Abstract
Objective
This study aims to evaluate the diagnostic value of human serum cysteine protease inhibitors (cystatin 4 [CST4]) in colorectal cancer (CRC) patients.
Methods
A total of 291 patients who were admitted to Zhuzhou Central Hospital for colonoscopy from January 2020 to December 2021 and met the inclusion criteria were selected. Serum samples of the patients were collected, and CST4 was detected by double-antibody sandwich enzyme-linked immunosorbent assay. Simultaneously, CEA and CA19-9 were detected, and the patients were divided into the CRC group, benign lesion group, and healthy control group. An attempt was made to construct a CRC prediction model including CST4 and draw a subject working characteristic curve as a diagnostic threshold for CRC prediction, and evaluate the diagnostic efficacy of the above indicators. At the same time, the expression analysis of CST4, CEA, and CA19-9 was verified by combining the data of CRC in the Tumor Genome Atlas (TCGA).
Results
In this study, the levels of serum CST4, CEA, and CA19-9 in the CRC group were higher than those in the colorectal benign lesion group and healthy control group, with statistical significance (
Conclusion
CST4 is a novel and improved diagnostic marker for CRC. The combined model based on CST4 has a certain potential value in terms of predicting the occurrence of intestinal cancer.
Introduction
Colorectal cancer(CRC) is the most common cancer-related cause of death in the world, mainly due to its high rate of metastasis and mortality. 1 Therefore, early detection of CRC is crucial for the implementation of curative surgery and the extension of patients’ survival. 2 China is a region with a high incidence of CRC, and the 5-year survival rate of CRC is significantly lower than that of Europe, the United States, Japan, Korea, and other countries. The main reason is that CRC is generally detected late in China, and most patients go to the hospital for treatment of middle and advanced tumors, and most patients have lost the best opportunity for treatment, while the treatment effect of middle and advanced colon cancer has not improved significantly in the past 50 years. Studies have shown that CRC has a clear “trilogy” of “precancerous lesions—early cancer—advanced cancer,” which makes the early prevention and treatment of CRC possible. Among various prevention and treatment strategies, digestive endoscopy is undoubtedly the core technology for the diagnosis and treatment of CRC, and the detection of tumor markers is an important screening and auxiliary diagnostic means. At present, common colorectal tumor markers in hospitals, such as CEA and CA19-9, are not specific antigens of CRC, and the sensitivity and specificity of detection are very low, so it is urgent to detect tumor markers with higher sensitivity and specificity.
Cysteine protease inhibitors (cystatins [CSTs]) were first isolated from egg white using affinity chromatography by Anastasi et al. 3 Studies have found that CSTs play an important role in tumor growth, angiogenesis, invasion, and metastasis and have the potential to be used as a marker for tumor diagnosis and prognosis evaluation.4–6 Serum cysteine protease inhibitors (CST4) are a member of the cysteine protease inhibitor superfamily, with a molecular weight of about 15 kDa, and have 2 special disulfide bonds at the C-terminal. 7 According to Akiba et al, 8 CST4 regulates cysteine protease activity by specifically combining with cysteine protease and it prevents hydrolysis of the extracellular matrix. Located in the cytoplasm, CST4 has the required characteristics of a blood biomarker (low molecular weight, secreted in blood, etc). 9 Previous studies have revealed that CST4 is significantly up-regulated in gastric cancer tissues, and its overexpression significantly promotes the proliferation, migration, and invasion of gastric cancer cells. 9 In addition, studies have confirmed that the overexpression of CST4 in CRC tissues and cell lines has high specificity and sensitivity and may become a new blood marker for gastric and CRC. 10 However, there are few in-depth studies on the diagnostic value of CST4 in CRC. Consequently, this study aims to explore the value of CST4 detection in the diagnosis of CRC and to provide new ideas and methods for its early diagnosis, treatment, and prevention.
Materials and Methods
Research Object
This performed a retrospective study. A total of 291 patients met the inclusion criteria in Zhuzhou Central Hospital for colonoscopy between January 2020 and December 2021. Their sera were collected and tested for CST4 using a double-antibody sandwich enzyme-linked immunosorbent assay (ELISA). CEA and CA19-9 were detected simultaneously. According to colonoscopy and pathological diagnosis results, 101 patients were identified as part of the CRC group, including 53 males and 48 females. In addition, there were 156 cases with benign colorectal lesions diagnosed by endoscopy and pathology, including 81 males and 75 females. There were 34 cases in the healthy control group, including 17 males and 17 females. The statistical analysis showed no significant difference in age and gender between the 2 groups (
Ethics Statement
This study was approved by the Institutional Research Ethics Board of Zhuzhou Central Hospital (reference number 201908069). The approval date was 12 September 2019.
Inclusion, Exclusion, and Rejection Criteria of Samples
Inclusion Criteria
Over 18 years of age; samples of CRC diagnosed by endoscopy and pathology; endoscopy and pathological diagnosis showed benign intestinal lesions; patients who gave consent to the trial.
Exclusion Criteria
Patients who are not suitable for colonoscopy; patients with acute infection or other tumor-related complications such as perforation, obstruction, and bleeding; patients with postoperative or chemotherapy of CRC; patients who were or are now accompanied by primary malignant tumors in other parts; patients who have undergone major organ transplantation; patients who are taking aspirin, clopidogrel, warfarin, and other anticoagulants, or have coagulation dysfunction; patients with severe heart, liver, and kidney dysfunction; pregnant or lactating women.
Rejection Criteria
Patients with poor sample quality (insufficient blood volume, fatty blood, hemolysis or expired, etc).
Research Methods
The reporting of this study conforms to STARD guidelines. 11 Two tubes of venous blood were collected from all subjects. One tube of venous blood was left at room temperature for 30 min, centrifuged at 3000 r/min for 10 min, and the serum was separated and stored in a −80 °C refrigerator for CST4 detection. Another tube of venous blood was used to detect CEA and CA19-9, and the detection was completed within 3 h of serum separation. CEA and CA19-9 were determined by an electrochemiluminescence immune analyzer and its supporting kit, and all operating steps were carried out in strict accordance with the operating specifications. Reference values of various indicators in our hospital: CEA ≤ 5.0 ng/mL and CA19-9 ≤ 37 U/mL. CST4 was detected by double-antibody sandwich ELISA, and the reagent was purchased from Shanghai Liangrun Biomedical Technology Co., Ltd.
Statistical Analysis
SPSS 25.0 statistical software was used for statistical analysis. The measurement data conformed to the normal distribution and were expressed as mean ± standard deviation. The nonnormal distribution data were expressed as median and interquartile spacing. The counting data were expressed by the rate or component ratio. Kruskal–Wallis test was used for data that did not conform to an analysis of variance. Mann–Whitney
Results
Comparison of CST4, CA19-9, and CEA Test Results in Each Group
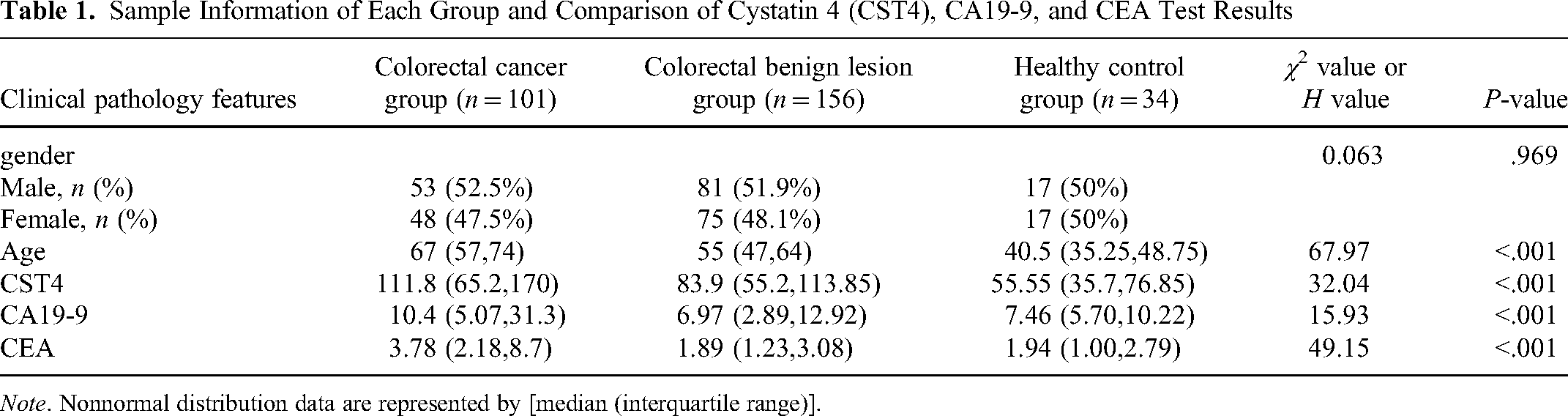
CST4, CA19-9, and CEA expressions were compared in the CRC group, benign lesion group, and healthy control group. The results showed that the level of CST4 in the CRC group was significantly higher than in the benign lesion group and healthy control group (

Comparison of CST4, CA19-9, and CEA test results in each group.
Sample Information of Each Group and Comparison of Cystatin 4 (CST4), CA19-9, and CEA Test Results
Comparison of serum CST4, CA19-9, and CEA in the Diagnosis of CRC
The receiver operating characteristic (ROC) curve is a technique for visualizing and providing a cutoff-independent method for the evaluation of diagnostic tests. ROC analysis can be used as a statistical method to evaluate the performance of diagnostic tests. In the present study, we used ROC analysis to visually and statistically assess the value of CST4, CA19-9, and CEA as tools for the diagnosis of predicting CRC. As described in Figure 2, the area under the ROC curve (AUC) of CST4 is 0.7739, obviously larger than that of CA19-9 and CEA. The results showed CST4 as a better predictor of CRC.
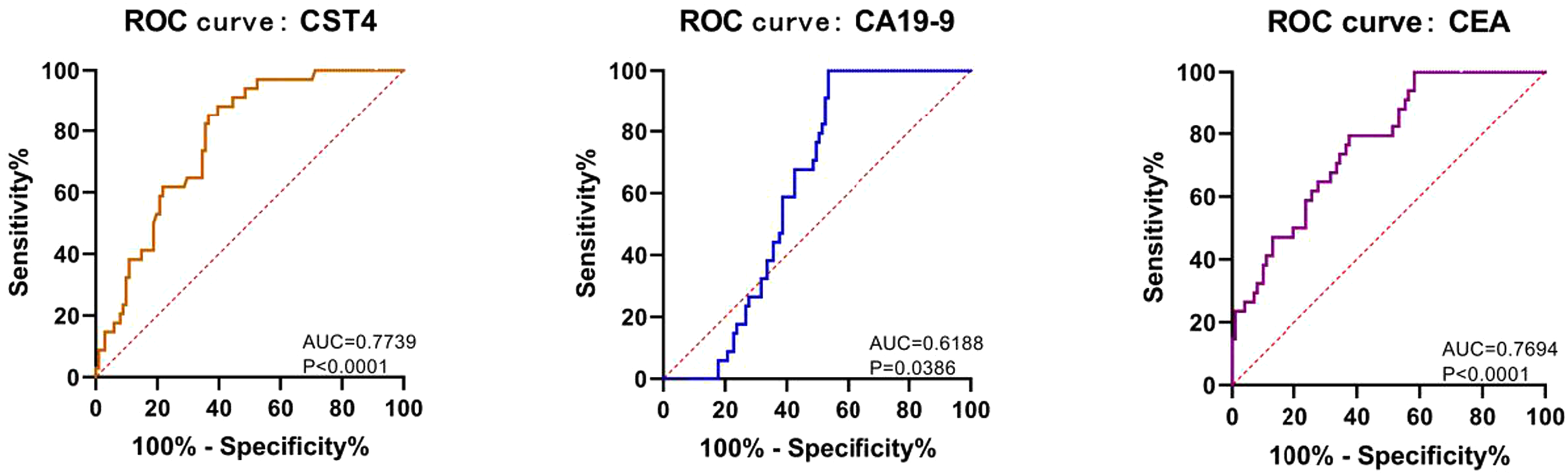
Comparison of diagnostic value of serum CST4, CA19-9, and CEA in CRC.
Expression Analysis of CST4, CEA, and CA19-9 in CRC Data in Tumor Genome Atlas Database
The CRC data were downloaded from the Tumor Genome Atlas (TCGA) database, and CST4, CEA, and CA19-9 expression differences in tumor patients and normal samples were analyzed and compared. As depicted in Figure 3A and C, the expression of CST4 in CRC patients was significantly higher than in normal samples (

Expression analysis of CST4 and CEA in CRC data in TCGA database. (A) Comparison of CST4 expression between rectal cancer patients and normal rectal samples in TCGA database (166 and 10 samples in tumor and normal groups, respectively, ***
Construction of CRC Prediction Model
According to the experimental data of this study and the above analysis, the expressions of CST4, CA19-9, and CEA in serum and patients’ age are related to CRC. In this regard, the 4 factors were further verified by LASSO analysis. The factors screened by LASSO analysis were used to make a nomogram (Figure 4C) as a “joint model” for predicting the occurrence of CRC. Model formula:





Construction of CRC prediction model. LASSO analysis was used to screen the factors related to the occurrence of CRC. (A) The vertical coordinate is the variable coefficient in LASSO analysis, the upper abscissa represents the number of variables, and the lower abscissa represents the parameter lambda. The figure shows that with the increase in punishment, the coefficient of each variable decreases. (B) The vertical coordinate is the LASSO analysis MSE, the upper abscissa represents the number of variables, and the lower abscissa represents the parameter lambda. The figure shows that MSE changes as lambda changes. (C) A line diagram of the joint model to predict the incidence of CRC (in the figure, PR (sample nature) is the predicted incidence of CRC). (D) ROC curve of joint model.
Simultaneously, the ROC curve of the combined model was analyzed (Figure 4D), and the area under the curve was 0.7851, larger than that under the separate ROC curve in result 2. This shows that the model made by the joint model has good prediction ability.
Discussion
The incidence and fatality rate of CRC remain high all the time, which is a serious threat to national health. Early detection and treatment are the most effective methods for the prevention and treatment of CRC. Digestive endoscopy is one of the main clinical examination methods for the diagnosis of CRC at present, but its invasive examination method can cause great pain to patients and has limitations in the extensive screening of the population. The detection of serum tumor markers is widely used in auxiliary diagnosis, disease monitoring, and prognosis evaluation because of its convenience, fast, and relatively noninvasive. However, the commonly used indicators of CRC, such as CA19-9 and CEA, have low sensitivity and specificity. Therefore, more sensitive and specific tumor marker detection items are urgently needed clinically.
The research on CST4 provides a new way to improve this situation. According to Akiba et al, 8 CST4 regulates cysteine protease activity and prevents extracellular matrix hydrolysis by specifically binding to cysteine protease. Furthermore, CST4 is located in the cytoplasm and has the characteristics required by blood biomarkers (low molecular weight, secreted in the blood, etc), making it a potential biomarker for gastrointestinal cancer. Previous research has shown that CST4 has high accuracy and specificity in gastric and CRC. 9
In this study, a human serum cysteine protease inhibitors ELISA kit was used to compare the difference in serum CST4 expression between the colorectal and non-CRC groups. The results showed that the CST4 level in the CRC group was significantly higher than that in the non-CRC group (
Since 1965, tumor marker CEA has been considered a glycoprotein, which can be detected in blood and adenocarcinoma cells.10,12,13 CEA is an intracellular adhesion molecule with a half-life of 1–3 days. It is produced in fetal intestinal tissue and epithelial tumor cells and contributes to angiogenesis.10,14 The serum CEA levels in malignant tumors such as colorectal, breast, gastric, lung, ovarian, and pancreatic cancer increased.10,12 Nevertheless, elevated CEA can also be observed in many nonmalignant diseases, such as smoking, alcoholism, chronic inflammatory bowel disease, diverticulitis, pancreatitis, and liver disease.15–17 CA19-9, another tumor marker discovered in 1979, is now used more frequently for the early detection of pancreatic cancer. CA19-9 is a monoclonal antibody and a ligand of E-selectin. 18 However, CA19-9 may be increased in both malignant and benign lesions, indicating that it may lack specificity.
In addition to CST4 results, we collected CEA and CA19-9 data from each group. A comprehensive analysis of CST4, CEA, and CA19-9 in CRC showed that CST4 had the highest sensitivity and specificity, and its prediction accuracy was 74.4%. Previous research has shown that CEA has a sensitivity range of 65%–74% in patients with CRC, whereas CA19-9 has a range of 26%–48%.14,19,20 This is similar to our results. However, after CEA and CA19-9 were evaluated based on patient characteristics, they were not recommended for early diagnosis and screening of CRC in a study. 21 This prompted us to conduct this study. Simultaneously, our results showed that the accuracy of CST4 as a predictor of CRC was higher than that of CEA and CA19-9, which also suggested that CST4 as a new blood biomarker had great potential in the clinical diagnosis of CRC. Furthermore, when the serum test results of patients collected by our research group were compared to the colon cancer data in the TCGA database, they were consistent with their results. Simultaneously, this study successfully constructed a combined model based on CST4 and discovered that the AUC of CRC was 0.7851, larger than the AUC of any single factor.
Conclusion
Based on the findings, CST4 test results are better indicators for diagnosing CRC, and serum CST4 may be a new marker for the early diagnosis of CRC. Furthermore, the CST4-based combined model has a certain potential value in terms of predicting the occurrence of intestinal cancer. However, this study still has some defects, such as a small sample size and no histological verification. Further study on the value of CST4 in the diagnosis of CRC needs to be done with multicenter, large sample size, and histological validation.
In the follow-up, our research team will study the pathogenesis of CST4 in CRC and develop a better CRC diagnosis and prediction model in conjunction with other clinical predictors.
Footnotes
Abbreviations
Author Contributions
Shuang Deng wrote the manuscript. Meiyuan Huang and Zhenyu Yang participated in the search strategy development. Taoli Wang and Yanqiong Zhan collected experimental data. Dongliang Chen and Wenxing Le prepare figures and tables. Shuang Deng and Meiyuan Huang double-checked the data. Jianqiang Ren reviewed the article when it was revised. All authors contributed to critical manuscript revision.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Innovative City Construction Project of Zhuzhou (Zhuzhou Branch Office [2022] No. 1).
