Abstract
Background
Although preoperative radiotherapy (PORT) is a promising therapeutic option for stage III non-small cell lung cancer (NSCLC), the efficacy of this treatment remains controversial. The positive lymph node ratio (PLNR) is recognized as an independent prognostic factor for survival. However, no previous studies have focused on the association between PLNR and PORT in stage III NSCLC.
Methods
Data were collected from the Surveillance, Epidemiology and End Results (SEER) database, and all patients enrolled in this analysis were diagnosed during 2010–2015. The primary endpoint was overall survival (OS). Univariate and multivariate Cox regression analysis was used to identify factors associated with survival before and after case-control matching. PLNR was defined as the ratio of the number of positive lymph nodes to the total number of retrieved or examined lymph nodes. A cutoff value for PLNR was calculated using an X-tile model.
Results
Overall, 391 patients with PORT and 2814 patients without PORT were enrolled in this study. The cohort after 1:1 case-control matching included 322 patients who received PORT and 322 patients without PORT. PORT was not associated with a significant effect on OS (HR = 1.14; 95% CI: 0.91–1.43; P = 0.825). Multivariate Cox regression analysis showed that PLNR (P < 0.001) was independently associated with OS in patients with stage III NSCLC. An X-tile model was used to identify a cutoff value for PLNR: the risk of death was significantly lower in patients with PLNR ≤0.41 who received PORT than in those with PLNR >0.41 who received PORT (HR = 0.59; 95% CI: 0.38–0.91; P = 0.015).
Conclusion
PLNR may be a prognostic factor for survival in patients with stage III NSCLC who undergo PORT. Lower PLNR is a predictor of better OS and thus warrants further study.
Keywords
Introduction
Lung cancer ranks second worldwide in terms of cancer incidence and is a leading cause of death, and the incidence and mortality rates for this disease are consistently rising.1,2 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancers, and the 5-year overall survival (OS) of NSCLC is only 26%. 3 Most patients with NSCLC are diagnosed at an advanced stage, and their prognosis is particularly poor. 4 Advanced stage NSCLC is heterogeneous and extremely difficult to treat, and the therapeutic options are limited.
The treatment of NSCLC depends on the tumor size, type and stage. The therapeutic options include surgery, radiotherapy, chemotherapy, targeted therapy and immunotherapy.5,6 Surgery is usually the first choice treatment, but preoperative neoadjuvant therapies are available. 7 Radiotherapy, alone or in combination with chemotherapy, makes an important contribution to the treatment of stage III NSCLC. 8 Several studies have demonstrated that high or standard doses of preoperative radiotherapy (PORT) can remarkably improve survival rates.9-12 However, the role of PORT in resectable stage III NSCLC is still unclear.
Prediction of the prognosis of NSCLC is largely dependent on the American Joint Committee on Cancer (AJCC) Tumor Node Metastasis (TNM) staging system, which recommends station-based lymph node sampling/excision and tumor staging.13,14 The degree of lymph node involvement, which includes the locations of the metastatic lymph nodes and the number of lymph nodes involved, is an important factor affecting the prognosis of patients with NSCLC after surgery and subsequent treatment decisions. 15 Recent studies have identified the positive lymph node ratio (PLNR) as an independent prognostic factor for survival.17-20 In lymph node-positive NSCLC cases, the PLNR is defined as the ratio of the number of positive lymph nodes to the total number of retrieved or examined lymph nodes. The PLNR has been suggested to be an efficient prognostic stratification tool. 21 Furthermore, the PLNR has been shown to have prognostic value in breast, esophageal, gastric, colorectal and bladder cancers. 16 Several previous studies have confirmed that a higher number of positive lymph nodes and a higher PLNR are correlated with worse survival results for patients with NSCLC.18,22,23
However, no relevant study has focused on the relationship between PLNR and PORT in patients with stage III NSCLC. It is important to establish whether all patients with stage III NSCLC would benefit from PORT, and if not, determine whether a subgroup of patients would benefit from PORT. The present study analyzed data for patients with resectable stage III NSCLC to evaluate whether PORT improved OS and to investigate the association between PLNR and prognosis.
Methods
Patient Selection
The Surveillance, Epidemiology, and End Results (SEER) database is a national-level cancer monitoring initiative that collects cancer-related data such as primary tumor site, stage at diagnosis, histology, therapy, staged reexamination and cause of death. 24 In this study, data for 3205 patients with primary stage III NSCLC diagnosed between 2010 and 2015 were obtained from the SEER database. Lung cancer was defined according to the third edition of the International Classification of Diseases for Oncology (ICDO-O-3). 25 The primary inclusion criteria were: 1) adult; 2) histological documentation of stage III NSCLC; 3) data available for tumor subsite, grade, size and laterality; and 4) cause of death was defined. Those with incomplete staging, unknown tumor grade, unknown survival time, missing or unknown cause of death, and who did not receive surgical treatment were excluded. Patients with insufficient data regarding whether they underwent radiotherapy or refused it were also excluded. The analysis did not distinguish between preoperative neoadjuvant chemotherapy and postoperative adjuvant chemotherapy because the SEER database did not provide any information as to whether chemotherapy was concurrent with or sequential to radiotherapy. Cases and controls were matched 1:1 according to the following confounding variables: age, race, sex, pathology, grade, laterality, stage and recorded chemotherapy. All the patients who met the criteria were divided into PORT and no-PORT groups (Figure 1).

Flowchart showing the selection of patients.
Construction of a Nomogram and Assessment of its Clinical Utility
All variables were analyzed using univariate analysis, and Cox regression models were used to identify independent predictors in multivariate analyses. A nomogram predicting 1-year, 3-year and 5-year survival was constructed based on the independent risk factors included in the final multivariate Cox model. Sensitivity and specificity between column line graphs and TNM staging were analyzed using receiver operating characteristic (ROC) and decision curve analysis (DCA) curves.
Statistical Analysis
The primary endpoint was OS, determined according to the data in the SEER database. OS was defined as the period from the date of diagnosis to the date of death. Prognostic factors were analyzed using a univariate Cox proportional hazards model. Comparisons of OS between different groups were made using Kaplan-Meier plots and log-rank tests. The confounding influences of baseline clinicopathological characteristics were accounted for by the application of case-control matching. SPSS 23.0 (IBM Corp, Armonk, NY, USA), Prism 8.3.0 (GraphPad Software, San Diego, CA, USA) and R-4.0.5 (R Core Team, Vienna, Austria) were used for the statistical analyses and construction of figures. An X-tile model was used to determine the cutoff value for the PLNR. P < 0.05 was considered statistically significant.
Results
Baseline Characteristics of the Patients
Before case-control matching, 3205 patients diagnosed with stage III NSCLC were enrolled in our study, including 1191 patients (37.2%) under the age of 65 years old and 2014 patients (62.8%) over 65 years old. The pathological type was adenocarcinoma in 1871 cases (58.4%) and squamous cell carcinoma in 972 cases (30.3%). PORT was performed in 391 of the 3205 patients, including 214 men (54.7%) and 177 women (45.3%). After matching, the PORT and no-PORT groups each comprised 332 patients. The matched case-control cohort consisted of 368 men (57.1%) and 276 women (42.9%), with 352 patients (54.7%) younger than 65 years old and 292 patients (45.3%) older than 65 years old. Adenocarcinoma was diagnosed in 362 cases (56.2%), and squamous cell carcinoma was diagnosed in 234 cases (36.3%) (Table 1).
Baseline Characteristics of the Patients in the PORT and no-PORT Groups Before and After Case-Control Matching.

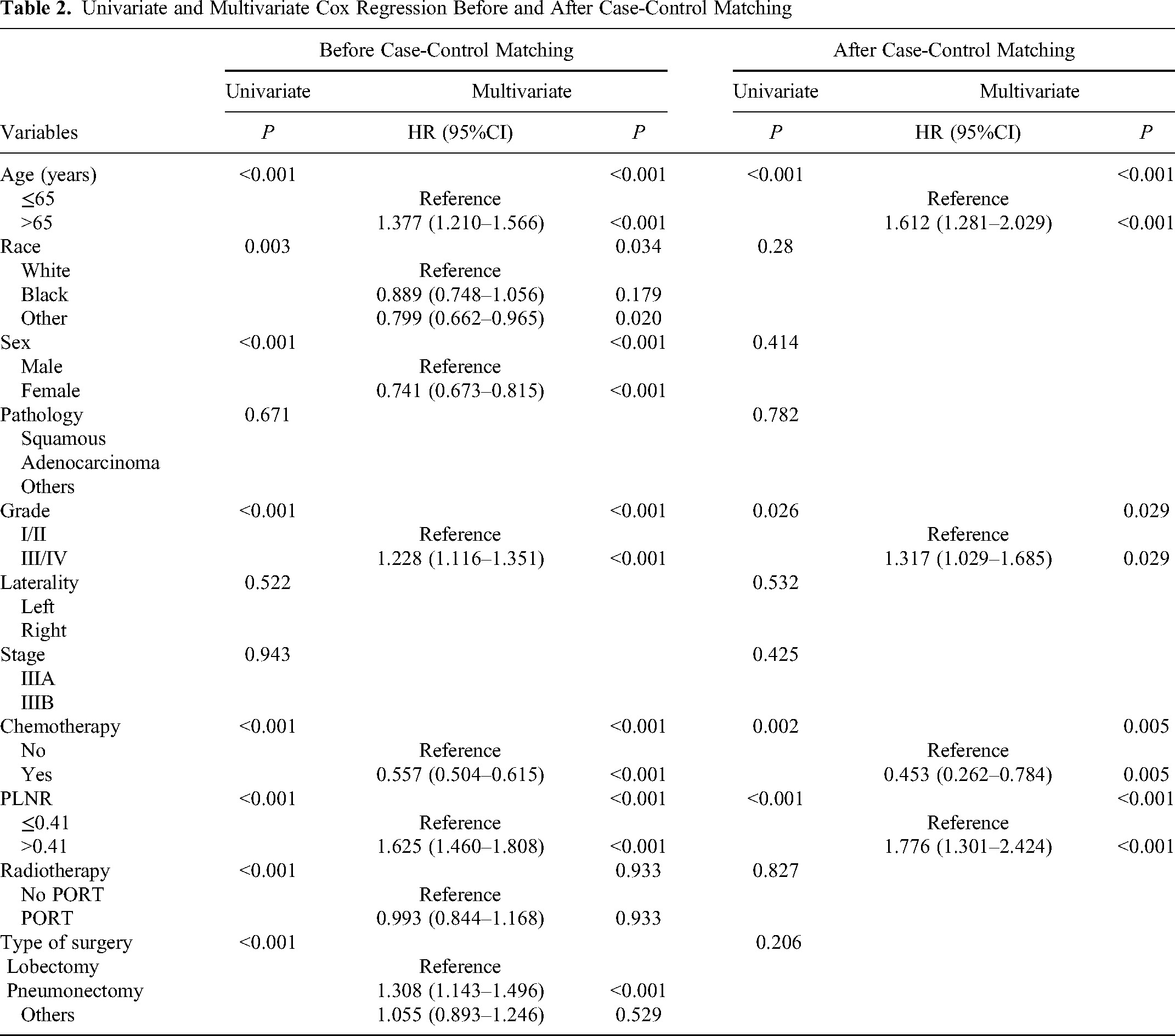
Age, ethnicity, sex, tumor pathology, tumor grade, tumor laterality, tumor stage, chemotherapy, type of surgery, PLNR and PORT were entered into the Cox analysis. Before case-control matching, univariable analysis revealed that age, ethnicity, sex, tumor grade, chemotherapy, type of surgery, PLNR and PORT were significant prognostic factors for OS (all P < 0.05). In the multivariable analysis, age (hazard ratio [HR] = 1.377; 95% confidence interval [CI]: 1.210–1.566; P < 0.001), sex (HR = 0.741; 95% CI: 0.673–0.815; P < 0.001), tumor grade (HR = 1.228; 95% CI 1.116–1.351; P < 0.001), chemotherapy (HR = 0.557; 95% CI: 0.504–0.615; P < 0.001), type of surgery (pneumonectomy vs lobectomy, HR = 1.308; 95% CI: 1.143–1.496; P < 0.001) and PLNR (HR = 1.625; 95% CI: 1.460–1.808; P < 0.001) were independent and significant prognostic factors for OS. After matching, univariable analysis revealed that age, tumor grade, chemotherapy and PLNR were significant prognostic factors for OS (all P < 0.05). In the multivariable analysis, age (HR = 1.612; 95% CI: 1.281–2.029; P < 0.001), tumor grade (HR = 1.317; 95% CI: 1.029–1.685; P = 0.029), chemotherapy (HR = 0.453; 95% CI: 0.2620.784; P = 0.005) and PLNR (HR = 1.776; 95% CI: 1.301–2.424; P < 0.001) were independent and significant prognostic factors for OS (Table 2). Before case-control matching, the PLNR value was significantly higher in patients who did not undergo preoperative radiotherapy than in those who underwent preoperative radiotherapy (Z = −14.213, P < 0.001, Mann-Whitney U test). A similar result was obtained after case-control matching (Z = −7.581, P < 0.001, Mann-Whitney U test) (Supplement Figure 1).
Univariate and Multivariate Cox Regression Before and After Case-Control Matching
OS of Patients Before and After Case-control Matching
Before case-control matching, patients who underwent PORT had better OS than patients who did not undergo PORT. The 1-year survival rates for the PORT and no-PORT groups were 83.8% and 75.9%, respectively. The 3-year survival rates for the PORT and no-PORT groups were 57.6% and 48.4%, respectively. The 5-year survival rates for the PORT and no-PORT groups were 43.8% and 35.8%, respectively. Median survival time was 44 months in the PORT group and 35 months in the no-PORT group (Figure 2A). Patients with PORT did not show a significant advantage in OS after case-control matching (P = 0.825). After matching, the 1-year survival rates for the PORT and no-PORT groups were 83.1% and 85.8%, respectively. The 3-year survival rates for the PORT and no-PORT groups after matching were 56.2% and 56.5%, respectively. The 5-year survival rates for the PORT and no-PORT groups after matching were 43.5% and 45.2%, respectively. The median survival time was 42 months for the PORT group and 48 months for the no-PORT group after matching (Figure 2B).

Kaplan-Meier curves for overall survival before case-control matching. (A) Overall survival of patients before propensity score matching (P = 0.0002). (B) Overall survival of patients after propensity score matching (P = 0.825). (C) Overall survival of patients receiving PORT (P = 0.015).
The X-tile mode determined that the appropriate cutoff for the PLNR was 0.41. Survival was further analyzed using Kaplan-Meier curves. The results showed that after PORT, patients with a PLNR ≤0.41 had a better OS than patients with a PLNR >0.41 (HR = 0.59; 95% CI: 0.38–0.91; P = 0.015). The median survival time was 46 months for the group with PLNR ≤0.41 and 27 months for the group with PLNR >0.41 (Figure 2C).
Establishment of a Nomogram for Patients with NSCLC
The nomogram ultimately comprised four prognostic factors: age, tumor grade, PLNR and chemotherapy. The scores for each factor were summed, and the total score was calculated. The 1-year, 3-year and 5-year survival rates were estimated by drawing a vertical straight line downward from the total score. The C-index measuring predictive accuracy was 0.614 (95% CI: 0.597–0.631) for OS, indicating good discriminatory ability (Figure 3A). A calibration plot was constructed to determine whether the predicted and observed survival probabilities were consistent. The calibration plots for 1-year, 3-year and 5-year OS showed that the probabilities were consistent between the standard curve and nomogram prediction. The calibration curves also had good predictive consistency (Figure 3B, 3C, 3D). The area under the curve (AUC) values for 1-year, 3-year and 5-year OS were 0.64 (P < 0.001), 0.63 (P < 0.001) and 0.64 (P < 0.001), respectively (Figure 4A). DCA curve analysis also demonstrated that the nomogram had utility in the prediction of OS both in the short term and long term (Figure 4B, 4C, 4D).

Construction of the nomogram. (A) Nomogram predicting the 1-year, 3-year and 5-year overall survival of patients with stage III NSCLC after case-control matching. (B) Calibration curve for the prediction of 1-year overall survival. (C) Calibration curve for the prediction of 3-year overall survival. (D) Calibration curve for the prediction of 5-year overall survival.

Receiver operating characteristic (ROC) curve and decision curve analysis (DCA) curve for overall survival
Discussion
The present study focused on patients with stage III NSCLC who underwent surgery with or without PORT. Radiotherapy makes an important contribution to the treatment of stage III NSCLC. 8 It has been suggested that lymph node metastasis status plays an important role as a prognostic factor for OS in this setting, and the PLNR is a useful indicator for predicting prognosis. However, to date, no relevant studies have described the relationship between PORT and PLNR in patients with stage III NSCLC. The purpose of our study was to assess the association between PORT and PLNR in patients with resectable stage III NSCLC.
Patients with stage III NSCLC are heterogeneous and should be treated individually. Reif et al concluded that surgery alone has a more limited role in the treatment of patients with stage III disease. 26 Several studies have shown that patients given preoperative therapy followed by surgery have better survival.27-31 The choice of the preoperative regimen remains an issue. Radiotherapy is often used as the primary local treatment, but preoperative radiotherapy suffers from shortcomings. The original tumor borders are unclear after radiotherapy, which may lead to inadequate surgical margins. The local tissue response to radiotherapy can complicate anatomical identification and increase the difficulty of the surgical procedure. PORT also has the potential to interfere with wound healing. These drawbacks of PORT have created uncertainty in its use. Nevertheless, the usefulness of PORT cannot be ignored despite its shortcomings. The advantages of preoperative radiotherapy are obvious. Radiotherapy leads to tumor shrinkage, which facilitates radical resection; moreover, radiotherapy can facilitate the complete resection of tumors that otherwise would be difficult or impossible to completely resect, thereby improving the success rate of surgery. In addition, radiotherapy can kill active tumor cells and subclinical lesions outside the tumor, reduce the viability of tumor cells, and decrease local recurrence and distant metastasis. Several clinical studies have shown that preoperative radiotherapy can improve the survival rate of patients with stage III NSCLC. Therefore, PORT is an important therapeutic strategy for stage III NSCLC, and its contribution has been increasingly recognized in recent years. Despite the increasing use of PORT in stage III NSCLC, it is still not routinely used. Several randomized trials and a retrospective analysis of the SEER database suggested that neoadjuvant radiotherapy can provide potential benefit to some patients with stage III NSCLC.32,33 However, the value of PORT for patients with resectable stage III NSCLC remains controversial. Chen et al 34 showed that PORT may improve the prognosis of patients with resectable stage IIIA/N2 NSCLC. However, in a systematic review and meta-analysis of 7 studies that met the criteria for analysis, 1 randomized control trial, 1 phase II study, 3 retrospective reviews, and 2 published abstracts of randomized controlled trials demonstrated that the addition of PORT to induction chemotherapy did not improve survival after surgery as compared with induction chemotherapy alone. 35 Our study retrospectively compared the prognosis of patients with stage III NSCLC between those who received preoperative radiotherapy and those who did not. Case-control matching was used to exclude the effects of differences in sample size and baseline characteristics between the two groups. The results showed that before case-control matching, PORT was associated with a survival benefit. In contrast, after matching, no benefit was derived from PORT. The results may have been influenced by the large difference in the number of patients receiving PORT and the number of patients not receiving PORT. We, therefore, explored whether a variable could be identified that might predict which patients would benefit from PORT.
We found that not all subgroups tended to receive preoperative radiotherapy. Therefore, it was necessary to select a variable that would indicate which patients would benefit from PORT. The survival of patients with stage III NSCLC is dependent on the number of involved lymph nodes. 36 The number of PLNs has been shown to have prognostic value in breast, esophageal, gastric, colorectal and bladder cancers. 16 Evidence from recent publications suggests that patients with a higher number of involved lymph nodes tend to have a poorer prognosis than patients with fewer involved lymph nodes. 37 In addition, the PLNR was reported to be a good predictor of prognosis in patients with NSCLC.17-20 A retrospective analysis by Bria et al 38 that used the seventh edition of the AJCC guidelines identified the PLNR as an independent prognostic factor in NSCLC. The findings of current study also suggest that the PLNR has a significant impact on prognosis. In this study, PLNR was an independent risk factor for OS in the univariate and multifactorial analyses both before and after case-control matching. In addition, patients with a low PLNR who underwent PORT had better OS than those with a high PLNR who underwent PORT. The X-tile method was used to establish an optimal cut-off value for PLNR to effectively define a subgroup of node-positive NSCLC cases that benefited from PORT. In addition, a nomogram that included the PLNR was created to help clinicians predict and assess the prognosis of patients with stage III NSCLC and to guide clinical treatment. The nomogram is a model that associates several factors to predict survival outcomes. Subsequently, we verified the sensitivity and specificity of the model using calibration curves as well as ROC and DCA curves, and the results showed that the model has good predictive ability.
A previous study has established a nomogram using SEER database and showed that the nomogram containing the PLNR can accurately predict survival in patients with T1−4N1−3M0 NSCLC, 39 which was similar with our results that PLNR might be a prognostic factor for survival in patients with stage III NSCLC who undergo PORT. Interestingly, it is considered that fit elderly patients benefit from concurrent radiochemotherapy and age alone should not exclude fit patients and deprive them of the standard treatment. 40 In current study, we found that age was independent and significant prognostic factors for OS, which was different from previous results. PLNR may be also used to assess the prognosis in elderly patients, which needs to be further evaluated. Meanwhile, when patients with stage III NSCLC present good prognostic factors and are considered candidates for surgical resection, robotic approach for locally advanced NSCLC is safe and feasible. 41 PLNR might be a key indicator used in the patient selection.
The current study has several limitations. Since the PLNR is determined after surgery following resection of the lymph nodes and pathological evaluation, it is not possible from a practical standpoint to evaluate the direct effects of preoperative radiotherapy on the PLNR. It might be expected that adjuvant radiotherapy would reduce the PLNR, for example by killing metastatic tumor cells in lymph nodes or inhibiting the metastasis of tumor cells to additional lymph nodes before surgery was performed. This would be consistent with our results, which showed that the PLNR was significantly lower in patients who underwent PORT than in those who did not, both before and after case-matching. Therefore, our observation that patients with PLNR ≤0.41 after PORT had a longer OS than those with PLNR >0.41 after PORT may in part reflect a better response to adjuvant radiotherapy. Nevertheless, from a clinical perspective, whether or not induction radiotherapy directly reduced PLNR does not alter the important finding that PLNR was a prognostic factor for OS in patients who had undergone radiotherapy before surgery. Another limitation is that, while the SEER database can provide a large number of cases for analysis, the accuracy and completeness of the database may limit the accuracy of our research. Due to the limited information available in the SEER database, data regarding smoking history, specific radiotherapy targets, radiotherapy doses, radiotherapy methods, chemotherapy regimens and other clinical information were not available, which may have affected the results. Recent years have seen important advances in radiotherapy techniques, including the development of stereotactic ablative radiotherapy, intensity-modulated radiation therapy, volumetric-modulated arc therapy, and proton therapy,42,43 but a lack of relevant information in the SEER database precluded an evaluation of whether the potential survival benefits differed between specific types of PORT. Moreover, it was not detailed in the SEER database whether chemotherapy was concurrent with or sequential to radiotherapy. Therefore, it was not possible to compare survival between patients who received preoperative neoadjuvant chemotherapy and those who received postoperative adjuvant chemotherapy. This may have been a confounding factor in the present study because although there is evidence for survival benefits of neoadjuvant chemotherapy and adjuvant chemotherapy, 44 there are limited data directly comparing preoperative versus postoperative chemotherapy. Similarly, it was not possible to make comparisons between patients who had adjuvant chemoradiotherapy and planned surgery with those who had radical chemoradiotherapy for unresectable stage III NSCLC followed by salvage surgery to achieve local control 45 ; this could be a confounding factor because patients with unresectable NSCLC who underwent radical chemoradiotherapy and salvage surgery would be expected to have a poorer prognosis than those who underwent adjuvant chemoradiotherapy and planned surgery. In addition, because this was a retrospective analysis, additional prospective randomized clinical trials and multicenter studies with large sample sizes are needed to further validate the reliability and generalizability of our findings. Finally, the lack of molecular analysis may have overlooked important prognostic clues.
Conclusion
Our findings suggest that PLNR may be a prognostic factor for PORT in stage III NSCLC. A lower PLNR was independently associated with better OS, which warrants further study.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231173498 - Supplemental material for Patients with Lower Positive Lymph Nodes Ratio May Benefit from Preoperative Radiotherapy in Stage III Non-Small Cell Lung Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338231173498 for Patients with Lower Positive Lymph Nodes Ratio May Benefit from Preoperative Radiotherapy in Stage III Non-Small Cell Lung Cancer by Ruiyang Wang, MB, Shijie Shang, MB, Xinyi Huang, MD, Yu Nie, MB, Fei Wang, PhD, Jinming Yu, PhD, MD, and Dawei Chen, MD in Technology in Cancer Research & Treatment
Footnotes
Data Accessibility
The data used for the analysis are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jinming Yu has received grants from the Academic Promotion Program of Shandong First Medical University (2019ZL002), Research Unit of Radiation Oncology, Chinese Academy of Medical Sciences (2019RU071), National Natural Science Foundation of China (81627901, 81972863 and 82030082), and Natural Science Foundation of Shandong (ZR20191104045).
Ethics Approval Statement
Consent to participate was waived because this was a retrospective observational study using anonymized data from the SEER database. All study authors signed the SEER database agreement and obtained a license to access the SEER information (accession username: 18157-Nov 2019).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
