Abstract
Introduction
Uterine sarcoma (US) is a rare tumor characterized by high aggressiveness, a tendency for recurrence and distant metastasis, and an extremely poor prognosis. In this study, we evaluated the expression of Orosomucoid 1 (ORM1) in different subtypes of US and the relationship between survival rates and clinicopathological features.
Method
A retrospective study was conducted on 50 cases diagnosed with US in our hospital from 2011 to 2023. Immunohistochemistry (IHC) was used to detect the expression levels of ORM1 in different subtypes of US.The chi-square test and Kaplan-Meier survival analysis were used to analyze the relationship between ORM1 expression and clinical parameters as well as prognosis. Cox analysis was employed to evaluate the relationships between prognosis and clinical parameters in US.
Result
Compared to normal proliferative endometrial tissue (NPE), the expression of ORM1 was downregulated in low-grade endometrial stromal sarcoma (LG-ESS), high-grade endometrial stromal sarcoma (HG-ESS), and undifferentiated uterine sarcoma (UUS) (P < .001,P < .001,and P < .001, respectively). Compared to normal uterine smooth muscle tissue (UNSM), the expression of ORM1 was upregulated in leiomyosarcoma (LMS) (P = .006). High ORM1 expression levels in US patients were associated with poor overall survival (OS) and progression-free survival (PFS) (P = .027 and P = .005, respectively). Multivariate COX analysis revealed that tumor stage and ORM1 expression are independent prognostic factors for US patients.
Conclusion
ORM1 is expressed at low levels in ESS and at high levels in LMS. ORM1 potentially serve as a novel biomarker for the diagnosis, classification, and prognosis of US.
Keywords
Introduction
Uterine sarcoma (US) is a rare, highly heterogeneous, and aggressive malignant tumor, accounting for approximately 1% of all female reproductive tract malignancies and 3%-7% of US.1,2 The clinical manifestations of US are nonspecific, often presenting as vaginal bleeding or abdominal pain, which resemble symptoms of uterine leiomyoma or adenomyosis. Some patients may not exhibit any clinical symptoms at all. 3 leading to frequent diagnosis at advanced stages of the disease. The prognosis of US is poor, with a high recurrence rate and a 5-year overall survival (OS) rate of approximately 40%-50%.4,5 Due to its rarity, histopathological diversity, and unfavorable prognosis, there is currently no consensus on the risk factors for poor prognosis or the optimal treatment strategies. 6 Therefore, it is important to discover new potential biomarkers to assist in diagnosis and predict patient prognosis.
Orosomucoid (ORM), also known as α-1 acid glycoprotein (AGP), is an acute phase protein that is synthesized mainly by hepatocytes and secreted into the blood. 7 It exists in two subtypes: ORM1 and ORM2, both of which increase under stress conditions such as inflammation, infection, injury, and burns.8-10 ORM1 plays a dual role in biological processes, and its expression varies depending on the tissue origin of different malignancies. For instance, in breast cancer, ORM1 is highly expressed and contributes to epirubicin resistance. 11 In hepatocellular carcinoma, ORM1 is generally expressed at low levels but is elevated in tissues with microvascular invasion. 12 Zhou et al indicated that higher serum ORM1 expression predicted inferior survival and lower response to chemotherapy in natural killer/T cell lymphoma. 13 However, the role of ORM1 in US remains unclear. In this study, to clarify the role of ORM1 in US, we explored the relationship between ORM1 expression and the prognosis of US.
Materials and Methods
Data Resources
A retrospective analysis was conducted on the clinicopathological information of 50 patients diagnosed with uterine sarcoma by the pathology department of our hospital from April 2011 to May 2023. This retrospective study was approved by the Biomedical Research Ethics Committee of the Affiliated Hospital of Zunyi Medical University (approval number: KLL-2023-556). Due to the retrospective nature of this study, the ethics committee of the hospital waived the informed consent of the patients and confirmed compliance with the Declaration of Helsinki and the confidentiality of the patient data. This research report complies with the REMARK guidelines. 14 All patient details have been de-identified.
The primary endpoint was OS, defined as the number of days from initial diagnosis to death or the last follow-up. Progression-free survival (PFS) was defined as the number of days from initial diagnosis to disease progression or death. The last follow-up was in May 2024. The median follow-up time was 35.6 months (range 2.9-159.3 months).
Inclusion Criteria and Exclusion Criteria
According to the 2019 International Federation of Gynecology and Obstetrics (FIGO) staging system, 15 US is classified into three main pathological types: leiomyosarcoma (LMS), endometrial stromal sarcoma (ESS), and adenosarcoma (AS). ESS is further subdivided into low-grade endometrial stromal sarcoma (LG-ESS), high-grade endometrial stromal sarcoma (HG-ESS), and undifferentiated uterine sarcoma (UUS). Inclusion criteria: Patients with a confirmed pathological diagnosis of LMS, LG-ESS, HG-ESS, or UUS were included. Patients with AS were excluded due to insufficient cases collected. Exclusion criteria: (1) patients without a definitive histologic diagnosis; (2) patients with metastatic sarcoma originating from other gynecological sites; (3) patients with endometrial carcinosarcoma; and (4) patients with incomplete pathological diagnosis, clinical manifestations, and follow-up information. A total of 50 patients were included.
Immunohistochemistry
In this study, we collected 50 cases diagnosed with US in our hospital from April 2011 to May 2023, including 32 cases of LG-ESS, 2 cases of HG-ESS, 4 cases of UUS, and 12 cases of LMS. Additionally, we randomly collected 10 cases of normal proliferative endometrium (NPE), 8 cases of normal uterine smooth muscle (UNSM), and 8 cases of uterine leiomyoma (ULM) as controls.
A rabbit anti-human ORM1 monoclonal antibody (EPR5605; ab134042; Abcam, Cambridge, UK) was used for immunohistochemistry. The tissue samples were fixed in 10% formalin and stored in paraffin. The paraffin-embedded blocks were sliced continuously to a thickness of 4 µm and fixed on glass slides for immunohistochemical staining. Paraffin-embedded sections were deparaffinized and then repaired by using ethylenediaminetetraacetic acid (EDTA) antigen recovery buffer. The primary antibody was incubated at room temperature for 2 h at a dilution of 1:300. An automated immunostaining instrument (VENTAN Bench Mark XT, Roche, Swiss) was used for secondary antibody detection, followed by counterstaining with hematoxylin. Normal human liver tissue was employed as a positive control. The higher the expression level and distribution density of ORM1 were, the stronger the positive results. The H-score was used to evaluate the expression of ORM1. The H-score is defined as the product of staining intensity and the percentage of positively stained cells. The staining intensity was recorded as 0, 1, 2, or 3. No staining was rated as 0 points, light-yellow staining was rated as 1 point, brownish yellow staining was rated as 2 points, and brown staining was rated as 3 points. The percentage of positive cells was also considered as follows: 0 points, 5% or less positive cells; 1 point, positive cells between 6% and 25%; 2 points, positive cells between 26% and 50%; 3 points, positive cells ranging from 51% to 75%; and 4 points, positive cells greater than 75%. The total score obtained was the product of the staining intensity and the percentage of positively stained cells, which was categorized as negative (0 points), weakly positive (1-4 points), moderately positive (5-8 points), or strongly positive (9-12 points). All the slides were reviewed and scored by two gynecological pathologists who were unaware of the patients’ clinical pathological information.
Statistical Analysis
Categorical variables are expressed herein as frequencies (percentages), while continuous variables were presented as means ± standard deviations (SDs) or medians and ranges. Statistical analyses were performed using IBM® SPSS® Statistics version 27 (IBM, Armonk, NY), R (version 4.3.3), and GraphPad Prism 10.1.2. Pearson's chi-square test was used to analyze categorical variables, and one-way analysis of variance (ANOVA) was applied for univariate analysis. Kaplan-Meier(K-M)analysis was conducted to evaluate the association between OS in US and ORM1 expression levels as well as pathological subtypes. Univariate and multivariate Cox proportional hazards regression analyses were employed to assess the role of ORM1 expression as an independent prognostic factor for US. The proportional hazards assumption of the Cox regression model was validated using Schoenfeld residual tests, which confirmed that the assumption was met (P > .05). In the multivariate analysis, adjustments were made for potential confounding factors, including ORM1 expression levels, tumor stage, serum CA125 levels, tumor size, lymphadenectomy, and adjuvant chemotherapy. These factors were selected based on their clinical relevance and support from existing literature. A P-value of < .05 was considered statistically significant.
Results
Baseline Characteristics
According to the guidelines of the FIGO, we analyzed 50 cases of US diagnosed by the pathology department of our hospital. Clinical data, including age at diagnosis, histological type, clinical and pathological staging, and adjuvant chemotherapy (CT) or radiotherapy (RT), were collected from patient medical records. Table 1 presents the baseline characteristics of the 50 US patients.
Baseline Characteristics of 50 Patients with US.

TH: total hysterectomy; TH + BSO: total hysterectomy + bilateral salpingo-oophorectomy; PLA: pelvic lymphadenectomy; omentectomy: omentectomy; appendectomy: appendectomy
The pathological types included LG-ESS (n = 32, 64.0%), HG-ESS (n = 2, 4.0%), UUS (n = 4, 8.0%), and LMS (n = 12, 24.0%). The average age of all participants was 45.56 ± 9.647 years (ranging from 28 to 67 years). With respect to menopausal status, 33 patients (66.0%) were premenopausal, and 17 patients (34.0%) were postmenopausal. Symptoms included vaginal bleeding (n = 29,58.0%), abdominal pain/bloating (n = 13, 26.0%), abdominal mass (n = 3, 6.0%), vaginal discharge (n = 1,2.0%), and no symptoms (n = 4, 8.0%). With respect to tumor size, 14 patients (36.36%) had tumors <5 cm, while 36 patients (63.64%) had tumors >5 cm . For CA125 levels, 23 patients (46.0%) had CA125 levels of <35 U/ml, 10 patients (20.0%) had CA125 levels of >35 U/ml, and 17 patients (34.0%) had unknown CA125 levels. FIGO staging included 26 cases (52.0%) in stage I, 12 cases (24.0%) in stage II, 3 cases (6.0%) in stage III, and 9 cases (18.0%) in stage IV. Surgical approaches included total hysterectomy (TH, n = 1, 2.0%), TH with bilateral salpingo-oophorectomy (BSO, n = 18, 36.0%), TH + BSO with pelvic lymphadenectomy (PLA, n = 22, 44.0%), TH + BSO + PLA with omentectomy (n = 1,2.0%), TH + BSO + PLA with omentectomy and appendectomy (n = 2, 4.0%), TH + BSO with omentectomy (n = 2, 4.0%), TH + BSO with appendectomy (n = 1, 2.0%), TH + BSO with omentectomy and appendectomy (n = 1, 2.0%), and simple mass resection (n = 2, 4.0%). Among all patients, 24 (48.0%) underwent lymphadenectomy, and none had lymph node metastasis.In addition, for adjuvant therapy, 15 patients (30.0%) received chemotherapy (CT) primarily based on doxorubicin and ifosfamide, while the remaining patients did not. Three patients (6.0%) received radiotherapy, and 47 patients (94.0%) did not. Regarding recurrence or metastasis,10 patients (20.0%) experienced recurrence or metastasis, with common sites being the pelvic cavity and lungs (both 50%). Based on the median ORM1 expression level, patients were divided into a high-expression and a low-expression group. The results showed that ORM1 was highly expressed in LMS and UUS (83.0% and 75.0%, respectively) andlowly expressed in LG-ESS and HG-ESS (69.0% and 100.0%,respectively),with a statistically significant difference (p = .006). However, ORM1 expression showed no significant differences in other clinical baseline characteristics (P > .05),such as age, menopausal status, symptoms,tumor size, staging, surgical approach, radiotherapy, and chemotherapy.
The Expression of ORM1 Protein in Different Subtypes of US
The expression of ORM1 was assessed using immunohistochemistry (IHC) in 50 samples of US tissue, 10 samples of NPE tissue, 8 samples of UNSM tissue, and 8 samples of ULM tissue. Positive expression of ORM1 protein was detected in the cytoplasm, appearing as light yellow, brownish-yellow, or dark brown granules. All US specimens exhibited varying degrees of positivity. LG-ESS and HG-ESS showed weak positivity, while UUS and LMS demonstrated moderate to strong positivity. In contrast, the control group NPE exhibited strong positivity, and UNSM and ULM displayed weak positivity.
As shown in Figure 1, ORM1 exhibited weak positive expression in LG-ESS and HG-ESS, strong positive expression in NPE, and moderate positive expression in UUS. Compared to the NPE group, the expression levels of ORM1 were significantly lower in LG-ESS, HG-ESS, and UUS (P < .001, P < .001, and P < .001, respectively). Although ORM1 expression was higher in UUS compared to LG-ESS and HG-ESS, the differences were not statistically significant (P = .248 and P = .524, respectively). ORM1 expression was lowest in LG-ESS and highest in NPE, and moderately expressed in UUS.
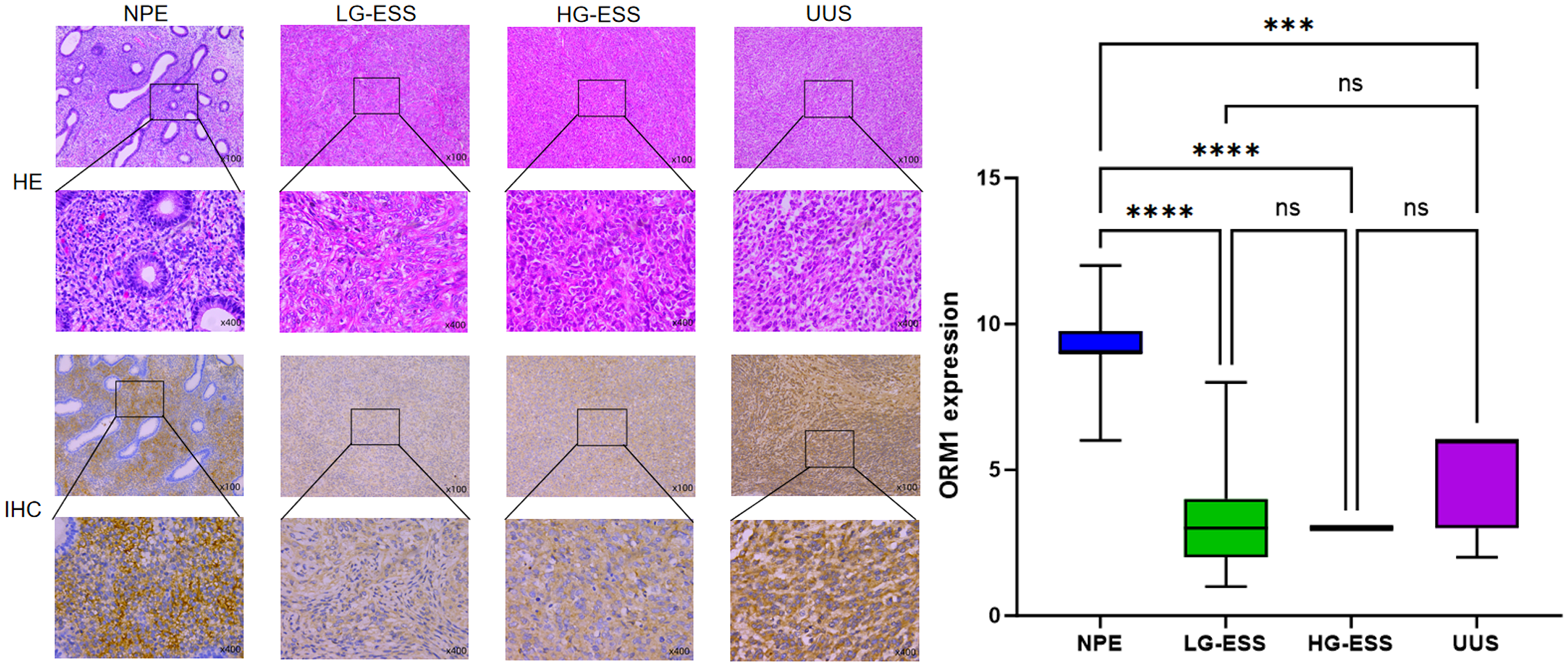
(A) IHC Staining Demonstrates The Expression of ORM1 in NPE, LG-ESS, HG-ESS, and UUS. ORM1 Showed Weak Positive Expression in LG-ESS and HG-ESS, Strong Positive Expression in NPE, and Moderate Positive Expression in UUS. (B) The Box Plot Illustrates the Differential Expression of ORM1 among 10 NPE Cases, 32 LG-ESS Cases, 2 HG-ESS Cases, and 4 UUS Cases. Compared with the NPE group, ORM1 Expression Levels were Significantly Decreased in LG-ESS, HG-ESS, and UUS.
As shown in Figure 2, ORM1 demonstrated moderate to strong positive expression in LMS, while weak positive expression was observed in UNSM and ULM. Compared to the UNSM and ULM groups, ORM1 expression was significantly elevated in LMS (P = .006 and P = .043, respectively). Although ORM1 expression was higher in ULM compared to UNSM, the difference was not statistically significant (P = .701).

(A) IHC Analysis Reveals ORM1 Expression in UNSM, ULM, and LMS. ORM1 Exhibited Moderate to Strong Positive Expression in LMS, While Showing Weak Positive Expression in UNSM and ULM. (B) The Box Plot Compares ORM1 Expression Across 8 UNSM Cases, 8 ULM Cases, and 12 LMS Cases. Compared with the UNSM and ULM Groups, ORM1 Expression was Significantly Elevated in LMS.
As shown in Figure 3, ORM1 expression in different subtypes of US varied, with weak positive expression observed in LG-ESS and HG-ESS, and moderate to strong positive expression in UUS and LMS. Compared to LMS, ORM1 expression was significantly lower in LG-ESS (P < .001), while it was also reduced in HG-ESS and UUS, though these differences were not statistically significant (P = .132 and P = .463, respectively). ORM1 expression was lowest in LG-ESS and highest in the LMS group.

(A) IHC Staining Demonstrates ORM1 Expression in LMS, LG-ESS, HG-ESS, and UUS. ORM1 Displayed Weak Positive Expression in LG-ESS and HG-ESS, and Moderate to Strong Positive Expression in UUS and LMS. (B) the box Plot Presents the Differential Expression of ORM1 among 12 LMS Cases, 32 LG-ESS Cases, 2 HG-ESS Cases, and 4 UUS Cases. Compared with the LMS Group, ORM1 Expression was Significantly Reduced in LG-ESS, HG-ESS, and UUS.
The Correlation Between ORM1 Expression and the Prognosis of US
Based on the median expression level of ORM1, patients were divided into high-expression and low-expression groups, and K-M analysis was performed on 50 US patients. The K-M analysis revealed that high ORM1 expression levels were associated with poor OS and PFS in US patients (P = .027, HR: 0.37, 95% CI: 0.15-0.92 and P = .005, HR: 0.29, 95% CI: 0.12-0.74, respectively; Figure 4A and B). The low ORM1 expression group demonstrated superior OS and PFS compared to the high-expression group, indicating better survival outcomes for patients with low ORM1 expression. The median OS and PFS in the low-expression group were not reached (NR), whereas the high-expression group had a median OS of 36.5 months and a median PFS of 26.2 months, showing statistically significant differences (P = .027 and P = .005, respectively). Additionally, we analyzed the OS and PFS of the 50 US patients across different pathological subtypes and found that LG-ESS had significantly better OS and PFS than other subtypes (P = .001, HR: 4.02, 95% CI: 1.43-11.27 and P = .004, HR: 3.29, 95% CI: 1.23-8.82, respectively; Figure 4C and D).

(A, B) K-M Analysis Shows the Relationship Between ORM1 Expression Levels and OS or PFS in US Patients. High ORM1 Expression Levels in US Patients are Associated with Poor OS and PFS. (C, D) K-M Analysis Illustrating OS and PFS between LG-ESS and Other Types of US Patients. LG-ESS Demonstrates Significantly Better OS and PFS Compared to Other Subtypes.
Univariate and Multivariate Cox Regression Analyses
We conducted univariate and multivariate Cox proportional hazards regression analyses on the OS and PFS of 50 US patients. As shown in Tables 2 and 3, the results of the univariate analysis revealed that ORM1 expression (HR = 0.36, P = .033, 95% CI: 0.14-0.92 for OS; HR = 0.29, P = .009, 95% CI: 0.11-0.73 for PFS) and tumor stage (HR = 0.36, P = .040, 95% CI: 0.14-0.95 for OS; HR = 0.29, P = .011, 95% CI: 0.11-0.75 for PFS) were significantly correlated with OS and PFS in US patients. Multivariate analysis further confirmed that ORM1 expression (HR = 0.37, P = .037,95% CI: 0.14-0.94 for OS; HR = 0.26, P = .004, 95% CI: 0.10-0.65 for PFS) and tumor stage (HR = 0.37, P = .044, 95% CI: 0.14-0.97 for OS; HR = 0.25, P = .005, 95% CI: 0.10-0.66 for PFS) were independent prognostic factors for both OS and PFS. In contrast, other clinical parameters—including tumor size, lymphadenectomy, CA125 level, and adjuvant chemotherapy—showed no significant correlation with OS or PFS (P > .05).
Univariate and Multivariate Cox Analyses of OS in the US Patients.

Univariate and Multivariate Cox Analyses of PFS in the US Patients.

Discussion
US is a rare mesenchymal malignant tumor, and its incidence increases with age. It has been reported that the incidence of US among women aged 50 and older in the United States is approximately 6.4 cases per 100 000 person-years. 16 The age of onset for US ranges from 28 to 63 years, with a mean age of 45 years. The majority of patients (71.0%) develop the disease during the perimenopausal period, which is consistent with previous studies.17-19 In our study, ESS was the most common pathological subtype (n = 38), which differs from some studies where LMS was reported as the most prevalent type, 20 but aligns with findings from other studies.1,17,19,21 The primary clinical manifestations included vaginal bleeding, abdominal pain, and bloating, with some patients being asymptomatic, consistent with previous research. 22
In this study, we investigated the relationship between ORM1 expression and the clinical characteristics and survival rates of patients with US. The results demonstrated that tumor stage is an independent prognostic factor for survival in US, which aligns with previous studies.5,17,19,23-25 Among the 12 LMS cases in our study, 7 were stage I, 2 were stage II, and 3 were stage IV. Eight patients had died, with a median survival time of 22.4 months. The 2-year OS rate was 50.0% (n = 6), and even early-stage LMS had a poor prognosis, consistent with prior research (the 5-year OS for stage I LMS does not exceed 51.0%, and for stage II, it is 25.0%).5,24 Among the 32 LG-ESS patients, the majority were stage I and II (n = 17 and n = 8, respectively), with a minority in stage III and IV (n = 3 and n = 4, respectively). Seven patients had died, including 4 with stage IV disease and 3 with early-stage disease. The 5-year OS rate was 68.18% (n = 17), which is largely consistent with previous findings (LG-ESS has a relatively favorable prognosis, with a 5-year OS of 90% for stage I and II patients, compared to 50% for stage III and IV patients). 22 Studies have shown that HG-ESS has a poor prognosis, with approximately 70% of patients diagnosed at stage III-IV, 23 while UUS has an extremely poor prognosis (most patients survive less than 2 years). 22 In our study, of the 2 HG-ESS patients, 1 died within a year of diagnosis, while the other remained alive after 6 years of follow-up. Among the 4 UUS patients, 3 had died, and the 3-year OS rate was 25% (n = 1), consistent with prior research. 22 However, the small number of HG-ESS (n = 2) and UUS (n = 4) cases may introduce bias into the results. This study found that LG-ESS had significantly better OS and PFS compared to other subtypes, consistent with previous findings (ESS has a higher 5-year survival rate than LMS, and LG-ESS has a better survival rate than LMS and HG-ESS).17,25 In this study, only 10 ESS patients (20.0%) experienced recurrence or metastasis, which differs from some studies reporting a 40%–50% recurrence rate for US 5 but aligns with Sucheth et al 19 (a recurrence rate of 23.40%). This discrepancy may be due to the predominance of ESS cases in our study, the exclusion of UCS cases, and the limited number of LMS cases (n = 12). Common sites of recurrence included the pelvis and lungs (both at 50.0%).
In this study, 80.0% (n = 40) of the patients underwent TH + BSO as part of their treatment. Among them, 24 patients (48.0%) received lymphadenectomy, 15 patients (30.0%) underwent chemotherapy (CT), and 3 patients (6.0%) received radiation therapy. However, neither lymphadenectomy nor chemotherapy was associated with OS or PFS, which partially contrasts with previous studies suggesting that lymphadenectomy may improve overall survival in patients with HG-ESS. 26 This discrepancy may be attributed to the limited number of HG-ESS cases in our study. Among the 24 patients who underwent lymphadenectomy, none had positive lymph nodes. Lymph node positivity has been identified as a significant independent risk factor, with a markedly reduced 5-year disease-specific survival (CSS) observed in all three histological types (LMS, ESS, and UAS) compared to those with negative lymph nodes1,27
TH + BSO is the gold standard treatment for all types of US. 26 Conservative treatment, such as myomectomy, is only suitable for highly selected patients who wish to preserve fertility and are willing to undergo intensive follow-up. 26 Systematic lymphadenectomy is not routinely recommended unless there is clinical or radiological suspicion of lymph node involvement or when the histological diagnosis confirms HG-ESS. 26 Studies have shown that surgery for recurrent US is associated with improved disease-specific survival (DSS), whereas chemotherapy and radiation therapy do not significantly improve prognosis. 21 Currently, laparoscopic myomectomy is widely used due to its numerous advantages, including shorter hospital stays, reduced blood loss, decreased use of postoperative analgesics, and the potential to preserve fertility. However, the use of laparoscopic myomectomy in undiagnosed US may lead to worse oncological outcomes, such as tumor upstaging from stage I to stage IV due to the spread of uterine malignancy. 28 Therefore, patients with US who wish to preserve fertility should be managed and counseled by a multidisciplinary team of experts to develop individualized surgical approaches. 26 Although the use of in-bag morcellation during minimally invasive myomectomy is recommended, there is currently no high-level evidence demonstrating that it reduces the risk of spillage of any potential occult sarcoma. 28 Additionally, preoperative hysteroscopy with targeted biopsy can help establish a definitive diagnosis, aiding in the selection of the most appropriate surgical approach. 28 However, due to the limitations of biopsy tissue, achieving a definitive preoperative diagnosis remains challenging. In summary, the treatment of US is complex and controversial, necessitating further research to identify optimal management strategies.
The rarity and heterogeneity of US pose significant challenges in identifying appropriate biomarkers and treatment modalities, leading to limited progress in survival rates over recent decades. 29 This study is the first to explore the expression of ORM1 in US and its relationship with patient prognosis. We found that ORM1 is expressed at low levels in LG-ESS andHG-ESS, while it is highly expressed in UUS and LMS. K-M analysis revealed that high ORM1 expression is significantly associated with poor OS and PFS, whereas patients with low ORM1 expression exhibited better prognosis. Cox regression analysis further demonstrated that ORM1 expression is an independent prognostic factor influencing the survival rate of US patients. These findings suggest that ORM1 may serve as a potential prognostic biomarker for US.
As an acute-phase protein, ORM1 has been studied in various cancers. Research indicates that ORM1 expression is increased in breast cancer, colon adenocarcinoma, prostate cancer, and oral squamous cell carcinoma, where it promotes tumor initiation and progression.11,12,30,31 Conversely, ORM1 expression is reduced in esophageal cancer, lung squamous cell carcinoma, hepatocellular carcinoma (HCC), and cholangiocarcinoma.12,30,31 In breast cancer, high ORM1 expression has been shown to promote epirubicin resistance by upregulating MMP-2 and MMP-9 expression and activating the AKT/ERK signaling pathway, suggesting its potential as a therapeutic target. 11 In HCC, ORM1 is generally expressed at low levels but is elevated in cases with microvascular invasion. ORM1 expression negatively correlates with tumor stage and grade in HCC, and its high expression increases vascular invasiveness and reduces sensitivity to sorafenib. 12 Additionally, ORM1 can be detected in plasma and other body fluids, making it an ideal candidate for dynamic monitoring of disease progression. Studies have found that ORM1 is highly expressed in colorectal cancer liver metastasis (CRLM) compared to primary colorectal cancer lesions, with elevated serum ORM1 levels observed in CRLM patients. Serum ORM1 levels significantly decrease after CRLM resection, indicating its potential diagnostic and predictive value for CRLM. 32 Furthermore, ORM1 may enhance the tumor immune microenvironment by mediating macrophage M2 polarization and increasing interleukin-10 (IL-10) expression, thereby promoting CRLM. Upregulation of ORM1 not only enhances the proliferation, migration, and invasion of colorectal cancer cells but also facilitates epithelial-mesenchymal transition (EMT) through the PI3 K/AKT pathway. 32 Thus, ORM1 could serve as a predictor for CRLM and a potential target for immunotherapy. 32 Other studies have suggested that serum ORM1 and TGF-β levels may be reliable clinical biomarkers for the early diagnosis of non-small cell lung cancer. 33 Our study is the first to reveal the expression pattern of ORM1 in US and its association with prognosis, suggesting that ORM1 has the potential to serve as a biomarker for the diagnosis, classification, and prognosis prediction of US. However, the molecular mechanisms underlying the role of ORM1 in US remain unclear and require further investigation.
However, this study has several limitations. First, the sample size was relatively small, with a median follow-up time of 35.6 months (range: 2.9-159.3 months). At the end of the study, the median PFS and OS in the low ORM1 expression group had not yet been reached, which may introduce bias into the conclusions. Second, as a retrospective study, there is a potential for selection bias. Additionally, due to sample limitations, we did not assess ORM1 mRNA expression levels. Future research should validate these findings in larger cohorts and further explore the molecular mechanisms of ORM1 in US using molecular biology techniques such as RNA sequencing and proteomics. Further investigation is also needed to determine whether ORM1 could serve as a potential therapeutic target for US.
Conclusion
ORM1 exhibits differential expression across various subtypes of US, with low expression in ESS and high expression in LMS. Patients with US and low ORM1 expression demonstrate better prognosis, and ORM1 expression level is an independent prognostic factor influencing patient survival rates. Therefore, ORM1 has the potential to become a novel biomarker for the diagnosis, classification, and prognosis of US. However, further large-scale studies and functional experiments are needed to confirm these findings.
Footnotes
Ethics and Patient Consent Statement
This retrospective study was approved by the Biomedical Research Ethics Committee of the Affiliated Hospital of Zunyi Medical University (approval number: KLL-2023-556). Due to the retrospective nature of this study, the ethics committee of the hospital waived the informed consent of the patients and confirmed compliance with the Declaration of Helsinki and the confidentiality of the patient data.
Author Contributions
Conceptualization and writing—original draft preparation, Dan Yuan, Jian-Guo Zhou;data curation, Yue Huang, Ying Cai; formal analysis, Chi Zhang; Writing—review and editing, Jin-Jing Wang, Jian-Guo Zhou. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Plan Project of Zunyi City,Zunshi Kehe HZ (2023) 234; The Guizhou Provincial Health Commission Science and Technology Fund Project for 2024 (Grant No. D596), and the Zunyi Medical University Affiliated Hospital Excellent Youth Talent Training Program Project (Grant No. rc220240423). Noncommunicable Chronic Diseases-National Science and Technology Major Project (Grant No. 2023ZD0502105), the National Natural Science Foundation of China (Grant No. 82060475), Chunhui program of the MOE (Ministry of Education in China) (Grant No. HZKY20220231), MOE Liberal Arts and Social Sciences Foundation (Grant No. 24YJCZH462), Youth Science and Technology Elite Talent Project of Guizhou Provincial Department of Education (Grant No.QJJ-2024-333), Excellent Young Talent Cultivation Project of Zunyi City (Zunshi Kehe HZ (2023) 142), Future Science and Technology Elite Talent Cultivation Project of Zunyi Medical University (ZYSE 2023-02), the Key Program of the Education Sciences Planning of Guizhou Province (Grant No.7), Collaborative Innovation Center of Chinese Ministry of Education (Grant No. 2020-39).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The images and tables in this manuscript are included in the main text. Due to necessary confidentiality, the clinical cohort data proposed in this article is not easily accessible as it is part of an ongoing study. The request to access the dataset should be sent directly to the corresponding author.
