Abstract
Keywords
Introduction
De novo stage IV breast cancer refers to patients with metastatic breast cancer at initial diagnosis, and has been reported that approximately 6% to 10% of newly diagnosed breast cancer patients present with distant metastases.1–3 This group of patients with de novo metastatic breast cancer (dnMBC) makes up about 3% to 8% of new breast cancer diagnoses in the United States and Western,1,4 and 3% to 5% of these patients in China. 5 The overall survival (OS) was approximately 16 to 29 months. 6 For several decades, treatment of metastatic breast cancer has been challenged by lack of reliable therapeutic options. As with other advanced cancers, it is considered an incurable disease and significantly longer OS with the best possible quality of life is the ultimate goal for the treatment.7,8 In the last few years, although the therapeutic strategy for metastatic breast cancer has undergone changes, systemic therapy has remained the cornerstone of treatment in patients with dnMBC. Similarly, although many articles have suggested progressive improvement in OS for patients with metastatic breast cancer, a recent analysis has shown that metastatic survival has barely budged in recent decades; 9 it appears from these results that we may be at a crossroads for dnMBC treatment and survival. 10 As a result, the association of primary site local therapy in these patients with survival has received a great deal of attention at the same time. However, whether locoregional therapy (LRT, surgery and/or radiotherapy) should be performed in this patient population has been debated to date.
The current practice guidelines for the role of breast surgery in stage IV disease from the NCCN Guidelines and the ESMO Guidelines recommended different points. 11 The former indicated that the primary treatment approach for patients with dnMBC is systemic therapy, with consideration of LRT after initial systemic treatment for those patients requiring palliation of symptoms or with impending complications, such as skin ulceration, bleeding, fungation, and pain. 12 The latter suggested that the true value of LRT is currently unknown and recommended that it could be considered in selected patients. In addition, ESMO (4th edition) guidelines for advanced breast cancer pointed out that until there was more supporting evidence, it was not advocated to routinely perform aggressive surgery in patients with dnMBC. 13 Nevertheless, a large number of retrospective data show that breast surgeons worldwide had done primary tumor surgery on patients who had dnMBC although there was no robust evidence. 14 The results of many previous studies have been contradictory: some researchers suggested that local therapy might improve the prognosis in patients with dnMBC.8,15–22 A recent publication by Soran et al 23 found that LRT was associated with an OS benefit in dnMBC patients. Other researchers observed that surgical resection of primary breast tumor did not improve outcomes of these patients.24–32 Several studies have similarly reported that certain patients, such as those who are younger and have bone-only metastasis, may benefit from LRT.33–36
While metastatic breast cancer is incurable, advances in systemic therapy have contributed to improved survival, prompting breast surgeons to question whether LRT is beneficial for clinical outcomes. Over the past few decades, multiple retrospective data from single institutions and large databases have suggested that LRT may have survival benefit, confirmed by 5 meta-analyses.21,37–40 Until recently, however, only 4 published randomized clinical trials assessed the impact of LRT in dnMBC;22,24–26 and these studies have shown conflicting results. The debate about LRT in patients with dnMBC persists. 41 Thus, we conducted a retrospective analysis to explore and evaluate the association of LRT in patients with dnMBC between the survival outcomes, in particular the association of surgical timings and surgical margins with OS in this patient population.
Methods
Study Design and Patients
We previously conducted a retrospective cohort study to investigate the clinicopathological profile and survival outcomes of local therapy based on surgical resection of the primary tumor in female patients with dnMBC. Details of the study were described previously, 19 and the information was supplemented and updated. The current analysis aimed to further assess the survival impact of LRT, focusing on the association of surgical timing and surgical margin with OS in these patients. Patients were included in the analysis if they met the following criteria: (1) had clear histological evidence at initial diagnosis; (2) identified with metastases by clinicopathological or radiological evidence at the time of being diagnosed with breast cancer; (3) metastases were identified according to the American Joint Commission of Cancer (AJCC) 7th edition staging criteria; (4) did not receive any prior treatment; (5) no other malignancies were present; and (6) had follow-up of more than 3 months. Patients with recurrent metastases and incomplete or refusal to provide follow-up information were excluded; patients who had received any previous treatment for advanced disease were not enrolled. We have de-identified all patient details. All patients have signed the consent prior to treatment. The reporting of this study conforms to STROBE guidelines. 42 This study was approved by the Cangzhou Clinical College of Integrated Traditional Chinese and Western Medicine of Hebei Medical University Research Ethics Board (2017-AF29-058). The study was conducted in accordance with Declaration of Helsinki.
Treatment information, including LRT and systemic therapy alone (STA), was recorded and analyzed. Surgical treatment of primary tumors, alone or in combination with radiotherapy, referred to LRT. What needs to be explained here was that LRT generally refers to LRT added on the basis of systemic treatment, rather than LRT alone. STA was defined as the use of any chemotherapy, endocrine therapy, and/or anti-HER2 target therapy. OS, as primary end point, was defined as days from diagnosis to last clinic visit day or day of death. OS results in this analysis are for all eligible patients with dnMBC. Secondary outcomes included timings of surgery, surgical margin involvement, and utilization of radiation therapy. In this study, the timing of surgery refers to the time relationship of surgery with the systemic treatment period (mainly the duration of systemic chemotherapy), including surgery before chemotherapy (SBC), surgery during chemotherapy (SDC), and surgery after chemotherapy (SAC). The duration of systemic chemotherapy was designed to be an average of 4.6 months (6 courses), which was based on the treatment experience of 2 Chinese tertiary hospitals. 19
Statistical Analysis
The primary analysis of OS was conducted on 153 patients (118 died) at the data-cutoff date of December 31, 2017. Descriptive statistics were used to examine the following baseline characteristics of the breast cancer patients: age, year of diagnosis, tumor size, regional node status, tumor grade, molecular subtype, types of surgery, and radiation. Continuous variables were reported as mean ± standard deviation, and comparisons between the LRT and STA groups were by the Mann–Whitney U-test since normality was not assumed. Categorical variables were analyzed by the chi-square test, while Fisher's exact test was used when the expected value was less than or equal to 5. All tests used 2-sided P-values of <.05 suggesting statistical significance unless otherwise stated. Univariable and multivariable Cox regression analyses were used to assess the association between LRT and OS. Kaplan-Meier method was used for survival analysis and the outcome variables were compared using the log-rank test. The hazard ratio (HR) for death in the analysis of OS was estimated with the use of a stratified Cox proportional-hazards model. Analyses were performed in the following subgroups: patients receiving surgery therapy, timing of the surgery, surgical margin and patients receiving radiation therapy. An additional exploratory analysis was carried out to assess the consistency of survival benefit in subgroups based on clinicopathologic characteristics. We used Cox proportional hazards analysis to calculate the adjusted mortality risks (HR) and the 95% CIs. All analyses were conducted with Stata 16.0 (Stata Corp LP).
Results
Patients and Treatment
From March 2007 through December 2017, a total of 153 female patients met the study criteria and were included in the analysis. The median age at diagnosis was 55.8 years (26-82 years). The median follow-up time was 33 months (range 5-128 months). Demographics and baseline characteristics of patients in LRT and STA groups are shown and compared in Table 1. The clinicopathological characteristics of patients undergoing surgery are indicated in Table 2. Drug therapy of enrolled patients is shown in Table 3.
Demographics and Baseline Characteristics.

Abbreviations: LRT, locoregional therapy; STA, systematic therapy alone; yr, year; HR, hormone receptor; HER-2, human epidermal growth factor receptor-2.
Ki-67 threshold is set to 20% instead of 14%.
Clinicopathological Characteristics of Patients Undergoing Surgery.

Abbreviations: SAC, surgery after chemotherapy; SBC, surgery before chemotherapy; SDC, surgery during chemotherapy; HR, hormone receptor; HER-2, human epidermal growth factor receptor-2; yr, year.
Ki-67 threshold is set to 20% instead of 14%.
Drug Therapy of Enrolled Patients.

Abbreviations: LRT, locoregional therapy; STA, systematic therapy alone.
Kaplan-Meier Survival Estimate
At the data cutoff date for the analysis of OS (Dec 31, 2017), 118 deaths had occurred: 64 among 87 patients (73.6%) in LRT cohort and 54 among 66 patients (81.8%) in STA cohort. Survival time was significantly improved in LRT cohort versus STA cohort, with an OS of 39 months (95% CI, 33.6 to 44.4 months) versus 24.6 months (95% CI, 20.6 to 28.6 months), respectively. OS calculated via the Kaplan-Meier method showed a significant difference between the LRT group and the STA group (P = .0001). Multivariate analysis using Cox's proportional hazards model in terms of clinicopathological factors showed that LRT was significantly associated with better OS, with a 38% difference in the relative risk of death (HR, 0.62; 95% CI, 0.39 to 0.99; Figure 1; Table 4).

Comparison of overall survival between LRT and STA for de novo metastatic breast cancer.
Multivariate Analysis of LRT on Overall Survival.

***P < .01, **P < .05, *P < .1.
Stratified Analyses Associated With OS
Subgroup analyses that were defined according to clinicopathological characteristics and treatment options were performed. Obviously, the benefits of surgery with respect to OS in the present study are noteworthy in the context of LRT for patients with dnMBC. OS calculated via the Kaplan-Meier method showed a significant difference between the surgery and the non-surgery group. The equality of these curves was confirmed by log-rank rest (P < .0001). The OS was 39.7 months (95% CI, 33.8 to 45.6 months) in patients who had surgery and 24.7 months (95% CI, 20.8 to 28.6 months) in patients who had non-surgery. Multivariate analysis showed that surgery was significantly correlated with better OS, with a 42% difference in the relative risk of death (HR, 0.58; 95% CI, 0.35 to 0.94; Figure 2; Table 5).

Overall survival in patients with de novo metastatic breast cancer were assessed to receive either surgery or non-surgery.
Multivariate Analysis of Surgery on Overall Survival.
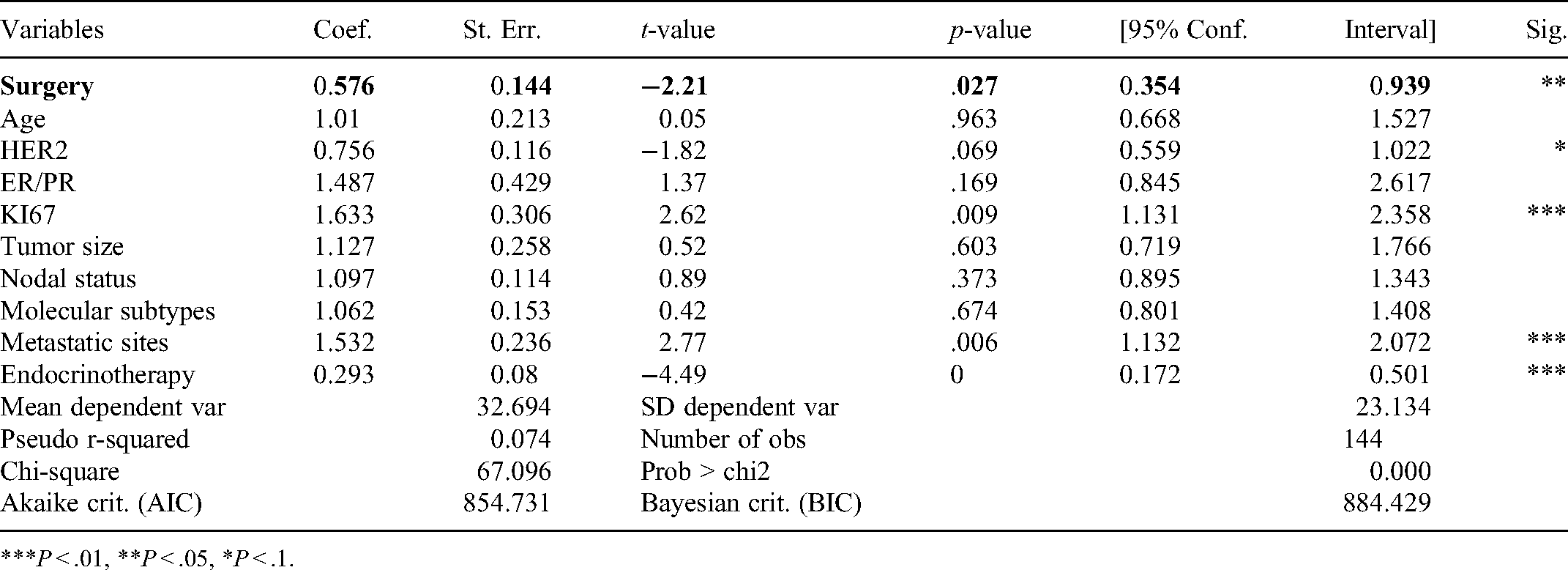
***P < .01, **P < .05, *P < .1.
For surgical patients, the OS was 50.1 months (95% CI, 40.5 to 60.2) in patients who underwent SAC and 29.4 months (95% CI, 23.2 to 35.6) in patients who underwent SBC; and the OS of patients receiving SDC was 36.7 months (95% CI, 23.2 to 50.3). Of these, only SAC had a higher OS than all patients who underwent surgery. OS calculated via the Kaplan-Meier method showed a significant difference between the 3 arms (P = .0001). Multivariate analysis showed that SAC was significantly correlated with better OS, with a 23% difference in the relative risk of death (HR, 0.77; 95% CI, 0.63 to 0.94; Figure 3).

The association of timings of surgery with survival in patients with de novo metastatic breast cancer.
Of the 77 patients received primary tumor surgery, 66 (85.7%) had lumpectomy and 11 (14.3%) had mastectomy. The surgical margins status of primary breast cancer was found to be an affecting factor on OS for patients who had surgery. OS calculated via the Kaplan-Meier method showed a significant difference between the 3 arms. The equality of these curves was confirmed by log-rank rest (P < .0001). As shown in Figure 4, OS was significantly improved in patients with negative margins versus patients with positive margins, with an OS of 49.2 months (95% CI, 41.8 to 56.7 months) versus 21.5 months (95% CI, 15.4 to 27.5 months), respectively, and a HR of 0.22 (95% CI, 0.13 to 0.37; P < .001).

The association of surgical margins status with survival in patients with de novo metastatic breast cancer.
As an alternative to surgery, the effects of radiotherapy on OS were also subgroup analyzed in the present study. The results were received for radiotherapy on survival with a P-value of .003 calculated by log-rank statistics. The OS in this subgroup was 41.8 months (95% CI, 33.4 to 50.1) versus 25.6 months (95% CI, 21.2 to 29.9), and a HR of 0.45 (95% CI, 0.27 to 0.75; P = .002; Figure 5). Of the total cohort, the majority receiving no radiotherapy and only 38 cases (ten patients received radiotherapy alone) being analyzed (a small sample size and limited number of events), thus the treatment effect HR in the subgroup of patients should be interpreted with caution.

Overall survival in patients with de novo metastatic breast cancer were assessed to receive either radiotherapy or non-radiotherapy.
The effect of LRT was also evaluated in the subgroup of patients with bone metastases only. Figure 6 shows the survival curve of patients with bone only metastases derived from the Cox model. The OS was 49.2 months (95% CI, 40.4 to 57.9) in patients who had LRT and 34.2 months (95% CI, 19.5 to 48.9) in patients who had STA; and the effect seems more evident for the patients who received LRT, although this difference was not statistically significant (HR, 0.51; 95% CI, 0.25 to 1.02; P = .05; Figure 6).

Comparison of overall survival between LRT and STA in patients with bone metastases only.
As shown in Figure 7, exploratory analyses demonstrated consistent treatment effects across specified subgroups. The results for OS in subgroups were generally consistent with those in the overall population.

Exploratory analyses of overall survival in subgroups. The solid vertical line represents the point of no effect, and the dashed vertical line represents a hazard ratio of 0.62. Hazard ratios were estimated according to a hierarchical Cox proportional hazards model. Subgroup data were based on case report forms. The hazard ratios for some subgroups had larger confidence intervals due to the small number of patients. (*)The sum of the percentages may not reach 100 due to unknown or missing data for some patients not included in the subgroup analysis.
In addition, results from the multivariate analysis of the impact of LRT on survival showed that these 3 factors (metastasis, Ki67, and endocrine therapy) as well as HER2 status were significant (Table 4), which was consistent with a multivariate analysis based on the survival effect of surgery alone (Table 5). The COX regression adjusted for other factors and found that these 4 factors were also important factors affecting survival.
Discussion
The retrospective cohort study demonstrated that LRT was associated with a significant benefit with respect to OS with a 38% relative difference in the risk of death as compared to STA in women with dnMBC. Subgroup analyses also showed a survival benefit in patients undergoing surgery in these patients, with an OS of 39.7 months (HR, 0.58). This OS benefit was generally consistent across patient subgroups, including those who received surgical resection of the primary tumor. In this study, the greatest benefits were seen among both patients who had surgery with negative margins and patients undergone SAC, with an average survival of 50.1 months, regardless of the site of their metastatic disease. Exploratory analyses also demonstrated consistent treatment effects across specified subgroups. Our findings show that LRT in patients with dnMBC had a positive impact on OS. This work will support and add evidence to the critical concepts in the treatment of patients with dnMBC.
With respect to the survival effects of LRT in patients with dnMBC, retrospective studies have shown the potential survival benefits,8,15,43–49 while prospective studies have revealed conflicting results. 35 Currently, 3 published prospective studies have assessed the impact of LRT in patients with dnMBC. A randomized controlled trial by Badwe et al 24 found no difference in OS based on LRT; another study by Soran et al 22 randomized 274 dnMBC patients to either LRT or SAT, in whom survival was similar at 36 months, but actually higher in LRT group versus STA group at 60 months; the ABCSG-28 POSYTIVE trial by Fitzal F et al 25 stopped early owing to poor recruitment and no definite conclusion was reached. In addition, a prospective registry study of 127 patients revealed no improvement in OS for those having resection of their primary tumor. To date, prospective trials failed to provide a definitive answer to the question of optimal utility of surgery in this patient population. 27 By contrast, several retrospective studies have revealed the potential survival benefits with LRT in patients with dnMBC. A recent meta-analysis by Ritika Gera et al, of 216066 patients found all forms of LRT resulted in a significant 31.8% reduction in mortality and surgical resection resulted in a significant 36.2% reduction in mortality. Ritika Gera therefore concluded that LRT of the primary tumor seems to improve OS in dnMBC. 21 Another study by Seo et al showed that LRT might be an important option for dnMBC patients. 50 Our findings are in accordance with these reports analyzing the impact of LRT on survival.
The latest research from the NCDB (2003-2012) demonstrated that among women alive one year after a diagnosis of metastatic breast cancer, primary tumor surgery was associated with improved survival, regardless of treatment sequence. 8 Another recent study based on the SEER database (2010-2015) proposed a survival advantage with surgical intervention (the OS, 43 months for surgery vs 27 months for non-surgery). 49 Similarly, in our study, the OS of the 2 groups was 39 months versus 24 months, respectively.
Most retrospective studies focused on the survival outcomes of surgical treatment after systemic chemotherapy. For example, a recent research by Lane WO et al from NCDB showed that the greatest survival benefit was seen in women treated with systemic therapy followed by surgery; the OS was 52.8 months compared to 37.5 months for those undergoing STA. 8 However, no studies discussed the survival of receiving surgical treatment at 3 different time periods (SAC, SDC, and SBC) in dnMBC patients. One of major strength of this study, we analyzed the survival outcomes of SBC, SDC, and SAC, rather than that of SAC alone.
In the present study, despite different timings of surgery that included SAC, SDC, and SBC, patients underwent resection of the primary tumor not only failed to increase the risk of death, but instead showed an improvement in OS compared to nonsurgical treatment during the follow-up period. Moreover, our study found a significant OS difference between patients in whom the 3 surgical options were SAC, SDC, and SBC. Compared with SBC and SDC, SAC have a survival advantage in these 3 different options (P = .0001). Our findings are in accordance with the report by Lane WO et al analyzing the impact of surgery on survival in those patients treated with systemic therapy followed by surgery. 8 The authors speculated that patients with SAC may have an opportunity to receive preoperative chemotherapy, and thus respond well to complete local remission of the tumor, leading to prolonged survival. While the mechanism of this phenomenon has yet to be confirmed, several previously published animal and human studies may help explain the association between the timings of surgery and OS.51–56
We also found that surgery with negative margin had a positive impact on OS for surgical patients. In our study, OS was significantly improved in patients with negative margins versus patients with positive margins, with an OS of 49 months versus 21 months (P < .0001), respectively. The results of this study show that the surgical removal of primary tumor in these patients does not impair survival but actually improves prognosis if done with negative surgical margins. Similar to the results of Rapiti E et al, primary tumor removal with negative margins significantly improved survival. 46 One possible explanation is that total tumor burden plays a central role in patient survival, and resection of the primary tumor reduces tumor burden, thereby increasing response to systemic therapy.48,57 Another factor is that primary tumors are a major source of new cancer cell clonal lines, which are involved in the emergence of treatment resistance and more aggressive disease phenotypes. 58 Consistent with this hypothesis, recent studies have found a strong correlation between the level of circulating tumor cells and prognosis of metastatic breast cancer.59,60 Surgical resection of primary tumor would disrupt this process.
As an alternative to surgery, the survival impact of radiotherapy for the primary tumor was also analyzed in the present study. The results were received for radiotherapy on survival with a P-value of .003 calculated by log-rank statistics. Indeed, our findings are consistent with the conclusions of contemporaneous cohorts from the literature.4,61–64 Nevertheless, the treatment effect HR in the subgroup of patients should be interpreted with caution due to the small sample size and limited number of events. Additionally, based on the results of LRT for patients with bone only metastases, we believe that LRT may bring survival benefits to patients with bone metastases only, although the difference is not statistically significant (small sample size or limited number of events as possible explanations). Our findings are supported by a recent prospective study suggesting that LRT extends survival in dnMBC patients with bone metastases only. 65
Some limitations in the interpretation of our findings should be acknowledged. This study's retrospective nature inevitably leads to certain limitations, such as potential selection bias, and the reliance on various clinicians’ documentation of indications for treatments. Some imbalances in the baseline characteristics of the cohort exist between groups. Moreover, the study includes a limited number of patients and it is not possible to derive definitive conclusions regarding survival outcomes, especially relative to LRT. Despite these limitations, our study has several strengths. Importantly, focusing on the impact of LRT on OS, across all subgroups, we have performed multivariate analysis using Cox's proportional hazards model in terms of clinicopathological factors and the contents of drug therapy in order to eliminate the effect of bias as much as possible. We tried to address some controversial and important issues in a field where limited data are available. First, few studies have compared the survival outcomes of dnMBC patients undergoing surgery at 3 different intervals between systemic chemotherapy and surgery. To the best of our knowledge, this is the first study that assessed the survival outcomes of these patients with a particular focus on the 3 different intervals associated with use of surgery, and concluded that SAC benefited significantly. Furthermore, whether surgical margins corresponding to the 3 surgical procedures have an impact on patient survival has not been previously studied. In the present study, we found a significant survival difference between patients in whom the surgical margins were negative and positive. The results showed that patient who had SAC, with negative surgical margins, were expected to have better survival. Incidentally, based on the results of multivariable analysis regarding LRT, we found that metastasis, Ki67 index, and HER2 status may be independent prognostic factors affecting survival. However, due to the above limitations, in our study, more researches should be conducted to confirm the hypothesis.
Conclusions
The current study demonstrates that LRT significantly improves survival in patients with dnMBC compared to STA. The benefits of surgery with respect to OS in our study are noteworthy in the context of LRT in this patient population, especially in patients undergoing SAC with negative margins. This study provides additional evidence that LRT may have survival benefit in this patient population. Well-designed prospective studies are needed to re-evaluate LRT strategy for patients with dnMBC.
Footnotes
Acknowledgments
We would like to acknowledge the Department of Breast Oncology, Tianjin Medical University Cancer Institute and Hospital, Tianjin Medical University.
Availability of Data and Materials
The datasets used and analyzed during the current study are stored by corresponding author of this paper and are available upon request.
Ethics Approval
Approval for this study was obtained from the Cangzhou Clinical College of Integrated Traditional Chinese and Western Medicine of Hebei Medical University Research Ethics Board (2017-AF29-058).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
