Abstract
Introduction
The role of radiotherapy (RT) in de novo metastatic breast cancer (dnMBC) patients undergoing surgery remains controversial due to limited evidence. This study aimed to evaluate the impact of postoperative radiotherapy on survival outcomes in this population.
Materials and methods
We retrospectively analyzed 102 dnMBC patients who underwent surgery at a provincial cancer hospital. Patients were grouped based on whether they received postoperative RT. Baseline characteristics were compared using the chi-square test. Kaplan-Meier analysis and Cox proportional hazards models were used to assess the prognostic impact of postoperative radiotherapy on local recurrence-free survival (LRFS), progression-free survival (PFS), and overall survival (OS).
Results
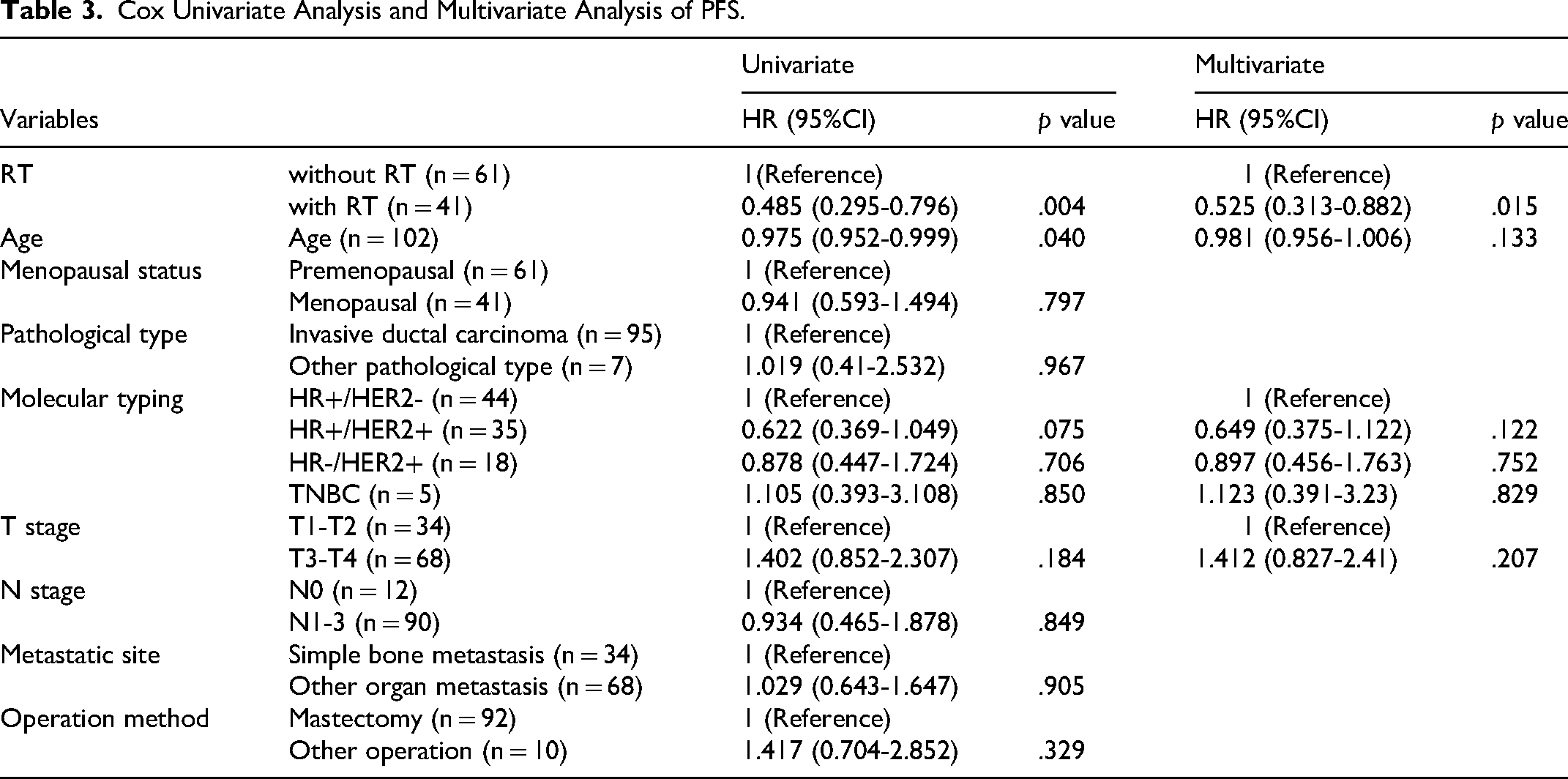
KM survival analysis showed that postoperative RT significantly improved LRFS (HR = 0.3634, 95%CI 0.1552-0.8508, p = .0197) and PFS (HR = 0.4903, 95%CI 0.3061-0.7855, p = .003) but had no significant effect on OS (HR = 0.7337, 95%CI 0.3514-1.508, p = .5395). Multivariate analysis identified postoperative RT as an independent protective factor for LRFS (HR = 0.265, 95%CI 0.088-0.795, p = .018) and PFS (HR = 0.525, 95%CI 0.313-0.882, p = .015). Subgroup analysis showed that for LRFS, RT had no significant interaction with different subgroup classification variables (all interaction p > .05). However, RT had a significant interaction with N stage for PFS (p = .016), specifically in N1-3 patients (HR = 0.384, 95% CI 0.221-0.668).
Conclusion
RT may improve disease control in selected dnMBC patients undergoing surgery, particularly those with lymph node metastasis. However, these findings still require further validation in larger, multicenter cohorts.
Introduction
Breast cancer is one of the most prevalent malignancies among women worldwide. 1 Approximately 3% to 8% of patients are diagnosed with distant metastasis at their initial presentation, defined as de novo metastatic breast cancer (dnMBC). 2 Prognosis for these patients is generally poor, with survival ranging from a few months to several years.3–5 Standard treatment for stage IV breast cancer emphasizes systemic therapy, with local treatments like surgery and radiotherapy applied selectively.
Emerging evidence suggests that timely surgical intervention may confer survival benefits in dnMBC patients.6–9 With the progress of systemic therapy and the deepening of statistical research, more and more evidence emphasizes the role of surgical treatment.10,11 However, the potential additional benefit of combining surgery and radiotherapy remains contentious due to inconsistent findings across studies.12–14 Although prospective randomized controlled trials (RCT) have evaluated the impact of local treatments on prognosis,15–17 the specific role of postoperative radiotherapy remains underexplored.
For instance, the EA2108 clinical trial demonstrated that locoregional progression was significantly less frequent in patients receiving locoregional therapy (3-year rate: 16.3% vs 39.8%, p < .001), yet no significant difference in overall survival (OS) was observed (3-year OS: 67.9% vs 68.4%, HR = 1.11, 90% CI 0.82-1.52, p = .57). 17 Similarly, another RCT reported a median OS of 19.2 months in the locoregional therapy group compared to 20.5 months in the non-locoregional therapy group (HR = 1.04, 95% CI 0.81-1.34, p = .79), indicating no significant survival benefit. 15 In contrast, the MF07-01 trial found a 34% reduction in the hazard of death in the locoregional therapy group (HR = 0.66, 95% CI 0.49-0.88, p = .005). 16 A meta-analysis focusing on local therapy in dnMBC patients further supported these findings, showing a significant 31.8% reduction in mortality (HR = 0.6823, 95% CI 0.6365-0.7314, p < .05). 18
Current research on de novo metastatic breast cancer (dnMBC) has primarily focused on local therapies, particularly surgery, while the potential benefits of combined postoperative radiotherapy remain unclear. Given China's unique patient population and disease characteristics, there is a pressing need to evaluate this treatment approach. Our study systematically assesses the impact of postoperative radiotherapy on locoregional recurrence-free survival (LRFS), progression-free survival (PFS), and overall survival (OS) in dnMBC patients, providing critical evidence for this multimodal strategy.
Methods
Clinical Data
The clinical data of dnMBC patients (n = 102) who treated at a provincial cancer hospital, between January 1, 2014, and January 1, 2021, were retrospective collected (Figure 1). Including age, menopausal status, TNM staging, molecular typing, site of metastasis at first treatment, surgical method, time of diagnosis, time of first local recurrence or progression, and overall survival time. This study was approved by the Ethics Committee of Hospital and the data has been anonymized, exempting informed consent. The reporting of this study conforms to STROBE guidelines. 19

A Flowchart Detailing the Process of Patient Screening.
Inclusion and Exclusion Criteria
Inclusion criteria:
1. Histopathological confirmation of breast cancer. 2. Presence of distant metastasis at initial diagnosis, confirmed through CT, MRI, PET-CT, bone ECT, or tissue biopsy. 3. Received surgical treatment. 4. Continuation of systemic therapy following surgery.
Exclusion criteria:
1. Incomplete medical records. 2. Lack of surgical intervention. 3. Presence of metastatic lesions originating from malignancies other than breast cancer, to ensure diagnostic accuracy and avoid confounding effects on survival analysis.
Follow-up
The follow-up items included whether local recurrence occurred, time to local recurrence, whether disease progression occurred, time to first progression, whether death occurred, and time of death. LRFS, PFS, and OS were calculated from the date of surgical intervention until the occurrence of the respective outcome or the end of follow-up. Disease recurrence or progression was determined through pathological examination or imaging studies. Follow-up data were collected through outpatient visits, inpatient check-ups, and regular telephone interviews, and were entered by trained personnel. To ensure data accuracy, all collected data were cross-checked with hospital records and patient medical records.
Patients who were lost to follow-up were treated as right-censored at their last known follow-up time. Their data were included in the survival analysis until the point of their last follow-up, ensuring that all available information was utilized while minimizing potential bias. This approach is consistent with standard practices in survival analysis and allows for the inclusion of valuable data from patients with incomplete follow-up. 20
Subgroup Analysis
Patients were stratified into subgroups by menopausal status, histopathological type, molecular subtype, T stage (T1-2 vs T3-4, consistent with conventional postoperative radiotherapy criteria for breast cancer), N stage (N0 vs N1-3), visceral metastasis status, and surgical approach (mastectomy vs breast-conserving surgery/reconstruction). Within each subgroup, we evaluated the prognostic impact of postoperative radiotherapy and examined its interactions with other variables using Cox proportional hazards models.
Statistical Methods
Statistical analyses were performed using R4.2.2, SPSS 25.0 and GraphPad Prism 10.1.2. Patients were divided into two groups: those who received postoperative radiotherapy targeting the primary focus (“with RT”) and those who did not (“without RT”). Baseline characteristics between the groups were compared using Chi-square tests. Local recurrence-free survival (LRFS), progression-free survival (PFS) and overall survival (OS) were analyzed using the Kaplan-Meier method, with group comparisons performed via the log-rank test. To account for multiple testing across the three endpoints (LRFS, PFS, and OS), we applied the Holm-Bonferroni correction. We performed post-hoc power analyses to evaluate the statistical power of our study based on the observed effect sizes. Evaluate Hazard ratios (HR) were calculated using Cox proportional hazards models. Variables with p < .2 in univariate analysis were included in multivariate analysis. A p < .05 was considered statistically significant.
Results
Patient Characteristics
A total of 102 patients were included: 41 in the “with RT” group and 61 in the “without RT” group. The follow-up durations for the radiotherapy and non-radiotherapy groups ranged from 13 to 101 months (median 53 months) and 13 to 100 months (median 52 months), respectively. Baseline characteristics such as age, menopausal status, pathological type, molecular subtype, primary tumor T stage, N stage, metastatic status at diagnosis, and surgical method showed no significant differences between the groups (p > .05) (Table 1), confirming comparability.
Clinical Characteristics of dnMBC.

Survival Analysis
During follow-up, 24 cases of disease progression (including 4 local recurrences) and 10 deaths occurred in the “with RT” group, compared to 51 cases of progression (including 18 local recurrences) and 20 deaths in the “without RT” group. Kaplan-Meier analysis revealed significant improvements in LRFS (HR = 0.3634, 95%CI 0.1552-0.8508, p = .0197) and PFS (HR = 0.4903, 95%CI 0.3061-0.7855, p = .003) with primary focus radiotherapy but no significant benefit in OS (HR = 0.7337, 95%CI 0.3514-1.508, p = .5395) (Figure 2). Specifically, the 5-year LRFS rate was 89.9% (95%CI 81.1%-99.8%) in the “with RT” group compared to 66.1% (95%CI 53.5%-81.8%) in the “without RT” group. Similarly, the 5-year PFS rate was 42.3% (95%CI 28.8%-62.0%) in the “with RT” group, significantly higher than the 12.9% (95%CI 6.13%-27.3%) observed in the “without RT” group. In contrast, the 5-year OS rate was 74.5% (95%CI 62.0%-89.5%) in the “with RT” group compared to 63.3% (95%CI 51.2%-78.2%) in the “without RT” group.

Kaplan-Meier Survival Curves of with RT Group and Without RT Group A. The with RT Group Improved the LRFS of De Novo IV Stage Breast Cancer (HR = 0.3634, 95%CI 0.1552-0.8508, p = .0197). B. The with RT Group Extended Progression Free Survival Compared to the Without RT Group (HR = 0.4903, 95%CI 0.3061-0.7855, p = .003). C. There Was No Significant Difference in OS Between the with RT Group and the Without RT Group (HR = 0.7337, 95%CI 0.3514-1.508, p = .5395).
Prognostic Influencing Factors Analysis
Univariate Cox analysis showed that RT, menopausal status, T stage, and surgical approach might be influencing factors for LRFS (p < .2). When these variables were included in the multivariate analysis, radiotherapy was identified as a protective factor for LRFS (HR = 0.265, 95% CI 0.088-0.795, p = .018), whereas postmenopausal status, higher T stage, breast-conserving surgery, and breast reconstruction surgery were risk factors (HR > 1, p < .05) (Table 2). Additionally, univariate Cox analysis indicated that radiotherapy, age, molecular subtype, and T stage might be influencing factors for PFS (p < .2). After incorporating these variables into the multivariate analysis, radiotherapy was found to be an independent protective factor for PFS (HR = 0.525, 95% CI 0.313-0.882, p = .015) (Table 3).
Cox Univariate Analysis and Multivariate Analysis of LRFS.

Cox Univariate Analysis and Multivariate Analysis of PFS.
Subgroup Analysis
Given the complexity of dnMBC and the substantial individual variations among patients, it is crucial to identify which patients may benefit from local radiotherapy. To address this issue, we employed COX proportional hazards models to analyze the interaction between radiotherapy and various classification variables, and evaluated the impact of postoperative radiotherapy on LRFS and PFS across different subgroups.
As shown in Figure 3, no significant interactions were observed between RT and menopausal status, molecular subtype, T stage, N stage, metastatic site, or surgical approach (all interaction p > .05). Thus, the effect of radiotherapy on LRFS may not differ significantly across subgroups.

Forest Plot of Subgroup Analysis for LRFS. HR and 95%CI of Radiotherapy (vs no Radiotherapy) Across Subgroups Stratified by Menstrual status, Molecular Subtype, T Stage, N Stage, Metastatic Site, and Surgical Method.
However, in Figure 4, the impact of RT on PFS varied by N stage (interaction p = .016), with more significant benefit observed in N1-3 patients (HR = 0.384, 95% CI 0.221-0.668), while no significant difference was found in N0 patients (HR = 2.113, 95% CI 0.501-8.912).

Forest Plot of Subgroup Analysis for PFS. HR and 95%CI of Radiotherapy (vs no radiotherapy) Across Subgroups Stratified by Menstrual Status, Molecular Subtype, T stage, N stage, Metastatic Site, and Surgical Method.
Discussion
The role of RT in the dnMBC patients undergoing primary tumor resection warrants further elucidation. Current literature primarily focuses on comparisons between local therapy (particularly surgery) and systemic therapy alone.15,17,18,21 Notably, there is a paucity of randomized controlled trials (RCTs) and meta-analyses directly evaluating the survival impact of postoperative RT versus no RT. Furthermore, existing database-derived retrospective studies have predominantly examined overall survival (OS) differences,18,22,23 with limited assessment of RT's effects on disease control endpoints such as LRFS and PFS. Distinct from prior research emphases, this study specifically investigates the influence of postoperative RT on disease control and survival outcomes in dnMBC patients, with particular attention to LRFS, PFS, and OS.
Our data demonstrate that postoperative RT significantly improves LRFS (HR = 0.3634, 95%CI 0.1552-0.8508, p = .0197) and PFS (HR = 0.4903, 95%CI 0.3061-0.7855, p = .003) in dnMBC patients, consistent with previous findings.12,16 For instance, they reported superior distant progression-free survival (DPFS) with surgery plus RT compared to surgery alone (32% vs 18%; p < .0001). 12 However, our analysis did not reveal statistically significant OS benefits (HR = 0.734, 95% CI 0.351-1.508, p = .540), mirroring results from the M07-01 trial where subgroup analysis showed no significant OS difference between RT and non-RT patients. 16 Contrastingly, large database retrospective studies and meta-analyses have documented significant OS improvements with adjuvant RT (HR = .74, 95% CI 0.69-0.80, p < .001; 22 HR = 0.744, 95% CI 0.628-0.881, p = .001). 23 These discrepancies may be attributable to our study's relatively limited sample size (n = 102) and shorter follow-up duration compared to these large-scale analyses.
Multivariate Cox regression analysis identified postoperative RT as an independent prognostic factor for both LRFS (HR = 0.265, 95%CI 0.088-0.795, p = .018) and PFS (HR = 0.525, 95%CI 0.313-0.882, p = .015), thereby providing novel evidence supporting the therapeutic value of adjuvant RT in dnMBC disease control. Notably, our subgroup analysis revealed that patients with lymph node involvement (N1-3) derived more substantial PFS benefit (p = .016) from RT (HR = 0.384, 95% CI 0.221-0.668), a finding that corroborates the SEER study's observation of enhanced therapeutic effect in node-positive patients. 23
To minimize potential biases, we implemented rigorous methodological safeguards. Baseline characteristics were balanced between groups as confirmed by chi-square testing (all p > .05), thereby reducing selection bias. Furthermore, we employed multivariate adjustment for potential confounding factors to enhance the validity of our findings. Nevertheless, as postoperative RT is not standard practice for dnMBC, potential confounding by indication (eg, selection of healthier patients or those with more favorable disease characteristics for RT) cannot be entirely excluded. Incorporation of comprehensive performance status metrics (eg, ECOG scores) in future studies would help address this limitation.
To ensure statistical robustness, we applied the Holm-Bonferroni method for multiple testing correction and conducted interaction analyses to identify potentially underpowered subgroup comparisons. As a retrospective study, our research was inherently limited by the availability of existing clinical data, precluding a priori sample size calculation. However, post-hoc power analysis demonstrated that our study achieved >99% statistical power for primary endpoints (LRFS, PFS, OS) using log-rank tests. Nevertheless, we acknowledge that certain subgroup analyses were underpowered due to small sample sizes in some categories, potentially compromising the robustness of these specific findings. These results should therefore be interpreted with caution and considered exploratory. Large-scale, prospective multicenter studies are warranted to validate these observations.
Conclusion
Postoperative radiotherapy appears to confer locoregional and progression-free survival advantages in selected dnMBC patients, with particularly pronounced benefits observed in those with lymph node metastases. However, given the limitations imposed by our study's sample size and follow-up duration, definitive conclusions regarding the role of RT in this setting await validation through larger, prospective multicenter trials. These future studies should incorporate comprehensive patient stratification and standardized outcome assessments to provide more robust evidence for clinical decision-making.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this work the authors used ChatGPT in order to polish the article, enhance academic rigor and improve sentence structure to meet academic standards. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Footnotes
Abbreviations
Ethical Considerations
The study was conducted in accordance with the Declaration of the World Medical Association in Helsinki. This study has been approved by the Ethics Committee of Guangxi Medical University Cancer Hospital, with ethics code KY2025035.
Author Contributions
YNJ and HWY contributed to conception and design. PCL and LNW acquired data. PCL analyzed data. PCL, LNW, YNJ and HWY wrote and revised the manuscript. YNJ and HWY contributed to supervision. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangxi Zhuang Autonomous Region, National Natural Science Foundation of China, (grant number 2020GXNSFBA297072, 81860464).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
