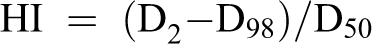Abstract
Objective:
To examine the accuracy and efficiency of breast radiotherapy after breast-conserving surgery of a novel 3-dimensional (3D) printing tissue compensator technology, the 3D-precise breast conformer, compared with a usual compensator and an unstructured compensator.
Methods:
This novel device is patented in China (patent No.: ZL2015 2 0259472.9). Thirty patients with breast cancer after breast-conserving surgery were randomly divided into 2 control groups (no compensator, NST group, and usual compensator, ST group) and 1 study group (3D-precise breast conformer, 3D-BCT group) (n = 10/group). Before radiotherapy, all patients were scanned in the same CT positioning conditions to prepare the treatment plans.
Results:
The 3D-BCT showed the best homogeneity index (HI) (0.08 ± 0.03) and conformity index (CI) (0.95 ± 0.03), while the NST group showed the worst HI (0.34 ± 0.07) and CI (0.78 ± 0.06), with the ST group between the 2 (HI: 0.15 ± 0.05; CI: 0.87 ± 0.04) (all P < 0.01). The common tissue compensation membrane could lead to 95-100% of the prescription dose covering 85-95% of the target volume, and the uniformity and conformability of the target dose were improved overall compared with the NST group. In the 3D-BCT group, 100% of the prescription dose covered the target volume of 95-100%.
Conclusion:
The 3D-precision breast conformal device had the highest individualization, uniformity, and conformity. The V95, V98, CI, and HI of PTV were optimal in the 3D-BCT group, and an ideal isodose curve distribution of the breast and clavicle upper and lower target areas was achieved. This device could improve the surface dose and the efficacy of radiotherapy after breast-conserving surgery.
Keywords
Introduction
Breast cancer is the most common malignant tumor in women 1,2 The estimated incidence of breast cancer was 2,088,849 new cases in 2018, for an age-adjusted incidence of 46.3 per 100,000 women and a cumulative lifetime risk of 5.0% [2]. Most (95%) of the cases occur in women >40 years of age. 3,4 About 62% of the patients are diagnosed at the early stage of the disease. 1 The management of breast cancer is multidisciplinary and involves surgery, chemotherapy, hormonal therapy, targeted therapy, and radiation therapy. 4 In recent years, the survival rate of the patients has greatly increased. 5
Breast-conserving surgery (BCS) is the surgical option of choice in eligible patients. 6 Radiation therapy after BCS is widely used. 4 At present, BCS, combined with postoperative radiotherapy, has become the standard treatment for early and mid-stage breast cancer. 4 Three-dimensional (3D) conformal radiotherapy (3D-CRT) and intensity-modulated radiotherapy (IMRT) have improved the efficacy and control probability of breast cancer. 7,8
Due to the gross shape of the breast, its poor fixation and reproducibility, and respiratory motion, it is easy to have an obvious target movement during radiotherapy. 9 Nowadays, there is no fixation technology that has been recognized by radiation experts. 10 In addition, due to the inherent characteristics of radiations, the subcutaneous tissues often receive an insufficient dose in the target area. Nowadays, radiotherapy tissue compensators can effectively increase the radiation dose covering the skin and subcutaneous tissue. 11 With the use of an appropriate tissue compensator, you can increase the 6-MV X-ray skin dose from 10-40% to nearly 100%. 12 The usual compensator is a square equivalent tissue compensation membrane with a predefined size, thickness, and softness, but the human body surface has an irregular curvature, and there might be air gaps between the membrane and the body surface, significantly reducing the surface dose of the target volume during radiotherapy. 12 Therefore, reducing the air gap should improve the dose uniformity of the target area and the accuracy of radiotherapy. 12 -14
3D laser scanning and printing technology was introduced in 1988; the first SLA250 additive metal production based on a photo-solid process produced by 3D systems belongs to non-traditional processing technology. 5 It directly processes materials into finished products through additive metal production. It is especially suitable for the fabrication of complex products with multiple surfaces, asymmetry, and exquisite internal structure required in medicine. 15 -17 With the development of digital medicine, 3D printing technology has been widely used in the fields of stomatology, surgery, and radioactive particle implantation. 15 -17
Therefore, the aim of the present pilot study was to examine the accuracy and efficiency of radiotherapy of a novel patented 3D printing tissue compensator technology, the 3D-precise breast conformer, compared with a usual compensator and an unstructured compensator.
Methods
Production of the New 3D-Precise Breast Conformal Device
This novel device is patented in China (patent No.: ZL2015 2 0259472.9). The conformer is composed of a breast, a sub-clavicle, and supra-clavicle radiotherapy target volume covering membrane (chest membrane) and a fixing device (tight-fitting garment). The process of making the chest membrane begins with obtaining the 3D data of the patient’s body surface using a hand-held 3D-laser scanner (Go Scan50, Creaform, Canada) to scan the bilateral breast and chest wall of the patients after BCS (Figure 1). Data processing and the mold design of the chest membrane are completed by computer-aided design. Finally, the chest membrane mold processing is completed using 3D laser printing technology. The mold is made by 3-dimensional scanning and sizing printing according to the size and contour of each patient’s breast and chest wall. The compensation material such as silica membrane or epoxy resin is dissolved and injected into the mold, and the thickness is controlled according to the clinical requirements, which achieves the precise production process of the individualized chest membrane for each patient.
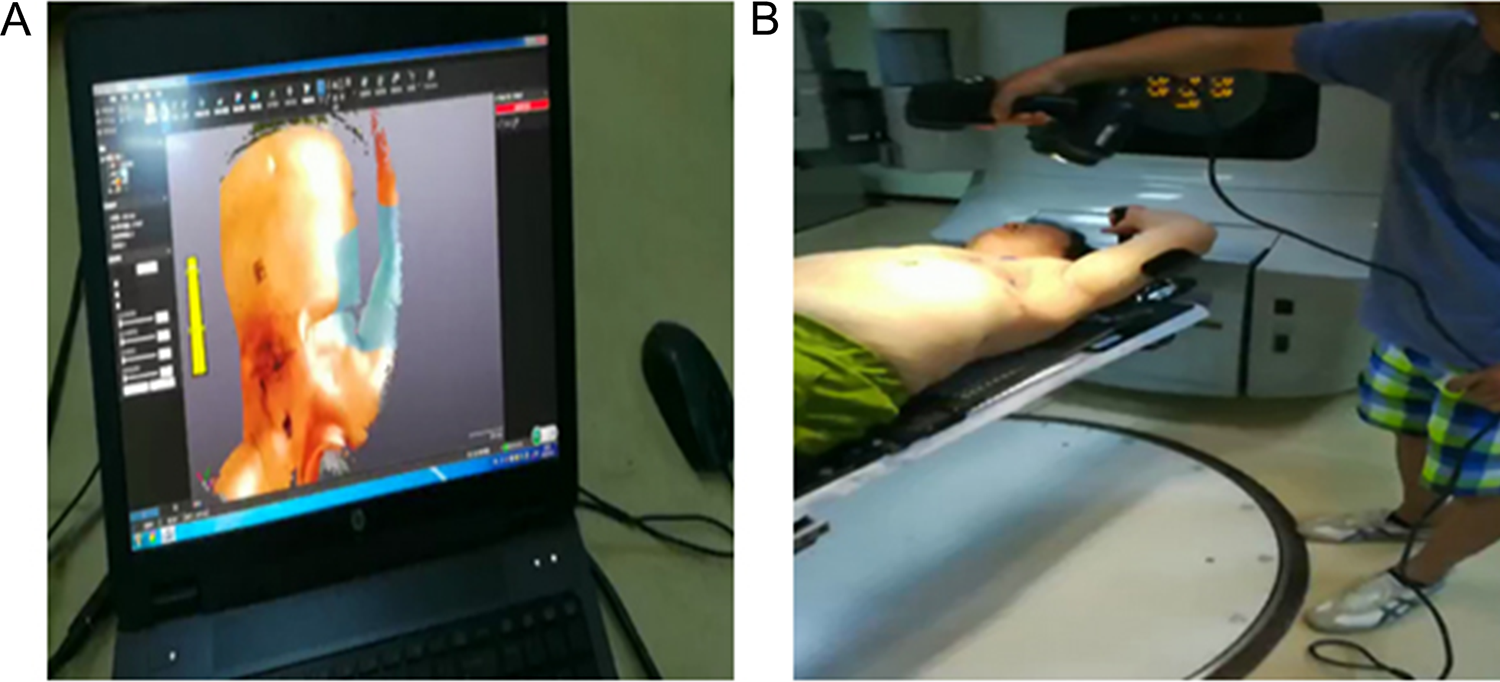
3D data image (A) of the 3D scan of a patient on a treatment bed (B).
Technical Material Selection and Tight-Fitting Garment Production
The device is manufactured by Kelary Co. (Guangzhou, China). The chest membrane is made of silica gel, epoxy resin, or paraffin (mixed) with a tissue-equivalent material density of 1.034 or 0.987 g/cm, 3 according to 3D laser scanning, shaping, and printing. A tissue compensator suitable for the profile of the patient’s breast and chest wall is fabricated, and the processed tissue compensator (chest membrane) is then placed under the treatment device (tight-fitting mammary garment) similar to female sports underwear made of a specific material (Figures 2 and 3). Before radiotherapy, the chest membrane was accurately placed to ensure that there was no gap between the mold and the skin. Finally, the zipper of the tight-fitting garment is pulled and sealed, and then the positioning and treatment can be started. The design and manufacture of the pleural fixation device (tight-fitting garment) is based on the measurement of each patient’s body surface, including parameters such as chest circumference, neck circumference, and length, and tailored to achieve individualized design and production. The characteristics of the nanomaterials are high elasticity, comfort, individuality, durability, easy to put on and take off, and self-disassembled chest membrane and tight-fitting garment. When the patient wears this personalized, tailored feminine underwear during treatment, the characteristics enable the chest membrane to fit the patient’s breast surface and chest wall closely and fit perfectly between the chest membrane and the breast and chest wall, so its conformation can reach 100%.
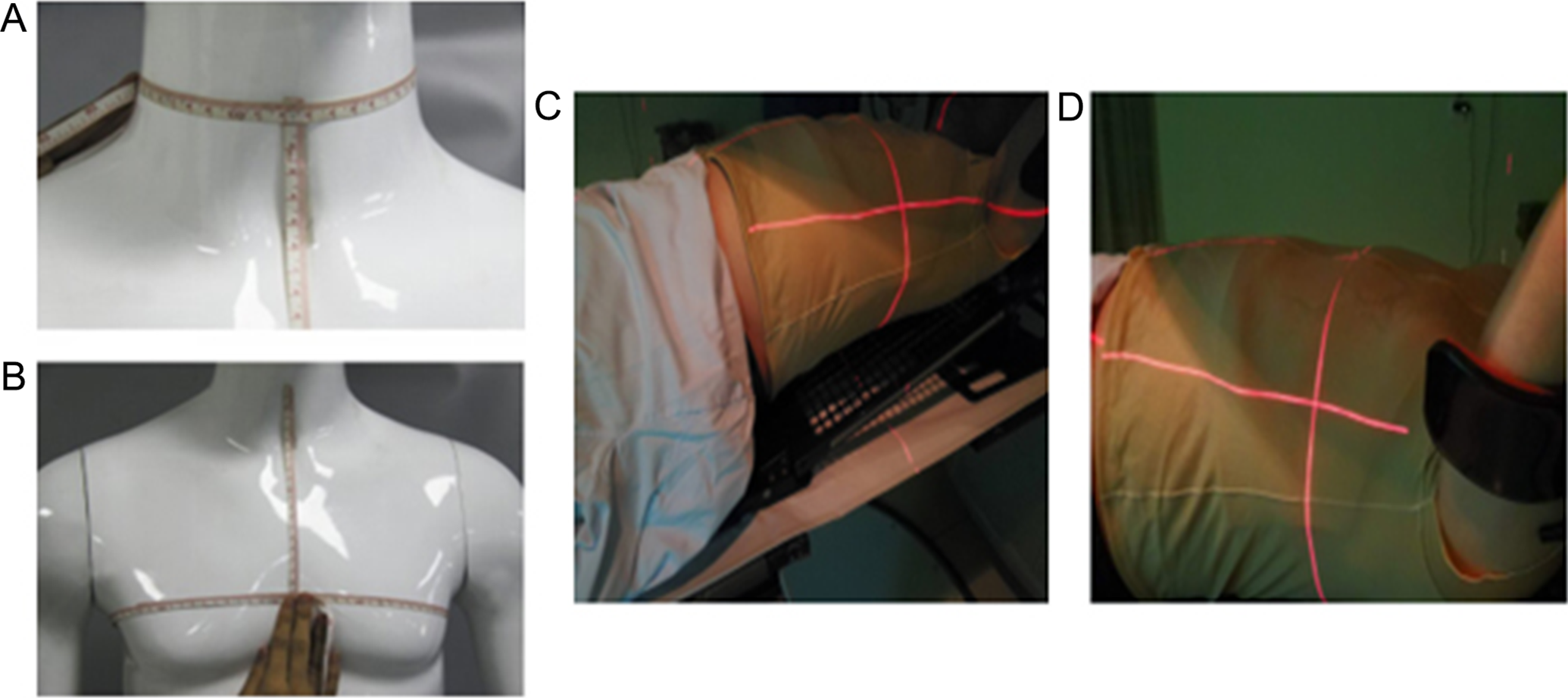
Neck (A) and chest circumference measurement (B) before the tight-fitting garment is made, tailored and placed on the patient’s individualized tight-fitting garment before treatment (C, D).

3D-scanning and printing based on the 3D scanning data of the whole breast and anterior chest wall (A) and the tight-fitting garment (B).
Study Design
Thirty patients with breast cancer who underwent BCS radiotherapy and at the Fourth Hospital of Harbin Medical University from January 2016 to February 2018 were enrolled. The study protocol was approved by the hospital ethics committee, and the patient’s signed an informed consent form prior to treatment.
The inclusion criteria were: 1) underwent BCS and radiotherapy; 2) postoperative pathology confirmed that there was no residual cancer at the surgical margin and that the number of axillary or sentinel lymph nodes was ≥1; 3) the irradiation range included the breast and supraclavicular and subclavicular areas and chest wall; 4) pathological stage I-III; and 5) KPS ≥80.
Placement Method
The first control group did not use any compensator (NST group). The second control group used the usual commercial block type tissue compensation membrane (ST group). The 3D-CBT group received the 3D-precise breast conformer. All patients were placed supine on a breast mount in a 64-row computed tomography (CT) positioning bed (Philips, Best, The Netherlands) to adjust the position of the headrest. The affected side was held by a handle, and the upper limb of the affected side was abducted. The angle, length, height, and position of the handle were adjusted according to the actual situation so that the axilla was fully exposed and to avoid the upper limb from being irradiated. Before positioning, the compensator was placed according to grouping. Finally, the position parameters were recorded to ensure the repeatability accuracy for each treatment. Then, with the help of the 3D laser positioning system (handyscan700, CREAFORM, Canada), the reference line of the anterior chest sagittal body surface (located in the midline of the body) and the reference line of the coronal line and the cross-section on both sides of the patient were marked on the body surface, that is, 3 lines from different dimension are marked on the patient’s body (cross “+”). CT scanning was performed from the cranial side of the cricothyroid membrane to 10 cm caudal from the sub-mammary fold, covering the whole assumed planning target volume (PTV) area and the whole lung volume.
Delineation of the Target Volume, Organ at Risk (OAR), and Radiotherapy Planning
The CT scan images from the ECLAPSE radiotherapy planning system (Varian Associates Inc., Palo alto, California, USA) were used to delineate the clinical target volume (CTV) in the mediastinum according, including the whole breast, sub- and supra-clavicle, and chest wall. The PTV was 5.0 mm underneath the CTV in the 3-dimensional direction; at least 95% of the PTV volume was subject to 95% of the prescription dose. The bilateral lung tissue, heart, liver, contralateral breast, ipsilateral humerus, and cervical spinal cord were defined as important critical tissues and organs. The tumor was given a single prescription dose of 2.0 Gy, and the total prescribed dose was 50 Gy/25 fractions. In the end, the tumor bed was boosted up to 60 Gy. The V20 of the affected lung tissue was less than 25%, the V40 of heart in patients with left breast cancer was less than 5%, and the average liver dose was less than 5 Gy.
PTV: maximum dose Dmax <55 Gy, minimum dose Dmin >48 Gy, 51 Gy dose volume V51 ≤4%, prescription dose volume V50 ≥95% PTV
Normal tissue: contralateral side V30 ≤8%, V20 ≤10%, V5 ≤50%, heart V40 ≤15%, V30 ≤20%, V20 ≤25%, V10 ≤30%, V5 ≤50%, the contralateral breast V3 ≤50%, V5 ≤10%, Dmax <6 Gy; spinal cord ≤40 Gy.
Ensure that the PTV uniformity is optimal.
Treatment Plan Evaluation
Using the “double 95” standard (95% isodose curve 47.5 Gy wrapped around 95% of the target volume), the optimal parameters and comparative analysis of the dose parameters of the PTV and organ at risk (OAR) are based on the Dose Volume Histogram (DVH). The target volume evaluation index CI was calculated as CI = (VTref/VT)×(VTref/Vref); HI = (D2-D98)/D50 , where D2 refers to the dose corresponding to 2% of the target volume on the DVH, and D98 refers to the dose corresponding to 98% of the target volume on the DVH, the prescribed dose is given by D50, PTV compares V95, V98, V105, V110, Dmean , and Dmax. The DVH curves were compared between the 3 planned PTVs.
In order to compare the target dose, the target dose CI and the homogeneity index (HI) were defined as follows:
CI is between 0-1. The larger the value, the better the conformality. The smaller the HI value, the better the uniformity
Statistical Methods
SPSS 22.0 (IBM, Armonk, NY, USA) was used for all analyses. The continuous data were tested for normal distribution using the Kolmogorov-Smirnov test and were presented as means ± standard deviations and analyzed using ANOVA with Tukey’s post hoc test (normal distribution) or with medians (ranges) and the Wilcoxon signed-rank test (skewed distribution). Categorical data were presented as numbers and frequencies and were analyzed using Fisher’s exact test. P-values <0.05 were considered statistically significant.
Results
Characteristics of the Patients
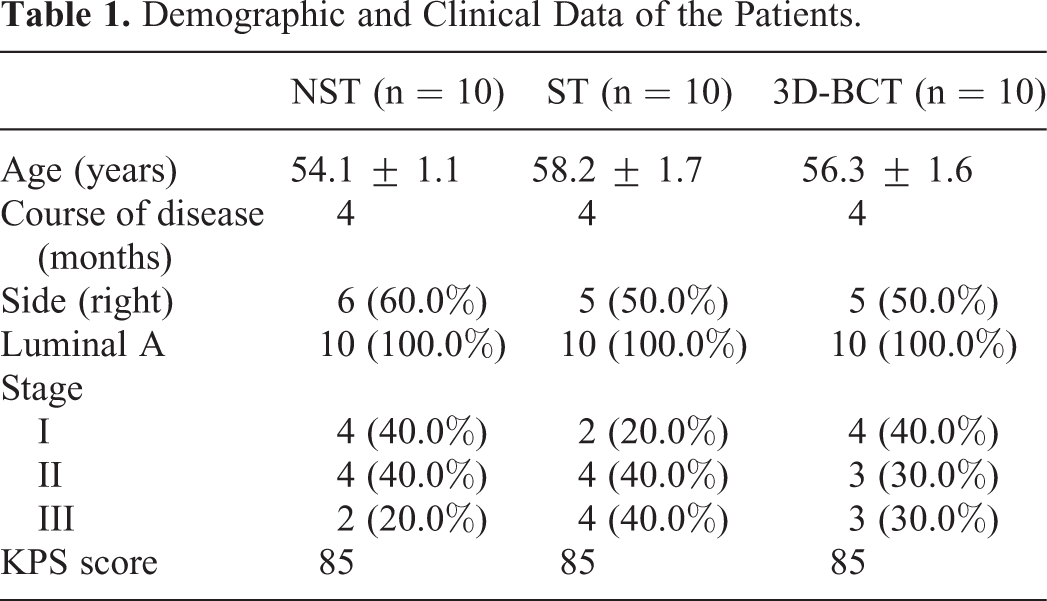
Table 1 presents the characteristics of the patients. The mean age of patients in the NST, ST and 3D-BCT groups were 54.1 ± 1.1, 58.2 ± 1.7, and 56.3 ± 1.6 years, respectively. All patients had luminal A breast cancer, stage I-III, with a disease course of 4 months. There was no statistical difference in demographic and clinical characteristics among the 3 groups.
Demographic and Clinical Data of the Patients.
Characteristics of the Treatment Plans
Table 2 shows that the target volume, average dose, minimum dose, and the highest dose of the target volume obtained by the optimization of the conformal intensity-modulation plan among the 3 techniques were significantly different (all P < 0.01). These differences lead to different CI and HI among the 3 groups (all P < 0.01) (Table 2). The HI was the smallest and CI the highest in the 3D-BCT group. The doses of the OAR in the 3 fixation technique were within the required specifications.
Comparison of 3 Planning Target Volume Parameters in 30 Breast Cancer Patients.
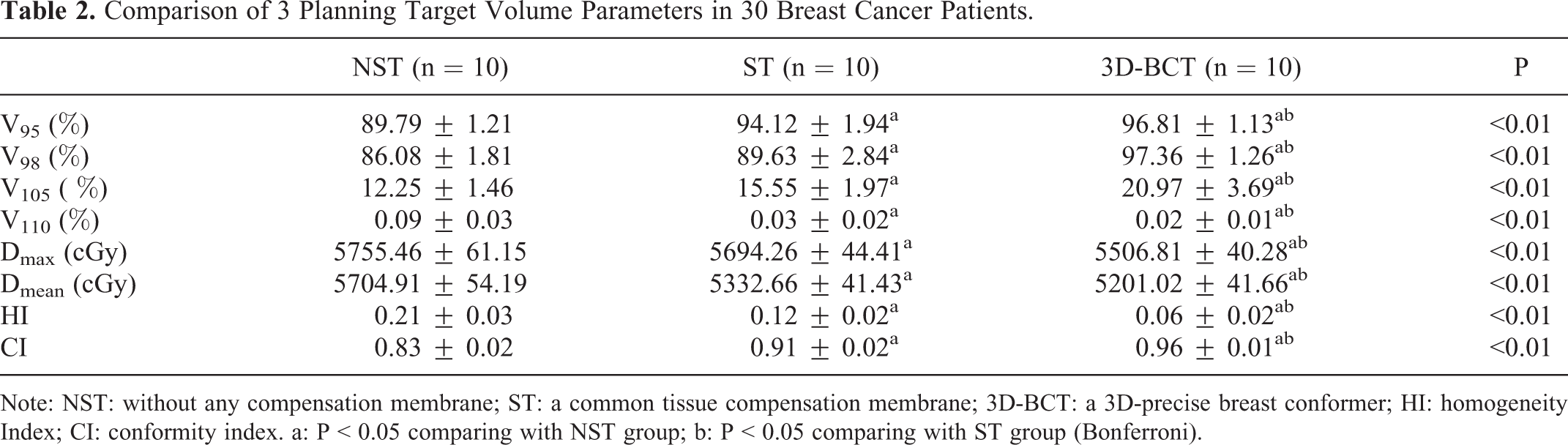
Note: NST: without any compensation membrane; ST: a common tissue compensation membrane; 3D-BCT: a 3D-precise breast conformer; HI: homogeneity Index; CI: conformity index. a: P < 0.05 comparing with NST group; b: P < 0.05 comparing with ST group (Bonferroni).
The dose uniformity and conformity of the target volume were the worst in the NST group, and the slope of the curve was the largest (Figure 4). The uniformity of breast dose distribution in 10 patients is 95% of the prescription dose only covering 80-90% of the target volume. The common tissue compensation membrane could lead to 95-100% of the prescription dose covering 85-95% of the target volume, and the uniformity and conformability of the target dose were improved overall compared with the NST group (Figure 5). In the 3D-BCT group, 100% of the prescription dose covered the target volume of 95-100% (Figure 6). It can be concluded that the average dose coverages obtained by the 3 schemes in the 3 modes are different under the same prescription dose. The target volume V95 is the most optimal for the 3D-BCT group.

The target dose of the planning target volume (PTV) in the dose-volume histogram (DVH) (B) of the patient without compensator (A) was only 85.87% of the target volume. The target volume planned to be PTV had poor uniformity (HI = 0.21) and conformality (CI = 0.84), and the curve slope was large.

In the second control group, a usual tissue compensator was used (A, B), and 97% of the prescription dose of the target planning target volume (PTV) in the dose-volume histogram (DVH) (C) reached the target volume of 95.64%. The PTV uniformity (HI = 0.12) and conformality (CI = 0.91) were better than in the first control group.

Using the new 3D-precise breast conformal device (A, B) to eliminate the gap between the membrane and the skin completely, 100% of the prescription dose of the planning target volume (PTV) in the dose-volume histogram (DVH) (C) could be wrapped around 97.68% of the target volume, and the PTV was evenly distributed. The homogeneity (HI = 0.06) and the conformality (CI = 0.96) were the best (the curve is steep).
Discussion
Available tissue compensators have the disadvantage of possible air gaps that reduce the efficiency of radiotherapy. 12 -14 In addition, reproducible positioning from 1 radiotherapy session to the next is a major issue because the breast is an exterior organ. 9 Therefore, this study aimed to examine the accuracy and efficiency of breast radiotherapy after breast-conserving surgery of a novel 3-dimensional (3D) printing tissue compensator technology, the 3D-precise breast conformer, compared with a usual compensator and an unstructured compensator. The results of this pilot study suggest that the 3D-precision breast conformal device had the highest individualization, uniformity, and conformity. The V95, V98, CI, and HI of PTV were optimal in the 3D-BCT group, and an ideal isodose curve distribution of the breast and clavicle upper and lower target areas was achieved. This device could improve the surface dose and the efficacy of radiotherapy after breast-conserving surgery.
In the present study, the individualized compensator was manufactured after the precise 3D scanning of the patient. Perkins et al. 18 examined individualized tissue compensators for electron beam irradiation after modified radical mastectomy for breast cancer and showed improved local dose distribution and reduced dose to the heart and lung. Park et al. 14 studied and evaluated the feasibility and advantages of a patient-specific breast compensator made by a 3D printer technique. They concluded that the 3D-printed solid compensator could reduce the uncertainty of the daily setup and help improve the dose discrepancy by unwanted air gaps in breast cancer radiation therapy. Park et al. 19 showed that 3D tissue compensator achieved better dose coverage in patients after total mastectomy. Craft et al. 20 examined the value of a 3D printed tissue compensator for electron beam radiotherapy and showed better dose coverage in phantoms. Importantly, the present study showed that a 3D printed tissue compensator designed from 3D scanning data achieved good outcomes in patients who underwent BCS, while the other previous studies 14,19,21 were performed in patients who underwent mastectomy. In an intrapatient study, Robar et al. 21 showed that a 3D printed tissue compensator achieved better planning outcomes than a standard vinyl gel sheet bolus in patients after mastectomy. After mastectomy, the chest is flatter than after BCS and the compensator is easier to be fitted than around a round mammary gland. Nevertheless, the present study indicates that it is indeed possible.
In the present study, the isodose curve of the target volume in the control group without tissue compensation membrane could not meet the basic principle of radiotherapy, that is, 95% of the prescription dose wrapped around 80-90% of the target volume. The control group, with the usual tissue compensation membrane, showed that 95-100% of the total prescribed dose reached 85-95% of the target volume. Wilks and Bliss. 22 showed that the use of a compensator could reduce dose inhomogeneity in radiotherapy of the breast. In the study group that used the novel 3D-precision breast conformer, the distribution curve showed that 100% of the total prescribed dose could encompass 95-100% of the target volume. Therefore, the patients who received the patented product (3D-precision breast conformer) achieved the most optimal parameters on the highest dose of the radiotherapy target volume (upper and lower breast and clavicle), the average dose of the target volume, the lowest dose of the target volume, the target CI, and the target HI, followed by the control group with the market-equivalent tissue compensation membrane, and the useless tissue compensation membrane group was the worst. Considering the improvements in 3D printing technologies and the ever reducing costs, such 3D printed compensators represent a good prospect. 23
Of course, this was only a pilot study in a small number of patients from a single center. Patient outcomes, toxicities, and ease of setup were not examined. Nevertheless, this pilot study could be considered a proof-of-concept study for the planning of larger multicenter studies.
In conclusion, the 3D-precision breast conformal device had the highest individualization, uniformity, and conformity. The V95, V98, CI, and HI of PTV were optimal in the 3D-BCT group, and an ideal isodose curve distribution of the breast and clavicle upper and lower target areas was achieved. This device could improve the surface dose and the efficacy of radiotherapy after breast-conserving surgery.
Footnotes
Authors’ Note
Our study was approved by Medical Ethics Committee of the Fourth Affiliated Hospital of Harbin Medical University (approval no. 2020-WZYSLLSC-02). All patients provided written informed consent prior to enrollment in the study. Chunbo He conceived and coordinated the study, designed, performed and analyzed the experiments, wrote the paper. Shilin Zhang, Lei Shi carried out the data collection, data analysis, and revised the paper. All authors reviewed the results and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.