Abstract
Introduction
Multiple targets with varying distances are common in radiotherapy. Reducing treatment time in the plan design helps minimize patient movement and discomfort during the treatment process. This retrospective study aimed to investigate the impact of varying intertarget distances (ITDs) on the dosimetric differences and delivery efficiency of two single-isocenter techniques.
Methods
ITDs for 15 patients with dual-site vertebral metastases undergoing volume-modulated arc therapy (VMAT) were modified using Matlab 2019a. Distances of 2, 4, 6, 8, and 10 cm were considered. The VMAT plans were designed with a prescription dose of 40 Gy/20f on Infinity Linac and Monaco 5.40.01. Single-isocenter with jaw tracking (VMAT1) and fixed jaw (VMAT2) were compared in terms of dosimetry and delivery efficiency under different ITDs.
Results
Results showed that both VMAT plans exhibited dosimetric parameters meeting clinical requirements. The conformity index (CI) of VMAT1 plans was smaller than that of VMAT2 at ITD = 4, 6, and 8 cm (P = 0.007, 0.020, and 0.039, respectively), with no significant differences in other planning target volume dosimetry parameters. In terms of delivery efficiency, the treatment time of VMAT1 increased significantly when ITD > 2 cm compared with that at ITD = 2 cm (P = 0.000). Conversely, VMAT2 exhibited no significant change in treatment time at different ITDs (P = 0.073). For ITD = 2 cm, the treatment time of VMAT1 was shorter than that of VMAT2, with a median difference of 77 s. For ITD > 2 cm, the treatment time of VMAT2 was shorter than that of VMAT1, with a median difference ranging from 65 s to 121 s.
Conclusion
The experimental results showed that the single-isocenter with jaw tracking is recommended in the planning design when ITDs are less than 2 cm. However, for ITDs greater than 2 cm, the single-isocenter with fixed jaw demonstrates high delivery efficiency.5075
Highlights
The impact of various intertarget distances (ITD) on single-isocenter radiotherapy plans with jaw-tracking and jaw-fixed techniques for vertebral metastases was considered.
Distances of 2 cm, 4 cm, 6 cm, 8 cm, and 10 cm were used. Results showed that the treatment time of VMAT1 was shorter than that of VMAT2 for ITD = 2 cm and the treatment time of VMAT2 was shorter than that of VMAT1 ITD > 2 cm.
Monaco and Mosaiq subfields were recorded to correspond to the delivery time.
Introduction
The vertebral body stands out as one of the most prevalent sites for bone metastasis during the advanced stages of malignant tumor treatment.1,2 Unfortunately, vertebral metastases often bring about severe pain, significantly diminishing the life quality of affected patients. In response, radiation therapy emerges as a common and effective approach for addressing vertebral metastases, providing relief from pain, locally controlling tumors, and mitigating the risk of pathological fractures. 3 Volume-modulated arc therapy (VMAT) has become a cornerstone in radiotherapy practices, renowned for its ability to diminish the exposure dose to surrounding organs at risk (OARs) while ensuring comprehensive coverage of the target. 4 Furthermore, VMAT has the added advantage of reducing treatment duration. 5
Vertebral metastases often manifest as multiple-site metastases, presenting a challenge in maintaining a fixed posture for an extended period during radiotherapy for patients with multiple metastatic tumors within the vertebral body. This challenge can potentially impact the efficacy of radiotherapy and, in severe cases, lead to interruptions due to patients’ inability to adhere to treatment. Consequently, the control of treatment duration is a crucial consideration in the design of radiotherapy plans. 6 In the context of multi-target plan design, especially when the size of the multi-target area does not exceed the dimensions of the Linac field, adopting a single-isocenter planning design proves to be a highly effective strategy. This approach eliminates the need for re-entry into the therapeutic room and repositioning during the treatment process, contributing significantly to the reduction of treatment time.7,8 For dual-target vertebral metastases, VMAT plans can be implemented in two distinctive ways: the single-isocenter with jaw-tracking and jaw-fixed functions.9,10 The VMAT plan with jaw tracking uses the multi-leaf collimator (MLC) to block the space between the two targets, creating an isolated field. Conversely, the VMAT plan with fixed jaw utilizes fixed-jaw technology to conformally treat the two sub-targets separately. 10
The current literature indicates that the treatment time for the VMAT plan with jaw tracking is shorter than that for the VMAT plan with fixed jaw. Lai et al 11 reported estimated overall treatment times ranging from 9.98 min to 11.38 min and from 12.39 min to 13.74 min for VMAT without and with fixed jaw, respectively. Similarly, Gao et al 12 found that the beam-out time in the jaw-tracking plan is significantly shorter than the fixed-jaw plan (3.31 min vs 9.75 min, P < 0.05). Notably, these reported times may refer to beam-on time (BOT); in clinical practice, the actual treatment time holds greater significance than the BOT. Empirical observations in our clinic have revealed that, particularly for some double-target plans with large intertarget distances (ITDs), there is a notable occurrence of MLC movement without beam delivery in the treatment of the jaw-tracking plans. This phenomenon results in a long delivery time. Thus, this article aimed to further explore the impact of ITD on the dosimetry and delivery efficiency of the two single-isocenter techniques.13,14 The retrospective findings seek to provide valuable insights for refining the radiotherapy plan design, especially in the context of clinical scenarios involving multiple vertebral metastases.
Materials and Methods
Patient Selection and Contouring
A retrospective selection included 15 patients with vertebral multiple metastatic tumors who underwent VMAT treatment at our hospital between January 2021 and December 2022. The cohort comprised 8 males and 7 females, aged 45–76 years, with a median age of 58 years. As per imaging diagnosis, all patients exhibited multiple vertebral metastases from malignant tumors. Inclusion criteria were as follows: (1) multiple vertebral metastases and undergoing radiotherapy; (2) the target area comprised isolated metastatic tumors at both ends, with the maximum distance between the ends of the two lesions being less than 38 cm; and (3) the patient could sustain the treatment time for 15 min. CT images were acquired using the Philips Brilliance Big Bore scanner, featuring an image matrix of 512 × 512, a pixel size of 0.975 mm, and a thickness of 0.25 cm. The CT data were transmitted to the Monaco 5.40.02 treatment planning system (TPS, version 5.40.02;Elekta Instrument AB, Stockholm, Sweden). Following ICRU 62 guidelines, 15 two senior physicians delineated the clinical target volume (CTV), distinguishing between upper (CTV1) and lower (CTV2) segments. The expansion of CTV1 and CTV2 by 0.5 cm yielded the planning target volume (PTV1 and PTV2, respectively), and the combination of PTV1 and PTV2 was denoted as PTV. OARs, including the lungs, heart, liver, kidneys, and others, were also contoured by the physicians. The patient information has been anonymized to ensure that the identity of any individual cannot be determined in any manner. 16
ITD Settings
To investigate the impact of ITD on the treatment time of radiotherapy plans, we established five groups with ITD values of 2, 4, 6, 8, and 10 cm in this study. Here, ITD refers to the spacing between the lower PTV1 and upper PTV2 bounds. The ITD settings were implemented as follows:
The outlines of PTV1 and PTV2 were exported in DICOM format. Using Matlab 2019a, we read the DICOM file outlines and modified the header file information. ITDs were set at 2, 4, 6, 8, and 10 cm based on the PTV center. This resulted in five PTV1 groups (PTV1_2, PTV1_4, PTV1_6, PTV1_8, and PTV1_10) and five PTV2 groups (PTV2_2, PTV2_4, PTV2_6, PTV2_8, and PTV2_10). Subsequently, Matlab 2019a was utilized to export the modified contour files into DICOM format and import them into the Monaco system.
VMAT Plan Design
Utilizing the Infinity Linac (Elekta Instrument AB, Stockholm, Sweden) and Monaco TPS, each patient underwent the creation of two single-isocenter VMAT plans: the jaw-tracking plan (VMAT1) and the jaw-fixed plan (VMAT2). The planned dose rate was set at 600 MU/min, with a calculated mesh size of 0.3 cm and 1% computational accuracy. The Monte Carlo algorithm served as the algorithm. The PTV prescription dose was established at 40 Gy/20 fx, ensuring that 95% of the PTV volume received the prescription dose. Naming conventions for VMAT plans included VMAT1_2, VMAT1_4, VMAT1_6, VMAT1_8, and VMAT1_10 and VMAT2_2, VMAT2_4, VMAT2_6, VMAT2_8, and VMAT2_10 when ITD equaled 4, 6, 8, and 10 cm.
For VMAT1 plans, a single field was designed with the following parameters: the isocenter located at the center of the PTV, gantry rotation from −180° to 180° and back to −180°, collimator angle set to 0°, and the field parameter in the beam set to Auto in Monaco.
VMAT2 plans comprised two fields, mirroring the VMAT1 design but with the distinction that the two fields conformed to PTV1 and PTV2 through fixed-jaw technology. The field parameter of the first field was set to PTV1, and the second field parameter was set to PTV2 in Monaco.
Dose constraints for VMAT1 and VMAT2 were identical. OAR dose evaluation criteria were established as follows: V5 of bilateral lung < 50% (V5 represents the percentage of the organ volume receiving ≥ 500 cGy, and so forth), V20 < 25%, and Dmean < 1200 cGy; Dmax of the spinal cord planning risk volume (PRV) < 4500 cGy; Dmean of the heart < 2000 cGy; V20 of the kidney < 20%; and Dmean < 1200 cGy (Dmax and Dmean represent the maximum and average doses, respectively).
Plan Quality and Delivery Efficiency Evaluation
Following ICRU report No. 83,
17
the dosimetric parameters of PTV were assessed, including D2 (minimum doses delivered to 2% of PTV, and so forth), D98, Dmean, homogeneity index (HI),
18
and conformity index (CI).
19
The formulas for calculating HI and CI are as follows:
Dosimetric parameters of OAR were evaluated, encompassing Dmean of bilateral lungs; Dmean of the heart, left kidney, and right kidney; V5 of the liver; and Dmax of the spinal cord PRV.
During measurements, discrepancies in the number of subfields between Monaco and Mosaiq were noted. Accordingly, we separately recorded the subfield numbers for evaluation. The indices for assessing delivery efficiency included monitor units (MUs), Monaco subfield numbers, Mosaiq subfield numbers, beam timer, and total delivery time. The beam timer represents the BOT recorded by the Infinity Linac. Notably, the switching field time was incorporated into the total treatment time for the VMAT2 plan.
Statistical Analysis
SPSS 20.0 (Chicago, IL, USA) software was employed, and the analysis data were expressed as the median (lower and upper quartiles). For paired data in VMAT1 and VMAT2 plans with the same ITD, a non-parametric Wilcoxon signed-rank test was utilized. In the case of dosimetry and delivery efficiency parameters involving different ITDs using the same VMAT plans, the non-parametric Friedman test method was applied between groups, and the Nemenyi algorithm was employed within groups. P < 0.05 was considered statistically significant.
Result
PTV Dosimetry Comparison
Table 1 presents a comparison of PTV dosimetry parameters between VMAT1 and VMAT2 across five ITD groups. The dosimetry parameters of VMAT1 and VMAT2 plans fulfilled clinical requirements, with no statistical differences observed in D2, D98, Dmean, and HI (P > 0.05). However, for CI, significant differences were noted at ITD intervals of 2, 4, and 6 cm, with median value differences ranging from 0.01 to 0.03. Overall, PTV dosimetry parameters for VMAT1 and VMAT2 were similar in the context of multiple vertebral metastases.
Comparison of PTV Dosimetry Parameters Between VMAT1 and VMAT2 at Five ITD Groups.

OAR Dosimetry Comparison
The comparison results for OAR dosimetry parameters between VMAT1 and VMAT2 across five ITD groups are presented in Table 2. All OAR dosimetry parameters met clinical requirements, with no significant differences observed, except for spinal cord PRV Dmax (P > 0.05). The median difference in spinal cord PRV Dmax ranged from 0.27 Gy to 0.72 Gy. In summary, OAR dosimetry parameters for VMAT1 and VMAT2 were similar in the context of multiple vertebral metastases.
Comparison of OAR Dosimetry Parameters Between VMAT1 and VMAT2 at Five ITD Groups.

Comparison of Delivery Efficiency Parameters
The results of delivery efficiency parameters in the VMAT1 plans at various ITDs are outlined in Table 3. The discrepancy in MUs demonstrated statistical significance (P = 0.001), with a notable difference between VMAT1_6 and VMAT1_2 in the Nemenyi algorithm result (P = 0.001). Significant variations were noted in the number of Mosaiq subfields (P = 0.014), and VMAT1_4 and VMAT1_6 exhibited statistically significant differences compared with VMAT1_2 in the Nemenyi algorithm results (P = 0.027 and 0.027). Conversely, no significant difference was observed in the number of Monaco subfields (P = 0.102). The difference in beam timer was statistically significant (P = 0.021), with VMAT1_8 differing significantly from VMAT1_10 in the Nemenyi algorithm result (P = 0.022). The total treatment time showed a significant difference (P = 0.000), and VMAT1_4, VMAT1_6, VMAT1_8, and VMAT1_10 exhibited statistically significant differences compared with VMAT1_2 in the Nemenyi algorithm results (P = 0.000, 0.000, 0.003, and 0.000).
Delivery Efficiency Parameters at Different ITDs in the VMAT1 Plans.

The results of delivery efficiency parameters at various ITDs in the VMAT2 plans are outlined in Table 4. A significant difference was observed in the number of MUs (P = 0.000), with VMAT2_6, VMAT2_8, and VMAT2_10 displaying statistically significant differences compared with VMAT2_2 in the Nemenyi algorithm results (P = 0.003, 0.000, and 0.000). Statistically significant differences were also noted in the number of Mosaiq subfields (P = 0.000), where VMAT2_6, VMAT2_8, and VMAT2_10 exhibited statistically significant differences compared with VMAT2_2 in the Nemenyi algorithm results (P = 0.012, 0.000, and 0.002). The difference between VMAT2_4 and VMAT2_8 was statistically significant (P = 0.012). The median difference between the Monaco subfield number and the Mosaiq subfield number was 4, with a statistical difference mirroring that of the Mosaiq subfield number. A significant difference was observed in beam timer (P = 0.000), and VMAT2_4, VMAT2_6, VMAT2_8, and VMAT2_10 displayed statistically significant differences compared with VMAT2_2 in the Nemenyi algorithm results (P = 0.018, 0.000, 0.000, and 0.000). However, we found no significant difference in actual treatment time (P = 0.073).
Delivery Efficiency Parameters at Different ITDs in the VMAT2 Plans.

Table 5 illustrates the comparison of treatment time between VMAT1 and VMAT2 in the five groups of ITDs. Notably, the treatment time of VMAT1 was shorter than that of VMAT2 when ITD = 2 cm (P = 0.001), whereas the treatment time of other VMAT1 plans was longer than that of VMAT2 (P < 0.05).
Comparison of Treatment Time Between VMAT1 and VMAT2 Plans in the Five Groups of ITDs.

Dose Distribution and Subfield Shape of VMAT1 and VMAT2 Plans
Figure 1 compares the dose distribution of VMAT1 and VMAT2 at ITDs of 2 and 6 cm to visually illustrate the dose distribution of the two kinds of plans. In (a) and (b), the dose distribution of the two plans was similar when ITD was 2 cm. Notably, the 10 Gy dose-line area of PTV1 in VMAT2_2 was relatively smaller than that of VMAT1_2, and the 5 Gy dose-line area between PTV1 and PTV2 was also smaller than that of VMAT1_2. However, when the ITD was 6 cm, the dose leakage between PTV1 and PTV2 became apparent, resulting in an evident 500 cGy dose line between PTV1 and PTV2 in the dose distribution (c) of VMAT1_6, in contrast to the dose distribution (d) of VMAT2_6.

Comparison of Dose Distributions of VMAT1 and VMAT2 Plans with ITD of 2 and 6 cm, (a), (b), (c), and (d) are the Coronal Dose Distributions of VMAT1_2, VMAT2_2, VMAT1_6, and VMAT2_6 Plans, Respectively. The blue translucent area is PTV1, and the Orange Translucent Area is PTV2.
Figure 2 illustrates the subfield shapes of the two plans at ITDs of 2 and 6 cm. In (d), the subfield shape of VMAT1_6 showed that the single-isocenter common field plan utilized MLC to shield the spaced part of the target area. As the MLC required a narrow slit to prevent collision, the area between PTV1 and PTV2 lacked additional jaw protection, resulting in the formation of a low-dose area (Figure 1(c)).
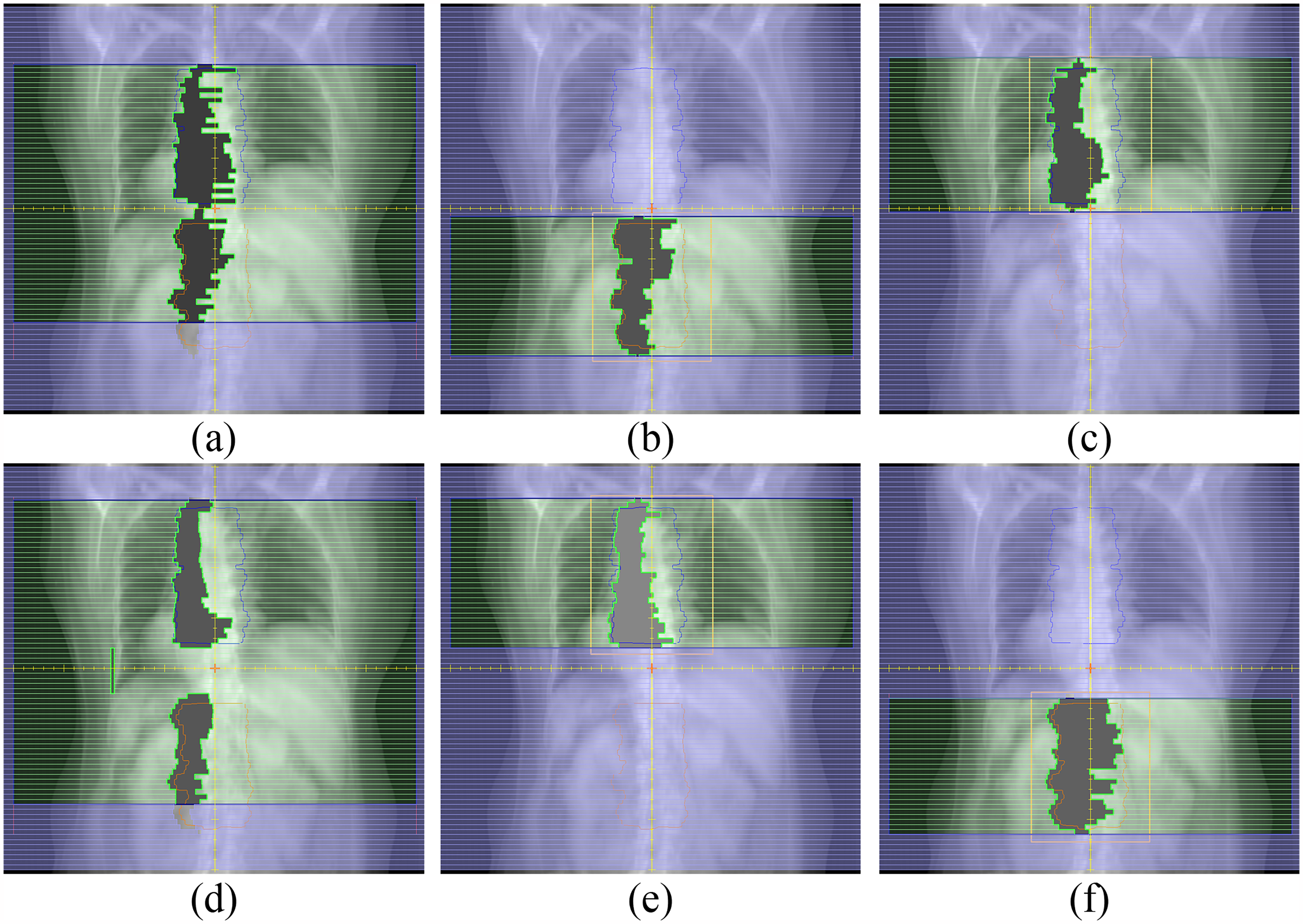
Comparison of the Subfield Shapes of VMAT1 and VMAT2 Plans with ITD of 2 and 6 cm, (a) is the Subfield Shape of VMAT1_2, and (b) and (c) are the Subfield Shapes of Two VMAT2_2 fields. (d) is the Subfield Shape of VMAT1_6, and (e) and (f) are the Subfield Shapes of Two Subfields of VMAT2_6. The thin Blue Line is PTV1, and the thin Orange Line is PTV2.
Discussion
Multiple vertebral metastases are frequently observed in patients with advanced malignancies, where radiotherapy primarily serves a palliative role to alleviate pain. Historically, concerns about significant toxicity from radiation therapy for bone metastases have been raised due to the large clinical target volumes involved. The most commonly reported toxicities include hematologic and gastrointestinal side effects. 20 However, with the advancement of modern radiation therapy techniques—such as intensity-modulated radiation therapy, VMAT, and stereotactic body radiation therapy—the protection of OARs has significantly improved, potentially reducing these adverse effects. 21
The challenge lies in maintaining a fixed position for an extended period to complete radiotherapy, especially for frail patients under pain. Therefore, finding ways to shorten the treatment time while ensuring the dose meets the therapeutic requirements is of crucial clinical significance. Compared with multi-center planning designs, the single-isocenter planning design offers the advantage of reducing the time technicians spend entering the machine room for setup, leading to a significant reduction in the actual treatment time. 22
In related studies, emphasis has been placed on the dosimetry difference between jaw-tracking and jaw-fixed techniques.9,11,23 Notably, Lai et al 11 observed no significant change in BOT and ITD between jaw-tracking and jaw-fixed plans. However, in clinical practice, the actual delivery time holds more significance than BOT. In this context, our research goes a step further by considering BOT and the actual delivery time.
In this article, we investigated the impact of different ITDs on the delivery time of single-isocenter plans for multiple metastatic tumors in the vertebral body. To explore this, we designed five sets of data with target area spacings of 2, 4, 6, 8, and 10 cm, creating two types of single-isocenter plans with jaw tracking and fixed jaw. The results revealed no significant differences in the dosimetry parameters of the PTV and OAR between VMAT1 and VMAT2, except for some CI values and spinal cord PRV Dmax. Overall, these parameters met the clinical requirements.24,25 Regarding delivery efficiency, when the ITD was 2 cm, the delivery time of VMAT1 was shorter than that of VMAT2. However, when the distance exceeded 2 cm, the delivery time of VMAT2 was smaller than that of VMAT1, accounting for the time spent on switching fields for tens of seconds. Some studies reported that the use of jaw tracking can lead to numerous subfields below 3 × 3 cm, potentially causing significant discrepancies between calculated and delivered doses.26–28 In the execution of VMAT1, a large number of MLC movements can occur without beam delivery or with a low dose rate. 11 Thus, the beam timer of VMAT1 exhibited minimal changes at different ITDs and was less than the delivery time.
From the perspective of control points, the number of Mosaiq subfields for VMAT1 exceeded the number of Monaco subfields by tens of points, and these additional control points had an output of 0 MU. For VMAT2, as the beam field conformed to PTV1 and PTV2 simultaneously, MLC motion and beam delivery occurred concurrently. Therefore, the beam timer was close to the actual delivery time, and the number of Mosaiq subfields was also close to the number of Monaco subfields. An exception occurred when ITD was 2 cm, possibly due to the absence of a dose gradient, allowing VMAT1 to avoid forming an MLC slit between PTV1 and PTV2. Additionally, the distance from the beam's-eye view perspective was less than 2 cm due to the specific size of the PTV when ITD = 2 cm.
Prior to the machine operating under normal conditions, we observed that during the implementation of the VMAT1 plan, when the MLC moved and the beam was emitted at a low dose rate, the machine was prone to errors, leading to treatment interruptions and necessitating machine resets. These events increased the overall treatment time.
For patients with vertebral metastases, the efficiency and dosimetric parameters of the radiotherapy plan are of equal importance and should be carefully considered in the daily planning design by medical physicists. Our study has provided preliminary evidence that, for the Infinity Linac and Monaco TPS system, given that dosimetry meets clinical requirements, the single-isocenter plan with jaw tracking is preferable when the ITD is under 2 cm. However, when the ITD exceeds 2 cm, the delivery efficiency of the single-isocenter with fixed jaw is high. Additionally, VMAT plans with fixed jaw demonstrate better protection of the tissue between PTV1 and PTV2 than those with jaw tracking.10,29
This study had some limitations. First, as an exploratory study, we selected 15 patients with vertebral metastases, and increasing the number of cases will help to obtain more reliable conclusions. Second, we only used the Infinity Linac and Monaco systems and did not conduct further experiments on different machines and TPS systems. Third, further segmentation between the spacing of 2 and 4 cm is helpful to explore the reasons for the impact of ITD on delivery efficiency. This limitation is also a problem that needs to be further addressed in our future work.
Conclusion
This study investigated the effect of different ITDs of multiple vertebral metastases on dosimetry and efficiency of single-isocenter plan with and without jaw tracking. Results showed that the dosimetry parameters of the two plans met the clinical requirements. At an ITD of 2 cm, the delivery efficiency of the single-isocenter plan with jaw tracking was higher than that without. When the ITD exceeded 2 cm, MLC movement with beam delivery at a low dose rate resulted in a treatment time that was longer than that of the jaw-fixed single-isocenter plan, with a median difference of 65–121 s.
Footnotes
Acknowledgments
None.
Ethics Considerations and Consent to Participate
The study was approved by the ethics committee of Changzhou No.2 People's Hospital with the exemption for patient consent because it is a retrospective study that would not bring extra risk to patient health and human rights (#2020KY154-01).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (No. 62371243), Changzhou Social Development Program (No. CE20235063), and Jiangsu Provincial Key Research and Development Program Social Development Project (No. BE2022720), Jiangsu Provincial Medical Key Discipline Cultivation Unit of Oncology Therapeutics (Radiotherapy) (No. JSDW202237), the National Natural Science Foundation of Jiangsu (No. BK20231190).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data presented in this study are available on request from the corresponding author.


