Abstract
Introduction
In 2020, there were more than 2.26 million new cases of breast cancer (BC) in women, and BC has surpassed lung cancer as the most common malignant tumor in the world. 1 With the improvement of medical level, the mortality rate of BC worldwide has gradually decreased, but distant metastasis of cancer cells is still the main factor in tumor death. Among them, triple-negative breast cancer (TNBC) is an aggressive breast cancer that is usually associated with increased metastasis, accounting for 12%-18% of the total number of BC patients, resulting in high mortality and poor prognosis. Patients with TNBC lack estrogen receptors (ER), progesterone receptors (PR), and epidermal growth factor receptor-2 (HER2) and are therefore not candidates for hormonal or anti-HER2 therapy, 2 Total breast resection is often used for treatment. 3 Although TNBC responds to existing chemotherapy drugs, the end result is unsatisfactory due to the rapid development of drug resistance. 4 In addition, almost all of these treatments cause mild to severe side effects, such as lymphedema. 5 Therefore, the discovery and development of synergistic anticancer drugs with few toxic side effects and strong targeting have become the main research goals, among which anti-cancer drugs from natural sources such as plants or bacteria have received more and more attention.
Antimicrobial peptides (AMPs) are small molecule polypeptides, encoded by specific genes, with the function of resisting foreign bacteria invasion and destroying mutant cells in the human body, and are a key component of the natural immune system of organisms, which can effectively protect the body from pathogens. Among them, cationic peptides (CAPs) have endogenous targets due to their low molecular weight, which can effectively penetrate tumor cells and prevent tumor angiogenesis and tumor growth and metastasis, which has become a hot spot in the field of cancer prevention and treatment. 6
The antimicrobial peptide LL-37 is the only member of the cathelicidin family of peptides in the body. It can be produced by neutrophils and various epithelial cells, etc, and participate in immune regulation, thereby promoting the body's immune function.
7
Many studies have shown that LL-37 not only plays an important role in antimicrobial,
8
immunology,
9
angiogenesis,
10
wound repair,
11
and bone tissue engineering,
12
but also can inhibit tumor occurrence and progression.LL-37 has great potential in anti-tumor treatment, but due to its long sequence, high cost in the process of chemical synthesis, greater possibility of error, complex effect on tumor cells, easy to be hydrolyzed by proteases,
13
therefore, the search for shorter and more active LL-37-derived peptides has become a current research hotspot. merecidin is the 14th-32nd amino acid formation short peptide in LL-37, which retains the most active effective fragment in LL-37, and has acetylation modification at the N-terminus, which improves the antitumor biological activity and stability of antimicrobial peptides, and can be considered as an alternative for the treatment of breast cancer. After the preliminary research of our group, merecidin can effectively control the proliferation and aggregation of
miRNA is a small RNA molecule that is generally between 18-22 nucleotides in size and non-coding. The vast majority of human miRNAs are encoded in introns, exons, exon ligations, or their own genes, and mature miRNAs can suppress gene expression by directing the relevant protein to a target site in the mRNA's 3’ UTR.
18
miRNAs regulate a variety of physiological processes, including growth, stress response, cell attachment, motility, inflammation, cell survival, aging, and apoptosis, all of which are important factors in tumorigenesis.
19
To date, more than 3000 miRNAs associated with tumorigenesis and progression have been identified (miRbase database). Studies have shown that different antimicrobial peptides can regulate different miRNAs in vitro and in vivo, affecting various biological functions of tumor cells. Chenlu L. Wu
Aggressive growth and distant metastasis are major features of cancer and major life-threatening risk factors. Therefore, it is believed that targeting aggressive growth, early stages of metastasis and potential therapeutic targets of breast cancer can reduce the development of secondary tumors and improve overall survival. Epithelial-mesenchymal transition (EMT) is significantly associated with tumor metastasis, which is a cell reprograming process that causes significant morphological changes and greater movement of epithelial cells, 23 during which cancer cells acquire mesenchymal characteristics. For example, the expression of N-cadherin and vimentin increased.At the same time, the expression of epithelial features, such as E-cadherin, is reduced and lost. 24 Among them, vimentin, as a cytoskeletal protein, is closely related to cell movement and plays a key role in EMT and metastasis, and has become a research hotspot in recent years. 25 The expression of vimentin is regulated by a variety of factors. For example: DNA methylation, 26 miRNA 27 and lncRNA, among which, miRNA, as a non-coding small molecule RNA, can regulate the expression of vimentin, thereby affecting the EMT. Therefore, whether the antimicrobial peptide merecidin can inhibit the migration and invasion of TNBC cells by inhibiting the vimentin and EMT is also the main concern of this study.
Therefore, this paper aims to explore the inhibitory effect of antimicrobial peptide merecidin on TNBC cells and further explore its inhibition of migration and invasion of cancer cells by regulating miR-30d-5p/vimentin, in order to provide more effective strategies and methods for clinical practice.
Materials and Methods
Cell Culture
Human TNBC cell MDA-MB-231 originated from the Laboratory of Molecular Diagnosis, School of Inspection, Ningxia Medical University; Human TNBC cell MDA-MB-468 was purchased from Pricella Company. In a constant temperature incubator at 37 °C, human TNBC cells (MDA-MB-231 and MDA-MB-468) were cultured in a 5% CO2 environment using H-DMEM medium (Hyclone®, Logan, USA) containing 10% FBS (PAN®,Adenbach,Germany), 1% penicillin mixture (Solarbio®,Beijing,China) and MDA-MB-468and adding Mmerecidin 24 h later. Conduct a follow-up experiment. All methods follow the BELMONT report. For ARRIVE: “The reporting of this study conforms to ARRIVE 2.0 guidelines.” 28
Cell Transfection and Drug Administration
MDA-MB-231 and MDA-MB-468 cells were transfected with miR-30d-5p mimic (50 nM), mimic negative control (NC), miR-30d-5p inhibitor (50 nM), inhibitor NC, si-vimentin (2 μg), si-NC, pcDNA3.1-vimentin (2 μg) or empty pcDNA3.1 (GenePharma®, Shanghai, China). Lipo2000 reagent (Thermo Fisher®, Waltham, USA) was used to facilitate the transfection. The following experiments were conducted 24 h after 3 repeated transfection experiments.
Merecidin (purity > 98%) was obtained from GL Biochem Co, Ltd (GL Biochem®, Shanghai, China). Cells were treated with merecidin (5 μmol/L, 10 μmol/L) 24 h after transfection. The experiments were conducted 24 h after the administration of merecidin.
Cell Counting Kit-8 Assay
The cell viability was assessed using the CCK8 proliferation detection kit (Dojindo®, Tokyo, Japan). Briefly, a total of approximately 5 × 103 cells were seeded in 96-well plates, after merecidin stimulation, 10 μL CCK-8 solution was added to each well and cultivated for 1.5 h. The absorbance of the reaction system was measured spectrophotometrically at 450 nm.
Plate Cloning Experiments
Collect MDA-MB-231 and MDA-MB-468 cell suspensions in the logarithmic growth phase, culture with medium at a concentration of merecidin 5 μmol/L, 10 μmol/L (treatment group) and medium without merecidin (control group) for 24 h at 37 °C, then digest the cells, dilute the appropriate folds and suspend them into a single-cell suspension, and seed 2200 cells per well in a six-well plate, respectively. Continue incubating for 2-3 weeks (depending on cell growth status), solidified using 4% paraformaldehyde, stained with 0.1% crystal violet, and observe and count cell colonies with more than 60 cells under low magnification.
Scratch Assay
A suspension of MDA-MB-231 and MDA-MB-468 cells grown at the logarithmic phase was transferred to 6-well plates, cultured with medium at a concentration of 5 μmol/L, 10 μmol/L of merecidin (treatment group) and medium withoutmerecidin (control group) at 37 °C for 24 h, three parallel lines were drawn in each well with a sterile pipette (100 μL) to create cell wounds, floating cells were washed away by discarding the medium, complete medium containing 10% serum was added, photographed and recorded under low magnification, and returned to the incubator after continuing to culture for 24 h (MDA-MB-231) and 48 h (MDA-MB-468), respectively, the area of cell scratches was measured by ImageJ and the mobility of cells was calculated. Mobility = (0 h width − 24 h width) / (0 h width) × 100%.
Transwell Assay
Gently spread the 1:5 ratio diluted Matrigel matrix glue(Corning Costar) into the transwell chamber(Corning Costar), wait 4 h, and after the gel solidifies, collect the MDA-MB-231 and MDA-MB-468 cell suspensions in the logarithmic growth phase [incubate sequentially with merecidin concentration of 5 μmol/L, 10 μmol/L medium (treatment group) and erecidin-free medium (control group) for 24 h at 37 °C, and add 1 × 105 cells to the upper chamber, 200 μL per well, add 700 μL of medium containing 10% serum to the lower chamber, put it into the incubator, incubate for 24 h, take out the transwell chamber, fix the cells with 4% paraformaldehyde, then stain with 0.1% crystal violet dye, and finally wash with PBS, and use a cotton swab to stain the cells that have not penetrated the upper chamber, take pictures and records under low magnification, and count the number of any ten field stained cells.
Western Blotting
Cells were added into lysis buffer (KeyGEN Bio TECH, Jiangsu, China) containing protease inhibitors and phosphatase inhibitors. The cell lysates were centrifuged at 6000 rpm, 4 °C for 5 min. Tumor tissue homogenates were lysed on ice for 30 min. The tissue lysates were transferred into 1.5 mL tubes and centrifuged at 12 000 rpm, 4 °C for 5 min. Protein samples were obtained after the centrifugation. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the loading control. Protein concentration was measured using a BCA kit (KeyGEN Bio TECH) to ensure the same loading amount of each protein sample. The mixture of proteins and loading buffer was heated in boiling water for 10 min before 10% SDS-PAGE. The separation gel was prepared using the SDS-PAGE gel preparation kit (KeyGEN Bio TECH). The electrophoresis was maintained for 1 to 2 h, during which the voltage was shifted from 80 V to 120 V when bromophenol blue entered the separation gel. The proteins were transferred onto a membrane in an ice bath (300 mA, 60 min). The membrane was then rinsed for 1 to 2 min and immersed in blocking buffer at room temperature for 60 min. Primary rabbit anti-human antibodies against GAPDH (5174S, 1:1000, Cell Signaling Technology, USA), E-cadherin (3195, 1:1000, Cell Signaling Technology), Snail (3879, 1:1000, Cell Signaling Technology), vimentin (5741, 1:1000, Cell Signaling Technology) were incubated with the membrane on a shaker at 4 °C overnight. The membrane was washed 3 × 6 min before the proteins were incubated with HRP conjugated secondary antibody (goat anti-rabbit IgG, 1:5000, Cell Signaling Technology) at room temperature for 1 h. The membrane was washed 3 × 10 min and added with chemiluminescence fluid. Protein expression was detected using a chemiluminescence imaging system (Bio-Rad, Hercules, CA, USA).
Quantitative Reverse-Transcriptase Polymerase Chain Reaction
Total RNA of tumors or cells from each group was extracted using total RNA small preparation kit (Corning Costar, USA), followed by concentration and purity tests. Qualified RNA samples were adjusted to an appropriate concentration and reverse-transcribed using a reverse transcription kit (TransGen Biotech, Shanghai, China) and random primers. Gene expression was detected by the Real-time polymerase chain reaction (PCR) system (Thermo Fisher, USA) under the conditions provided by the PCR kit (SYBR Green Mix, TransGen Biotech). The template DNA was predenatured at 94 °C for 30 s, followed by 40 cycles of denaturation (94 °C, 5 s), annealing (61.5 °C, 15 s) and extension (72 °C, 10 s). Each RNA sample had 3 duplicates. The internal reference genes for miRNA and mRNA were U6 and GAPDH, respectively. The ΔΔ/CT analysis protocol was used for data analysis. Primers used in the PCR are listed in Table 1.
Primary Primer Sequences.

Dual-Luciferase Reporter Assay
The binding sites between miR-30d-5p and VIM mRNA were predicted by targetscan (http://www.targetscan.org/vert_72/). Wild and mutant sequences of the binding site on VIM (wt-VIM and mut-VIM) were designed and synthesized according to the information provided by targetscan. The luciferase reporter vectors (pGL3-Promoter, RiboBio, Guangzhou, China) were inserted with wt-VIM or mut-VIM and transfected into MDA-MB-231 cells with 50 nM miR-30d-5p mimic or mimic NC. The cells were accordingly designated as mimic + mut-VIM, mimic + wt-VIM, mimic NC + mut-VIM and mimic NC + wt-VIM groups. The luciferase activity was detected using a dual-luciferase reporter assay kit (TransGen Biotech) and averaged after 3 repeats.
Laser Confocal Microscopy to Observe Positioning
The collected MDA-MB-231 cell suspension was seeded on a cell climbing tablet of a 24-well plate, incubated with medium containing 10 μmol/L FITC-merecidin (treatment group) and medium containing 10 μmol/L FITC-RI-10 (control group) for 12 h at 37 °C, then discarded the medium, carefully cut out the cell climbing sheet, and under dark conditions, take 5 μL of anti-fluorescence attenuation mounting agent (containing DAPI) drop on a clean slide, and lightly cover the cell climbing sheet. Allow the cells to fully contact the mounting solution and subsequently observe by laser confocal microscopy.
Lactate Dehydrogenase (LDH) Experiment
The collected MDA-MB-231 cell suspension was seeded in a 96-well plate with 200 μL per well, 1.5 × 104 cells, incubated overnight at 37 °C, and then merecidin at a concentration of 5 μmol/L, 10 μmol/L, 15 μmol/L was added sequentially as the experimental group, the cell group without merecidin was the control group, the highest enzyme activity control group, and the cell-free culture group was used as the blank group, each group had 3 complex wells, and incubated at 37 °C, 5% CO2 for 12 h. Take out the 96-well plate from the incubator 1 h in advance, add 20 μL of Lactate Dehydrogenase (LDH) release reagent to the control group with the highest enzyme activity, mix well, then place it in the incubator, continue to culture for 1 h, take out the 96-well plate, centrifuge 400 g for 5 min, add 120 μL of supernatant to a new 96-well plate, and add 60 μL of LDH detection working solution, mix well, incubate at room temperature in the dark for 30 min, and then measure the absorbance value at 490 nm. Dual-wavelength assays were performed at 600 nm as the reference band to calculate the relative activity of lactate dehydrogenase. Lactate dehydrogenase relative activity = (treatment-blank) / (highest enzyme activity control−blank) × 100%.
Pull Down Assay
The Protein Pull-Down Kit (Thermo Fisher, USA) was used for this assay. Biotin-merecidin (1 mg/mL) 200 μL was added into Streptavidin reagent, After mixing, incubate at 4 °C for 3 h, centrifuge at 1250 × g for 1 min in a low-speed shaker. Add 250 μL of biotin blocking solution to the spin column, invert the column for 3-5 times to mix, stand at room temperature for 5 min, centrifuge at 1250 × g for 1 min, Add 200 μL of MDA-MB-231 cell protein (0.6 mg/mL) to the spin column, add the same volume buffer to the centrifuge column designated as the positive control, mix well and incubate at 4 °C in a low-speed shaker for 4 h, centrifuge at 1250 × g for 1 min, Add 250 μL of wash buffer to the spin column, gently invert the column for 5-7 times to mix, let stand at room temperature for 1 min, centrifuge at 1250 × g for 1 min, repeat 3 times, and collect the effluent. Add 50 μL of elution buffer to the centrifuge column, add 2 μL of neutralization buffer in advance to the corresponding collection tube, then gently flip the column, mix 5-7 times, and finally stand at room temperature for 5 min, centrifuge at 1250 × g for 1 min, recover the effluent and label “Elution C”. Repeat 1 time and collect the effluent labeled “Elution D”. The Elution was detected by SDS-PAGE electrophoresis. Banded strips were digested by mass spectrometry.
Animal Experiments
Specific pathogen-free (SPF) BALB/c nude mice (n = 28, 4 weeks, 16 ± 2 g) were purchased from Beijing HFK Bioscience Co, Ltd (Beijing, China). All animals were raised with standard food and water in a SPF laminar flow room at 22 to 26 °C with 55% ± 5% humidity for at least 1 week. The raising environment was provided with 12/12-h light-dark cycles. All animal experiments com plied with the regulations and ethic requirements for management of laboratory animals.
The mice were randomized into 3 groups: PBS, paclitaxel and merecidin groups (4 mice per group). MDA-MB-231 cells (1 × 106) were suspended in 100 μL of PBS-Matrigel (1:1, v/v) and subcutaneously injected in the right axilla of nude mice to establish subcutaneous tumor model of TNBC. The syringe was slowly pulled out after injection and the injection area was pressed with an alcohol cotton ball for 10 s to prevent effusion of cell suspension. The merecidin group was given paratumoral injection of merecidin (200 mg/kg) daily until the implanted tumor grew to 100 mm3. The paclitaxel group was given paratumoral injection of toxal (100 mg/kg) daily. The PBS group was injected with equal volume of normal PBS. The tumor volume was measured every 7 days (tumor volume = 1/2 × long diameter × short diameter2). The mice were sacrificed 4 weeks after the injection. The tumor weight was measured and the tumors were photographed.
Immunohistochemistry
Tumor tissues were fixed in 4% paraformaldehyde for 48 h and made into paraffin sections (4 μm). The sections were roasted for 20 min, dewaxed with routine xylene and washed with distilled water. After being washed 3 times with PBS, the sections were incubated with 3% H2O2 at room temperature for 10 min and washed 3 times with PBS, followed by heat-induced antigen retrieval and another round of PBS washing. Normal goat serum was used for blocking nonspecific binding at room temperature for 20 min before the sections were incubated with anti-vimentin antibody (ab16667, 1:200, Abcam, Cambridge, MA, USA) at 4 °C overnight. The sections were washed 3 times with PBS and incubated with secondary antibody at room temperature for 1 h. Following PBS washing, the sections were stained in DAB solution for 1 to 3 min and in hematoxylin for 3 min. The sections were dehydrated, transparentized, and mounted for observation of vimentin positive cells under a microscope.
Statistical Analysis
Data were analyzed in GraphPad 7.0 and shown in a form of mean ± SD. A t test and one-way analysis of variance were used for 2-group and multigroup comparisons, respectively. Significant statistics were represented by
Results
Merecidin Inhibits Proliferation, Migration and Invasion of TNBC Cells
The toxic effect of different concentrations of merecidin on two TNBC cells was detected by CCK-8 method (Figure 1A), and when the concentration of merecidin increased, the inhibitory effect of the two TNBC cells was also enhanced (

Merecidin inhibits proliferation, migration and invasion of TNBC cells. (A) Inhibition of different concentrations of merecidin on proliferation of MDA-MB-231 and MDA-MB-468 cells; (B) inhibition of different concentrations of merecidin on colony formation in MDA-MB-231 and MDA-MB-468 cells; (C) inhibition of different concentrations of merecidin on the migration of MDA-MB-231 and MDA-MB-468 cells; (D) inhibition of different concentrations of merecidin on the Invasion of MDA-MB-231 and MDA-MB-468 cells; (E) different concentrations of merecidin on vimentin and EMT-related proteins of MDA-MB-231 and MDA-MB-468 cells (Compared to the Control group, *
The effect of different concentrations of merecidin on the migration and invasion ability of two TNBC cells was detected by scratch experiment and transwell experiment (Figure 1C, D), and compared with the control group (0 μmol/L), the migration and invasion ability of cells was significantly reduced after 24 h treatment with 5 and 10 μmol/L merecidin (
Merecidin Upregulates the Expression of miR-30d-5p in TNBC Cells and Binds to Vimentin In Vitro
With MDA-MB-231 cell as the main research object, laser confocal microscopy was used to observe the localization of merecidin in TNBC cells (Figure 2A). The cytoplasm of MDA-MB-231 cells is filled with merecidin, indicating that merecidin is mainly present in the cytoplasm of TNBC cells. When the membrane structure is disrupted, enzymes in the cell plasma are released into the culture medium in large quantities, and LDH activity is the most stable, so its production can be used as an indicator of cell membrane integrity. Therefore, the destructive effect of merecidin on cell membranes can be judged by detecting the LDH activity released by MDA-MB-231 cell into the culture medium after the action of merecidin (Figure 2B). Compared with the control group (0 μmol/L), after 10 μmol/L and 15 μmol/L merecidin action for 12 h, the LDH released in the culture medium gradually increased (

merecidin upregulates the expression of miR-30d-5p in TNBC cells and binds to vimentin in vitro. (A) Localization of the merecidin in MDA-MB-231 cell; (B) effect of different concentrations of merecidin on LDH in MDA-MB-231 cell culture medium; (C) expression of miR-30d-5p in tTNBC cells; (D) effects of different concentrations of merecidin on miR-30d-5p expression in MDA-MB-231 and MDA-MB-468 cells; (E) the SDS-PAGE electrophoresis of Pull-down experiment; (F) Western-Blot validates the results of pull-down experimental; (G) mass spectrometry analysis of base peak plots. (Compared to the Control group, *
In addition, the expression of miR-30d-5p in TNBC cells was detected by qRT-PCR in normal breast epithelial cells MCF-10A as the control group (Figure 2C). Compared with MCF-10A, the expression of miR-30d-5p in TNBC cells (MDA-MB-231 and MDA-MB-468) was significantly downregulated (
At the same time, the interaction proteins of merecidin with MDA-MB-231 cell were identified by pull-down experiment, and SDS-PAGE electrophoresis was performed on each sample protein in the experiment (Figure 2E). The protein bands in the sample collection tubes C and D in lanes 5 and 6 were faintly visible between 55-70 kD, which were proteins that bound to Biotin-merecidin and the protein of MDA-MB-231 cell, indicating that merecidin interacted with MDA-MB-231 cell proteins
The protein bands in the range of 55-70 kD obtained by cutting the pull down assay can be clearly seen in the base peak mass spectrum (Figure 2F) after tandem mass spectrometry analysis, and the signal and noise are low, so they can be used for protein database search. After the database Proteome Discoverer 2.4 search, 44 protein components were matched, the main protein components are shown in Table 2, and it was found that the proteins bound to merecidin in MDA-MB-231 cell were mainly structural proteins and immune proteins, of which vimentin was used as a cytoskeletal protein and an important marker of EMT, which could be used as a follow-up research object. In order to verify the results of the pull down experiment, the samples obtained from the pull down experiment were subjected to the Western-Blot (Figure 2G), and likewise, vimentin was expressed in the D effluent of the sample collection tube to be tested. It was further confirmed that merecidin interacted with vimentin in TNBC cells
Primary Protein Information Matched in the Protein Database.

Protein molecular weight (kD): It is an important indicator of protein size and can be used to study the structure and function of proteins. Sequence coverage: The proportion of the sequence obtained by sequencing to the whole target sequence such as the genome sequence protein score: It is used to evaluate the degree of agreement between a mass spectrum data and the data in the database. The higher the score, the higher the probability that the fragment printed by the mass spectrum is the fragment recorded in the database.
miR-30d-5p Inhibits the Migration and Invasion of TNBC Cells
Transfect MDA-MB-231 and MDA-MB-468 cells with miR-30d-5p mimics/NC and miR-30d-5p inhibitor/NC (Figure 3A). Transfection of miR-30d-5p mimics significantly upregulated the expression of miR-30d-5p in cells (

miR-30d-5p inhibits the migration and invasion of TNBC cells. (A) Transfection efficiency of miR-30d-5p in MDA-MB-231 and MDA-MB-468 cells; (B) inhibition of miR-30d-5p on the migration of MDA-MB-231 and MDA-MB-468 cells; (C) inhibition of miR-30d-5p on the invasion of MDA-MB-231 and MDA-MB-468 cells. (Compared to the Control group, *
Up-regulation or down-regulation of miR-30d-5p, the effect of miR-30d-5p on the migration and invasion ability of TNBC cells was detected by scratch experiment and transwell experiment (Figure 3B, C), compared with the control group (NC), upregulating miR-30d-5p reduced the migration and invasion ability of cells (
miR-30d-5p Targets to Vimentin and Negatively Regulates Vimentin
The target genes predicted by miR-30d-5p were analyzed by bioinformatics analysis software starBase, microT, miRanda, TICA, and TargetScan, and at the same time, Wayne plots were drawn using Bioinformatics & Evolutionary Genomics software (Figure 4A), and finally 10 mRNAs were screened. Further research reports on miR-30d-5p in BC were reviewed through Pubmed, and vimentin, which was related to EMT and metastasis process, was selected as the follow-up research subject. After that, the expression of miR-30d-5p and vimentin was analyzed in starBase (Figure 4B), and miR-30d-5p was negatively correlated with vimentin expression in 1085 breast cancer tissue samples (
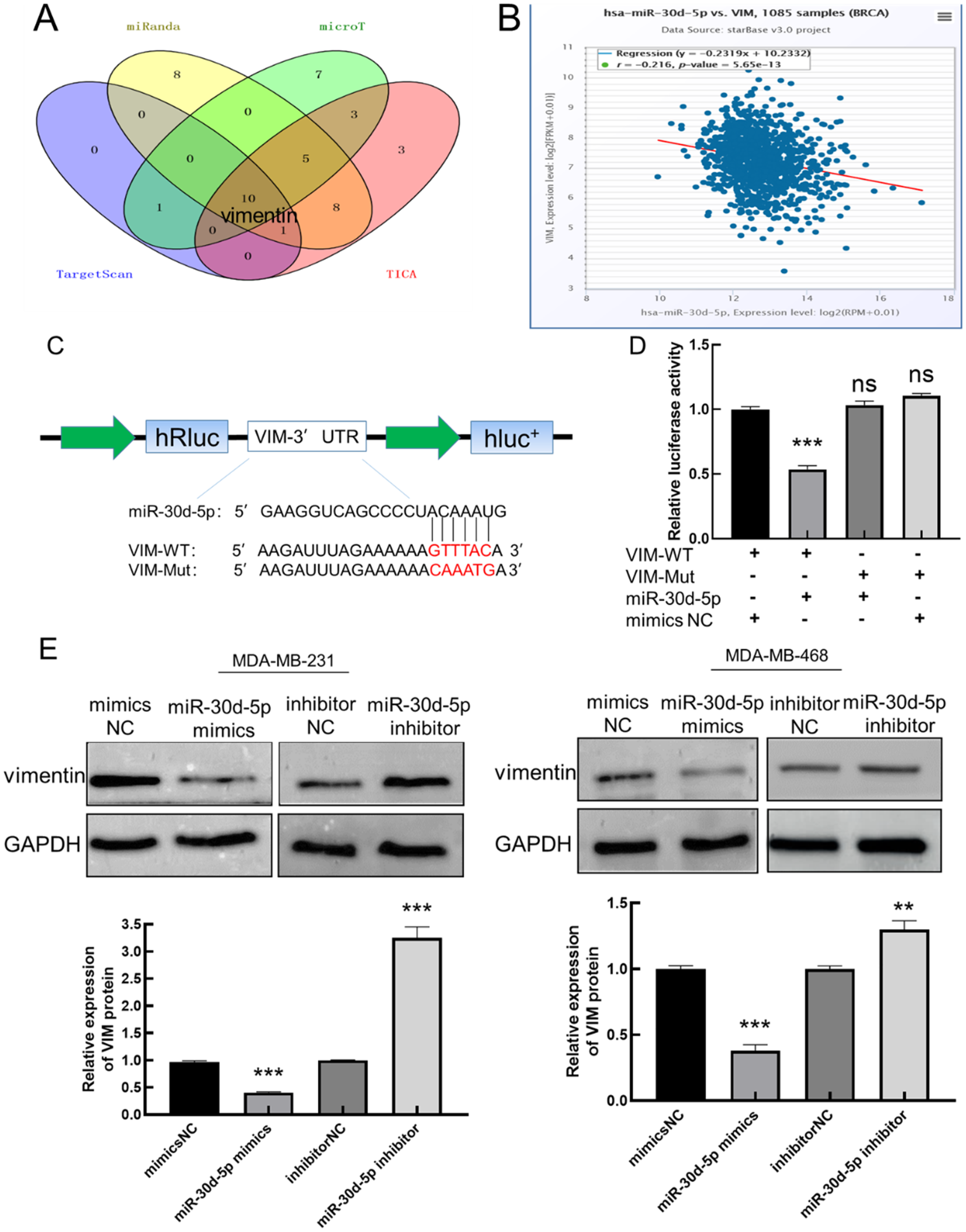
miR-30d-5p targets to vimentin and negatively regulates vimentin. (A) The Wayne diagram shows that miR-30d-5p-regulated mRNAs were screened; (B) in BC tissue samples, miR-30d-5p was negatively correlated with vimentin expression; (C) VIM 3UTR wild-type (VIM-WT) and mutant (VIM-Mut) Dual-Luciferase reporter plasmids; (D) effect of transfection mimics NC/miR-30d-5p mimics on VIM-WT/VIM-Mut luciferase activity; (E) effect of miR-30d-5p on vimentin expression in MDA-MB-231 and MDA-MB-468 cells. (Compared with NC, **
In order to further determine the targeting relationship between miR-30d-5p and vimentin, the binding sites between miR-30d-5p and vimentin mRNA were predicted by three databases: Targetscan, starBase and microRNAorg, and the wild body and mutant sequences of vimentin binding sites were designed and synthesized according to the predicted binding sites. The luciferin reporter carrier is inserted into wt-vimentin and mut-vimentin (Figure 4C). The miR-30d-5p mimics NC, luciferase reporter plasmid (containing VIM-WT/VIM-Mut) and miR-30d-5p mimics were then transfected into MDA-MB-231 cells in groups, and after 48 h, fluorescence values were determined and analyzed (Figure 4D). The results showed that the luciferase activity of the co-transfected VIM-WT and miR-30d-5p group decreased significantly (
Detect the expression of vimentin by up-regulating or down-regulating miR-30d-5p (Figure 4E). The results showed that when miR-30d-5p was upregulated, the expression of vimentin was significantly reduced (
Vimentin Is Positively Correlated with the Migration and Invasion of TNBC Cells
Transfect si-NC/siRNA and pcDNA3.1/pcDNA-vimentin in cells to verify their transfection efficiency (Figure 5A). Compared with the control group (NC), the expression of vimentin in si-vimentin-1764 group was significantly reduced (

Vimentin is positively correlated with the migration and invasion of TNBC cells. (A) Interferes with or overexpresses vimentin in MDA-MB-231 and MDA-MB-468 cells; (B) effects of vimentin on cell migration of MDA-MB-231 and MDA-MB-468; (C) effects of vimentin on cell invasion of MDA-MB-231 and MDA-MB-468 (Compared with NC, *
Interfering with or overexpressing the expression of vimentin in MDA-MB-231 and MDA-MB-468 cells, the effect of vimentin on cell migration and invasion was detected by scratch assay and transwell assay (Figure 5B, C). Compared with the control group (NC), interference vimentin, decreased migration and invasion of cells (
Downregulation of miR-30d-5p or Overexpression of Vimentin Counteracts the Inhibitory Effect of Merecidin on the Migration and Invasion in TNBC Cells
The effect of merecidin on the migration and invasion of TNBC cells was detected by scratch experiment and transwell assay (Figure 6A, B), Compared with the NC group, after 24 h of action with merecidin alone, the migration and invasion of cells was reduced (
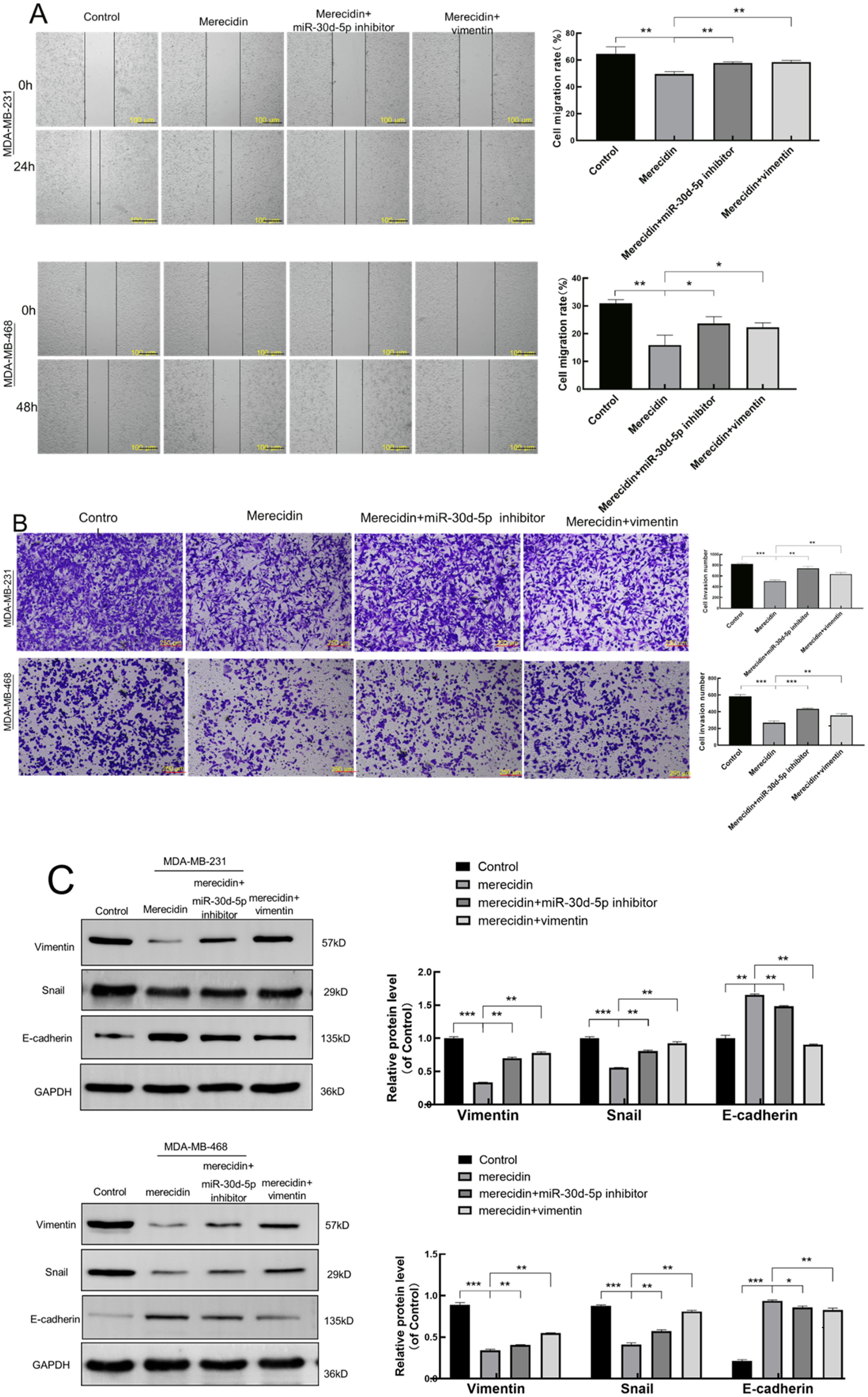
Downregulation of miR-30d-5p or overexpression of vimentin counteracts the inhibitory effect of merecidin on the migration and invasion in TNBC cells. (A) Downregulation of miR-30d-5p or overexpression of vimentin counteracts the inhibition of merecidin on migration in MDA-MB-231 and MDA-MB-468 cells; (B) downregulation of miR-30d-5p or overexpression of vimentin counteracts the inhibition of merecidin on invasion in MDA-MB-231 and MDA-MB-468 cells; (C) downregulation of miR-30d-5p or overexpression of vimentin counteracts the inhibition of merecidin on EMT in MDA-MB-231 and MDA-MB-468 cells. (Compared to the Control group, *
Similarly, in MDA-MB-231 and MDA-MB-468 cells, down-regulation of miR-30d-5p or overexpression of vimentin while merecidin was added for 24 h, and the effect of merecidin on EMT-associated protein in TNBC cells was detected by Western-blot (Figure 6C), compared with the NC group, after 24 h of merecidin action alone, The expression of the mesenchymal marker vimentin, snail decreases, the expression of the epithelial marker E-cadherin increases (
Merecidin Can Inhibit the EMT Process of TNBC Cells by Down-Regulating miR-30d-5p in Mice
Nude mice were injected with MDA-MB-231 cells and injected subcutaneous injection (SQ) with merecidin (200 mg/kg) or peritoneally with taxol (100 mg/kg) or equal volume PBS every other day. Two weeks later, the tumor volume and weight were measured and the tumor was isolated (Figure 7A). The tumor volume and weight in merecidin group were lower than those in PBS group (
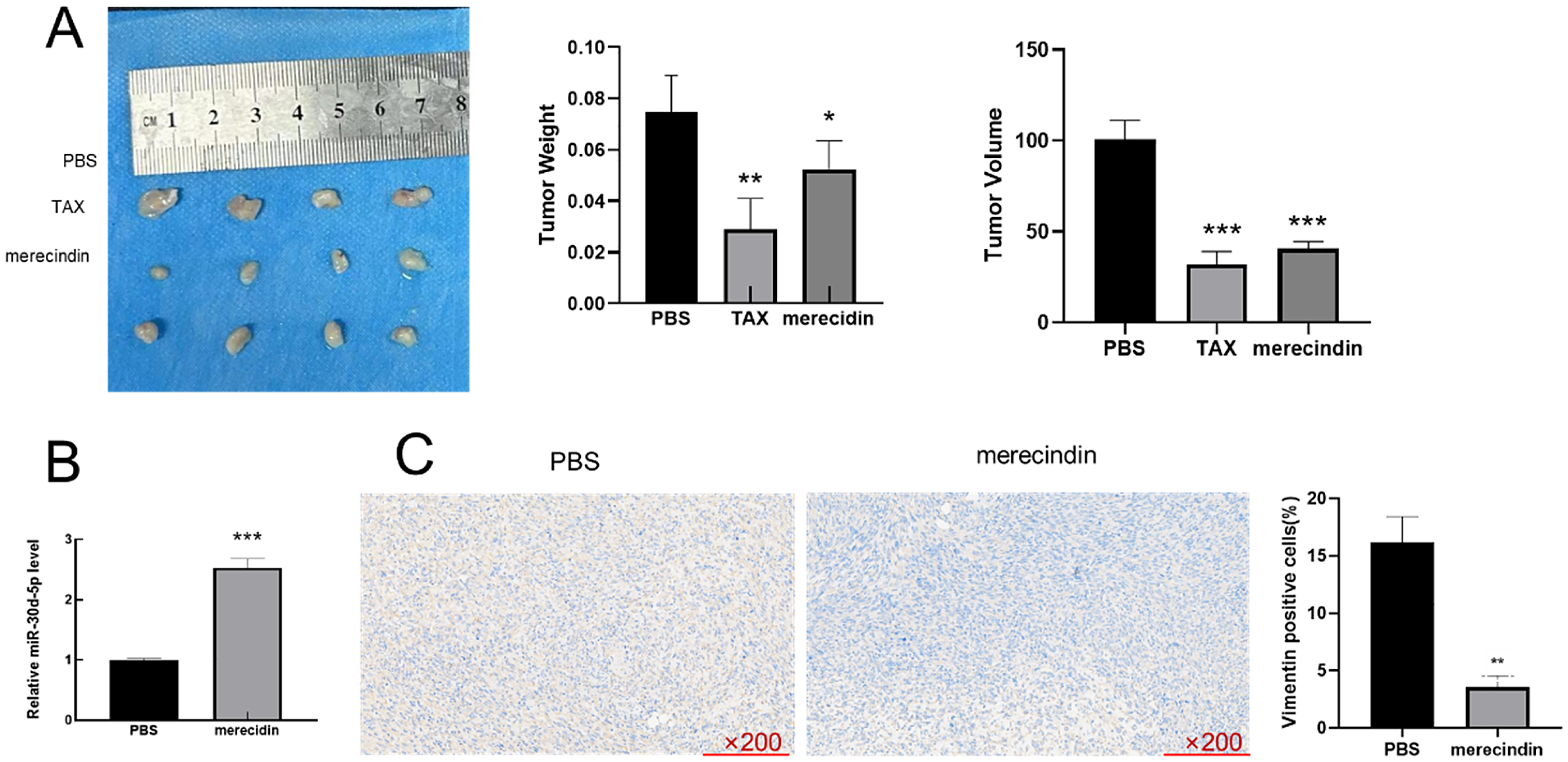
Merecidin can inhibit the EMT process of TNBC cells by down-regulating miR-30d-5p. Nude mice were injected with MDA-MB-231 cells and given either merecidin (200 mg/kg) or a single intrabitoneal injection of taxol (100 mg/kg) or equal volume PBS (n = 4). (A) The volume and weight of the tumor; (B) miR-30d-5p was detected by qRT-PCR; (C) the expression of vimentin in tumor tissues was analyzed by IHC. (*
Discussion
In recent decades, targeted therapies, including stratified medicine and personalized medicine, have been suggested to improve the treatment of cancers. 29 The development of precision medicine requires the characterization of molecular biomarkers. This study demonstrated the involvement of the miR-30d-5p/vimentin axis in the therapeutic effect of merecidin in TNBC.
Cell proliferation of cancer is an important part of tumor development. Kuroda
Tumor metastasis is an important cause of death in patients with TNBC. Tumor cells can be regulated by multiple mechanisms, go through multiple stages and steps, migrate from the primary location to other organs or tissues, and continue to proliferate and grow, eventually forming metastases with the same characteristics. 33 ADG-2e, a derivative of the antimicrobial peptide AZT, has been found to inhibit breast cancer cell migration through multidirectional plate adipogenesis, indicating its anti-metastatic potential. 34 Therefore, through scratch experiments and transwell experiments, merecidin was found to be able to effectively inhibit the migration and invasion of TNBC cells.
Epithelial-mesenchymal transition (EMT) plays an important role in metastasis. EMT is a binary process with two distinct cell populations, epithelial and mesenchymal, and is typically manifested by loss of expression of the epithelial marker E-cadherin and acquisition of the mesenchymal marker vimentin. EMT is closely related to tumor initiation and progression, stem cell nature of tumors, migration of tumor cells, hemovasation, and drug resistance.
35
During the EMT process, the epithelial cytoskeleton is restructured, resulting in the loss of its polar connection to the basement membrane and cells, resulting in increased cell transfer capacity.
36
Zhang
Disruption of cell membranes is an indispensable part of the antimicrobial peptide antitumor mechanism. The destruction of membrane structure will cause the release of LDH from the cell plasma into the culture medium, so the activity of LDH becomes an important basis for evaluating the integrity of the cell membrane. Zhou
Further, q-PCR results showed that miR-30d-5p was poorly expressed in TNBC cells, and after treatment with merecidin, the expression of miR-30d-5p in TNBC cells increased, and the interaction protein between merecidin and TNBC cells was detected by pull down experiment, and it was found that merecidin and vimentin interacted in vitro. The above evidence indicated that miR-30d-5p and vimentin were implicated in the progression of TNBC, which provoked investigations into the function of miR-30d-5p and vimentin and their interactions with merecidin.
As non-coding RNA, miRNAs are essential in the development of tumors. Studies have shown that miR-30d-5p is closely related to a variety of human tumors such as lung cancer, 42 colorectal cancer, 43 prostate cancer 44 and breast cancer, 45 reflecting the important role of miR-30d-5p in tumor prognosis. miR-30d-5p can act both as an inhibitor that hinders tumor development 46 and as a promoter that accelerates tumorigenesis. 47 In addition, miR-30d-5p also plays an important role in cell proliferation, migration, 48 apoptosis, autophagy, 49 tumorigenesis, 50 and chemotherapy resistance. 51 The multiple roles of miR-30d-5p in human cancer suggest broad feasibility as a biomarker and therapeutic target. In this study, miR-30d-5p was shown to have inhibitory effect on TNBC cell migration and invasion by scratch experiment and transwell invasion experiment. In order to further clarify the relationship between vimentin and miR-30d-5p, the regulatory relationship between miR-30d-5p and vimentin was detected by q-PCR and Western-blot, the results showed that the expression of vimentin was negatively correlated with the expression of miR-30d-5p. Dual-Luciferase reporter also confirmed that miR-30d-5p targets vimentin 3 ´UTR. The above results can show that miR-30d-5p and vimentin effectively inhibit the metastasis and invasion of TNBC cells by targeting and negatively regulating vimentin.
Vimentin, a key member of the intermediate filament (IF) protein family, is widely expressed in normal mesenchymal cells, is able to maintain cellular integrity and provide resistance to stress, and is a protein marker for EMT reprograming.
24
In colorectal cancer, breast cancer, gastric cancer,
52
and liver cancer,
53
vimentin expression is increased, and high expression of vimentin is associated with aggressiveness and poor clinical outcomes in many types of cancer.
54
Kim TW
It has been found that the LL-37 antimicrobial peptide analog FF/CAP18 inhibits the growth of colorectal cancer cells by directly targeting cancer cells and indirectly acting through exosomes, inhibiting intercellular communication. 57 In order to study the inhibitory effect of merecidin on TNBC cells through miRNA, a recovery experiment was designed to explore whether merecidin could play its main role on the basis of reverse regulation of miR-30d-5p or vimentin. Recovery experiments confirmed that whether it was down-regulating miR-30d-5p or overexpressing vimentin, it could counteract the effect of merecidin in inhibiting TNBC cell migration, invasion and EMT to a certain extent. That is, the antibacterial peptide merecidin hinders the EMT throughmiR-30d-5p/vimentin, which can effectively inhibit the spread and invasion of TNBC cells.
Furthermore,
Conclusions
In summary, the antimicrobial peptide merecidin can effectively inhibit the proliferation, migration and invasion of TNBC cells, and localized in the cytoplasm, it blocks the EMT process by regulating miR-30d-5p/vimentin, and it was able to stop TNBC tumorigenesis.
Footnotes
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Authors contributions
Song JinXuan conducted in vitro experiments, Ma Fei participated in animal experiments, He Min wrote the manuscript and Wang Xiuqing conceived and supervised the entire study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declarations
For ARRIVE: ‘The reporting of this study conforms to ARRIVE 2.0 guidelines, and all experimental protocols were approved by Ningxia Medical University ethics committees. The Tab of Animal Experimental Ethical Inspection, No.IACUC-2023-005, the paper ethics check label, No.2024-3763.
Ethical Statements
Ethical approval of all procedures performed in the current study was approved by Ningxia Medical University ethics committees (IACUC-2023-005).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ningxia Natural Science Foundation (2022AAC03185).
Data Availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
