Abstract
Purpose
Hippocampal-avoidance whole-brain radiotherapy (HA-WBRT) planning can present challenges. This study examines the influence of head tilt angles on the dosimetric characteristics of target and organs at risk (OARs), aiming to identify the optimal tilt angle that yields optimal dosimetric outcomes using tomotherapy (TOMO).
Methods
Eight patients diagnosed with brain metastases underwent CT scans at five tilt angles: [0°, 10°), [10°, 20°), [20°, 30°), [30°, 40°), and [40°, 45°]. Treatment plans were generated using TOMO and volumetric modulated arc therapy (VMAT). Dosimetric parameters including conformity index (CI), homogeneity index (HI), D2cc, D98%, and Dmean of PTV, as well as Dmax, and Dmean of OARs were analyzed. Furthermore, a comparison was made between the dosimetric parameters of TOMO and VMAT plans. Finally, delivery efficiency of TOMO plans were assessed.
Results
For the PTV, [40°, 45°] tilt angle demonstrated significantly better conformity, homogeneity, lower D2cc, and lower Dmean for the PTV. Regarding the OARs, the [40°, 45°] head tilt angle demonstrated significantly lower Dmax and Dmean in hippocampus, eyes, optic chiasm, and optic nerves. The [40°, 45°] tilt angle also showed significantly lower Dmax for brainstem and cochleas, as well as a lower Dmean for lens. In the [40°,45°] tilt angle for HA-WBRT, TOMO showed superior performance over VMAT for the PTV. TOMO achieved lower Dmax for brainstem, cochleas, optic nerves, and optic chiasm, as well as a lower Dmean for hippocampus. Furthermore, a significant correlation was found between delivery time and the PTV projection length in the sagittal plane.
Conclusion
The TOMO plan utilizing a tilt angle range of [40°, 45°] demonstrated superior PTV conformity and uniformity, along with enhanced OARs sparing. Furthermore, it exhibited a dosimetric advantage over VMAT for PTV and most OARs at the same angle range.
Introduction
Whole-brain radiotherapy (WBRT) is a common treatment for brain metastases and as a prophylactic measure.1,2 However, WBRT can damage the hippocampus, a critical structure for memory and cognitive function.3–9 The RTOG 0933 phase II clinical trial demonstrated that hippocampal-avoidance WBRT (HA-WBRT) can reduce the risk of neurocognitive decline. 10
Advancements in radiotherapy technology have led to the adoption of various modalities for HA-WBRT, including intensity-modulated radiation therapy (IMRT), 11 volumetric-modulated arc therapy (VMAT),12,13 and tomotherapy (TOMO). 14 These techniques aim to minimize radiation dose to the hippocampus while maintaining effective treatment of the whole brain.
To further minimize radiation dose to sensitive structures like the hippocampus and organs at risk (OARs), researchers have introduced the “head tilt” technique in radiation therapy procedures. This innovative approach involves tilting the patient's head at a specific angle during treatment.
LINAC-based studies on “head tilt” for HA-WBRT have demonstrated promising results.15–18 Moon et al 15 conducted a comparative analysis of dosimetric characteristics, exploring the differences between a 30° head tilt and no head tilt, employing both IMRT and VMAT techniques. Oh et al 16 compared the dosimetric characteristics of a 40° head tilt with no head tilt, utilizing the VMAT technique. Their investigation was limited to a single, specific tilt angle, and the analysis focused on the dose distributions within the target, hippocampus, cochleas, eyes, lens, and parotid glands. Other studies used virtual CT to simulate different head tilt angles. Siglin et al 17 utilized MIM software to artificially generate CT scans by rotating the non-tilt CT images 30° forward along the pitch axis. Lin et al 18 rotated the couch to simulate a range of virtual head tilt angles, spanning from 0° to 40°. However, this approach could introduce inaccuracies in the doses received by OARs, stemming from variations in head tilt angles and alterations in CT cross-sectional scanning.
Several researchers have reported advantages of TOMO compared to IMRT and VMAT for HA-WBRT.19–21 These advantages include superior dose conformity, improved dose homogeneity throughout the target area, and enhanced preservation of the hippocampus. Some scholars combined the advantages of both TOMO and head tilt techniques for HA-WBRT, achieving promising outcomes. Miura et al 22 explored the utilization of a head-tilting base plate to generate specific tilt angle for each patient in HA-WBRT using TOMO, demonstrating its potential for dose optimization. Sengul et al 23 investigated the influence of varying board angles on PTV and OARs during HA-WBRT with TOMO using a virtual phantom.
This study breaks new ground in several key areas, offering a comprehensive and novel approach to optimizing HA-WBRT using TOMO and head tilt techniques. Unlike previous research that relied on virtual simulations or single tilt angles, we utilized actual CT scans at multiple angles for each patient, providing more accurate and realistic dosimetric assessments. Our study was the first to systematically investigate the use of different tilt angles for each patient to determine the optimal head angles for HA-WBRT. We conducted a unique comparison between TOMO and VMAT at different head tilt angles, specifically evaluating TOMO's potential advantages in dose distribution and OAR sparing. Furthermore, this research goes beyond dosimetric analysis to investigate factors affecting the duration of TOMO treatment, providing valuable insights into the practical aspects of treatment delivery. Our primary objective was to identify the optimal head tilt angle in HA-WBRT to achieve optimized clinical outcomes. To this end, we conducted a study on a cohort of patients who received head-tilted HA-WBRT, with each patient scanned at five different angles. TOMO treatment plans were designed to compare the dosimetric outcomes, and the optimal head tilt angle was identified based on these comparisons. VMAT treatment plans were also generated to determine which technique is more effective for delivering HA-WBRT at the optimal head tilt angle. We also evaluated the delivery efficiency of TOMO and performed correlation analyses. These novel aspects collectively contribute to a more comprehensive understanding of HA-WBRT optimization using TOMO and head tilt techniques, potentially leading to improved treatment outcomes for patients undergoing WBRT.
Material and Method
Patient Selection and CT Simulation
This prospective, single-center clinical study was conducted with the informed consent of all participants. Prior to enrollment, each participant received comprehensive information about the research, including its background, objectives, design, voluntary participation, confidentiality, and contact information for inquiries. This information was presented in a written consent form, which participants signed after understanding and agreeing to participate. The present study consisted of a cohort of eight male patients diagnosed with brain metastases that originated from primary tumors located in the bronchi and lungs. The median age of the study population was 57.5 years, with an age range of 35 to 78 years. This study was conducted in accordance with ethical principles and received approval from the Institutional Review Board (IRB) of our cancer hospital (Ethical approval number: JS2023-9-1). The inclusion criteria for the study are listed as follows: (a) Age 18 years or older, regardless of gender; (b) Histologically confirmed primary solid malignant neoplasm (any type); (c) Karnofsky Performance Status (KPS) of 70 or higher; (d) Patients with prior surgical treatment or SBRT/SRS radiotherapy for brain metastases are eligible. Individuals meeting any of the following criteria will be excluded from this study: (a) Patients with radiologically confirmed leptomeningeal metastases; (b) Patients with prior WBRT; (c) Lesions within a 5 mm expansion zone surrounding the hippocampus; (d) Patients with radiologically confirmed obstructive hydrocephalus or significant structural brain deformation secondary to prior surgery or other benign or malignant brain diseases; (e) Patients with severe cervical spondylotic myelopathy or other cervical spine disorders precluding treatment at the necessary head frame angle; (f) Patients with significant underlying medical comorbidities (including hypertension, diabetes, and cardiovascular disease) or those in an acute phase of a disease who are not medically suitable for radiotherapy. Table 1 presents the clinical information of the patients enrolled in the study.
Patient Clinical Information

Patient Clinical Information

During computed tomography (CT) simulation, all patients were positioned in a head-first-supine position with arms akimbo to ensure optimal immobilization and comfort. The Klarity Optek System (R630-SHCF) was employed to provide stability and comfort for the upper body of the patients. A thermoplastic mask was used to immobilize patients’ heads and necks, thereby minimizing potential movement artifacts. CT images were acquired using a GE CT simulator (Discovery 590 RT, GE Healthcare, Waukesha, WI, USA), with the scan range encompassing the region from the vertex of the skull to the sternoclavicular joint. The images were reconstructed with a slice thickness of 2.5 mm to facilitate accurate tumor localization and treatment planning.
CT simulation was performed for five patients at incremental head tilt angles of 0°, 10°, 20°, 30°, and 40°, while the remaining three patients underwent CT simulation at head tilt angles of 5°, 15°, 25°, 35°, and 45°. The head tilt angles were divided into 10-degree intervals. All patients were stratified into five distinct groups based on their head tilt angles, specifically: [0°, 10°), [10°, 20°), [20°, 30°), [30°, 40°), and [40°, 45°]. Figure 1 presents a representative example of CT simulated images from each of the five groups, illustrating the varying head tilt angles.

The representative CT simulated images depicted in panels (a) through (e) correspond to the following head tilt angle groups, respectively: (a) 5°, (b) 15°, (c) 25°, (d) 35°, and (e) 45°.
According to the RTOG 0933 guideline, 10 the whole brain radiotherapy in this study was prophylactic cranial irradiation of the whole brain, without any defined GTV. The CTV was defined as the whole brain parenchyma. The PTV was defined as the CTV, excluding the hippocampal avoidance regions. The hippocampal avoidance regions were generated by three-dimensionally expanding the hippocampal contours by 5 mm. This protocol provides explicit criteria and guidelines for the delineation of the hippocampal avoidance region, which is recognized as a critical structure to spare to minimize radiation-induced neurocognitive dysfunction in patients undergoing brain radiation therapy.
AccuContour™ software (Manteia Medical Technologies Co. Ltd, Xiamen, China) was employed for the automated contouring of OARs. This advanced software leverages a deep learning-based algorithm to enable accurate and efficient delineation of anatomical structures. The model embedded within AccuContour™ has been clinically commissioned and validated for use in our center, ensuring reliability and consistency in contouring. Its automated and unattended control features streamline the contouring workflow, significantly reducing physician workload. In this study, the software was used to contour various OARs, including the hippocampus, brainstem, eyes, lens, optic chiasm, optic nerves, and pituitary gland. The entire contouring process took approximately one minute per image set. To achieve accurate hippocampus contouring, CT-MRI fusion was employed. Axial T1-weighted MRI sequences, considered optimal for delineating the hippocampus, 24 were imported into Eclipse™ using the “auto matching” mode for image registration. The entire brain was included in the region of interest for image fusion. Clinical oncologists reviewed the hippocampus contouring based on this fused image, adhering to RTOG 0933 guideline, and made any necessary corrections. All the OAR delineations were reviewed and verified by senior oncologists to ensure their accuracy and validity.
Treatment Planning
All acquired CT images and radiation therapy (RT) structures were imported into the TOMO planning station (Precision, version 5.1.1.6, Accuray, Sunnyvale, CA). The TOMO machine (Accuray, Sunnyvale, CA) was selected as the delivery platform for the treatment plan. During the TOMO plan optimization process, the planning parameters were established and defined as follows: a prescribed dose of 30 Gy, delivered in 3.0 Gy per fraction, was specified; a fine dose calculation grid was utilized; a field width of 2.512 cm was set, with jaws positioned at (1,-1) in fixed jaw mode; a modulation factor of 2.7 was employed; a pitch of 0.287 was selected; a total of 200 interactions were performed to ensure optimal plan convergence. PTV dose coverage was normalized to 92% for all TOMO plans. In clinical practice, PTV dose coverage typically ranges from 92% to 95%, with variations depending on individual patient anatomy.6,10 To maintain consistent and meaningful comparisons across all treatment plans in this study, target dose coverage was normalized to 92% of the prescribed dose. Both the dose and structure files of each treatment plan were imported into a house-developed software and precisely scaled to achieve the target dose coverage of 92%. This process was carried out using Matlab software (R2021a, MathWorks, Natick, MA).
VMAT planning was performed using Pinnacle 3 version 16.2.1 (Philips Medical Systems, Fitchburg, WI). In all VMAT plans generated in Pinnacle, 3 the following beam parameters were consistently applied: an Elekta Infinity machine (Elekta Instrument AB, Stockholm, Sweden) with a photon modality and a 6MV flattening filter-free (FFF) energy were employed; the beam type utilized was dynamic arc, with a dose rate of 1400 MU/min; two arcs were used, with one arc commencing at 179.9° and terminating at 180.1°, while the other arc oriented in the opposite direction; the collimator angle was set to 5°, and the couch angle was maintained at 0°; the final gantry spacing was set to 3°, and a leaf motion constraint of 1 cm/deg was imposed to ensure the optimal plan quality; the prescribed dose for the VMAT plans was identical to that of the TOMO plans, with a goal of delivering 30Gy in 3Gy per fraction. Furthermore, PTV dose coverage was normalized to 92% for all VMAT plans as well. Subsequently, a comprehensive comparison of dosimetric parameters of the PTV and OARs was performed between the TOMO and VMAT plans to evaluate their relative efficacy.
Planning Evaluation
A thorough evaluation was conducted to assess the PTV using a range of metrics, including the conformity index (CI), homogeneity index (HI), D2cc, D98%, and mean dose (Dmean). Additionally, a detailed analysis of the maximum dose (Dmax) and mean dose (Dmean) was performed specifically for the OARs.
The CI was calculated as the ratio of the volume of the prescription isodose line (VPIL) to the volume of PTV (VPTV), expressed mathematically as
25
:
The HI was determined by dividing the dose received by 5% of the PTV (D5%) by the dose received by 95% of the PTV (D95%), calculated as
26
:
The D2cc metric represents the maximum dose received by a 2 cubic centimeter (cc) volume within the PTV. D98% metric denotes the minimum dose received by at least 98% of the PTV, highlighting the dose coverage required for the majority of the target volume. The mean dose (Dmean) represents the average dose received by the PTV.
To investigate the differences among the various plans, a paired, two-tailed non-parametric Wilcoxon signed-rank test was utilized to compare the paired data. A p-value of less than 0.05 was considered statistically significant, indicating a significant difference between the plans. Furthermore, an analysis of variance (ANOVA) was performed to examine the differences between the groups, with a significance level (two-tailed) of less than 0.05 considered indicative of statistical significance. MATLAB software (R2021a, MathWorks, Natick, MA) was used to perform the statistical analysis.
Result
Comparison of Dosimetric Parameters for TOMO Plans in Different Tilt Angles
PTV
Figure 2 presents a comparative analysis of the CI, HI, D2cc, D98%, and Dmean of PTV across the five groups. Notably, group 5 demonstrated a significantly higher level of conformity compared to group 1 (p = 0.01) and group 2 (p = 0.02). Group 5 exhibited a significantly higher level of homogeneity compared to group 1 (p = 0.01) and group 2 (p = 0.02). The hot spot of group 5 was significantly lower than that of group 1 (p = 0.01). No significant differences were observed among the groups for target coverage D98%. The Dmean of group 5 was significantly lower than that of group 1 (p = 0.01), group 2 (p = 0.02), and group 3 (p = 0.02). In summary, group 5 exhibited a significantly higher level of conformity and homogeneity compared to group 1 and group 2. Additionally, group 5 had a significantly lower D2cc compared to group 1, and lower Dmean compared to group 1, group 2, and group 3.

The comparison of (a) PTV conformity index, (b) PTV homogeneity index, (c) PTV D2cc, (d) PTV D98%, and (e) PTV mean dose among five groups for TOMO. Note: Bold values indicate significance at P < 0.05.
With regard to OARs, the Dmax and Dmean of hippocampus, brainstem, cochleas, eyes, lens, optical nerves, optic chiasm, and pituitary were evaluated.
Notably, the Dmax values for the hippocampus in group 5 were significantly lower than group 1 (p = 0.04) (Figure 3). The Dmean values for the hippocampus in group 5 were significantly lower than group 1 (p = 0.01), group 2 (p = 0.01), and group 3 (p = 0.01). The hippocampus in group 5 received significantly lower maximum and mean doses compared to group 1, suggesting a potential reduction in radiation-induced toxicity.

Comparison of the Dmax and Dmean of hippocampus for TOMO plans in various angles ((a) the Dmax of hippocampus, (b) the Dmean of hippocampus). Note: Bold values indicate significance at P < 0.05.
A comparative analysis of the Dmax values of brainstem revealed that group 5 exhibited significantly higher values compared to group 4 (p = 0.02) (Table 2). In terms of the cochleas, group 5 exhibited significantly lower Dmax values compared to group 1 (p = 0.01) and group 2 (p = 0.04). Regarding the eyes, it was found that group 5 exhibited significantly lower Dmax and Dmean values compared to group 1 (p = 0.02). In the case of the lens, a statistical analysis revealed that group 5 exhibited significantly lower Dmean values compared to group 1 (p = 0.01) and group 2 (p = 0.01) . Regarding the optic chiasm, group 5 had significantly lower Dmax and Dmean values compared to group 1 (p = 0.01). In terms of optical nerves, group 5 demonstrated significantly reduced Dmax values when compared to group 1 (p = 0.02) and group 2 (p = 0.02). Additionally, group 5 exhibited notably lower Dmean values than group 1 (p = 0.02). No statistically significant differences were observed among the groups in terms of the Dmax values for the lens and pituitary gland. Similarly, there were no statistically significant differences identified in the Dmean values for the brainstem, cochleas, and pituitary gland.
Dosimetric Comparison of OARs for Different Tilt Angles in TOMO
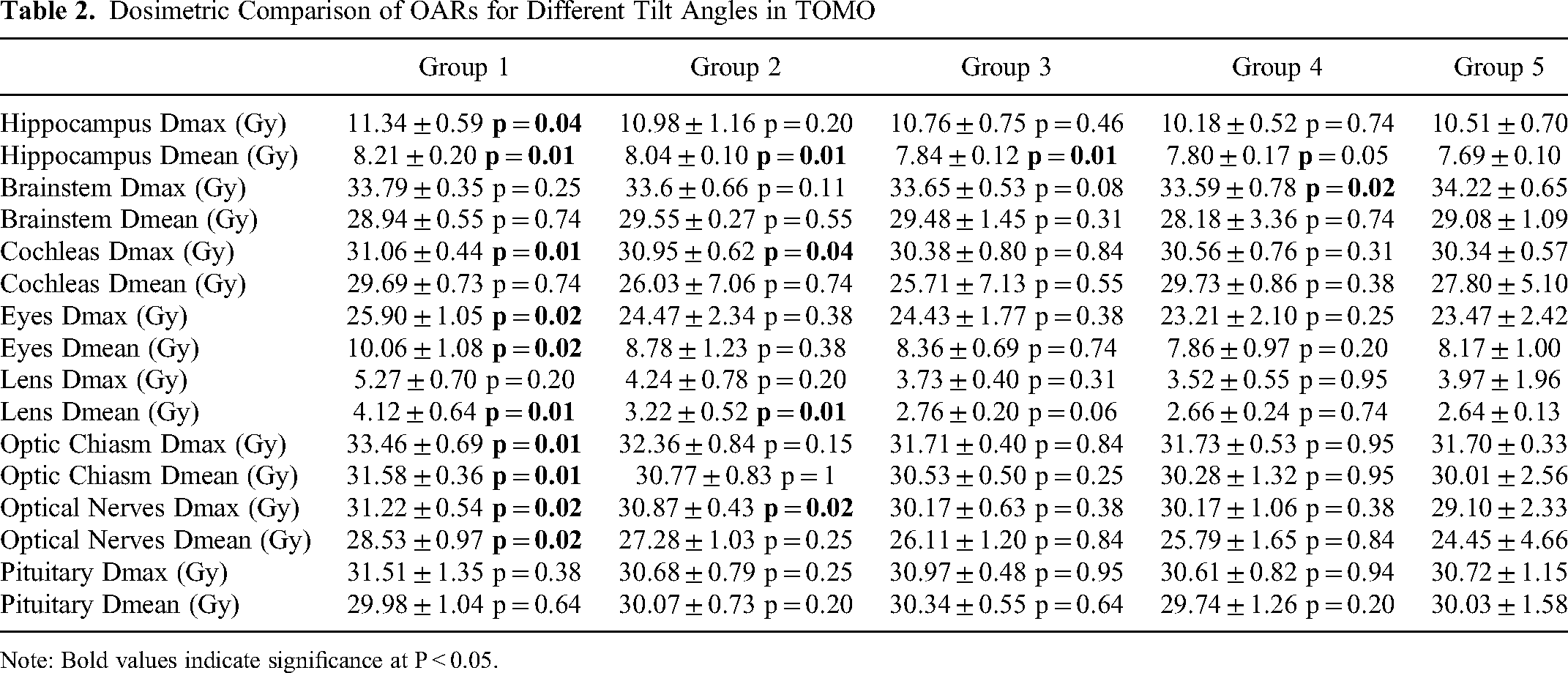
Note: Bold values indicate significance at P < 0.05.
Figure 4 presents a visual representation of the dose distribution in TOMO plans for different tilt angles. It provides clear evidence that an increase in the tilt angle results in a decrease in the size of the hot spots within the PTV. This observation suggests a potential correlation between the tilt angle and the distribution of high-dose regions. Through a comparative analysis of TOMO plans with different tilt angles, it was observed that group 5 exhibited notable dosimetric advantages in relation to both the PTV and OARs. These findings indicate that adopting the specific tilt angle associated with group 5 may result in improved dose distribution within the PTV while effectively minimizing radiation exposure to most of critical OARs.

An illustration showcasing the dose distribution in the sagittal view for different tilt angles. The tilt angles of (a-e) were 5°, 15°, 25°, 35°, and 45°, respectively. The red arrows indicate regions with high dose (hot spots), while the blue arrows indicate regions with low dose (cold spots).
Adopting the optimal head tilt angle as group 5, a comprehensive comparative analysis was undertaken to evaluate the treatment plans generated by VMAT technique against those derived from the TOMO modality.
Using the head tilt angle corresponding to group 5, the TOMO technique demonstrated superior performance compared to VMAT across all evaluated dosimetric parameters for the PTV. As illustrated in Figure 5, the CI, HI were significantly better than those of VMAT plans. Additionally, the D2cc, D98%, and Dmean of TOMO technique were significantly lower than those of VMAT plans. These findings indicate that TOMO technique provided superior dosimetric outcomes compared to VMAT plans in terms of the evaluated parameters at the head tilt angle corresponding to group 5, as evidenced by the statistical significance differences observed.
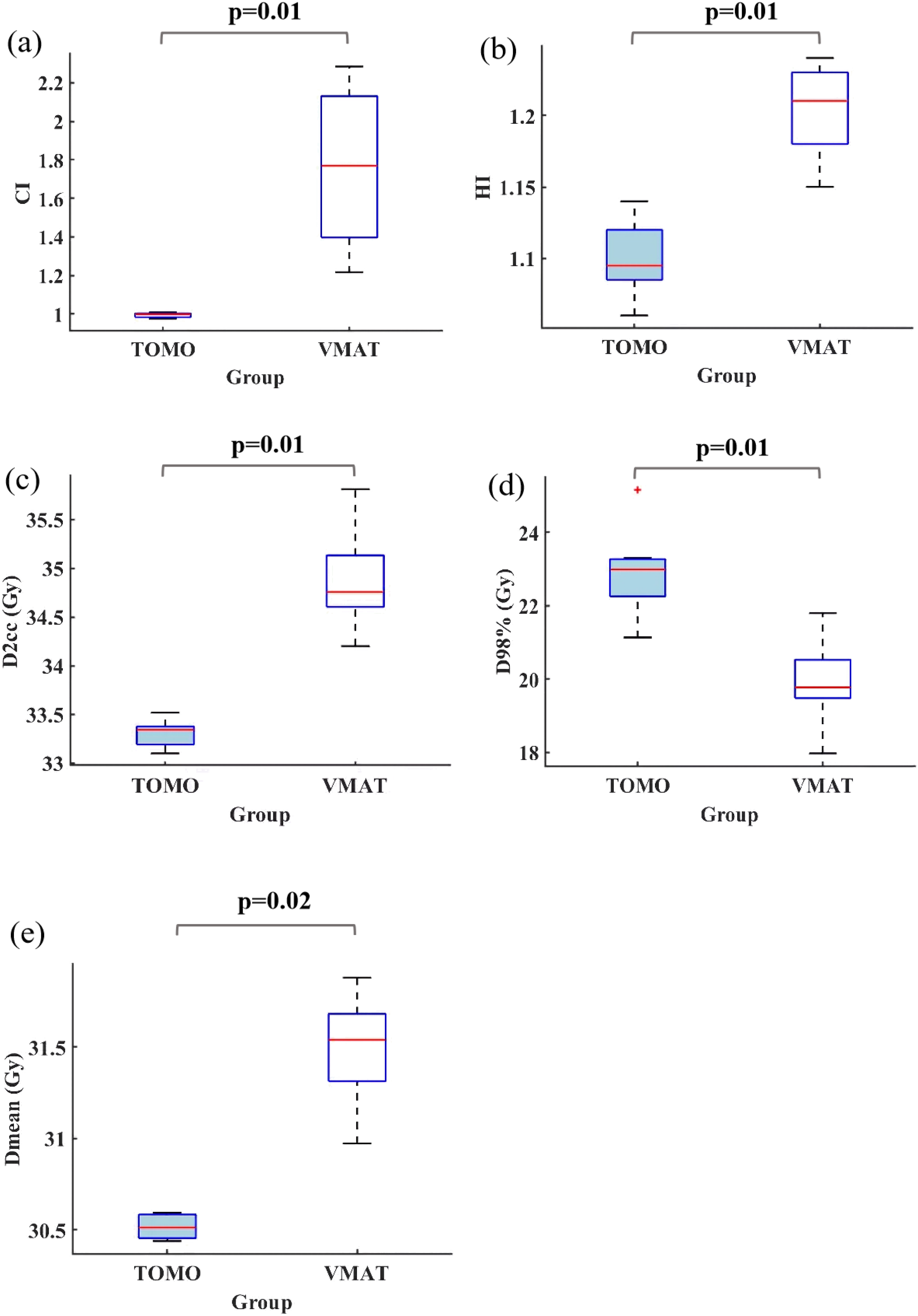
The comparison of (a) PTV conformity index, (b) PTV homogeneity index, (c) PTV D2cc, (d) PTV D98%, and (e) PTV Dmean for TOMO and VMAT plans. Note: Bold values indicate significance at P < 0.05.
In terms of OAR sparing for the treatment plans corresponding to group 5, the TOMO technique exhibited significantly lower Dmax values for the brainstem, cochleas, optic nerves, and optic chiasm (Table 3). Furthermore, TOMO plans exhibited a significantly lower Dmean values to the hippocampus compared to VMAT plans. However, TOMO plans had significantly higher Dmax values for the lens, as well as higher Dmean values for the eyes and lens compared to VMAT plans. No significant differences were observed for the Dmax values of the hippocampus, eyes, and pituitary gland, as well as the Dmean values of the brainstem, cochleas, optic nerves, optic chiasm, and pituitary gland.
The Comparison of the Dmax and Dmean for OARs Between TOMO and VMAT Plans at the Head Tilt Angle of Group 5

Note: Bold values indicate significance at P < 0.05.
Despite the higher Dmean values observed for the eyes and lens, as well as the higher Dmax value for the lens in TOMO plans compared to VMAT plans, these dosimetric values remained within the dose constraints set by clinical guidelines.
An evaluation of the delivery time for TOMO plans was conducted across different groups. The measured delivery time for groups 1 to 5 were reported as 9.25 ± 0.45 min, 8.99 ± 0.64 min, 8.60 ± 0.45 min, 8.76 ± 0.56 min, and 9.09 ± 0.58 min, respectively. Notably, group 5 exhibited a significantly longer delivery time compared to group 3 (p = 0.02).
In terms of the projection length of the PTV in the sagittal plane, the measured values for groups 1 to 5 (Figure 6) were 14.84 ± 0.46, 14.16 ± 0.44, 13.41 ± 0.33, 13.34 ± 0.40, and 13.94 ± 0.48, respectively. Group 5 demonstrated a significantly shorter PTV length compared to both group 1 (p = 0.02) and group 3 (p = 0.03).

Comparison of (a) delivery time, (b) PTV projection length in the sagittal plane, (c) hippocampus projection length in the sagittal plane, and (d) actual modulation factor among five groups. Note: Bold values indicate significance at P < 0.05.
Regarding the projection length of the hippocampus in the sagittal plane, the measurements in groups 1 to 5 were reported as 2.78 ± 0.41, 2.94 ± 0.22, 3.09 ± 0.57, 3.63 ± 0.48, and 3.38 ± 0.55, respectively. Group 5 exhibited a significantly shorter hippocampus length compared to group 1 (p = 0.02). Regarding the actual modulation factor, the values for groups 1 to 5 were 2.34 ± 0.02, 2.35 ± 0.01, 2.36 ± 0.04, 2.37 ± 0.02, and 2.41 ± 0.03, respectively. Group 5 had a significantly larger actual modulation factor compared to group 1 (p = 0.02), group 2 (p = 0.01), group 3 (p = 0.01), and group 4 (p = 0.02).
We further investigated the correlated factors influencing the delivery time of TOMO plans. Three treatment planning parameters were considered, including the PTV projection length in the sagittal plane, the hippocampus projection length in the sagittal plane, and the actual modulation factor. The results illustrated the correlation and statistical significance of these three factors in relation to delivery time (Figure 6). A significance level (two-tailed) of less than 0.05 was considered indicative of statistical relevance. Furthermore, a higher Pearson correlation coefficient suggests a stronger association between a particular factor and delivery time. A significant correlation was observed between delivery time and the PTV projection length in the sagittal plane (r = 0.53, p < 0.01). However, the analysis revealed no significant correlation between delivery time and the hippocampus projection length in the sagittal plane (r = -0.05, p = 0.75), as well as the actual modulation factor (r = -0.17, p = = 0.30).
A comparative analysis was conducted to evaluate the delivery time of TOMO and VMAT plans for group 5. The delivery time of TOMO and VMAT plans for group 5 exhibited significant differences, with mean values of 9.09 ± 0.58 min and 4.41 ± 0.76 min, respectively (p = 0.01). The results indicate that the delivery time of TOMO plans was considerably longer in comparison to their VMAT counterparts.
The evolution of radiotherapy technology has significantly advanced HA-WBRT, progressing from IMRT to VMAT and TOMO. Each advancement has improved dose distribution and sparing of critical structures, particularly the hippocampus. Building on this progress, our study investigated the dosimetric impact of various head tilt angles in HA-WBRT using TOMO. We identified the [40°, 45°] angle range as optimal, demonstrating superior PTV conformity and homogeneity, along with enhanced sparing of OARs compared to other angles. At this optimal angle, TOMO exhibited dosimetric advantages over VMAT, achieving lower doses to critical structures such as the brainstem, cochleas, optic nerves, and optic chiasm. This research represents a significant step forward in HA-WBRT techniques. By combining the benefits of TOMO with optimized head tilt angles, we've demonstrated the potential for improved treatment precision and reduced risk to critical brain structures. Additionally, our study revealed a significant correlation between treatment delivery time and PTV projection length in the sagittal plane, offering valuable insights for optimizing treatment efficiency. These findings contribute to the ongoing refinement of HA-WBRT techniques, potentially improving treatment outcomes and quality of life for patients with brain metastases.
The technological development route for HA-WBRT has seen significant advancements, progressing through several key innovations. This evolution began with the introduction of IMRT by Gondi (2010) 6 and Kendall (2018), 27 which effectively spared the hippocampus while delivering radiation to the whole brain. The next major advancement came with VMAT, pioneered by Lee (2015), 28 Rong (2015), 29 Kendall (2018) 27 and Deepsikha (2023), 30 which improved upon IMRT by achieving a more homogeneous dose distribution throughout the target volume and reducing the maximum point dose to the target. TOMO, introduced by Gondi (2010) 6 and further developed by Rong (2015), 29 emerged as a leading technique for HA-WBRT, delivering a lower dose to the hippocampus compared to IMRT while maintaining acceptable target coverage and homogeneity. Subsequent studies by Wang (2020), 20 Zhang (2020), 21 and Rong (2015) 29 demonstrated TOMO's superior sparing of critical anatomical structures, including the hippocampus and other OARs, compared to both IMRT and VMAT. These technological advancements have significantly enhanced the ability to deliver precise radiation therapy while minimizing damage to critical structures, marking a continuous improvement in the field of HA-WBRT and offering patients more effective treatment options with reduced risk of cognitive side effects.
Building upon these advancements, researchers have explored additional techniques to further enhance hippocampal sparing and overall treatment efficacy in HA-WBRT. One such advancement is the non-coplanar technique, which aims to improve target coverage and OAR sparing by utilizing radiation beams that are not all in the same plane. Non-coplanar IMRT (Sharma et al, 31 2021) and non-coplanar VMAT (Chen et al, 32 2021; Xue et al, 13 2023) have been investigated and implemented in HA-WBRT. Studies comparing non-coplanar techniques to their coplanar counterparts have shown promising results. Sharma et al 31 found that non-coplanar IMRT and VMAT exhibited superior target coverage and homogeneity index compared to coplanar IMRT. Chen et al 32 reported that non-coplanar VMAT significantly reduced Dmax for the lens compared to coplanar plans, with no significant differences observed in doses to the hippocampus or other OARs. Xue et al 13 demonstrated that non-coplanar VMAT significantly improved target dose homogeneity, reduced D50% of the brain, and decreased D2% of the hippocampus, lens, and optic nerves. Despite the dosimetric advantages offered by the non-coplanar technique in HA-WBRT, its implementation increases planning complexity, treatment time, and poses a risk of collisions between the LINAC and the patient or immobilization devices.
While non-coplanar techniques offer potential dosimetric advantages, their practical challenges, including increased planning complexity and treatment time, have prompted researchers to explore alternative approaches for enhancing hippocampal sparing in HA-WBRT. One such promising alternative is the head-tilting technique, which aims to leverage dosimetric benefits while addressing the implementation challenges associated with non-coplanar methods. Early research utilizing virtual CT simulations (Siglin et al, 17 2017; Lin et al, 18 2018) explored the optimal head tilt angles for HA-WBRT. Siglin et al 17 (2017) concluded that a 30° head tilt facilitated coplanar IMRT planning that met all dosimetric constraints, while Lin et al 18 (2018) found that VMAT plans with a head tilt of at least 15° met dose constraints, with a tilt of 25° or greater offering superior coverage and uniformity for the PTV, alongside reduced OAR doses. However, the utilization of virtual CTs or phantoms might limit the generalizability of these findings to real-world patient scenarios. Subsequent studies using actual patient CT data (Moon et al, 15 2015; Oh et al, 16 2016) further explored the impact of head tilt on hippocampal sparing during WBRT. Moon et al 15 highlighted a significant reduction in hippocampal dose with a 30°head tilt, particularly notable with VMAT. Oh et al 16 observed improvements in conformity and homogeneity indices with a 40° head tilt angle, alongside reduced mean doses to critical organs such as the hippocampus, parotid gland, and lens. While these studies provide valuable insights, they were constrained to a singular, predetermined tilt angle for each patient, limiting the analysis to dose distributions within the target, hippocampus, and lens. Based on these findings in IMRT and VMAT, researchers have begun to explore the potential of applying head-tilting techniques to TOMO for HA-WBRT, aiming to overcome the mechanical limitations of the TOMO couch while leveraging the dosimetric benefits observed in other modalities. Recent studies have shown encouraging results in this direction. Miura et al 22 investigated the use of a head-tilting baseplate during TOMO for HA-WBRT, demonstrating improved dose distribution for the PTV, reduced radiation exposure to OARs (notably the hippocampus and lens), and a substantial decrease in treatment duration by over 10%. Sengul et al 23 examined the impact of head tilt angles on TOMO plans using a RANDO phantom, finding that a 30° head tilt angle provided optimal protection for OARs, particularly the hippocampus, while ensuring adequate target coverage and reducing treatment time.
Despite these promising findings, research on head-tilting techniques for HA-WBRT using TOMO remains limited. Several critical questions persist, including the determination of the optimal head tilt angle, comprehensive dosimetric comparisons, and delivery efficiency assessments between TOMO and VMAT techniques when employing the head tilt method. These knowledge gaps underscore the need for further investigation to fully realize the potential of head-tilting techniques in TOMO for HA-WBRT, potentially enhancing both dosimetric outcomes and overall treatment efficacy. To address these knowledge gaps and contribute to the research on head-tilting techniques in TOMO for HA-WBRT, our study aimed to provide a comprehensive analysis of dosimetric outcomes across a range of head tilt angles, while also comparing TOMO and VMAT techniques and assessing delivery efficiency. In TOMO treatment planning, the field width corresponds to the longitudinal thickness of the treatment field at the machine's isocenter. The modulation factor (MF) plays a pivotal role in balancing plan efficiency and the optimizer's ability to adjust beamlet intensities to meet treatment objectives. TOMO offers MF options ranging from 1.0 to 5.0, along with three predefined field width selections: 1.048 cm, 2.512 cm, and 5.048 cm. Notably, considerable variability exists in field width and MF value settings across different institutions. 33 While prior studies (Gondi et al, 6 Gondhowiardjo et al, 34 Miura et al, 22 Yokoyama et al 35 ) have advocated for a higher MF of 3.0 and a narrower 1.048 cm field width to enhance plan quality, consistent with the RTOG 0933 protocol recommendations, our study opted for a 2.512 cm field width and an MF of 2.7 to reduce delivery time while maintaining plan quality.
Given the significantly longer delivery time of TOMO compared to VMAT plans, as reported by Wang et al 20 and Rong et al, 29 addressing treatment duration is crucial to mitigate uncertainties such as patient movement and anatomical changes. While our study demonstrated dosimetric advantages for head tilt angles between 40° and 45°, it was important to note that the patient comfort or treatment reproducibility concerned. The increased head tilt angle, combined with longer treatment times, may lead to inadequate neck support and patient discomfort (Figure 1), potentially affecting positioning reproducibility. To address these concerns, we suggest several mitigation strategies: using supportive materials like foam padding under the patient's neck, exploring alternative TOMO parameters (eg, larger field width, lower modulation factor) to shorten treatment duration, and implementing comfort measures such as music or therapy dogs to alleviate patient stress and anxiety. Future studies should incorporate these considerations to provide a more comprehensive evaluation of treatment efficacy, patient comfort, and overall experience, ultimately optimizing HA-WBRT techniques for improved patient outcomes.
The study presents several limitations that warrant consideration. First, the sample size was relatively small, comprising only 8 patients, potentially constraining the generalizability of the findings. Moreover, in the TOMO planning process, the selection of field width, modulation factor, and pitch values relied on the expertise and experience of our center. However, it is crucial to acknowledge that these chosen parameters may not universally represent the optimal settings across all cases and centers. While our study has identified promising results within the [40°, 45°] range, a more granular analysis could yield even more precise recommendations, including smaller increments within this range, evaluation of setup reproducibility, and long-term clinical outcomes. By conducting these detailed studies, we will establish more definitive guidelines for tilt angle selection in HA-WBRT.
Conclusions
This study demonstrated that TOMO plans with head tilt angles met all dose constraints for HA-WBRT, with the [40°, 45°] range showing significantly improved conformity, homogeneity, and reduced doses to most OARs. As the first comparison of TOMO and VMAT using the head tilt technique for HA-WBRT, our findings support the feasibility of implementing a [40°, 45°] head tilt angle with TOMO in clinical treatment. This approach allows for hippocampal sparing and potential reduction of cognitive impairment, which may improve patients’ quality of life. The results provide a foundation for optimizing HA-WBRT techniques and warrant further clinical investigation.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241281326 - Supplemental material for Comparison of Different Head Tilt Angles in Tomotherapy and Volumetric Modulated Arc Therapy for Hippocampal-Avoidance Whole-Brain Radiotherapy
Supplemental material, sj-docx-1-tct-10.1177_15330338241281326 for Comparison of Different Head Tilt Angles in Tomotherapy and Volumetric Modulated Arc Therapy for Hippocampal-Avoidance Whole-Brain Radiotherapy by Yang Li, Cuiyun Yuan, Dongjie Chen, Sisi Xu, Wei Jiang, Jiaxin Huang, Shanshan Ye, Yin Zhang, Jun Liang and Chenbin Liu in Technology in Cancer Research & Treatment
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was conducted in accordance with ethical principles and guidelines, and received approval from the Institutional Review Board (IRB) of our cancer hospital (Ethical approval number: JS2023-9-1).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (No. 12005301), Guangdong Basic and Applied Basic Research Foundation (No.2021B1515120065, 2022A1515012456), Natural Science Foundation of Shenzhen City (No: JCYJ20230807150502006), Shenzhen High-level Hospital Construction Fund, Shenzhen Key Medical Discipline Construction Fund (No. SZXK013), Sanming Project of Medicine in Shenzhen (No. SZSM201612063), Shenzhen High-level Hospital Construction Fund, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Shenzhen (No. SZ2020MS002, E010321015, E010322028, SZ2020ZD002), and Shenzhen Clinical Research Center for Cancer (No.[2021]287).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
