Abstract
Introduction
The incidence and mortality of lung cancer have decreased significantly due to the decrease in the number of smokers and advances in the early detection and treatment of lung cancer, but lung cancer is still the main cause of cancer deaths worldwide. 1 The poor specificity of LDCT in lung cancer diagnosis leads to a high rate of false-positive results. Patients with indeterminate pulmonary nodules who are followed with serial CT scans receive excess radiation exposure.2,3 To address the above issues, hematological biomarkers have been used to facilitate the diagnosis and early screening of lung cancer, including small molecule DNA/RNA, tumor markers, and autoantibodies.4–8 Liquid biopsy has been proven to have clinical value in the diagnosis and early screening of lung cancer through previous studies.9,10 Currently, tumor markers (carcinoembryonic antigen, cytokeratin 19 fragment, neuron-specific enolase, squamous cell carcinoma antigen) and seven autoantibodies are widely used clinically to screen for lung cancer. Previous studies11,12 have shown that tumor markers, which are mainly used to monitor the response to chemotherapy, have low sensitivity and are of unproven clinical value in early screening for lung cancer, while seven autoantibodies have better accuracy.
The human immune system plays an important role in the occurrence and development of malignant tumors, including cellular and humoral immune responses, which can produce a large number of antibodies. AABs is a corresponding antibody produced by the immune system after the release of early related antigens that recognize the occurrence and development of tumor cells. Thus, highly expressed autoantibodies can be detected in the early stage of patients with malignant tumors. 13 The early CDT lung detection kit, which is composed of seven autoantibodies ([7- AABs], anti-p53, -PGP9.5, -SOX2, -GAGE7, -GBU4-5, -MAGEA1, and -CAGE), has good diagnostic value for lung cancer with a sensitivity of 41.7% and a specificity of 91.7%. 14 In 2015, China approved the clinical use of seven autoantibody detection kits; however, the value of 7-AABs in the screening and early detection of lung cancer still needs to be confirmed. This article mainly discusses the clinical value of seven cancer antibodies and tumor markers in the diagnosis and screening of lung cancer through retrospective analysis of real-world data from a single center.
Materials and Methods
Clinical Characteristics of Patients
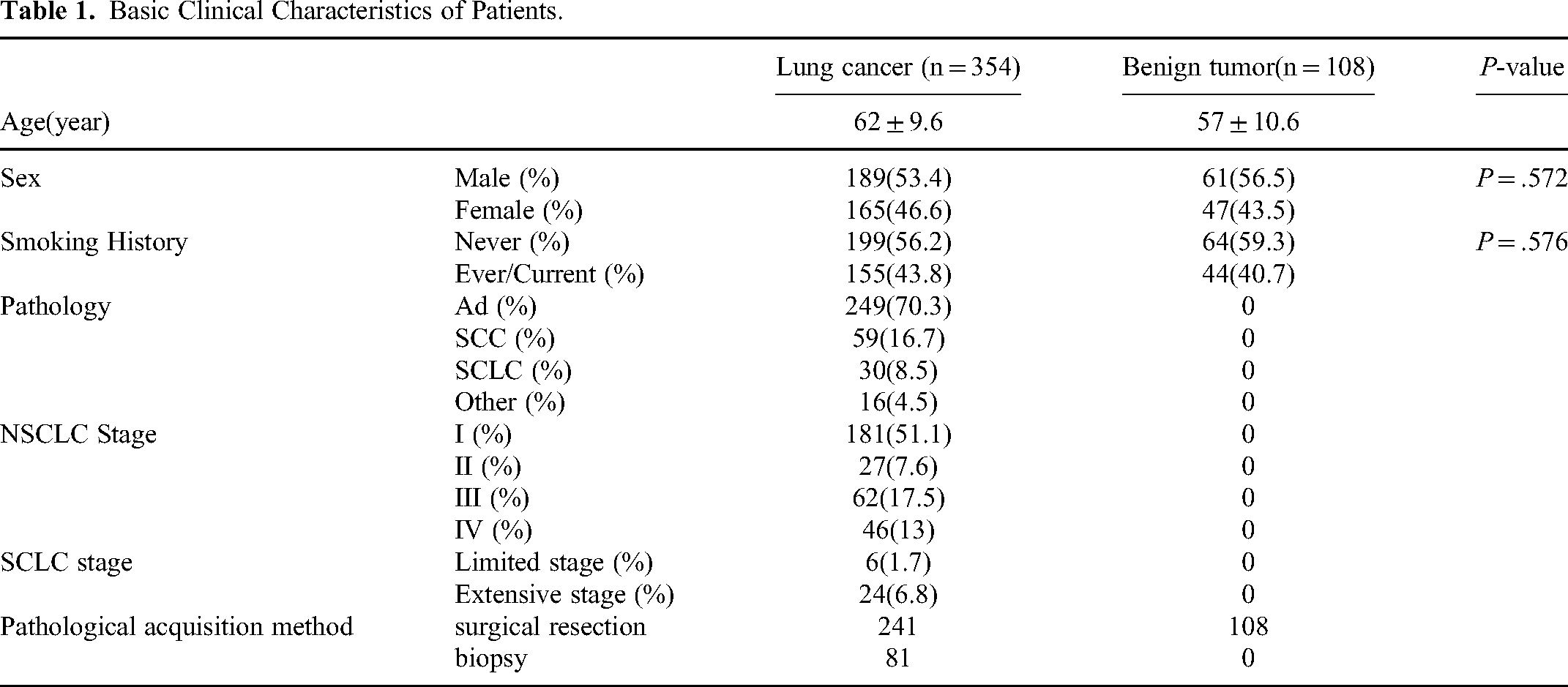
The study collected and analyzed 462 patients with pulmonary nodules, including250 males and 212 females, detected by chest CT in the Department of Lung Oncology at General Hospital of Tianjin Medical University from December 2018 to August 2019. There were 354 patients with lung cancer, including 249 patients with adenocarcinoma (AD), 59 patients with squamous cell carcinoma (SCC), 30 patients with small cell lung cancer (SCLC), and 15 patients with other lung malignant tumors (atypical carcinoid, carcinoid, sarcoma, sarcomatoid carcinoma, synovial sarcoma, and pleomorphic carcinoma). In the non-small cell lung cancer (NSCLC) group, patients with stage I-IV accounted for 51.1%, 16.7%, 8.5%, and 4.5%, respectively. The extensive and local stages of SCLC accounted for 6.8% and 1.7% of patients, respectively. All patients with lung cancer and no history of lung or other tumors and no history of treatment with antineoplastic therapy, radiotherapy, chemotherapy, or targeted therapy were staged according to the eighth edition staging criteria. There were 108 patients with benign lesions, including infectious lesions, tuberculosis, inflammatory pseudotumors, and hamartomas. The diagnoses were confirmed by pathologic evaluation of a surgical resection specimen or biopsy. Detailed information is shown in Table 1.
Basic Clinical Characteristics of Patients.
Sample Collection
Venous blood samples (10 mL and 3 mL) were been collected from the patients in the morning and sent to the Laboratory Department of the General Hospital of Tianjin Medical University for testing. The 10 mL-venous blood sample was used to detect 7-AABs. The 7-AAB detection procedure was performed strictly according to the kit instructions, and the absorbance value of the enzyme-linked detector at 450 nm (A450 nm) was read. A p53 ≥ 13.1 U·mL−1, PGP9.5 ≥ 11.1 U·mL−1, SOX2 ≥ 10.3 U·mL−1, GAGE7 ≥ 14.4 U·mL−1, GBU4-5 ≥ 7.0 U·mL−1, MAGE ≥ 11.9 U·mL−1, and CAGE ≥ 7.2 U·mL−1 were considered positive; otherwise, the autoantibody titers were negative. Tumor markers were detected using a chemiluminescence instrument and related reagents from 3-mL venous blood samples. Cy-fra21-1, squamous cell carcinoma antigen (SCC), carcinoembryonic antigen (CEA), gastrin releasing peptide precursor (ProGRP), and neuron specific enolase (NSE) were detected by enzyme-linked immunosorbent assay. A CEA ≥ 5.00 ng/mL, CYFRA21-1 ≥ 3.30 ng/mL, SCC ≥ 1.5 µg/L, ProGRP ≥ 63 pg/mL, and NSE ≥ 16.30 µg/L were considered positive; otherwise, the tumor markers were negative. If combined analysis is performed on tumor markers or 7-AABs, any single value above the specified value is defined as positive.
Statistical Analysis
Statistics were performed using SPSS22.0 software. Continuous variables with a non-normal distribution are represented by the median (25th percentile, 75th percentile). The normal distribution and variance homogeneity test were performed before the test, the Mann-Whitney U test was used to compare the two groups that did not conform to the normal distribution, and the Kruskal Wallis test was used for multiple independent sample data. The counting data are expressed by n (%) based on a chi- square test and Fisher's exact probability method. The clinical value was evaluated by a ROC). P < .05 indicates that the difference was statistically significant.
Results
Expression of 7-AABs and Tumor Markers
Four hundred sixty-two patients with pulmonary nodules were divided into two groups based on the pathologic results: lung cancer; and benign disease. Propensity matching analysis showed that there was no statistical difference between the two groups with respect to smoking history, gender, age, and other basic factors. The results of the analysis are shown in Figure 1. There was no significant difference in the positive expression rate of individual autoantibodies between the lung cancer and benign disease groups with the exception of GAGE7, but the positive expression rate in patients with lung cancer was significantly higher than patients with benign diseases when the seven individual autoantibodies were combined (30.1% vs 15.7%, P < .05). The positive expression rates of the tumor markers (CEA (27.2%), CYFRA21-1 (29.9%), ProGRP (8.3%), NSE (37.4%)) in lung cancer group were significantly higher than the benign disease group (P < .05). The positive expression rate in the lung cancer group was 68.8% when the tumor markers were combined with the 7-AABs, which was significantly higher than the benign disease group (P < .05).

Positive expression rates of 7-AABs and tumor markers.
The lung cancer group was further divided into NSCLC and SCLC groups based on the pathological results to further analyze the level of AAB expression. The levels of SOX2 (1.3 vs 0.9) and GBU4-5 expression (0.9 vs 0.8) in patients with NSCLC were significantly higher than the levels in the benign disease group (P < .05). The levels of GUB4-5 (1.2 vs 0.8) and CAGE expression (0.4 vs 0.1) in patients with SCLC were significantly higher than the levels in the benign disease group (P < .05). The levels of CEA, CYFRA21-1, ProGRP, and NSE expression in patients with lung cancer were higher than the levels in the benign disease group, while the levels of ProGRP (243.47 vs 30.40) and NSE expression (22.01 vs 14.41) in patients with SCLC were significantly higher than the levels in patients with NSCLC (P < .05; Table 2). The levels of GUB4-5 (0.9 vs 1.2) and CAGE expression (0.1 vs 0.4) in patients with SCLC were significantly higher than the levels in the NSCLC group (P < .05).
Expression Levels of 7-AABs and Tumor Markers in Lung Cancer and Benign Lesions.

NSCLC versus Benign P = .031.
SCLC versus Benign P = .016, NSCLC versus SCLC P = .046.
NSCLC versus SCLC P = .005, SCLC versus Benign P = .012
NSCLC versus Benign P = P = .048, SCLC versus Benign P = .000, NSCLC versus SCLC P = .002.
NSCLC versus Benign P = .000, SCLC versus Benign P = .000.
NSCLC versus Benign P = .000, SCLC versus Benign P = .000.
NSCLC versus Benign P = .000, SCLC versus Benign P = .000.
Diagnostic Efficacy of 7-AABs and Tumor Markers
A ROC curve was drawn to analyze the diagnostic efficacy of the 7-AABs or tumor markers in patients with lung cancer. The sensitivity of individual autoantibody was <10%, the specificity was >90%, and the area under each ROC curve was >0.50. The sensitivity, specificity, positive predictive value (PPV), and the area under the ROC curve of 7-AABs were 30.0%, 84.3%, 86.3%, and 0.61, respectively. The sensitivity and specificity of the individual tumor markers fluctuated widely; NSE had the highest sensitivity (37.4%) and ProGRP have the highest specificity (98.1%). The sensitivity of the combined tumor markers was 61.4%, the specificity was 56.7%, the PPV was 82.7%, and the area under the ROC curve was 0.69. The sensitivity of the 7-AABs combined with the tumor markers was 68.6%, the specificity was 52.8%, the PPV was 82.7%, and the area under the ROC curve was 0.72 (Figure 2).

Diagnostic efficacy of 7-AABs and tumor markers. A. Sensitivity of 7-AABs and tumor markers; B. Specificity of 7-AABs and tumor markers; C. PPV of 7-AABs and tumor markers; D. ROC curve of 7-AABs and tumor markers.
Analysis of Clinicopathological Characteristics with 7-AABs and Tumor Markers
There were different positive expression rates of the 7-AABs and tumor markers among the patients with NSCLC in different clinical stages; patients with early lung cancer had the lowest positive expression rate. According to the eighth edition staging criteria, the positive expression rates of AABs from stage I-IV NSCLC patients were17.1%, 44.4%, 46.8%, and 40.4%, respectively (P < .05). The more advanced the stage, the higher the positive expression rate of tumor markers (43.6%-91%; P < .05). When the two methods were combined, the positive rates increased to 50.3%,76.9%, 82.3%, and 93.6%, respectively (P < .05). But in SCLC, the positive expression rate of the 7-AABs compared to the 7-AABs combined with tumor markers was not significantly different between patients with limited- and extensive- stage disease (P > .05; Table 3).
Relationship Between 7-AABs and Tumor Markers with Clinical Features, Clinical Stages or Pathological Features.

The positive expression rates of 7-AABs varied among patients with cancer compared to benign disease as a function of clinical features. The positive expression rates of 7-AABs was high in male elderly smokers (age, 32.9% vs 23.8%; sex,35.1% vs 24.8%; and smoking history, 40.6% vs 22.2%) When combined with tumor markers, the positive expression rate was more easily affected by age (57% vs71%), sex (79.7% vs 52.7%) and smoking history (54% vs 83.8%; P < .05). Among the different pathological types, the positive expression rate of the 7-AABsin patients with SCLC was the highest. There were significant differences among the positive expression rates in patients with AD (26.1%), SCC (37.3%), and SCLC (53.3%; P < .05). When the 7-AABs were combined with tumor markers, the positive expression rates of AD, SCC, and SCLC were 70.8%, 84.7%, and 96.6%, respectively (P < .05).
Relationship Between the 7-AABs and Pathological Classification or Diameter of Adenocarcinomas
The positive expression rates of the 7-AABs varied as a function of the adenocarcinoma pathologic type. Specifically, the positive expression rate of soild adenocarcinomas was the highest (38.46%), followed by mucinous adenocarcinomas (37.5%). The positive expression rate of 7-AABs among the adenocarcinoma subgroups gradually increased as the tumor diameter increased, but there was no statistical difference between the positive expression rate of 7-AABs and the pathological types or tumor diameter of adenocarcinomas because of the small sample size (Table 4).
Relationship Between 7-AABs and Pathological Subtypes of Adenocarcinoma or maximum Tumor Diameter.

Discussion
Most patients with lung cancer are diagnosed at an advanced stage, losing the opportunity for surgical treatment and reducing survival time. The 5-year survival rate of patients with stage IV lung cancer is approximately 2%, and the 5-year survival rate of early lung cancer is significantly higher than advanced lung cancer, 15 thus early detection and early treatment of lung cancer is an effective way to improve the survival rate. Currently, early screening for lung cancer mainly depends on low dose computed tomography (LDCT), which has high sensitivity. Two meta- analyses2,16 concluded that LDCT is more sensitive than chest X-ray or no screening in the detection of early lung cancer, and reduced lung cancer mortality by 20%.
The early detection and diagnosis of lung cancer remains a challenge. In this study, we reviewed and analyzed the diagnostic efficacy of 7-AABs and tumor markers in 462 patients with pulmonary nodules using real-world data. The 7-AABs have high specificity as a diagnostic tool for tumors and have thus recently received considerable attention. The specificity of individual autoantibodies was >90% and the specificity of 7- AABs was 84.3%, which was significantly higher than tumor marker (56.7%). In addition, the PPV of the 7-AABs was 86.3% and the area under the ROC curve was 0.61, suggesting that the 7-AABs had clinical significance in the diagnosis of lung cancer. The sensitivity of the 7-AABs in the clinical setting was 30.0%, which is significantly lower than previously reported.7,16 There is a significant gap between the positive expression rates of 7-AABs in patients with lung cancer and benign tumors, but the positive expression rates of the 7-AABs were not as high as previously reported3,7,16 A previous study 7 showed that tumor markers were valuable in the diagnosis of lung cancer, but were easily affected by infection, benign tumors, pregnancy, and other factors, resulting in false-positive results. Combining the 7-AABs with the tumor markers, the increased the sensitivity to 68.6%, decreased the specificity to 52.8%, increased the area under the ROC curve to 0.72, and significantly increased the positive expression rate in patients with lung cancer, which indicated that 7-AABs combined with tumor markers had better diagnostic value in patients with lung cancer. For patients with pulmonary nodules, the detection of 7-AABs combined with tumor markers has high diagnostic value in determining the malignancy of pulmonary nodules.
The presence of AABs may provide an aid to early detection of lung cancer, particularly in early-stage disease, which is potentially curable; 17 however, the positive expression rate of 7-AABs was shown to differ in the current study as a function of NSCLE clinical stage; the positive expression rate of 7-AABs was lowest in stage I lung cancer (17.1%). Among the different pathological types, the positive expression rate of 7-AABs in patients SCLC was the highest, and there were significant differences among patients with AD, SCC, and SCLC. CT imaging combined with 7-AABs can preliminarily differentiate SCC. As is well known, the prognosis of SCC is poor and requires the attention of clinical doctors. The positive rate of 7-AABs increased with the adenocarcinoma diameter. The positive expression rate of adenocarcinoma with a poor prognosis was higher than the other pathologic types. Taken together, the positive expression rate of 7-AABs was affected by clinical features, pathological types, or clinical stages, especially in early-stage lung cancer. Furthermore, the sensitivity of the 7-AABs was low, thus 7-AABs, as early screening indicators of lung cancer, warrant further study. In contrast, when combining 7-AAbs with tumor markers for analysis, the sensitivity increased, but the specificity decreased, thus increasing the false-positive and positive expression rates of the combination, which were also affected by clinical features, pathologic type, and tumor stages. As a result, the effectiveness of AABs as an indicator of lung cancer screening was reduced. In addition, the positive expression rate of AABs and the combination of AABs and tumor markers were high in patients with SCLC, and there was no statistical difference between limited- and extensive-stage lung cancer, which suggested that AABs or the combination of AABs and tumor markers may have distinguishing efficiency in early-stage SCLC. These can provide a warning to patients with SCLC.
The limitation of the current study was the focus on the analysis of 7-AABs and the combination of 7-AABs and tumor markers, due to partial missing data related to tumor markers of retrospective research. There was no statistical difference in the analysis of adenocarcinoma subgroups because of the small number of adenocarcinoma patients with detailed pathologic results. Because the data were collected after a comprehensive clinical evaluation and bias existed, the effectiveness of screening could not be confirmed. Given the limitations of combining seven lung cancer antibodies with tumor markers in early screening of lung cancer, further RCT studies may be combined with intelligent imaging AI technology to improve the sensitivity and specificity of lung cancer screening.
In conclusion, the 7-AABs, when combined with tumor markers, contributed to the diagnosis of lung cancer; however, the efficacy of the 7-AABs combined with tumor markers in the screening and early detection of lung cancer warrants further investigation.
Footnotes
Abbreviations
Acknowledgments
We would like to thank all the subjects and family members who gave their consent to present the data in this study, as well as the investigators and researchers at all relevant hospitals and study sites. Furthermore, the abstract of this manuscript has been presented in conference proceedings at the Chinese Academic Conference on Tumor Biomarker 2021.
Author Contributions
F. R: Data curation, Formal Analysis, Investigation, Methodology, Writing – original draft. F. C: Formal Analysis, Methodology, Writing – original draft, Writing – review & editing. X. X and H. N: Investigation, Methodology, Software, Writing – original draft. T. L, D. R, Z. S and G. C: Data curation, Supervision, Validation, Writing – review & editing. J. C and S. X: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Writing – review & editing, Project administration.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was conducted in accordance with the Helsinki declaration and with approval from the Ethics Committee of Tianjin Medical University General Hospital. (IRB2024-YX-045-01)All data comes from the clinical database, and patients who enter the database have signed the relevant informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was funded by the National Natural Science Foundation of China (82172776), Tianjin Science and Technology Plan Project (19ZXDBSY00060), Tianjin Medical University General Hospital Incubation Fund (ZYYFY2018019), Natural Science Foundation of Tianjin (NO.20JCYBJC01350).and Tianjin Key Medical Discipline (Specialty) Construction Project.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
