugation on Encapsulation Efficiency. F1
Structure and Morphology of Nanoliposomes Loaded with Apt-RNE (Transmission Electron Microscopy)
The TEM analysis revealed the successful formulation of nanoliposomes loaded with Apt-RNEs, expressed in uniform spherical shapes. The data suggest an average particle size of approximately 62.5 ± 25.5 nm (n = 20). Remarkably, the images clearly show that all nanolipoomes were thoroughly encapsulated with the drugs. The core contains Etoricoxib-β-cyclodextrin, while the bilayer encapsulates Raloxifene and Naringin, distinguishing them as dark nanolipoomes (Figure 5).

Structure and Morphology of Nanoliposomes Loaded with Apt-RNE.
Stability Studies
Size, Charge, and PDI Tests Were Performed to Assess the Effect of Lyophilization and low Temperature (2 °C). on RNE-F1 and RNE-F2
Lyophylization
The particle size (d.nm), PDI, and charge (mV) of the constructed nanoliposomes in both formulations remained within the optimal range. For the RNE-F1 formula, a significant increase in both size and PDI value was recorded after lyophilization (

(A) Comparison of Size (nm), Polydispersity Index (PDI), and Charge (mV) between Nanoliposomes Loaded with RNE (F1) Before and After Lyophilization, (B) Comparison of Size (nm), PDI, and Charge (mV) between Nanoliposomes Loaded with Apt-RNE (F2) Before and After Lyophilization.
For the RNE-f2 formula, the particle size (nm), PDI, and charge (mV) of the constructed nanoliposomes in both formulas were within the optimal range. Significant differences were observed in the size, PDI, and charge of the aptamer-conjugated formulations before and after lyophilization (
Low Temperature (2 °C)
The particle size (nm), PDI, and charge (mV) for the constructed nanoliposomes in both formulas remained within the acceptable range for 5 days in RNE-f1 and 2 days in aptamer-RNE-f2 after preparation. The repeated measures ANOVA analysis for both formulas revealed significant size differences 48 h post-storage (

Changes in the RNE-f1 Characteristics (Size, PDI, and Charge) During Storage at 2 °C Over the Experiment Time.
Effects of Aptamer Conjugation and Lyophilization on EE of Each Therapeutic Agent
The stability of the EE following lyophilization of RNE-loaded nanoliposomes was assessed using a paired t-test. Significant differences were observed in the EE of Naringin before and after lyophilization (39.26 ± 0.74 and 31.32 ± 0.20, respectively;

Stability of Encapsulation Efficiency After Lyophilization of Nanoliposomes Loaded with RNE.

Stability of Encapsulation Efficiency After Lyophilization of Nanoliposomes Loaded with Apt-RNE.
Thermography Analysis of Nanoliposomes Loaded with Apt-RNE
The TGA conducted at temperatures exceeding 400 °C provided valuable insights into the thermal stability of the Apt-RNE-loaded nanoliposomes compared with their RNE-loaded nanoliposomes, as shown in Figure 10. The co-loaded system exhibited enhanced thermal stability compared with the single components loaded system, their potential suitability for high-temperature applications. The observed enhancement in thermal stability underscores the potential benefits of the encapsulation approach in preserving the integrity of the incorporated therapeutic agents, as shown in Figure 10.
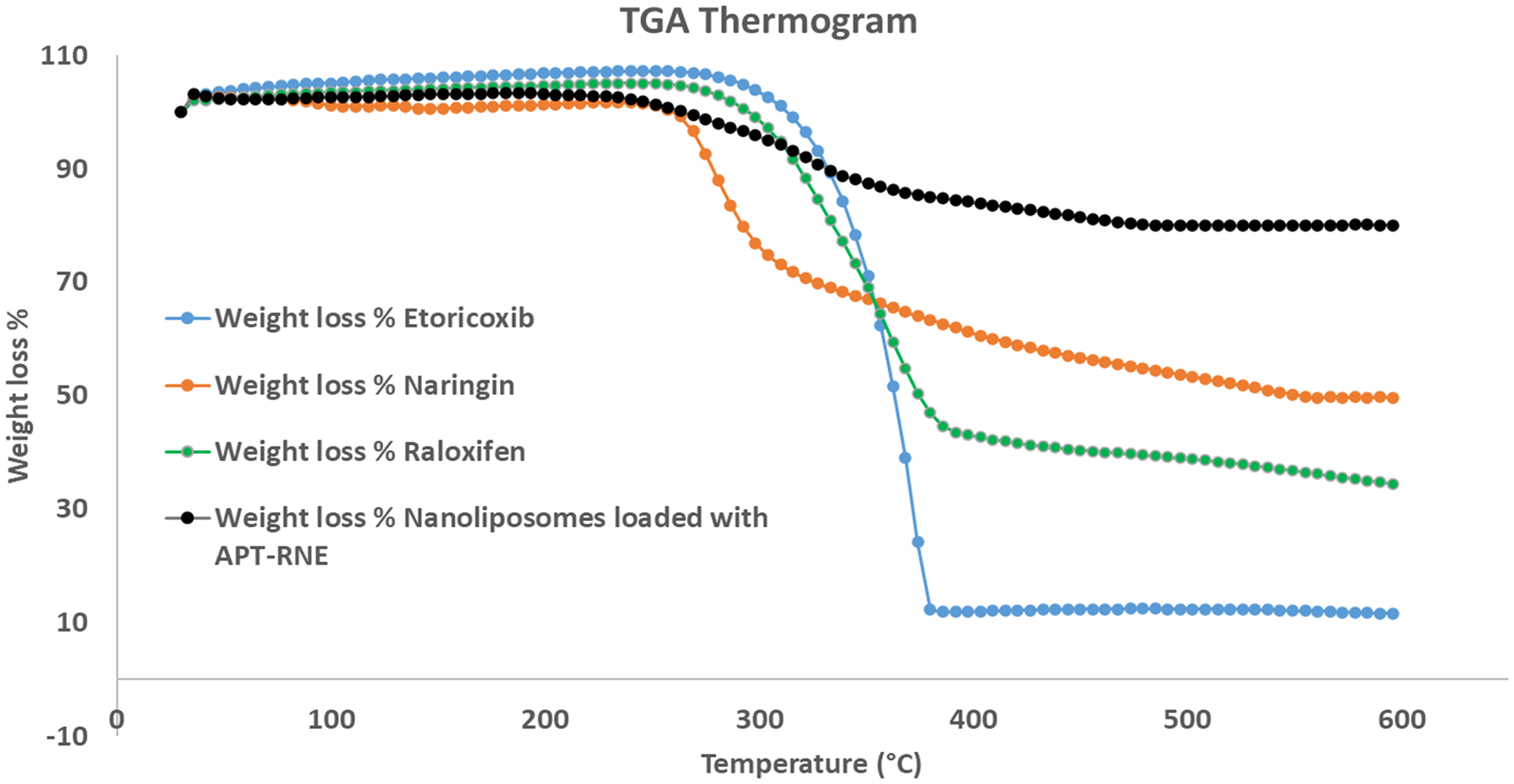
TGA of Nanoliposomes Loaded with Apt-RNE, Raloxifene, Naringin, and Etoricoxib.
In Vitro Release
The

in Vitro Release Profiles of Drug-Loaded Nanoliposomes (F1) and Drug-Loaded with Aptamer Conjugated-Nanoliposome at 37 °C.
Biological Activity
Viability Assay
The cytotoxicity of the tested formulations in MCF7 and A549 cells was evaluated at a range of concentrations high concentrations over 48 h. Different response patterns were obtained: the percentage inhibition of HT-29 cells was significantly lower for free Etoricoxib than for the nanoformula (
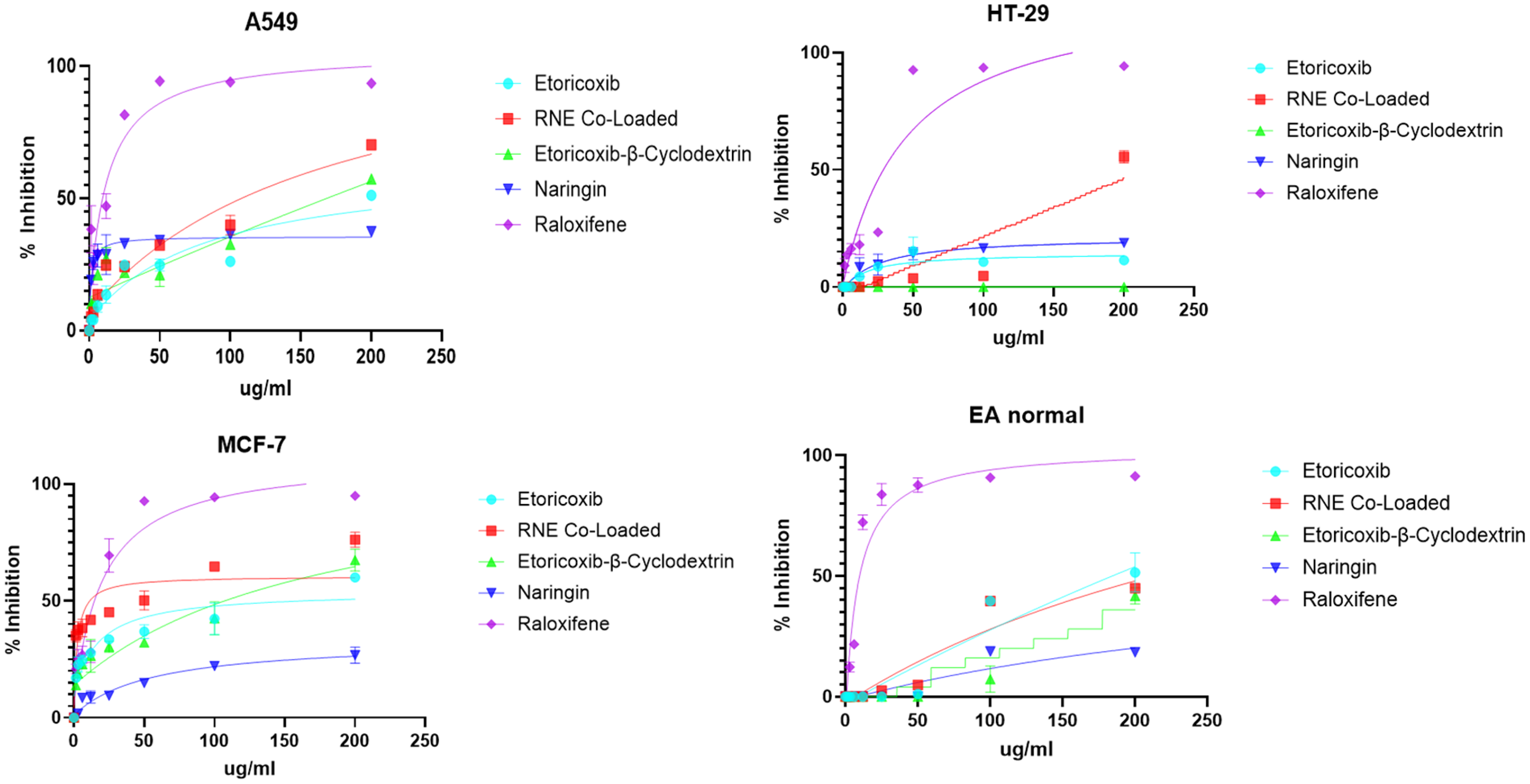
Antiproliferative Activities of Different Formulas at a Range of Concentrations on Selected Cancer Cell Lines. Values are Represented by the Mean of the Percent of Inhibition of Three Dependent Replicates ± SD.
The tested agents (Etoricoxib, Naringin, Etoricoxib-β-cyclodextrin, and nanoliposomes-RNE) exhibited negative cytotoxic effects on EA cells (Table 1). Moreover, significant cytotoxicity of these formulations was observed against HT29, MCF7, and A549 cell lines after 48 h of incubation. The nanoliposome-RNE formulation demonstrated a significant decrease in A549 cell viability compared with free Etoricoxib and Naringin (
IC50 Values (µg/mL) of in Vitro Antiproliferative Activity of Etoricoxib, Naringin, Etoricoxib-β-Cyclodextrin, Raloxifene, and RNE co-Loaded on Different Cancer Cell Lines.

Results mean ± SD (n = 3 independent replicates). IC50 values (concentration at which 50% inhibition of cell proliferation took place in comparison to non-induced basal 48 h incubations).
Combination index (CI) Analysis
The combination index (CI) analysis based on IC₅₀ values from MTT assays revealed distinct interaction patterns across different cancer cell lines. In MCF-7 cells, the CI for the RNE co-loaded formulation (containing Raloxifene, Naringin, and Etoricoxib) was 0.11, indicating strong synergism among the three drugs. In contrast, the CI value in A549 cells was 45.38, suggesting a strong antagonistic effect, likely due to the disproportionate sensitivity of the cells to Naringin when administered alone. No CI could be calculated for HT29 cells, as the co-loaded formulation did not achieve 50% inhibition, indicating insufficient efficacy. These findings highlight the importance of cell line-specific responses in combination therapies and suggest that while the RNE formulation may be highly effective against certain cancer types like breast cancer (MCF-7), its efficacy and drug interactions vary significantly in others.
Effect of Aptamer Conjugation on Selectivity and Cytotoxicity
Cytotoxicity of the prepared formulations was tested against various cell lines including MDA-MB-231, MCF7, and EA normal cells (human dermal fibroblasts). The results revealed a slight increase in Apt-RNE co-loaded activity against MDA-MB-231 cells at lower concentrations over 24 h compared with the RNE co-loaded formula. A significant increase in activity was observed at higher concentrations. The aptamer-containing formula showed a notable increase in activity against MCF7 cancer cells compared with the aptamer-free formula. Interestingly, the addition of the aptamer had no significant effect against human dermal fibroblasts, indicating selective cytotoxicity against cancerous cells. The mean percentage inhibition at various concentrations for each formula against the tested cell lines is shown in Figure 13.

Impact of various Treatments on the Antiproliferative Activity Against the MDA-MB-231 Cancer Cell Line. Cells Were Subjected to Varying Concentrations of RNE co-Loaded and Apt-RNE co-Loaded. Values are Represented by the Mean Percentage of Inhibition from Three Independent Replicates ± SD.
Discussion
In the present study, we successfully developed a triple-loaded nanoliposomal system incorporating Raloxifene, Naringin, and Etoricoxib using the thin-film hydration method, followed by extrusion to achieve size uniformity. A key focus of this work was to evaluate the impact of aptamer (AS1411) conjugation on the physicochemical and biological performance of the liposomal formulation.
The comparative analysis between aptamer-functionalized and non-functionalized nanoliposomes revealed a significant increase in particle size upon aptamer incorporation. This enlargement is attributed to the additional hydrodynamic layer introduced by the aptamer strands, in agreement with previous findings. 55 The PDI also increased, suggesting a broader size distribution and potential heterogeneity in the aptamer-containing formulation. This shift in PDI is a common observation in complex surface-modified systems and may indicate aggregation tendencies, although the overall size remained within a pharmaceutically acceptable range. 56 Interestingly, no significant change in zeta potential was observed post-aptamer conjugation, indicating that the surface charge and associated electrostatic repulsion—critical for colloidal stability—were preserved.57–59 These findings confirm that the aptamer attachment did not compromise the system's stability, thereby maintaining its potential for targeted delivery.
Thermogravimetric analysis (TGA) highlighted the improved thermal stability of the co-loaded nanoliposomes compared to individual components, supporting the protective role of encapsulation during processing and storage. 60 Furthermore, the inclusion of cyclodextrins—specifically HP-β-CD—significantly improved the solubility of Etoricoxib, a poorly water-soluble drug (Class II), which enhanced its incorporation efficiency and controlled release behavior.27,61
These findings align with prior work advocating cyclodextrins as valuable excipients for enhancing the solubility and bioavailability of hydrophobic drugs.25,27
Etoricoxib was complexed with HP-β-CD to improve its solubility and bioavailability, facilitating incorporation into nanoliposomes and ensuring controlled release and preventing rapid drug release.
62
Both formulations (F1 and F2) exhibited sustained release of Etoricoxib, with no significant differences in the maximum release after 24 h (96.58 ± 2.91% for F1 and 95.28 ± 1.58% for F2;
The absence of significant differences in drug release between F1 (RNE-loaded nanoliposomes) and F2 (Apt-RNE-loaded nanoliposomes) suggests that aptamer conjugation did not notably impact the release behavior. This is likely due to the dialysis membrane method used in the in vitro release study, which may have controlled the diffusion rate and overshadowed any subtle effects of aptamer addition. 64 Naringin and Raloxifene both demonstrated a biphasic release profile, consisting of an initial burst release attributed to surface-associated drug, followed by a slower release phase likely driven by diffusion or liposomal degradation. In contrast, Etoricoxib showed a steady and nearly complete release in both formulations, which can be attributed to its higher solubility and efficient diffusion. Overall, the release kinetics were consistent across F1 and F2 for all three drugs, indicating that the aptamer modification did not alter the fundamental release mechanisms under the given experimental conditions.
The cytotoxicity assays revealed a dose-dependent response for all formulations. Notably, the triple-loaded liposomes significantly reduced cell viability compared to individual agents, likely due to the synergistic effects of the combined drugs and enhanced cellular uptake. While free drugs showed faster action, potentially due to passive diffusion, the liposomal formulations provided sustained and targeted delivery.65,66
Raloxifene is known for its antiproliferative activity through estrogen receptor modulation and apoptosis induction, 67 while Naringin contributes antioxidant effects by reducing reactive oxygen species (ROS), 68 and Etoricoxib adds anti-inflammatory and antiangiogenic activity via COX-2 inhibition. 69 Their co-delivery within a single liposomal carrier enables a multifaceted attack on cancer cells, addressing different biological targets.
The RNE-co-loaded formula exhibited a significant reduction in cell viability compared with the individual therapeutic agents. Interpretation of the data obtained was based on previous findings. Etoricoxib initiates antiproliferative activity and induces the apoptosis of angiogenic endothelial cells. 23
The incorporation of AS1411 aptamer further enhanced the cytotoxic effect of the triple formula. AS1411's known mechanisms—such as Bcl-2 destabilization, inhibition of NF-κB-mediated survival pathways, and reduction of nucleolin-dependent miRNA—may contribute to this heightened efficacy. 70
These findings correlate with previously published reports. Raloxifene loaded into phospholipids with based vesicles with melittin has been shown to have an enhanced cytotoxic activity against pancreatic cancer cells by the interception of the cell cycle in the G2-M phase, increased apoptosis, elevated expression of TNF-α and Bax, and a significant reduction in Bcl-2 and NF-kB expression. 71 , A COX-2 inhibitor synergized with a Cholecystokinin receptor-2 (CCK-2) antagonist in pancreatic cancer cells and impeded cell proliferation while influencing apoptosis and angiogenesis. 72 Similarly, the anti-breast cancer effects of Raloxifene were enhanced after formulation into emulsomes, and formulated etoricoxib has been assessed for activity against breast, colon, and skin cancerous cells.11,20 Flavonoids, including Naringin, modulate chronic diseases and cancers by reducing the production of reactive oxygen species and exerting anantioxidant activity.48,73 Our findings align with previous reports where aptamer-functionalized nanocarriers, such as those encapsulating Apigenin or Curcumin, showed improved cellular uptake and cytotoxicity against various cancer cell lines.74,75
In comparison, our study combined three different therapeutic agents, Raloxifene, Naringin, and Etoricoxib, into a single nanoliposome. The EE of 88.63% for Raloxifene and the moderate efficiencies for Naringin (41.73%) and Etoricoxib (39.26%) were high for the triple-drug formulation, demonstrating the successful development of a multi-drug liposomal system with superior stability and enhanced drug encapsulation, compared with typical dual-drug formulations.
The encapsulation efficiency of Naringin and Etoricoxib (∼40-50%) was moderate but consistent with values reported for similar multi-drug liposomal systems. Their lower EE compared to Raloxifene may be due to limited lipid solubility. Optimization of lipid composition, pH conditions, or loading methods could improve EE in future formulations. 76
Taurin et al developed a styrene-co-maleic acid (SMA) micelle to encapsulate Raloxifene. SMA-Raloxifene micelles showed higher cytotoxicity against PC3 and DU 145 prostate cell lines (132% and 140%, respectively). SMA-Raloxifene effectively inhibited cell cycle progression, increased apoptosis, and altered the integrity of the tumor spheroid models.
77
Additionally, Alhalmi et al developed a dual-drug-loaded nanostructured lipid carrier consisting of Raloxifene and Naringin (RLX/NRG NLCs) for breast cancer. The optimized RLX/NRG NLCs exhibited a particle size of 137.12 nm, PDI of 0.266, zeta potential (ZP) of 25.9 mV, and entrapment efficiency of 91.05% and 85.07% for Raloxifene) and Naringin, respectively.
Our study presents the first combination of Raloxifene, Naringin, and Etoricoxib into a single nanoliposomal system, each addressing different aspects of cancer biology including estrogen receptor signaling, oxidative stress, and COX-2-mediated inflammation. This work pioneers a trimodal nanomedicine that fuses three complementary anticancer mechanisms into a single, precisely guided vehicle. Raloxifene blocks estrogen-dependent signalling, naringin augments apoptosis and counters oxidative stress, and the Etoricoxib/β-cyclodextrin complex suppresses COX-2-driven inflammation while the cyclodextrin ring boosts the drug's aqueous solubility and protects it from rapid metabolism. By co-encapsulating these agents in the bilayer of long-circulating PEGylated nanolipoomes, we ensure synchronous, sustained release at the tumour site and shield each molecule from premature degradation. Decorating the liposome surface with the AS1411 aptamer adds a third layer of innovation: the oligonucleotide locks onto nucleolin receptors that are abundant on many cancer cells but scarce on healthy tissue, so the carrier is pulled preferentially into malignant cells by receptor-mediated endocytosis. In vitro, this targeting step magnified cytotoxicity well beyond that of non-targeted nanolipoomes or any single drug, confirming a genuine synergistic effect rather than simple additivity. The net result is a formulation that can deliver lower individual doses yet achieve stronger tumour kill, thereby diminishing the dose-limiting cardiotoxicity of raloxifene, the gastrointestinal risks of etoricoxib, and the systemic oxidative stress sometimes associated with high naringin levels. Taken together, these attributes position our aptamer-conjugated, tri-drug liposome as a next-generation platform that could transform combination chemotherapy by uniting multi-pathway attack, smart targeting and pharmacokinetic enhancement in one clinically translatable package.However, a limitation of this study is that the
Conclusions
We created the first PEGylated nanoliposome that co-loads raloxifene, naringin and an etoricoxib/β-cyclodextrin complex and decorates its surface with the AS1411 aptamer, a design that simultaneously boosts the solubility and stability of all three drugs, exploits their complementary anticancer pathways, and guides the carrier directly to nucleolin-rich tumour cells for controlled, sustained release; this one-step fusion of multi-drug synergy, cyclodextrin-assisted loading, and aptamer targeting has not been reported before and promises higher efficacy with lower off-target toxicity, meriting in-vivo validation.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251356548 - Supplemental material for Novel Anticancer Triple Formula Based on Aptamer-Conjugated PEGylated Nanoliposomes
Supplemental material, sj-docx-1-tct-10.1177_15330338251356548 for Novel Anticancer Triple Formula Based on Aptamer-Conjugated PEGylated Nanoliposomes by Ali Al-Samydai, Hamdi Nsairat, Moath Alqaraleh, Maha N. Abu Hajleh, Areej Jaber, Lidia Al-Halaseh, Hanan Azzam, Qasim Khalid Alazzawi, Israa Al-Ani, Simone Carradori and Walhan Alshaer in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338251356548 - Supplemental material for Novel Anticancer Triple Formula Based on Aptamer-Conjugated PEGylated Nanoliposomes
Supplemental material, sj-docx-2-tct-10.1177_15330338251356548 for Novel Anticancer Triple Formula Based on Aptamer-Conjugated PEGylated Nanoliposomes by Ali Al-Samydai, Hamdi Nsairat, Moath Alqaraleh, Maha N. Abu Hajleh, Areej Jaber, Lidia Al-Halaseh, Hanan Azzam, Qasim Khalid Alazzawi, Israa Al-Ani, Simone Carradori and Walhan Alshaer in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
Only the listed authors contributed to this work and no other individuals or collaborators were involved in this work.
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Research Ethics and Patient Consent
This study did not involve any experiment on animal or human subjects.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
