Abstract
Background
Abnormal mitochondrial pyruvate carrier 1 (MPC1) expression plays a key role in tumor metabolic reprogramming and progression. Understanding its significance in non-small cell lung cancer (NSCLC) is crucial for identifying therapeutic targets.
Methods
TIMER 2.0 was utilized to assess the expression of MPC1 in both normal and cancer tissues in pan-cancer. Overall survival (OS) differences between high and low MPC1 expression were analyzed in NSCLC using the Cancer Genome Atlas (TCGA) datasets. We also examined the expression of MPC1 in NSCLC cell lines using western blotting and quantitative reverse transcriptase polymerase chain reaction (qRT-PCR). In addition, the tissue samples and clinical information of 80 patients with NSCLC from our hospital were collected. Immunohistochemistry (IHC) was used to assess MPC1 expression, and OS was evaluated using Kaplan-Meier curves and the log-rank test. Univariate and multivariate Cox regression analyses were conducted to evaluate the prognostic values of the clinical characteristics and MPC1expression
Results
Analysis of public databases suggested that MPC1 was downregulated in NSCLC compared to that in normal lung tissue and predicted poor prognosis. In addition, the expression of MPC1 in NSCLC cell lines was lower than that in human bronchial epithelial (HBE) cells at both protein and mRNA levels. Further clinical analysis suggested that MPC1 expression was correlated with age, tumor T stage, and TNM stage. Kaplan-Meier analysis revealed that NSCLC patients with high MPC1 expression had a better prognosis, particularly in lung adenocarcinoma (LUAD), whereas no survival benefit was observed in lung squamous cell carcinoma (LUSC). Univariate and multivariate analyses suggested that MPC1 was an independent prognostic factor for patients with NSCLC.
Conclusions
MPC1 is poorly expressed in NSCLC, particularly in LUAD, which predicts a poor prognosis and may serve as an independent prognostic factor. Further studies on MPC1 may reveal new targets for the treatment of NSCLC.
Keywords
Highlight box
Key Findings
MPC1 expression is down-regulated and predicts poor prognosis in NSCLC patients.
What is Known and What is new?
MPC1 was lowly expressed in NSCLC tissue and cell lines. NSCLC patients with low MPC1 expression had a poor prognosis, especially those with LUAD.
What is the Implication, and What Should Change now?
Our study demonstrated that low MPC1 expression in NSCLC was associated with poor prognosis and could be used as an independent prognostic factor in NSCLC patients. Further studies of the potential molecular mechanism of MPC1 in NSCLC may provide new ideas for the prognosis and treatment of NSCLC patients.
Abbreviations
MPC1, Mitochondrial pyruvate carrier 1; NSCLC, non-small cell lung cancer; OS, Overall survival; TCGA, The Cancer Genome Atlas; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; IHC, Immunohistochemistry; HBE, human bronchial epithelial; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; EGFR, epidermal growth factor receptor; ALK, anaplastic lymphoma kinase; PD-L1, programmed cell death 1 ligand 1; GAPDH, Glyceraldehyde-3-phosphate dehydrogenase; PMSF, phenylmethanesulfonyl fluoride; OXPHOS, oxidative phosphorylation; erPCC, erlotinib- resistant persistent head and neck cancer cells; KDM5A, lysine (K) demethylase 5A; EMT, epithelial-mesenchymal transition; CRC, colorectal cancer; WB, Western blotting; M ± SD, mean ± standard deviation; BLCA, bladder cancer; BRCA, breast cancer; CESC, cervical squamous cell carcinoma and endocervical adenocarcinoma; GBM, glioblastoma multiforme; KIRC, kidney clear cell carcinoma; STATs, signal transducer and activator of transcription.
Introduction
Lung cancer is the most common morbidity and deadly cancer in China, and its incidence has increased in recent years. 1 Histopathological classification distinguishes two main types of lung cancer: small cell lung cancer and non-small cell lung cancer (NSCLC). NSCLC accounts for 85% of all lung cancers, mainly consisting of lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC). 2 Most patients with NSCLC are at an advanced stage at initial diagnosis because of inconspicuous early symptoms, and the 5-year survival rate is poor. 3 Current targeted therapies and immunotherapy based on molecular markers, such as epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), recombinant C-Ros oncogene 1, receptor tyrosine kinase (ROS1), and programmed cell death 1 ligand 1 (PD-L1) have improved the treatment prospects for NSCLC and achieved certain clinical benefits. However, the molecular mechanism of NSCLC progression remains unclear, and the clinical therapeutic effect is unsatisfactory.4-6 Thus, key target molecules must be identified for clinical prognosis and new therapeutic strategies for treating NSCLC.
Metabolic reprogramming is an important characteristic of tumor cell immortalization that satisfies the energy requirements and cellular macromolecular demands. 7 Tumor cells produce lactic acid through glycolysis even in the presence of sufficient oxygen for oxidative phosphorylation, which is known as the Warburg effect. 8 Several studies have shown that mitochondrial dysfunction plays a critical role in the malignant transformation of cells, and is closely related to tumor metabolic reprogramming. 9 Mitochondrial pyruvate carrier protein 1 (MPC1) is a key mitochondrial inner membrane protein that transfers the glycolysis product, pyruvate, to the mitochondrial matrix to enter the tricarboxylic acid cycle. 10 Previous studies have reported that MPC1 is lost or downregulated in multiple cancers, such as myeloma, gastric cancer, colorectal cancer, and prostate cancer, and is associated with poor prognosis.11-14 Our previous study have verified that MPC1 displays distinct expression patterns in lung cancer tissue compared to normal lung tissue. Notably, MPC1 plays a significant role in the progression t of lung cancer, particularly in the context of signal transducer and activator of transcription (STATs) signaling. 15 However, the expression of MPC1 in NSCLC and its clinical prognostic relevance have not yet been clearly elucidated. In this study, we investigated the expression of MPC1 in NSCLC, analyzed the correlation between MPC1 expression and the clinical characteristics and prognosis of patients with NSCLC, and evaluated the clinical significance of MPC1 as a potential therapeutic target in NSCLC.
Methods
Dataset analyses
MPC1 RNA expression in pan-cancer and adjacent normal tissues was retrieved from TIMER2.0 (http://timer.cistrome.org/). 16 The expression levels of MPC1 in carcinoma and adjacent tissues in lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC) were derived from the UALCAN database (https://ualcan.path.uab.edu/index.html), which is based on the Cancer Genome Atlas (TCGA) database. Gene expression levels were shown using a log2 (TPM + 1) scale, where TPM refers transcripts per million. 17 Kaplan-Meier Plotter website tools (http://kmplot.com/analysis/index) were used to construct survival curves and evaluate the prognostic potential of MPC1 in NSCLC, especially in LUAD and LUSC. 18
Cell culture and reagents
Human NSCLC cell lines A549 (accession number: SCSP-503), H1975 (accession number: SCSP-597), and SK-MES-1(accession number: SCSP-5010) were purchased from the Shanghai Cell Bank of the Chinese Academy of Science. Human normal bronchial epithelial cells (HBE) were obtained from the Department of Pathology of Southwest Hospital Affiliated to Army Military Medical University. The cells were cultured in Dulbecco's modified Eagle's medium (DMEM) (HyClone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS) (HyClone, Logan, UT, USA) and 100 U/mL penicillin–streptomycin (HyClone, Logan, UT, USA). The cells were maintained in a humidified 37 °C incubator with 5% CO2.
Quantitative reverse transcriptase polymerase chain reaction (qRT-PCR)
Total RNA was extracted from NSCLC and HBE cells using an RNA fast200 kit (Fastagen, China) and reverse transcribed to cDNA using Prime Script TM RT Master Mix (TaKaRa, Japan). Quantitative PCR was performed using SYBR® Premix ExTaqTM (TaKaRa, Japan). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control. The detailed steps were performed according to the manufacturer's instructions. The data were calculated using the 2-ΔΔCt method, 19 and each cDNA sample was tested at least in triplicate. The primer sequences were as follows: MPC1 (forward: 5′-TGGCTAAAGGAGCAGAGGAA-3′, reverse: 5′-ATGACCACATCACGGCTACA-3′); GAPDH (forward: 5′-TGTTCGTCATGGGTGTGAAC-3′, reverse: 5′-ATGGCATGGACTGTGGTCAT-3′).
Western blotting (WB)
Cells were harvested when the confluence reached 80%, and total protein was extracted using radio immunoprecipitation assay (RIPA) lysis buffer (Beyotime, China) containing the protease inhibitor phenylmethanesulfonyl fluoride (PMSF) (Beyotime, China). A BCA assay (Beyotime, China) was used for protein quantification. The WB procedure was performed according to the manufacturer's instructions and described previously. 20 The primary antibodies used were: anti-MPC1 (Abcam, ab74871, 1:1000) and anti-GAPDH (CST, #2118, 1:1000). Horseradish peroxidase-conjugated IgG (CST, #7074, 1:1000) was used as the secondary antibody.
Patients and tissue specimens
Eighty patients diagnosed with NSCLC from June 2019 to June 2020 were enrolled. The study was approval by the Institutional Ethics Committee, and all patients provided written informed consent. Patients who met the following criteria were included: 1) pathologically confirmed diagnosis of LUAD or LUSC, 2) available paraffin sections for subsequent immunohistochemistry, and 3) available information on clinical features (sex, age, smoking, pathological type, lymph node invasion, T staging, metastasis, and TNM stage) and follow-up information. Patients were followed up for a mean of 36 months. The final follow-up was conducted in July 2023. The clinical data are presented in Table 1.
Association of MPC1 Expression with Clinicopathological Parameters of Patients with NSCLC.
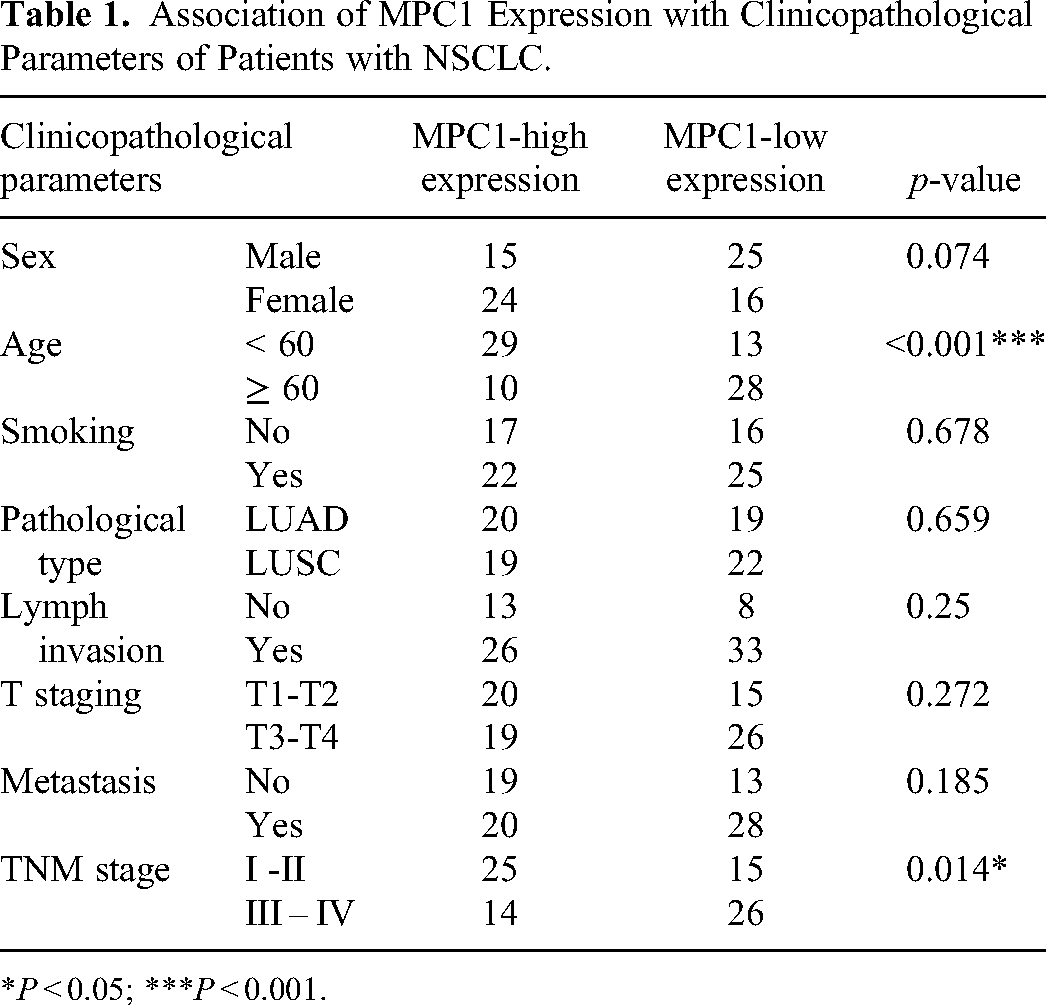
*P < 0.05; ***P < 0.001.
Immunohistochemistry (IHC) and scoring
All 80 patients included in this study had their cancer tissues subjected to immunohistochemistry staining. Paraffin-embedded 4-µm-thick lung cancer tissue sections were dewaxed, hydrated, antigen repaired, inactivated with endogenous peroxidase, blocked, and incubated with primary antibodies (rabbit polyclonal anti-MPC1, Abcam, ab74871, 1:250;) at 4 °C overnight. Sections were then incubated with a secondary antibody (avidin-biotin-peroxidase; DAKO), and diaminobenzidine was used for color development. The detailed steps follow a previously described procedure. 21 All images were captured by a microscope (OLYMPUS) with 20×, 40× objective lens. Two pathology professors independently scored the MPC1 staining in each section as follows: 1) percentage of positive tumor cells: Score 0, 0%–5% positive cells; Score 1, 5–25% positive cells; Score 2, 25%–50% positive cells; Score 3, 50%–75% positive cells; and Score 4, 75%–100% positive cells. 2) The staining intensity was scored as follows: Score 0, no positive staining; Score 1, weak staining; Score 2, moderate staining; Score 3, strong staining. Final scores were obtained by multiplying the percentage of positive tumor cells by the staining intensity scores. They were analyzed using statistical X-tile software, with high and low MPC1 expression defined based on a cutoff score of 4. 22
Statistical analysis
SPSS 27.0.1 (Chicago, IL, USA) and GraphPad 6.0 software (San Diego, CA, USA) were used for the data analysis and processing. Continuous variables were expressed as the mean ± standard deviation (M ± SD). Student's t-test or one-way ANOVA was used for comparison between groups. Pearson's chi-square test was used to analyze the correlation between MPC1 expression and the clinical features. The Kaplan–Meier method was used to investigate the prognostic value of MPC1 in NSCLC. Univariate and multivariate Cox regression analyses were used to identify independent prognostic factors. Differences were considered significant if P values were lower than 0.05. All significant differences were defined as * P < 0.05, ** P < 0.01, and *** P < 0.001.
Results
MPC1 is downregulated in NSCLC and is associated with a favorable prognosis
In our pan-cancer investigation using TIMER2.0, the expression of MPC1 was downregulation in tumor tissues compared to that in normal adjuncts across various cancer types, including bladder cancer (BLCA), breast cancer (BRCA), cervical squamous cell carcinoma and endocervical adenocarcinoma (CESC), glioblastoma multiforme (GBM), kidney clear cell carcinoma (KIRC), LUAD, and LUSC (Figure 1A). The expression trend was further verified in LUAD and LUSC by UALCAN based on the TCGA database, confirming lower MPC1 expression in tumor tissues. (Figure 1B-C). A subsequent assessment of NSCLC cell lines (A549, H1975, SK-MES-1) and normal bronchial epithelial cells (HBE) using qRT-PCR and western blotting revealed consistently decreased MPC1 levels in NSCLC cell lines, both at the mRNA and protein levels, compared to HBE cells (Figure 1D-F). Additionally, Kaplan-Meier Plotter was used to evaluate the prognostic value of MPC1 expression in patients with NSCLC. Strikingly, NSCLC patients with high MPC1 expression levels demonstrated significantly improved overall survival (OS) compared to those with low MPC1 expression levels (Figure 1G). When LUAD and LUSC were separated, we found that the survival benefit was particularly prominent in LUAD patients with higher MPC1 expression (Figure 1H). In contrast, no significant difference in OS was observed between LUSC patients with high and low MPC1 expression (Figure 1I). These results indicated that the expression of MPC1 is lower in NSCLC tissues than in normal lung tissues and has prognostic value, suggesting its potential as a biomarker for predicting poor prognosis in NSCLC patients, particularly in LUAD cases.
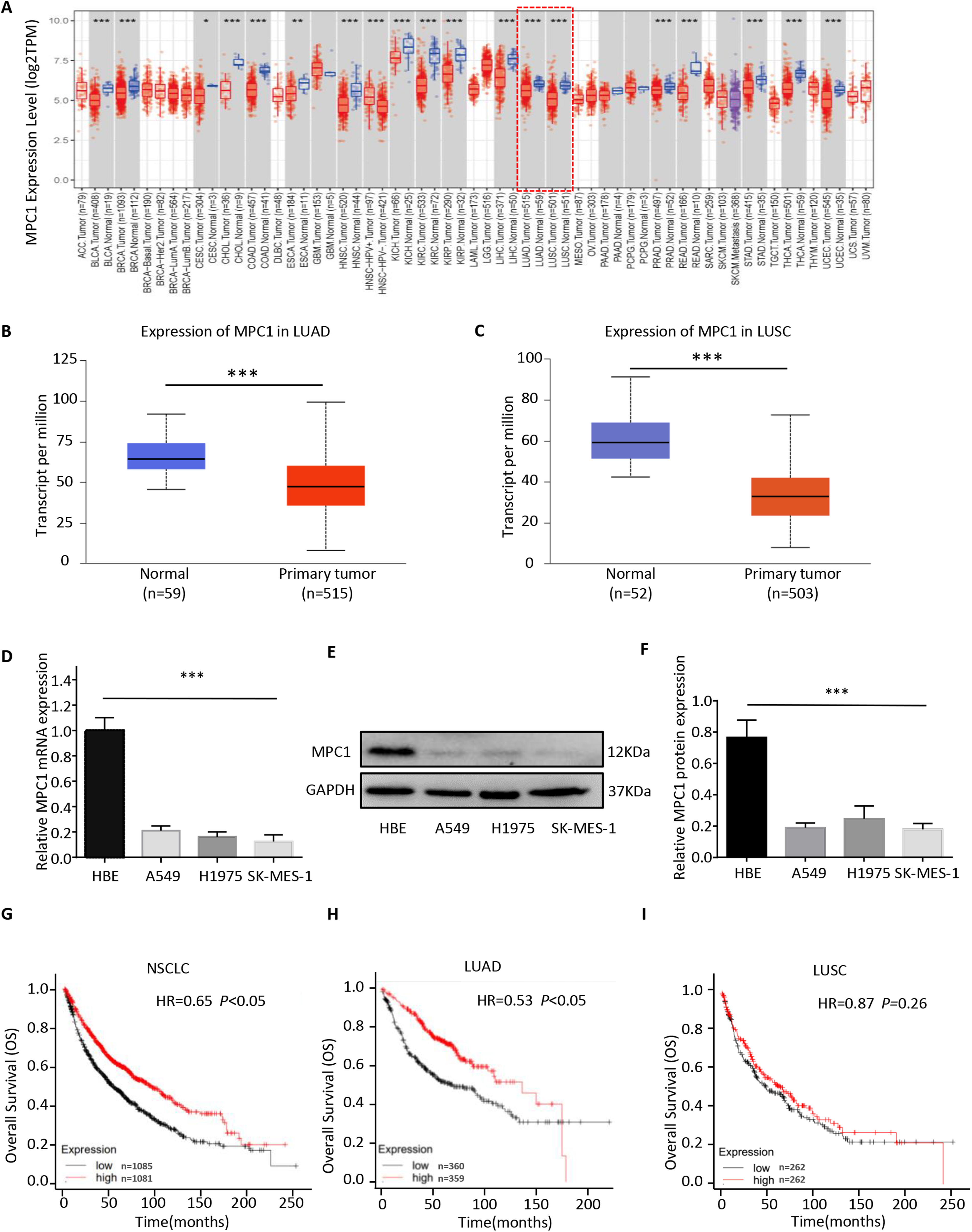
MPC1 is poorly expressed and predicts poor prognosis in NSCLC. (A) MPC1 mRNA expression levels in pan-cancer and adjunct normal tissues according to the TIMER2.0 analysis of TCGA data; (B-C) MPC1 protein levels in LUAD (B) and LUSC (C) in the UALCAN database; (D-E) MPC1 expression in human NSCLC cell lines and HBE cells as assessed by qRT-PCR (D) and western blotting (E), with GAPDH served as loading control; (F) The relative MPC1 protein expression was quantified by western blot. (G-I) Kaplan-Meier curves for OS of patients with NSCLC (G), LUAD (H) and LUSC (I) stratified by MPC1 expression levels as assessed using the Kaplan-Meier plotter database. (*P < 0.05, **P < 0.01, ***P < 0.001).
Associations of MPC1 expression with clinicopathological features of patients with NSCLC
Next, to verify the correlation between the expression of MPC1 and the clinical features of patients with NSCLC, 80 patients with pathologically diagnosed NSCLC from our hospital were included with an average age of 58 years (range, 34-76 years). The clinical features of the patients are presented in Table 1. Using IHC, patients were divided into a high MPC1 expression group (n = 39) and a low MPC1 expression group (n = 41) based on the MPC1 IHC score in the tumor tissues. Representative IHC images showed that MPC1 was mainly located in the cytoplasm of both LUAD and LUSC (Figure 2). Furthermore, correlation analyses using the Pearson chi-square test revealed that MPC1 expression was negatively correlated with patient age and TNM stage, indicating that patients with advanced age and TNM stage had lower MPC1 expression levels. However, there were no significant correlations between MPC1 expression and sex, smoking history, histological type, lymph node invasion, tumor size (T), or metastasis (Table 1).

Typical immunohistochemical images of low and high MPC1 expression levels in LUAD and LUSC tumor tissues. Low and high MPC1 expression levels in LUAD (A) and LUSC (B). (Scale bar, 200 μm).
Prognostic Significance of MPC1 Expression in NSCLC Patients
Kaplan-Meier curves and the log-rank test were used to assess the impact of MPC1 expression on OS in patients with NSCLC. The patients with high MPC1 expression exhibited a significantly better OS than those with low MPC1 expression (hazard ratio [HR] = 0.48, P = 0.008). Further analysis in LUAD patients showed that high MPC1 expression levels were indicative of a favorable prognosis (HR = 0.47, P = 0.041). In contrast, there was no significant difference in OS between LUSC patients with high and low MPC1 expression levels (HR = 0.36, P = 0.27) (Figure 3). Univariate and multivariate Cox regression analyses were used to identify the prognostic value of clinical features. The univariate analysis demonstrated that female sex, no smoking, LUAD, lower tumor (T) stage and TNM stage, no lymph node invasion, no metastasis, and high MPC1 expression were significantly associated with better OS in patients with NSCLC. Considering these significant clinical features in the multivariate analysis, T stage (HR = 3.447, 95% CI: 1.367-8.691, P = 0.009), TNM stage (HR = 3.497, 95% CI: 1.214-10.074, P = 0.02), and MPC1 expression (HR =2.036, 95% CI: 1.364-2.655, P = 0.011) were significant independent prognostic factors for NSCLC. Notably, patients with high MPC1 expression exhibited significant survival benefits (Table 2).

Correlation between MPC1 expression and the prognosis of NSCLC patients. (A-C) Kaplan-Meier survival curves of OS between high and low MPC1 expression in patients with NSCLC (A), LUAD (B), LUSC (C).
The Correlation Between Clinicopathological Parameters and Survival Rates was Analyzed with Univariate and Multivariate Analysis.

*P < 0.05; **P < 0.01; ***P < 0.001.
Abbreviations: CI, confidence interval; HR, hazard ratio.
Discussion
In recent years, the incidence and mortality rates of NSCLC has notable increased, with a 5-year survival rate is less than 20%. 23 Although extensive research has focused on the pathogenesis, early screening, diagnosis, and treatment of NSCLC, the intricate evolutionary mechanisms underlying NSCLC remain insufficiently elucidated. 24 Unraveling the biological intricacies governing tumor development, and identifying characteristic biomarkers with prognostic significance is important for the advancement of effective NSCLC treatments.
Tumor metabolic reprogramming, a prominent aspect among the ten molecular features of tumor cells, plays a pivotal role in adapting to dynamic requirements to meet the needs of tumor homeostasis and rapid growth. 25 The Warburg effect demonstrates that tumor cells preferentially utilize glucose for glycolysis rather than oxidative phosphorylation (OXPHOS), even under abundant oxygen conditions, resulting in a large amount of lactic acid. 26 Increasing evidence suggests that tumor metabolic reprogramming based on the Warburg effect is involved in tumor metabolism, proliferation, progression, distant metastasis, and chemotherapy resistance. 8 MPC is crucially linked to cytosolic glycolysis and mitochondrial OXPHOS metabolism, and is positioned at the intersection of the two processes.27,28 MPC1, an important functional protein in the inner mitochondrial membrane, plays a pivotal role in the transport of pyruvate from the cytoplasm to the mitochondria to enter the tricarboxylic acid cycle for oxidative decomposition and energy supply. MPC1 dysregulation may lead to compromised mitochondrial pyruvate uptake and impaired pyruvate oxidation in multiple human tumor cells. 12
Previous studies indicated that low MPC1 expression in many cancer tissues, including BRCA, KIRC, GBM, and colorectal cancer (CRC), contributes to tumor growth, invasion, and poor prognosis. 12 Based on comprehensive database analyses, we observed downregulation of MPC1 expression across various tumors, which has been previously reported. Of note, the expression of MPC1 is significantly lower in both LUAD and LUSC tissues than adjacent normal tissues. Consist with this, we further verified that the expression of MPC1 in NSCLC cell lines (A549, H1975, SK-MES-1) was downregulated compared to that in HEB cells at both the mRNA and protein levels. We have identified that low MPC1 expression was significantly correlated with the advanced age and TNM stage of NSCLC patients, indicating that patients with low MPC1 expression may have a higher risk of malignancy and tumor lymph node metastasis. Similarly, Zhou et al demonstrated that lower MPC1 expression is associated with advanced tumor stage, greater invasion depth, and lymph node metastasis in gastric cancer. 29 In a study on CRC, the expression of MPC1 decreased in CRC metastasis compared to that in primary CRC tissues. 30 MPC1 inhibits the phosphorylation and nuclear translocation of STAT3 by interacting with STAT3 to anchor it in the mitochondria, ultimately preventing the expression of tumor stem genes and invasion- and metastasis-related genes in LUAD. In LUAD cells with low MPC1 expression, STAT3 is released from the mitochondria and is activated by phosphorylation to promote the transcription of target genes, thereby promoting the dryness, invasion, and metastasis of tumor cells. 15 MPC1 has been shown to inhibit the invasion of renal cell carcinoma (RCC) cells in vitro and reduce tumor growth in vivo, potentially via suppression of MMP7 and MMP9. Further research revealed that hypoxia-induced loss of MPC1 in RCC cells was negatively correlated with HIF1α expression in both cells and patient samples. Overexpression of MPC1 can significantly decrease the average weight and volume of RCC compared with MPC1-deficient groups.31 These results imply that low MPC1 expression promotes tumor progression and can be used as a potential target for tumor therapy.
Several studies have suggested that MPC1 may be a key molecule in tumor metabolic reprogramming, corresponding to tumor prognosis. You et al 32 found that MPC1 is expressed at low levels in erlotinib- resistant persistent head and neck cancer cells (erPCC), and that high expression of lysine (K) demethylase 5A(KDM5A) promotes erlotinib-resistant persistent head and neck cancer cell epithelial–mesenchymal transition and increases susceptibility to iron death by inhibiting MPC1 expression. Yi et al 33 demonstrated low MPC1 expression in GBM, which is closely related to temozolomide resistance and poor prognosis. Tian et al 30 found that MPC1 was significantly downregulated in metastatic CRC and activated the Wnt/β-catenin pathway through nuclear translocation of β-catenin, thereby mediating the expression of MMP7, E-cadherin, Snail1, and myc to promote poor prognosis in CRC. Consistent with previous studies, in our analysis of the UALCAN database, we found that high expression of MPC1 in NSCLC patients, especially in patients with LUAD, was remarkably associated with a favorable prognosis. In contrast, in LUSC, there was no significant difference in OS between patients with high and low MPC1 expression. Similar results were found in our clinical study, low expression of MPC1 was closely associated with poor prognosis in NSCLC, especially in LUAD. Univariate and multivariate Cox regression analyses suggested that MPC1 could be used as an independent prognostic factor for NSCLC. These results suggest that MPC1 expression is downregulated in NSCLC tissues, highlighting its potential as a predictive marker for the prognosis of patients with NSCLC, especially those with LUAD. Further studies elucidating the molecular mechanism of action of MPC1 in the malignant phenotype of LUAD may provide new treatment strategies for NSCLC. The difference between LUAD and LUSC may be due to the sharply distinct origin, histopathology, clinicopathological features, driver genetic changes, transcriptomic levels, control of cellular networks. 34
In this study, an integrated analysis encompassing online databases, NSCLC cell lines, and tissue samples revealed notable downregulation of MPC1 expression in NSCLC. This decrease is inversely correlated with both age and TNM stage, indicating a potential association with disease progression. Strikingly, high MPC1 expression, particularly patients with LUAD, is associated with improved survival outcomes. Further substantiating its significance, Cox regression analysis identified MPC1 as an independent prognostic factor for NSCLC. However, the specific role of MPC1 in driving malignant phenotypes, including its involvement in the metabolic reprogramming of NSCLC cells, remains unclear. Future investigations will focus on unraveling the precise contribution of MPC1 to the progression of NSCLC to provide valuable insights into potential therapeutic avenues.
Conclusion
Our study conclusively establishes that diminished MPC1 expression is intimately linked to unfavorable prognosis and holds promise as an independent prognostic factor, particularly in LUAD patients among those with NSCLC. Delving deeper into the underlying molecular mechanisms of MPC1 in NSCLC may yield novel perspectives and strategies for improving prognosis and refining treatment modalities for patients afflicted with this condition.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241282080 - Supplemental material for Mitochondrial Pyruvate Carrier 1 as a Novel Prognostic Biomarker in Non-Small Cell Lung Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338241282080 for Mitochondrial Pyruvate Carrier 1 as a Novel Prognostic Biomarker in Non-Small Cell Lung Cancer by Hongbo Zou, Yunfei Yin, Kai Xiong, Xuelian Luo, Zhongju Sun, Bijing Mao, Qichao Xie, Mei Tan and Rui Kong in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
Author Contributions
(I) Conception and design: HZ and RK; (II) Administrative support: RK and QX; (III) Provision of study materials or patients: HZ, YY, XL, BM and MT; (IV) Collection and assembly of data: HZ, ZS and KX; (V) Data analysis and interpretation: HZ, RK and Y.Y.; (VI) Manuscript writing: All authors; (VII) Final approval of the manuscript: All authors.
Data Availability Statement
The data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Ethics Committee of the Third Affiliated Hospital of Chongqing Medical University(NO:201915 and NO:202306) and informed consent was obtained from all individual participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Third Affiliated Hospital of Chongqing Medical University Scientific Research Incubation Project Youth Project, (grant number KY19030).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
