Abstract
Introduction
Advances in and the widespread use of low-dose CT for screening lung cancer have helped significantly increase the detection of ground-glass nodules (GGNs). 1 Studies have shown that pure ground-glass nodules (pGGNs) mostly exhibit indolent biological characteristics, with a long window of progression; the results of surgical pathology of pGGNs often indicate a diagnosis of T1-stage lung adenocarcinoma or precancerous lesions.2,3 Among T1-stage lung adenocarcinomas presenting as pGGNs, the main pathological type is lepidic-predominant growth adenocarcinoma, which is characterized by low malignancy, a good prognosis, and a low rate of lymph node metastasis. Surgical methods mostly include segmentectomy or wedge resection, while systematic mediastinal lymph node dissection is less frequently employed. 4 According to the 2015 World Health Organization classification of lung adenocarcinoma prognostic risk levels, the lepidic-predominant type is classified as low risk, having the best prognosis. 5 Additionally, in the 2020 revised lung adenocarcinoma prognostic risk classification, lung adenocarcinoma is divided into three grades based on its pathological components, with lepidic-predominant and high-risk components (complex glandular type, solid or micropapillary predominant adenocarcinoma, MPA) accounting less than 20% classified as Grade 1, which also has the best prognosis. 6 Therefore, accurate preoperative assessment of the pathological subtype of pGGNs is highly important for adjusting follow-up strategies and formulating surgical plans for treating the patient.
Recent research has focused mainly on evaluating the conventional imaging features of GGNs on CT to further predict their postoperative pathological subtype.7–9 However, conventional imaging is limited to the morphological evaluation of pGGNs, making it difficult to conduct a multidimensional, in-depth analysis of their texture characteristics. Moreover, the results heavily depend on the radiologist's diagnostic experience, introducing significant subjectivity. Radiomics is a technique that uses computer algorithms to analyze medical imaging data and extract numerous image features, allowing for a more objective assessment of properties of the lesion, quantification of lesion characteristics, and prediction of disease progression and prognosis. It has extensive applications in the diagnosis, treatment strategy selection, and prognosis evaluation of lung nodules. 10 Furthermore, considering the potential for lung cancer to infiltrate surrounding lung parenchyma, which may alter the peritumoral microenvironment, the peritumoral characteristics has potential to serve as important tools for assessing tumor growth, cell migration, inflammation, and microvessel formation. 11
This study builds on previous research by extracting radiomic features of the tumor and peritumoral regions of pGGNs and collecting their clinical imaging data to establish a predictive model for the pathological subtype of pGGNs. The model aims to enhance diagnostic accuracy at an earlier preoperative stage, enabling clinicians to identify pathologic subtypes more clearly and to determine appropriate patient management strategies and treatment options in a shorter period of time, which will help reduce the chance of disease progression.
Materials and Methods
The retrospective studies involving human participants were reviewed and approved by the Ethics Committee of the Universal Cloud Medical Imaging Diagnostic Center, Shanghai University (No. 2024KY056). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements. In this study, informed consent was obtained from all study participants. The informed consent was verbal. The patients consented to participate in the research study and for the use of their data for scientific analysis and publication while ensuring the confidentiality and anonymity of their personal information. The reporting of this study conforms to TRIPOD guidelines. 12
Patient Selection
A retrospective analysis was conducted on T1-stage lung cancer patients who were diagnosed with pGGNs by CT and underwent surgical resection from June 2020 to June 2023 and identified according to strict inclusion criteria as follows: i. postoperative pathology confirming a diagnosis of T1-stage lung adenocarcinoma (T1N0M0); ii. preoperative chest CT images showing pGGNs; and iii. no history of radiotherapy, chemotherapy, immunotherapy, or other surgeries before the CT examination. The exclusion criteria were as follows: i. no preoperative CT volumetric scan (slice thickness: 0.625 mm–1.25 mm); ii. poor image quality due to motion or respiratory artifacts, affecting subsequent tumor delineation and image processing; and iii. maximum pGGN diameter on CT volumetric images larger than 30 mm or smaller than 5 mm. According to the Fleischner Society's recommendations, pGGNs are defined as hazy patches on lung windows with visible lung texture and faintly discernible lung parenchyma. 1 In this study, the enrolled patients were divided at a 2:1 ratio into a training cohort and a validation cohort. The data of the training cohort were used for model training, and those of the validation cohort were used for model validation.
CT Image Acquisition
The CT scans in this study were performed with a GE Revolution CT scanning system. During the lung CT scans, patients were instructed to take a shallow breath and hold it for 5 s. The scanning range extended from the lung apices to the bilateral adrenal glands. The scanning parameters of the system were as follows: tube voltage of 120 kV, tube current of 100–150 mA, pitch of 0.828–1.2, matrix size of 512 × 512, standard kernel algorithm, and reconstruction with a slice thickness of 0.625–1.250 mm and an interslice distance of 0.625–1.250 mm. The completed scans were automatically transmitted to the picture archiving and communication system.
Segmentation, Feature Extraction, and Selection
Two senior radiologists with over 10 years of experience in chest imaging analysis interpreted the conventional CT radiological features of the lung nodules while being blinded to the pathological results. When the conclusions between the doctors differed, they reached a consensus through discussion. The assessed conventional features included boundaries, lobulation, spiculation, vascular convergence, vacuole signs, and pleural retraction signs. The pGGNs of the lung were segmented with the open-source platform 3D Slicer (v4.9.0, www.slicer.org) and its “segment editor” module. Semiautomatic segmentation techniques, namely, “level tracing” and “smoothing”, were employed to delineate the tumor contour according to the appearance of the tumor on CT images in the axial, sagittal, and coronal planes, including the entire tumor (VOI entire) and the tumor boundary extended to the 2 mm peritumoral region (VOI +2 mm) (Figure 1). The segmentation was subsequently reviewed by radiologists and thoracic surgeons, with additional adjustments made in case of differing opinions.
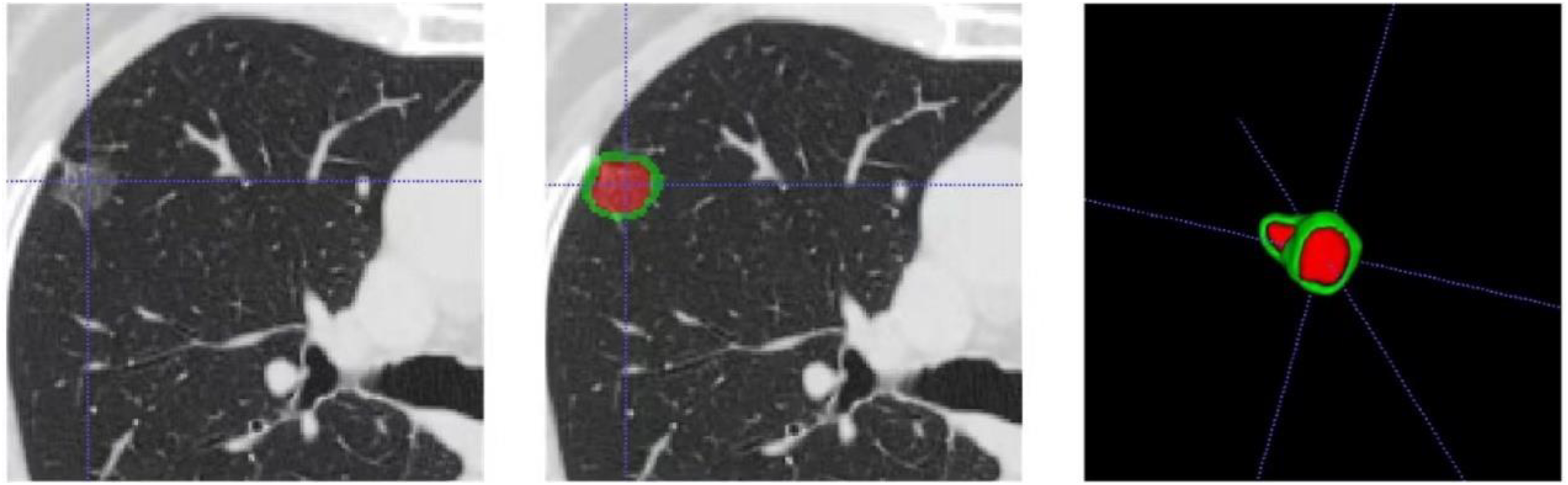
Process of VOI entire and VOI +2 mm lesion segmentation.
To extract radiomic features of the tumor and peritumoral regions from CT images, the PyRadiomics plugin 3.0, an open-source Python package for radiomic feature extraction, was employed. 13
The radiomic features obtained in this study covered multiple categories, including first-order statistical features, gray-level histogram statistical features, 2D and 3D morphological features, and texture features (such as gray-level dependence matrix and gray-level size zone matrix features).
Intra- and Peritumoral-Based Radiomic Model Development
Before dimensionality reduction, the patient feature values were standardized by calculating their z scores as follows: z score = (x - μ)/σ, where x represents the feature value, μ is the mean of the feature values across all patients, and σ is the corresponding standard deviation; this equation transforms the data to have a mean of 0 and a variance of 1. Subsequently, Pearson or Spearman correlation redundancy analysis was performed on the standardized radiomic features, and features with a correlation coefficient exceeding 0.9 were identified. Finally, least absolute shrinkage and selection operator (LASSO) and 10-fold cross-validation were used to reduce the dimensionality of the remaining radiomic features (Figure 2), retaining those with nonzero coefficients, which were then summed to obtain the Rad score.

Least absolute shrinkage and selection operator regression model with 10-fold cross-validation.
Statistical Analysis
Statistical analysis was performed with R.4.3.1, and the diagnostic performance for the pathological subtype of pGGNs in the lungs was assessed with receiver operating characteristic (ROC) curve analysis separately for models constructed with VOI entire, VOI +2 mm, and conventional features, including calculation of the area under the curve (AUC), sensitivity, specificity, and accuracy.
Results
Comparison of Baseline Data
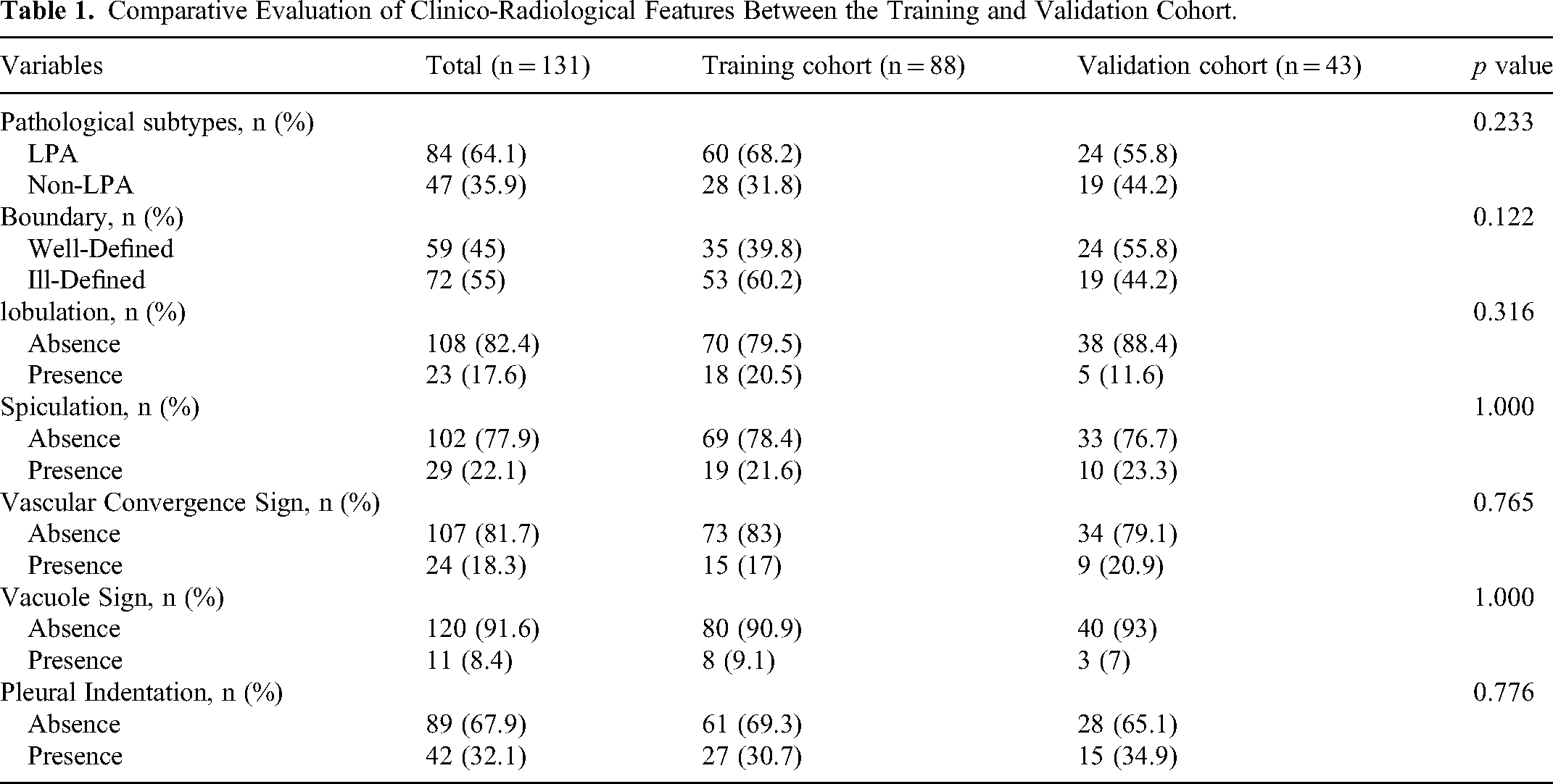
A total of 131 patients were included in the study after application of the predefined inclusion and exclusion criteria. Among them, 84 patients were diagnosed with the lepidic-predominant subtype, whereas the remaining 47 patients were diagnosed with the non-lepidic-predominant subtype. Univariate analysis (Table 1) between the training set, which consisted of the data of 2/3 (n = 88) of the patients, and the validation set, which consisted of the data of 1/3 (n = 43) of the patients, revealed no statistically significant differences in the baseline characteristics (all P > 0.05).
Comparative Evaluation of Clinico-Radiological Features Between the Training and Validation Cohort.
Evaluation of Different Models for Predicting the Pathological Subtype of Pure Ground-Glass Lung Nodules
Univariate and multivariate logistic regression analyses were performed on the conventional characteristics of pGGNs of the lung in the predominantly attached and nonattached groups. The univariate analysis results revealed that lobulation and spiculation were associated with the pathological subtype, whereas the multivariate results indicated that only spiculation was an independent subtype predictor (Table 2).
Univariate and Multivariate Logistic Analysis for Distinguishing Histological Subtypes in Clinical Stage T1 pGGNs Lung Adenocarcinoma.

Evaluation of Lesion and Perilesional Radiomic Features in Predicting the Pathological Subtype of Pure Ground-Glass Lung Nodules
In the selection of lesional and perilesional radiomic features for predicting the pathological subtype of pGGNs of the lung, LASSO dimensionality reduction resulted in the final identification of 5 and 9 radiomic predictors for VOI entire and VOI +2 mm, respectively (Figure 3). In a comparison of the Rad-scores constructed from the VOI entire and VOI +2 mm features, the Rad-scores of the lepidic-predominant patients were lower than those of the non lepidic-predominant patients (both P < 0.05) (Figure 4).

Radiomics signature was built for VOI entire and VOI +2 mm lesion.

Violin diagrams comparing Radscore between LPA and non-LPA in the training and validation cohort.
Using conventional features as well as the VOI entire and VOI +2 mm features to diagnose the pathological subtype of lung pGGNs, ROC curve analysis (Figure 5) revealed that the diagnostic performance of the VOI entire- and VOI +2 mm-based models was superior to that of conventional characteristics. Among them, the VOI +2 mm model had the best diagnostic performance: the AUC, sensitivity, specificity, and accuracy in the training set were 0.883, 0.964, 0.667, and 0.761, respectively, and those in the validation set were 0.886, 0.895, 0.750, and 0.815, respectively (Table 3).

ROC curve comparing performance across image finding, VOI entire, and VOI +2 mm in the training and validation cohort.
Evaluating Diagnostic Performance Across Models in Distinguishing LPA.

Discussion
This study conducted radiomic analysis on CT images of pGGNs in the lungs to explore the diagnostic efficacy of conventional features, VOI entire features, and VOI +2 mm for the pathological subtype of pGGNs. The results showed that all three methods effectively identified the pathological subtype of lepidic-predominant GGNs, with radiomics models (VOI entire, VOI +2 mm) outperforming the conventional feature models, and the VOI +2 mm model having the highest diagnostic efficacy.
Among these subtypes, the lepidic-predominant subtype has the better prognosis.5,6 Previous studies5–9 have indicated that for lung pGGNs, the most common pathological subtype is the lepidic-predominant subtype, characterized by relatively low malignancy and a low lymph node metastasis rate. Sublobar resection is frequently recommended for treating these pGGNs, while systematic mediastinal lymph node dissection is less frequently suggested. Therefore, preoperative evaluation of the pathological subtype of pGGN in the lungs is highly valuable for follow-up management, surgical timing and planning, postoperative monitoring, and prognosis assessment. However, the preoperative CT diagnosis of the pathological subtype also presents new challenges for radiologists. In previous studies, preoperative evaluations of the pathological subtypes of GGNs in the lungs mainly consisted of the analysis of conventional CT features, which include spiculation, lobulation, pleural retraction, bubble-like lucency, and vascular convergence signs.7–9 However, owing to the significant subjectivity of these signs, given their large dependence on the radiologist's diagnostic experience, their consistency is poor, limiting their generalizability.
Radiomics can delve deeper into conventional images, mapping image pixels to the tumor's biological microenvironment and thereby extracting multidimensional features that are not observable by the naked eye. 10 The value of CT radiomics in pGGNs has been reported with promising results.14–17 Xiong et al 18 demonstrated that radiomic features based on CT improved the recognition performance of pathological subtypes of pGNNs lung adenocarcinoma, and achieved a good prediction performance of 0.792 AUC. Currently, most studies focus only on intra-tumor features, ignoring the fact that peri-tumor features contain equally important information. And CT radiomic features of the peritumoral region has long been shown to be a very valuable factor to study.19–21 Considering the potential for tumor infiltration, which may alter the peritumoral lung tissue microenvironment, peritumoral features are also important for evaluating tumor growth, cell migration, inflammation, and microvascular formation. 11 Therefore, it is necessary to consider peritumoral features when assessing pGGNs of the lung. Wu et al 11 reported that when evaluating the invasiveness of pGGNs, the diagnostic efficacy of features of the peritumoral region 2 mm beyond the tumor boundary was not greater than the diagnostic efficacy of the features of the tumor region itself.
Based on this finding, two models were employed in this study, one based on features from the entire VOI and another based on the features from the entire VOI with a 2-mm peritumoral region, for the radiomic analysis of pGGNs in the lungs following dimensionality reduction to select optimal radiomic features and construct the Rad-score. The Rad-score can quantify tumor heterogeneity, the biological characteristics of malignancies, and the tumor microenvironment. The study results revealed that for lesions with a postoperative pathological subtype of lepidic-predominant, the Rad score was lower than that of the other pathological subtypes, corresponding to the lower degree of malignancy of lepidic-predominant lung adenocarcinoma. In addition, the results of the present study fully exposed the great potential of radiomics features of the peritumoral region in diagnosing the pathological subtypes of pGGNs, reaching an AUC of 0.886 on the validation group, which was superior to the conventional features of 0.768 and the whole VOI of 0.833. In contrast to other related studies that have utilized only tumor-area radiomic features, this study provides a valid comparison regarding the predictive effects of peritumoral and tumor-area radiomic features as well as conventional features, and achieves good predictive results.
This study has certain limitations: first, it is a single-center study and lacks external validation, and the generalizability of the model needs to be improved and it is prone to poor generalization of the model to data from external centers; second, it is a retrospective study with a small sample size, which made it vulnerable to selection bias and information bias, and the small sample size may have led to an increase in chance outcomes. Finally, our failure to conduct long-term follow-ups in our study may render us susceptible to bias. Future studies will consider multicenter, prospective data collection and larger sample sizes to improve the validity and stability of results. It will also aim to enhance the clinical application value of the model.
Conclusion
In summary, prediction models were constructed for the pathological subtype of pGGNs in the lungs with radiomic features extracted from the tumor and peritumoral regions. The results revealed that both the VOI entire model and the VOI +2 mm model outperformed the conventional feature model, with the VOI +2 mm model showing the best performance, highlighting the importance of the peritumoral microenvironment in evaluating the biological characteristics of tumors.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241305432 - Supplemental material for Intra- and Peritumoral-Based Radiomics for Preoperatively Assessing the Pathological Subtype of T1-Stage Lung Adenocarcinoma Presenting as Pure Ground-Glass Nodules
Supplemental material, sj-docx-1-tct-10.1177_15330338241305432 for Intra- and Peritumoral-Based Radiomics for Preoperatively Assessing the Pathological Subtype of T1-Stage Lung Adenocarcinoma Presenting as Pure Ground-Glass Nodules by Wenting Jiang, Tingting Qu, Weiran Liu, Huazheng Shi and Yali Zhang in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
None.
Author Contributions
The literature search, analysis, data explanation, and manuscript draft were finished by WJ, TQ, and WL. WL and HS are responsible for the analysis and explanation of the radiomics imaging features data. WJ, TQ, and WL and YZ acquired the clinical information and imaging. YZ designed the study, explained the data, and made multiple revisions to the manuscript. All authors contributed to the article and approved the submitted version.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
Ethics Statement
The retrospective studies involving human participants were reviewed and approved by the Ethics Committee of the Universal Cloud Medical Imaging Diagnostic Center, Shanghai University (No. 2024KY056). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements. In this study, informed consent was obtained from all study participants. The informed consent was verbal. The patients consented to participate in the research study and for the use of their data for scientific analysis and publication while ensuring the confidentiality and anonymity of their personal information.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Municipal Health Commission, (grant number No.202340225).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
