Abstract
Purpose
Research on bone metastasis in esophageal cancer (EC) is relatively limited. Once bone metastasis occurs in patients, their prognosis is poor, and it severely affects their quality of life. Currently, there is a lack of convenient tumor markers for early identification of bone metastasis in EC. Our research aims to explore whether neutrophil-lymphocyte ratio (NLR) can predict bone metastasis in patients with EC.
Methods
Retrospective analysis of clinical indicators was performed on 604 patients with EC. They were divided into groups based on whether or not there was bone metastasis, and the patients’ coagulation-related tests, blood routine, tumor markers and other indicators were collected. The receiver operating characteristic curve (ROC) were used to determine the predictive ability of parameters such as NLR for bone metastasis in EC, and univariate and multivariate logistic regression analyses were conducted to determine the impact of each indicator on bone metastasis. Using binary logistic regression to obtain the predictive probability of NLR combined with tumor markers.
Results
ROC curves analysis suggested that the area under the curve (AUC) of the NLR was 0.681, with a sensitivity of 79.2% and a specificity of 52.6%, which can be used as a predictive factor for bone metastasis in EC. Multivariate logistic regression analysis showed that high NLR (odds ratio [OR]: 2.608, 95% confidence interval [CI]: 1.395-4.874,
Conclusion
NLR, as a convenient, non-invasive, and cost-effective inflammatory indicator, could predict bone metastasis in EC. Combining NLR with tumor markers can significantly improve the diagnostic accuracy of bone metastasis in EC.
Introduction
Esophageal cancer (EC) is among the most prevalent malignant tumors of the digestive tract globally and currently ranking as the tenth most common cancer. Every year, there are over 600,000 new cases. 1 Due to the challenge of detecting early-stage EC, patients typically receive a diagnosis in later stages, resulting in a high mortality rate.
The overall 5-year survival rate for EC is 15% to 25%, 2 with the main causes of death being recurrence and distant metastasis. Compared to important metastatic organs such as the liver and lungs, the incidence of bone metastasis in EC is relatively low at 7.7%, 3 which results in less research being conducted on bone metastasis. Currently, the main methods for screening bone metastasis in clinical practice include PET-CT, 4 ECT, 5 and other methods. However, due to the invasive and high cost of these examinations, they result in higher economic burden and waste of medical resources. Therefore, discovering a new, non-invasive, and convenient tumor marker for indicating bone metastasis is crucial for the early detection and treatment of bone metastasis.
Previous studies have shown that there is a close relationship between inflammation and the occurrence and development of tumors.6,7 Chronic inflammation plays an important role in the initiation, promotion, malignant transformation, invasion, and metastasis of cancer. 8 Neutrophils secrete various cytokines to promote tumor development and formation of an immunosuppressive tumor microenvironment. 9 As the type of granulocyte with the highest content in human blood, neutrophils promote tumor progression and immune evasion through various mechanisms. The MMP-9 released by neutrophils infiltrating the tumor is the main source of MMP-9 highly induced angiogenesis in the tumor microenvironment. 10 Neutrophils can recruit macrophages and regulatory T cells by secreting chemotactic factors such as CCL2 and CCL17, thus inhibiting tumor growth. 11 On the contrary, lymphocytes, especially cytotoxic T lymphocytes, inhibit tumor growth by secreting a large quantity of cytotoxic proteins and inflammatory factors, such as tumor necrosis factor α (TNF-α), perforin, interferon gamma (IFN-γ), granzyme A/B, and various interleukins. 12 The neutrophil-lymphocyte ratio (NLR), as a cost-effective inflammatory index, is closely related to the prognosis of cancer patients, 13 but its role in diagnosing bone metastasis in EC has not been studied. Therefore, we aim to investigate the diagnostic effectiveness of NLR on bone metastasis in EC.
Methods
Patients
Retrospective data was collected from January 2019 to December 2022. The data was collected from electronic medical records. Informed consent was obtained from all participants and/or their legal guardians. All methods were carried out in accordance with relevant guidelines and regulations. This study was approved by the Ethics Committee in 2015 (approval number: 2015-046). We recorded patients’ gender, age, prothrombin time (PT), prothrombin ratio (PTR), international normalized ratio (INR), APTT, thrombin time (TT), fibrinogen, D-dimer and fibrin degradation products (FDP), CEA, CA199, CA724, erythrocyte, hemoglobin, leukocyte, neutrophil, lymphocyte, monocyte and platelet.
Inclusion and Exclusion Criteria
Inclusion criteria: diagnosed with EC or bone metastasis of EC through pathological or imaging examination.
Exclusion criteria: (1) Patients with thrombotic disease, active bleeding, or coagulation dysfunction may receive anticoagulant or antiplatelet therapy; (2) Patients with a history of other tumors; (3) Patients with other severe organic diseases; (4) Laboratory examination data are incomplete; (5) Infection; (6) History of blood transfusion or hematological disease in the last three months.
Of the 727 consecutive patients, 53 patients had no pre-treatment hematological tests, 20 patients had a recent history of blood transfusion, 34 patients had a combination of serious infections on admission, and 16 patients had a history of other tumors, and ultimately 604 patients with a first diagnosis of EC were enrolled in this study.
All data were collected before any treatment was received.
Test Methods
Blood samples were collected and immediately analyzed for hematological parameters using a hematology analyzer (The Sysmex HST302 Automatic assembly line for hematology analysis, Sysmex Shanghai Ltd, China). Coagulation parameters were tested using an automated hemagglutination instrument (STA Compact Max, Stago, China). Tumor markers were measured using Roche kits (Roche, Roche R&D (China) Co., LTD).
Statistical Analysis
The data is divided into a control group and a metastasis group based on whether EC has metastasized, and ROC curves are used to determine the ability of NLR and other parameters to predict bone metastasis in EC. The Youden index is used to obtain the optimal cutoff value for each parameter. Univariate logistic regression is conducted for each parameter, and parameters with
Results
Characteristics of Patients with EC
604 patients (447 males and 157 females) were enrolled in this study. The median age was 59 years (range 51-67). The tumor markers of the metastasis group (CEA, CA199, and CA724) have significantly higher than those of the non-metastasis group; Coagulation-related indicators (PT, PTR, INR, APTT, FIB, D-dimer, FDP, except for TT) were all higher in the metastasis group than in the non- metastasis group; Besides, absolute neutrophil count increased and absolute lymphocyte count decreased, leading to a significant increase in NLR. (Table 1).
Baseline Data for Patients.

Data are shown as number of cases and percentage or median and interquartile range. CEA, carcinoembryonic antigen; CA199, carbohydrate antigen 199; CA724, carbohydrate antigen 724; PT, prothrombin time; PTR, prothrombin ratio; INR, international normalized ratio; APTT, activated partial prothrombin time; TT, thrombin time; FIB, fibrinogen; FDP, fibrin degradation products.
NLR can Better Predict Bone Metastasis in EC Patients
The ROC curve was used to predict the diagnostic effectiveness of NLR in diagnosing bone metastasis in EC patients. The best cut-off values (sensitivity, specificity, and Youden index) for CEA were 3.4 (70.3%, 66.1%, and 0.364), CA199 was 16.91 (56.3%, 73.9%, and 0.302), CA724 was 7.79 (48.0%, 79.4%, and 0.274), Neutrophil was 4.23 (47.0%, 76.0%, and 0.230), Lymphocyte was 1.425 (55.6%, 63.1%, and 0.187), and NLR was 2.043 (79.2%, 52.6%, and 0.318). The AUC for predicting bone metastasis of CEA was 0.694 (95%CI: 0.645-0.744,

ROC analysis for the prediction of bone metastasis. AUC indicates the diagnostic power of (a) NLR, (b) PT, (c) APTT, (d) FDP, (e) CEA, (f) CA724, (g) hemoglobin, (h) platelet, (i)Pre1 and Pre2, the predicted probability obtained by combining NLR and tumor markers through binary logistic regression.
Diagnostic Parameters at the maximum of Youden index for Bone Metastasis.
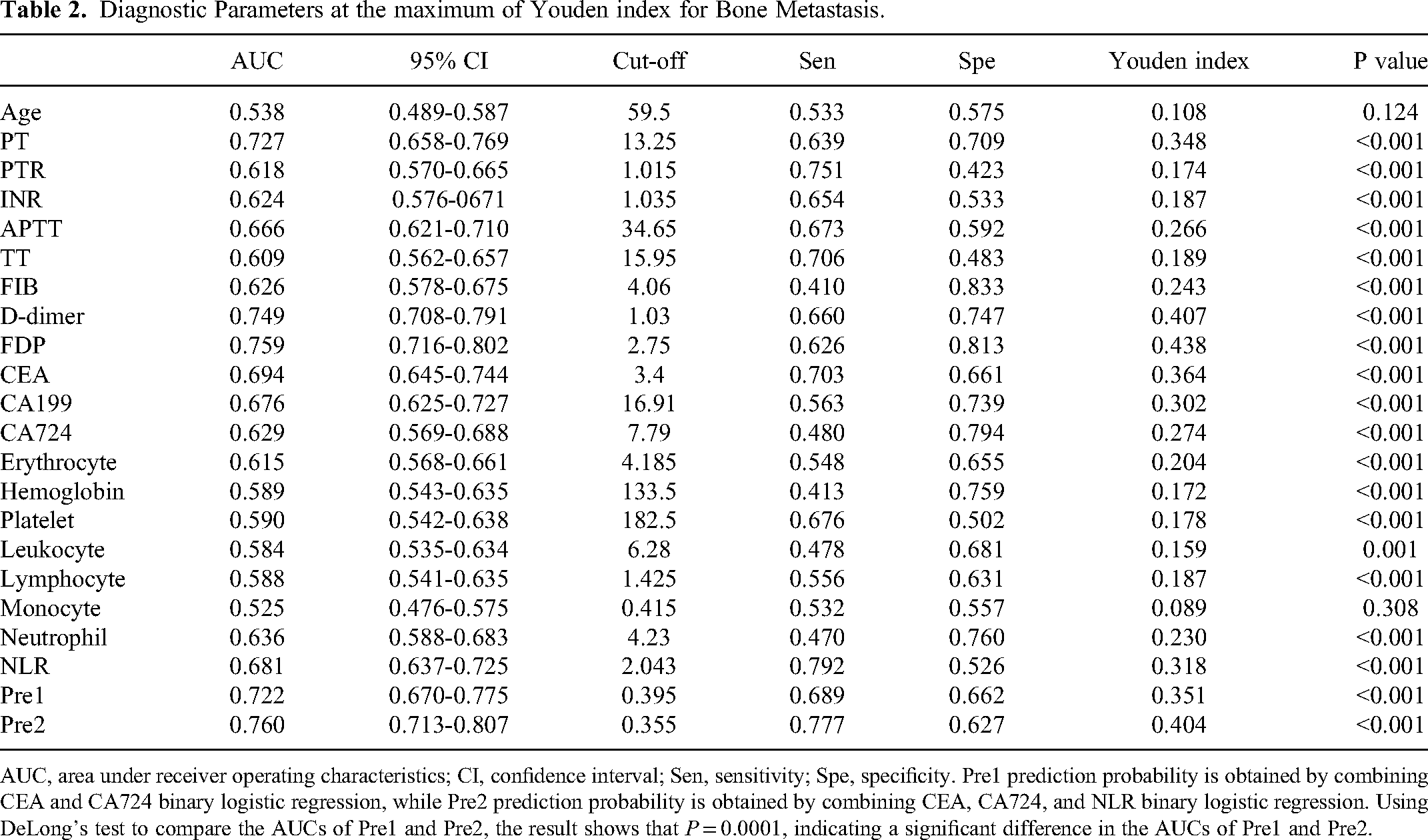
AUC, area under receiver operating characteristics; CI, confidence interval; Sen, sensitivity; Spe, specificity. Pre1 prediction probability is obtained by combining CEA and CA724 binary logistic regression, while Pre2 prediction probability is obtained by combining CEA, CA724, and NLR binary logistic regression. Using DeLong's test to compare the AUCs of Pre1 and Pre2, the result shows that
Additionally, through binary logistic regression, we obtained the predictive probabilities of NLR combined with tumor markers. Pre1 was obtained by combining CEA and CA724, while Pre2 was obtained by combining CEA, CA724 and NLR. The best cut-off values for Pre1 were 0.395 (sensitivity: 68.9%, specificity: 66.2%). The AUC for Pre1 was 0.722 (95%CI: 0.670-0.775,
NLR can be an Independent Risk Factor
We divided the clinical indicators into two groups - high and low - based on the optimal cutoff value, and the results of the multiple logistic regression showed: high NLR (OR: 2.608, 95%CI: 1.395-4.874,
Univariate and Multivariate Binary Logistic Regression Analyses of Variables for Bone Metastasis.

The reference of parameters is set to be less than their cutoff value, respectively.
Discussion
Compared to liver and lung metastasis, bone metastasis in EC is less common. 14 With the diversity of treatment options available, the overall survival of EC patients has gradually increased. 15 Additionally, due to the popularity of enhanced CT, PET-CT, bone scans and other diagnostic methods, there is an increasing number of patients with diagnosed EC bone metastasis. When bone metastasis occurs, it can cause severe bone pain, pathological fractures and other bone-related events that significantly affect the patient's quality of life, accelerating the progression of the disease. 16 Therefore, early screening methods for bone metastasis in EC patients are particularly important in extending patient survival and improving their quality of life.
In this study, we evaluated 604 patients with EC, among whom were 207 patients with bone metastasis. We found that NLR was able to predict bone metastasis in these patients with higher diagnostic accuracy than tumor markers CA199 and CA724, and a similar level of accuracy as tumor marker CEA. Combining NLR with tumor markers can significantly improve the diagnosis of bone metastasis in patients with EC. Since NLR is a non-invasive and cost-effective indicator, it can assist medical practitioners in effectively screening for bone metastasis in EC patients.
In recent years, an increasing number of studies have shown that the systemic inflammatory status of cancer is closely related to the immune cells infiltrating the local tumor environment. Many cytokines and other inflammatory cells migrate to the local tumor tissue through the bloodstream. They can promote or inhibit the development of tumors by affecting the local microenvironment of the tumor.17,18 This also indicates that the number of inflammatory cells in a patient's circulation may indicate whether the patient has tumor metastasis and their prognosis. As the neutrophil with the highest content in peripheral blood, it promotes tumor progression through multiple mechanisms. 9 The ROS produced by neutrophils can amplify cell DNA damage when exposed to carcinogens, thereby promoting tumor development. 19 In the tumor microenvironment, the crosstalk between neutrophils and tumor cells is very important for tumor progression. Studies have found that neutrophils inhibit T cell homing and anti-PD1 immunotherapy, and promote epithelial-mesenchymal transition of lung cancer cells, increasing the secretion of tumor CXCL2, which in turn promotes neutrophil infiltration and maintains the immune-exclusion tumor microenvironment. 20 Furthermore, it has been proven that tumor-associated neutrophils can promote extracellular matrix remodeling, triggering the release of growth factors such as alkaline fibroblast growth factor and vascular endothelial growth factor, thereby promoting angiogenesis and dissociation of tumor cells.10,21
When combined with lymphocyte count reduction, the predictive role of tumor progression may be further enhanced. 22 The progression of many malignant tumors is related to impaired lymphocyte function and reduced lymphocyte counts. 23 In breast cancer, the absolute lymphocyte count gradually decreases with the progression of metastatic breast cancer. Monitoring changes in lymphocyte count in peripheral blood is a potential indicator for predicting recurrence. 24 In addition, peripheral blood lymphocyte counts are significantly positively correlated with Tumor-infiltrating lymphocytes infiltrating tumor tissue locally. When the lymphocyte count in peripheral blood of EC patients decreases, Tumor-infiltrating lymphocytes significantly decrease as well, and the overall survival rate of patients decreases, suggesting that systemic immune capacity may affect the prognosis of patients through local immune responses. 25
The mechanism underlying the association between high NLR and bone metastasis in tumor patients is still poorly understood. Due to the discovery that tumors often originate from chronic inflammatory sites, the connection between cancer and inflammation is gradually being emphasized. 26 In some cancers, long-term chronic inflammatory stimulation promotes tissue carcinogenesis, while in other cancers, tumor infiltrating inflammatory cells can promote angiogenesis, stimulate tumor cell proliferation, and support cancer cell metastasis and spread. 27 Although further research is needed to explore the complex mechanisms between cancer and circulating inflammation, multiple studies have supported the close correlation between high NLR and tumor metastasis.28,29
In our study, we found that NLR > 2.043 is an independent predictor for bone metastasis in EC, with a sensitivity of 79.2% and a specificity of 52.6%. Many studies have explored the impact of NLR on the metastasis and prognosis of EC, and the optimal cutoff value varies from paper to paper. Zhiwei Zheng et al 30 found that NLR > 2.20 is an independent predictor for disease-free survival in resectable squamous cell carcinoma patients. Fuyan Han et al 31 found that pretreatment NLR > 1.88 is a reliable biological marker for diagnosing and predicting prognosis of EC. Despite variations in patient characteristics and optimal cutoff values of NLR in different reports, multiple studies have shown that NLR can serve as an independent predictor for diagnosing EC metastasis and prognosis.30,31 However, previous studies have not yet combined NLR with traditional tumor markers to further explore the predictive value of combined markers for tumor metastasis. In our study, we found that the diagnostic efficiency of combining NLR with tumor markers (NLR, CEA, CA724) was significantly higher than that of combining only tumor markers (CEA, CA724) (P = 0.0001). Therefore, when the critical value is > 2.043, we should be cautious about bone metastasis in EC, especially the occurrence of micro metastasis, and actively combine other biomarkers such as CEA and CA724 to detect bone metastasis in EC in the early stage.
The following limitations of our research should be taken into consideration. Firstly, this is a retrospective study. In manually entered data, we excluded data from incomplete laboratory tests, patients with multiple types of cancer, and patients with severe organic lesions, which may pose a risk of patient selection bias; Secondly, as a single center study, although our research results have certain limitations, all consecutive patients with EC who visited our department during the above period were included in the analysis and strictly followed the inclusion and exclusion criteria, and our sample size is sufficient. Therefore, it is concluded that NLR combined with tumor markers (CEA and CA724) has predictive significance for bone metastasis in EC and could guide clinical application. Finally, we included fewer factors in our analysis, and should have further included the results of patients’ TNM stage, degree of differentiation, and so on, which would have allowed us to understand whether the NLR is indicative of the progressive state of EC and whether it gradually rises as the tumor progresses. This study does not account for gender-specific variations, which can affect diagnostic methods. Future research should include gender-specific data to enhance the accuracy and applicability of the prognostic method.
Conclusion
Overall, our research found that NLR, as a convenient, non-invasive, and cost-effective inflammatory indicator, could predict bone metastasis in EC. Moreover, when NLR>2.043, further examinations such as tumor markers and imaging examination should be combined to detect bone metastasis of EC as early as possible.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241272043 - Supplemental material for Predictive Role of Elevated Neutrophil-Lymphocyte Ratio for Bone Metastasis in Esophageal Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338241272043 for Predictive Role of Elevated Neutrophil-Lymphocyte Ratio for Bone Metastasis in Esophageal Cancer by Xuan Wang, Ying Gao, Junyang Wang, Ling Chen, Xin Zhang, Min Chen, Ning Lan, Wenyang Li, Fang Wu and Juan Ren in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
Thanks to Doctor Dongmei Diao from the First Affiliated Hospital of Xi’an Jiaotong University for her guidance on our research.
Data Availability Statement
Dataset available from the corresponding author at<869491533@qq.com>.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This study was approved by the Ethics Committee of First Affiliated Hospital of Xi’an Jiaotong University (Approval No. 2015-046).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript is supported by the Scientific and Technological Research Foundation of Shaanxi Province, Key Research and Development Project, General project (JR, 2023-YBSF-666); supported by the Shaanxi Provincial Key Research and Development Plan Project (Key support projects Category A) (JR, 2021A011); supported by the Basic and Clinical Integration Innovation Project of Xi’an Jiao tong University (JR, YXJLRH2022006, YG, YXJLRH 2022004); Nanjing Tianqing Scientific Research Fund of First Affliated Hospital of Xi’an Jiaotong University (JR, 2023, TQ202205).
Informed Consent/Patient Consent
Informed consent was obtained from all participants and/or their legal guardians.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
