Abstract
Objective:
We compared radiotherapy plans among helical tomotherapy (HT), volumetric-modulated arc therapy (VMAT), and intensity-modulated proton therapy (IMPT) for angiosarcoma of the scalp (AS).
Methods:
We conducted a planning study for 19 patients with AS. The clinical target volume (CTV) 1 and CTV2 were defined as the gross tumor volume with a specific margin and total scalp, respectively. For HT and VMAT, the planning target volume (PTV) 1 and PTV2 were defined as CTV1 and CTV2 with 0.5-cm margins, respectively. For IMPT, robust optimization was used instead of a CTV-PTV margin (i.e. CTV robust). The targets of the HT and VMAT plans were the PTV, whereas the IMPT plans targeted the CTV robust. In total, 70 Gy and 56 Gy were prescribed as the D95% (i.e. dose to 95% volume) of PTV1 (or CTV1 robust) and PTV2 (or CTV2 robust), respectively, using the simultaneous integrated boost (SIB) technique. Other constraint goals were also defined for the target and organs at risk (OAR).
Results:
All dose constraint parameters for the target and OAR met the goals within the acceptable ranges for the 3 techniques. The coverage of the targets replaced by D95% and D98% were almost equivalent among the 3 techniques. The homogeneity index of PTV1 or CTV1 robust was equivalent among the 3 techniques, whereas that of PTV2 or CTV2 robust was significantly higher in the IMPT plans than in the other plans. IMPT reduced the Dmean of the brain and hippocampus by 49% to 95%, and the Dmax of the spinal cord, brainstem, and optic pathway by 70% to 92% compared with the other techniques.
Conclusion:
The 3 techniques with SIB methods provided sufficient coverage and satisfactory homogeneity for the targets, but IMPT achieved the best OAR sparing.
Background
Angiosarcoma of the scalp (AS) is the most common type of angiosarcoma and has a markedly unfavorable prognosis. 1 -5 Local recurrence is one of the main causes of treatment failure, in addition to lung metastasis. 6,7 Radiotherapy (RT) with or without tumor excision is an integral component of AS treatment. 7 -9
Prophylactic total scalp irradiation was reported to be a reasonable option for curative intent 10 because AS often has multifocal lesions with poor margins. High doses over 70 Gy may be required for tumor control. 6,7,10 -14 On the other hand, there are many organs at risk (OAR) around the planning target volume (PTV) of AS. The common conventional field arrangement for total scalp irradiation is the combination technique of 2 lateral X-ray and electron fields sharing the same central axis. 10,15 However, this conventional technique requires a complex procedure and it is technically difficult to successfully match electron fields with X-ray fields. 15 Inappropriate matching between X-ray and electron fields may result in an insufficient and inhomogeneous dose to the PTV.
Over the past decades, intensity-modulated radiation therapy (IMRT) has improved clinical outcomes for several cancers. 16 -18 Helical tomotherapy (HT) and volumetric-modulated arc therapy (VMAT) are novel IMRT techniques. 19 -21 Proton beam RT also achieves a more conformal target dose distribution due to the Bragg peak than photon RT. 22 In particular, the potential benefits of intensity-modulated proton therapy (IMPT) 23 enable an excellent dose distribution for AS.
Our hypotheses were (1) photon IMRT plans can provide sufficient target coverage while limiting the dose to the surrounding OAR when the target covers all macroscopic lesions (i.e. total scalp), and (2) IMPT plans resulting in excellent target coverage and OAR avoidance can be generated compared with photon IMRT techniques. In order to generate sufficient dosimetric data for these hypotheses, we created IMRT plans using HT, VMAT, and IMPT for AS patients, and conducted a planning study comparing the 3 plans.
Methods
Patient Characteristics
This study was performed after approval by the institutional review board. Twenty-two consecutive patients with AS were treated by RT between 2008 and 2018 at our institution. Of these, 19 patients who underwent computed tomography (CT) simulation were selected. The primary tumor originated in the scalp and histological classification was performed for all patients. The patient characteristics are shown in Table 1. Eight patients (42%) had at least 2 (i.e., multifocal) lesions. All described HT, VMAT, and IMPT plans were not actual clinical plans used in the treatments.
Patient Characteristics.

Treatment Volumes and Normal Structures
All patients were immobilized with a shell in a supine position and simulated by CT with a 2.5-mm slice thickness. All simulation data were imported into the RayStation treatment planning system (RaySearch Medical Laboratories AB, Stockholm, Sweden). The gross tumor volume (GTV) was defined as all known gross diseases according to clinical information, CT, magnetic resonance imaging (MRI), and/or 18 F-fluorodeoxyglucose positron emission tomography (FDG-PET). The clinical target volume (CTV) 1 included the GTV with an additional margin of 3.0 cm not to exceed the periosteum of the cranium. This margin was allowed to be as low as 1.0 cm for CTV1 close to the critical structures such as the eyes and optic pathway. CTV2 included the total scalp other than CTV1. For HT and VMAT, PTV1 was defined as CTV1 plus a 0.5-cm margin and PTV2 was defined as CTV2 plus a 0.5-cm margin excluding PTV1. For IMPT, no CTV-PTV margin was defined, but robust optimization was used to set the target (i.e. CTV robust). The robustness settings are described in the section of HT, VMAT, and IMPT planning. For comparison of the 3 techniques, the targets of the HT and VMAT plans were the PTV, whereas the IMPT plans targeted the CTV robust. The PTV and CTV robust are described hereafter as follows: PTV1 or CTV1 robust, target 1; PTV2 or CTV2 robust, target 2. As the purpose of this study was the comparison of 3 techniques for scalp irradiation, the cervical nodes were not included in the target.
The OAR included the spinal cord, brain, hippocampus, brainstem, chiasm, optic nerves, eyes, lenses, parotid glands, and inner ear. The planning organs at risk volume (PRV) was also used. The spinal cord PRV and brainstem PRV were defined as the corresponding structure plus 3-dimensional 0.5-cm and 0.3-cm margins, respectively. The optic pathway PRV was defined as the volume 0.3-cm larger in all directions than the chiasm and optic nerves.
Prescribed Dose and Goal of RT Planning
The prescription doses of target 1 (i.e. PTV1 or CTV1 robust) and target 2 (i.e. PTV2 or CTV2 robust) were 70 and 56 Gy, respectively, in 35 fractions over 7 weeks using the simultaneous integrated boost (SIB) technique. The calculation grid was common at 2.0 × 2.0 mm for all techniques. The plans were optimized to achieve a target coverage with 95% (D95%) of target 1 and target 2 receiving 100% of each prescription dose, and to provide as homogenous of a dose distribution as possible within the target. In all treatment modalities, when constraints were met, the plans were optimized using dose-volume histogram (DVH) dose constraints to reduce the dose to the OAR to as low as possible while maintaining target coverage. The goal and acceptable range of common dose constraints are shown in Table 2. All HT, VMAT, and IMPT plans were not normalized. When sufficient coverage of the target was not achieved without deviation from the dose constraint for the OAR, deviations to the acceptable range were allowed.
Dose Constraints for Target and Organs at Risk.

Target1 means PTV1 in photon IMRT plans and CTV1 robust in IMPT plans.
Target2 means PTV2 in photon IMRT plans and CTV2 robust in IMPT plans.
Abbreviations: CTV, clinical target volume; Dmax, maximum dose; Dmean, mean dose; D1, 2, 95, 98%, dose to 1, 2, 95, 98% volume of the target or organs at risk; IMRT, intensity-modulated radiation therapy; IMPT, intensity-modulated proton therapy; PRV, planning organ at risk volume; PTV, planning target volume.
HT, VMAT, and IMPT Planning
HT plans were generated using 6-MV X-ray beams of Radixact X9 (Accuray, Sunnyvale, USA). 24 The Precision Treatment Planning System (Accuray, Sunnyvale, USA) was used with the superposition algorithm for plan calculation. Regarding the HT treatment conditions, a 2.5-cm field width, pitch of 0.287, and modulation factor of 2.0 were used.
VMAT plans were generated using 6-MV X-ray beams of Trilogy (Varian Medical Systems, Palo Alto, USA). For plan calculation, the RayStation treatment planning system was used with the superposition algorithm. One arc was rotated from 179° to 181°, whereas another arc was rotated from 181° to 179°. The collimator angles were set to 90° for each arc and the multi-leaf collimator margin was 0.5 cm from the target. For VMAT plans, a 1.0-cm thick virtual bolus was used to achieve the dose constraint goals shown in Table 2.
IMPT plans were generated using scanning pencil beams of Proteus® One (Ion Beam Applications S.A., Louvain-La-Neuve, Belgium). For plan calculation, the RayStation treatment planning system was used with a pencil beam dose algorithm. Regarding IMPT plans, a setup robustness of ± 5 mm and range robustness of ± 3.5% were set. The robustness of all the treatment plans was evaluated in 12 scenarios using a setup robustness of ± 5 mm and range robustness of ± 3.5%. The worst-case-scenario achieved sufficient target coverage and avoided the OAR, with at least 90% of the CTV robust receiving the prescription dose and variation in the dose to the brain within ± 5%. In general, one non-coplanar and 4 coplanar beam directions were selected considering plan robustness, e.g. beams avoiding high-density gradients and beams with short path lengths to the target. The range shifter had a water equivalent thickness of 6.5 cm with 7.5 g/cm2 and was composed of Lexan. A range shifter was interposed into the beam injection site to enable delivery of the full dose to the skin surface. 25 CTV-based robust optimized IMPT plans were created using the same planning goals as for photon IMRT techniques. During IMPT optimization, auto-spot spacing was used to distribute the spots in the lateral direction and depth. A treatment machine was used with a spot size (in air at the isocenter) of 3 mm (r) at 230 MeV and a minimum energy of 70 MeV. All dose parameters are reported in Gy (RBE), assuming an RBE of 1.1 for protons.
Plan Comparison Criteria
The treatment plan comparison metrics were as follows: (1) target 1 (i.e. PTV1 or CTV1 robust) and target 2 (i.e. PTV2 or CTV2 robust): D2%, D95%, D98%, mean dose (Dmean), homogeneity index (HI), and conformity index (CI); (2) OAR: D1% and Dmean for all OAR, and maximum dose (Dmax) for the spinal cord PRV, hippocampus, brainstem PRV, and optic pathway PRV. HI is a homogeneity measure defined as D1% divided by D99%. 26 CI is a measure to evaluate how well the target fits the prescription isodose volume. 26 CI is defined as VPTV/TVPV × VTV/TVPV (VPTV, target volume; VTV, treatment volume of the prescribed isodose lines; TVPV, volume of VPTV within the VTV). The beam-on times for each simulated scenario were estimated from each treatment planning system. The beam-on times did not include setup times. ANOVA for repeated measures was employed to compare dosimetric parameters among the 3 techniques. Thereafter, post-hoc Bonferroni tests were performed to compare the groups pairwise. All statistical analyses were performed using EZR, 27 which is a graphical user interface for R (version 3.4.1; R Foundation for Statistical Computing, Vienna, Austria). A P-value of < 0.05 was defined as significant.
Results
Target Coverage, Homogeneity, and Conformity
The representative dose distribution of the 3 techniques for a patient with the primary tumor limited to the right sincipital area is shown in Figure 1. The spread of low doses to the brain was more restricted in the IMPT plans than in the other plans, and the dose conformity in the IMPT plans was superior to that in the other plans. The results of quantitative analysis of target 1 and target 2 in the 3 techniques are shown in Table 3. The D95% and D98% of target 1 were slightly lower in the HT plans than in the other plans, whereas the D95% and D98% of target 2 were similar among the 3 techniques. The D2% of target 1 and target 2 was significantly lower in the HT plans than in the other plans within the range of the preset constraints. The HI of target 1 was equivalent among the 3 techniques, whereas that of target 2 was significantly higher in the IMPT plans than in the other plans. The CI of target 1 in the IMPT plans was significantly lower than that in the HT plans, whereas that of target 2 was not significantly different among the 3 techniques.

Example of the dose distribution using helical tomotherapy (HT) (left), volumetric-modulated arc therapy (VMAT) (middle), and intensity-modulated proton therapy (IMPT) (right) for a patient with the primary tumor limited to the right sincipital area. For the VMAT plan, a 1.0-cm thick virtual bolus was used.
Target Coverage, Homogeneity, and Conformity.
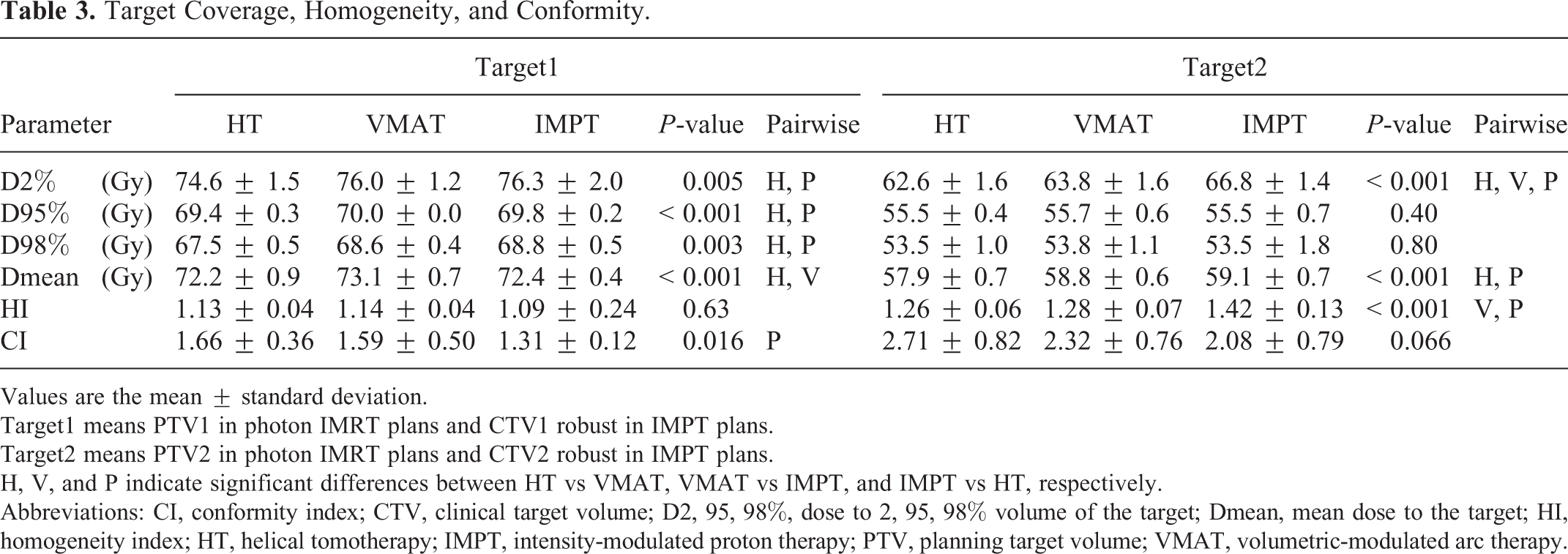
Values are the mean ± standard deviation.
Target1 means PTV1 in photon IMRT plans and CTV1 robust in IMPT plans.
Target2 means PTV2 in photon IMRT plans and CTV2 robust in IMPT plans.
H, V, and P indicate significant differences between HT vs VMAT, VMAT vs IMPT, and IMPT vs HT, respectively.
Abbreviations: CI, conformity index; CTV, clinical target volume; D2, 95, 98%, dose to 2, 95, 98% volume of the target; Dmean, mean dose to the target; HI, homogeneity index; HT, helical tomotherapy; IMPT, intensity-modulated proton therapy; PTV, planning target volume; VMAT, volumetric-modulated arc therapy.
In summary, both target 1 and target 2 coverage met the constraint goals within acceptable ranges in the 3 techniques. The target coverage was almost equivalent among the 3 techniques. The homogeneity of target 2 in the IMPT plans was inferior to that in the other plans, whereas the conformity of both targets in the IMPT plans was slightly superior to that in the other plans. DVH comparison of target 1 and target 2 among the 3 techniques is shown in Figure 2A.

Dose-volume histogram (DVH) comparison of target 1 (PTV1 or CTV1 robust) and target 2 (PTV2 or CTV2 robust) (A), the brain (B), hippocampus (C), left eye (D), left lens (E), and left parotid gland (F) among helical tomotherapy (HT, purple), volumetric-modulated arc therapy (VMAT, aqua), and intensity-modulated proton therapy (IMPT, orange). The DVH was calculated from the averaged data of 19 patients with angiosarcoma of the scalp.
OAR Sparing
The results of quantitative analysis of the OAR in the 3 techniques are shown in Table 4. All OAR met the dose constraints within acceptable ranges in the 3 techniques. IMPT reduced the Dmax of the spinal cord PRV, hippocampus, brainstem PRV, and optic pathway PRV by 70-92% compared with HT and VMAT. The Dmax of the spinal cord PRV, hippocampus, and optic pathway PRV was significantly lower in the VMAT plans than in the HT plans. DVH comparison of the brain (Figure 2B), hippocampus (Figure 2C), left eye (Figure 2D), left lens (Figure 2E), and left parotid gland (Figure 2F) among the 3 techniques is shown in Figure 2B-F.
Dose to Organs at Risk.

Values are the mean ± standard deviation.
H, V, and P indicate significant differences between HT vs VMAT, VMAT vs IMPT, and IMPT vs HT, respectively.
Abbreviations: Dmax, maximum dose; Dmean, mean dose; D1%, dose to 1% volume of organs at risk; HT, helical tomotherapy; IMPT, intensity-modulated proton therapy; Lt, left; Rt, right; VMAT, volumetric-modulated arc therapy.
IMPT reduced the D1% of the spinal cord PRV, hippocampus, brainstem PRV, optic pathway PRV, and both inner ears by 78%-91%, that of the brain by 19%, that of both lenses by 23%-34%, and that of both eyes by 24%-55% compared with HT and VMAT. The D1% of the hippocampus, optic pathway PRV, bilateral eyes, and bilateral lens was significantly lower in the VMAT plans than in the HT plans.
The Dmean of most OAR except the bilateral parotid glands in the IMPT plans was significantly lower than that in the other plans. IMPT reduced the Dmean of the spinal cord PRV, hippocampus, brainstem PRV, optic pathway PRV, and both inner ears by 88%-97%, that of the brain by 59% and 49%, that of both lenses by 41%-48%, and that of both eyes by 41%-65% compared with HT and VMAT, respectively. The Dmean of bilateral parotid glands in the HT plans was significantly lower than that in the other plans.
Beam-On Time
The beam-on times per fraction were 5.1 ± 0.3 min for the HT plans, 2.7 ± 0.1 min for the VMAT plans, and 5.8 ± 0.7 min for the IMPT plans. The beam-on time for the VMAT plans was significantly shorter than that for the HT plans, and that for the HT plans was significantly shorter than that for the IMPT plans (both P < 0.001).
Discussion
This was the first pilot study comparing photon IMRT and IMPT for AS patients. RT field and the total dose were applied according to the recommendations by other retrospective studies and a literature review. 6,7,10 -14 As a result, all 3 techniques provided sufficient coverage for the targets, as shown in Figure 2A. Homogeneity of prophylactic total scalp area (i.e. target 2) was slightly poorer in the IMPT plans than in the other plans, but it was within the acceptable range. This was caused by differences in plan quality depending on the physician rather than the difference in the 3 techniques because the IMPT plans reduced doses to most OAR. In particular, IMPT reduced the Dmean of the brain by 59% and 49% compared with HT and VMAT, respectively. IMPT also reduced the Dmax of the spinal cord PRV, brainstem PRV, and optic pathway PRV by 70%-92% compared with the other techniques. Our study confirmed the expected advantages of IMPT over photon IMRT, demonstrating significant dose reductions in the OAR without compromising target coverage. In addition, our current study confirmed that photon IMRT plans provide sufficient coverage of the PTV while limiting the surrounding OAR to the preset dose constraints when 70 Gy and 56 Gy are prescribed to PTV1 and PTV2, respectively. However, the Dmean of the brain was 37.4 Gy in the HT plans and 29.7 Gy in the VMAT plans, whereas it was only 15.2 Gy in the IMPT plans. As doses to the normal brain may exceed 30 Gy in the photon IMRT plans, RT-associated adverse effects on the brain are of concern with photon IMRT. Similarly, the Dmean of the hippocampus was 25.8 Gy in the HT plans and 16.1 Gy in the VMAT plans, whereas it was only 1.4 Gy in the IMPT plans. When patients with brain metastases receive whole-brain RT, early neurocognitive decline, such as short-term memory loss and verbal dysfunction, may develop within 4 months after RT, 28 -30 although the mini-mental state examination score may be unchanged. 31 As doses to the hippocampus are regarded as a major factor for the development of neurocognitive decline, 32 IMPT is advantageous for hippocampal sparing compared with photon IMRT.
In this study, we focused on AS, which is often treated by the combination of surgery and adjuvant RT with or without chemotherapy, 33 resulting in unfavorable outcomes. 3 -9,11 -14,16 Several problems remained unclear due to this rare aggressive malignancy such as the satisfactory radiation field and appropriate RT technique. The RT technique may be associated with the high frequency of local recurrence because the common conventional combination technique of X-rays and electron beams for total scalp irradiation can result in an insufficient dose to the target and complex procedures. Thus, total scalp irradiation using this conventional technique has not always been employed for AS patients and the extended local field with electron beams alone was often used. 11,13,34,35 The local control rate was 86% at 3 years in AS patients who received total scalp irradiation, 14 whereas it was 57-71% at 1-2.5 years in those who received only local irradiation. 11,34,35 Total scalp irradiation may improve local control for AS patients. Total scalp irradiation consists of the 3 features: many OARs around the PTV, a helmet-like shape, and superficial lesions. As it is challenging to create a satisfactory RT plan for total scalp irradiation, it is essential for improvement of the outcome to clarify the best RT technique. Our current study demonstrated differences between photon IMRT and IMPT. IMPT may improve outcomes for AS patients due to its excellent tumor coverage and OAR avoidance.
For fair comparison of different planning strategies, it is essential that all plans are of high quality, and generated using a similar approach regarding target coverage and OAR sparing. Therefore, we set common dose constraints and acceptable ranges for the target and OAR, and conducted manual planning. One limitation of this study was the difference in the target definition between photon IMRT plans and IMPT plans. For IMPT, robust optimization was used instead of a CTV-PTV margin. 36 On the other hand, the addition of a geometric margin to the CTV is insufficient in the proton plans because it cannot respond to changes in density along the beam path, upon which protons are highly dependent. As a result, this increases the distorted dose distribution in the proton plans, especially in IMPT because non-uniform fields are used to produce the desired dose distribution. Thus, it is necessary in the proton plans to account for both range uncertainty and setup uncertainty. Considering these uncertainties, the IMPT plans were constructed by the CTV robust optimization using worst-case scenario. If the PTV was defined as the CTV plus a 0.5-cm margin in IMPT and in HT and VMAT, adding another robustness margin beyond the PTV doubly accounts for uncertainties. Another limitation was the use of a pencil beam dose algorithm. The RayStation treatment planning system employs pencil beam and Monte Carlo algorithms for proton dose calculations. We noted superior dose prediction accuracy of a Monte Carlo algorithm over a pencil beam algorithm. Several studies 37,38 highlighted the limitations of a pencil beam algorithm within RayStation for dose calculation, especially in the presence of a range shifter. Shirey and Wu 38 reported better accuracy of a Monte Carlo algorithm than of a pencil beam algorithm when treatment involves the range shifter and superficial lesions. As the adoption of Monte Carlo algorithms is slow in our country due to the late acceptance of the pharmaceutical affairs law, we were unable to use a Monte Carlo algorithm in this study.
Our findings are consistent with those of previous studies, which reported an improvement in OAR sparing with IMPT compared with photon IMRT using comparative dosimetric evaluations for several types of cancer. 39 -43 On the other hand, the beam-on times for the IMPT plans were 2.1- and 1.1-times longer than those for the VMAT and HT plans, respectively. As one non-coplanar and 4 coplanar beam directions were needed considering plan robustness, IMPT plans require a longer time than photon IMRT plans excluding beam-on times.
On comparison of HT with VMAT, VMAT demonstrated better sparing of many OAR, such as the Dmax of the spinal cord PRV and the Dmean of the brain, than HT. However, HT demonstrated better sparing of the Dmean of both bilateral parotid glands. This was caused by the plan quality, which depended on the physician, as mentioned above. The plans with or without a bolus may also be associated with the difference in target coverage and OAR sparing between HT and VMAT. Song et al. 15 reported that HT plans with and without a bolus did not differ markedly, and a well-designed HT plan can be obtained, even for regions close to the surface, without the use of a bolus. This can be explained by the high number of tangential beams, which is unique to HT. 44 Thus, no bolus was used for the HT plans in our study. Unlike HT, the no-bolus VMAT plans were unable to cover the PTV sufficiently and D95% of the PTV achieved at most 90-95% of the prescription dose. As HT plans do not necessarily require the use of a bolus, this is a great advantage of HT and IMPT plans.
In conclusion, the 3 techniques of HT, VMAT, and IMPT provided sufficient coverage and satisfactory homogeneity for the target when 70 Gy and 56 Gy were prescribed to target 1 based on the GTV and target 2 based on the total scalp, respectively. Although longer delivery times were necessary in IMPT, it has the advantage of not requiring a bolus. IMPT achieved the best overall OAR sparing, with low doses to the brain and hippocampus in particular.
Footnotes
Abbreviations
Authors’ Note
This study was performed after approval by the institutional review board of Nagoya City University Graduate School of Medical Sciences (approval number: 60-19-0181). The requirement for written informed consent was waived due to the retrospective nature of this study in line with the Ethical Guidelines for Medical and Health Research Involving Human Subjects in our country. Therefore, its content was disclosed in the form of opt-out on the website.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 19K08183.
