Abstract
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide with non-small cell lung cancer (NSCLC) accounting for 85% of all lung cancers. 1 Surgery is the most effective treatment for patients with resectable NSCLC. However, only 20% to 35% can be treated through resection. 2 Compared with surgery alone, adjuvant platinum-based chemotherapy can further reduce the risk of disease recurrence and prolong survival in patients with stage II-IIIA or partial high recurrence risk stage IB NSCLC after complete resection. Evidence suggests that platinum-based dual chemotherapies can increase the 5-year overall survival (OS) rate by approximately 5%. 3 Thus, new modes of perioperative therapy need to be explored urgently.
Perioperative targeted therapy involves primarily locally advanced NSCLC with epidermal growth factor receptor (EGFR) mutations and can be divided into adjuvant targeted therapy and neoadjuvant targeted therapy. 4 Among these, more research exists on adjuvant targeted therapy, and EGFR mutation-positive (EGFRm) patients are the main target population for adjuvant targeted therapy. Osimertinib, an adjuvant therapy in the ADAURA trial focusing on stage IB-IIIA patients with EGFRm NSCLC, has received FDA approval. 5 New adjuvant targeted therapies have limited data available, and the phase II randomized study EMERGING-CTONG1103 is the largest published trial for neoadjuvant tyrosine kinase inhibitor therapy. 6
Perioperative immunotherapy has been incorporated into the NCCN Clinical Practice Guidelines for Oncology (NCCN Guidelines) and is categorized into adjuvant and neoadjuvant immunotherapy approaches. 7 Immunotherapy research is focused primarily on immune checkpoint inhibitors (ICIs), such as the programmed death 1 (PD-1)/programmed death ligand 1 (PD-L1) inhibitors. Current and completed trials for adjuvant immunotherapy specifically target each major ICI. A notable large-scale adjuvant immunotherapy trial is the Phase III IMpower010 trial. Although the OS results were unavailable at the time of publication, atezolizumab adjuvant therapy might receive approval for treating stage II-IIIA NSCLC patients with PD-L1 expression ≥1%. 8 Neoadjuvant immunotherapy represents a significant advancement in perioperative immunotherapy. Its primary goal is to shrink tumors, increase the likelihood of complete resection, and target micrometastases. We divided our research on new adjuvant immunotherapies into single-arm trials and randomized controlled trials (RCTs). Among these, the CheckMate816 trial is the first to receive FDA approval for a new adjuvant immunotherapy and is suitable for resectable NSCLC patients with any PD-L1 status. However, most benefits have been observed in stage IIIA or patients with PD-L1 expression ≥50%.9–12
Our study included single-arm trials and RCTs involving adjuvant and new adjuvant immunotherapies. The differences observed are summarized in measurements such as major pathological response (MPR), pathological complete response (pCR), OS, disease-free survival (DFS), event-free survival (EFS), and treatment-related adverse events (TRAEs).
Materials and Methods
The present study was conducted according to the 2020 Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. 13
Search Strategy
Our data sources included PubMed, OVID, Web of SCI, Embase, Cochrane Library, and the Chinese National Knowledge Infrastructure databases, as well as ASCO conferences and the European Society for Medical Oncology (ESMO) conferences. The detailed search strategy of each database was shown in Supplemental Table 1 (Table S1). The main search terms included “NSCLC,” “neoadjuvant therapy,” “adjuvant therapy,” and “immunotherapy” with the restriction of “clinical trials” and “RCTs.” Literature published within the last 10 years was searched. We screened the reference lists of all retrieved full-text articles to identify potentially relevant studies. For literature presented at ASCO and ESMO conferences, we extracted data from immunotherapy-related publications presented at these conferences.
Study Selection
The inclusion criteria for the selected studies were as follows: (1) patients with pathologically confirmed stage I-III NSCLC who underwent surgical resection in the trials; (2) RCTs or single-arm trials involving perioperative neoadjuvant or adjuvant immunotherapies; (3) sufficient data available for quantitative meta-analysis of at least 1 outcome measure; (4) summaries of the latest research findings reported at ASCO and ESMO conferences. For each study included, relevant data were extracted, including trial name, publication year or conference report, primary author, trial phase, number of participants, pathological staging, patients who underwent resection, and outcomes such as MPR, pCR, TRAE, OS, DFS, and EFS. Two researchers (YMW and LH) independently reviewed each retrieved article and conducted data extraction. One researcher performed the data analysis, and the other handled the graphical work.
The exclusion criteria were as follows: (1) letters, case reports, reviews, editorials, commentaries, duplicates, observational studies, or nonclinical studies; (2) studies with irretrievable or insufficient data for statistical analysis; (3) studies written in languages other than English; and (4) low sample size (n < 10).
Data Extraction
We extracted the corresponding information for each trial, including the trial name, year of publication or conference presentation, first author, trial phase, number of participants, treatment regimen, and outcomes (such as survival and toxicity).
Statistical Analysis
We used Stata software (version 16.0) for data analysis. For the included studies, which consisted of either RCTs or single-arm trials, we analyzed data related to patients’ OS, DFS, and EFS using hazard ratios (HR) and 95% confidence intervals (CIs). We summarized and analyzed the MPR, pCR, and TRAE data using odds ratio (OR) and the 95% CI. We conducted an I2 test to assess the heterogeneity, where I2 statistics estimate the percentage of total variation across studies due to heterogeneity rather than chance. If P > 0.1 or I2 < 50%, assuming no heterogeneity, we used a fixed-effects model (Mantel-Haenszel method). Conversely, if there was heterogeneity, we employed a random-effects model. Thresholds for low, moderate, and high heterogeneity based on I2 values were 0% to 49%, 50% to 74%, and ≥75%, respectively. A P-value ≤.05 was considered statistically significant. Subsequently, we conducted bias tests using funnel plots and analyzed the reasons for any observed bias. Finally, we performed sensitivity analyses to assess the impact of each study on the overall ES.
Results
Literature Search Results
The detailed study selection process is shown in Figure 1. In total, 3079 references were identified with the search strategy. After the screening, 991 references required comprehensive evaluation. After further assessment, 935 non-single-arm tests or non-RCTs were excluded. Finally, 17 studies were included,14–30 which consisted of 11 RCTs and 6 single-arm trials. Table 1 summarizes the characteristics of the patients, tumors, and treatment regimens for each trial. Table 2 is a summary of the population baseline based on published experimental data.
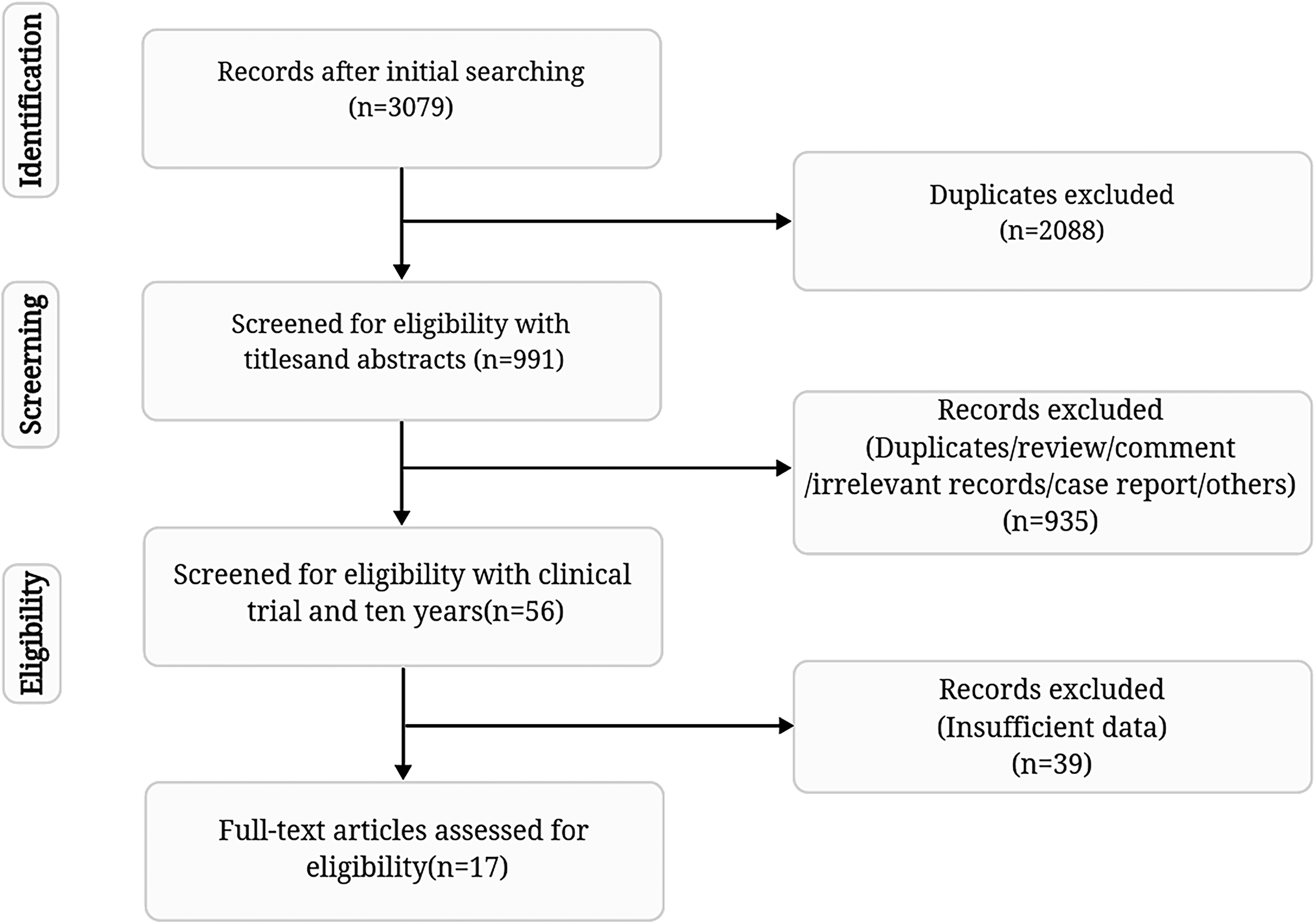
Overview of the study search and selection.
The Characteristics of the Patients, Tumors, and Treatment Regimens for Each Trial.

Summary of the Population Baseline.

Quantity and Quality of the Literature
Two authors (YMW and LH) independently extracted data and assessed the quality of the included RCTs using the Cochrane Risk of Bias (RoB) 2 tool. Discrepancies were resolved by consensus. The results are shown in Figure 2A and B.

Literature risk assessment chart (A and B).
Efficacy Analysis
Adjuvant Immunotherapy
DFS
An analysis of the DFS data showed that compared to the placebo group, adjuvant immunotherapy reduced the risk of disease progression or death by 22% (HR = 0.78; 95% CI, 0.69-0.90; P < .001; I2 = 0.0%) (Figure 3A).
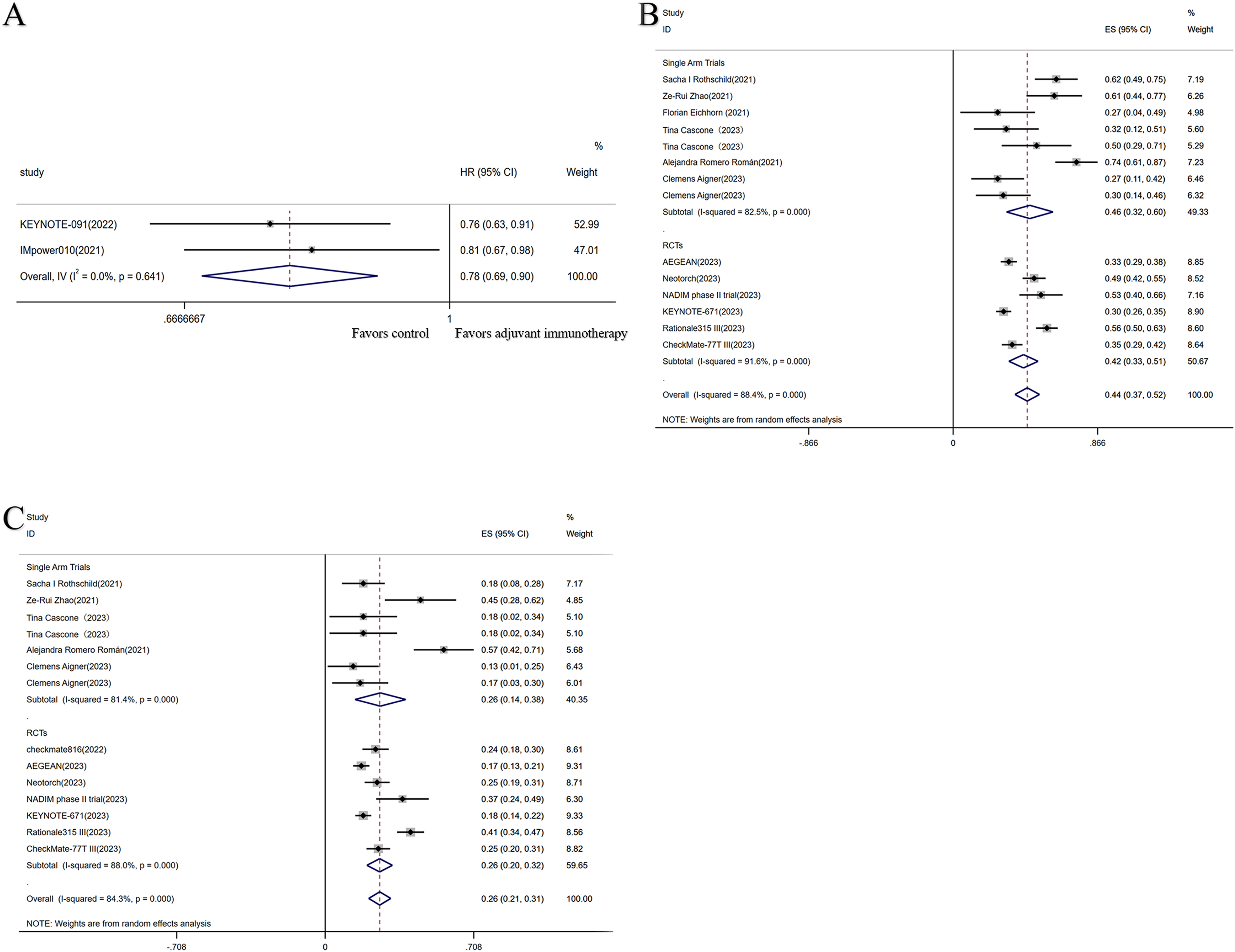
Efficacy analysis of adjuvant and neoadjuvant therapies. Forest plot of HR for DFS (A) of adjuvant immunotherapy in perioperative patients with NSCLC; forest plot of MPR (B) and pCR (C) in RCTs and single-arm studies of neoadjuvant immunotherapy.
Neoadjuvant Immunotherapy
Overall
Surgical resection rate
The results from 12 studies showed an overall resection rate of 85% (95% CI, 0.82-0.88). Among these, the pooled resection rate for the single-arm trials was 90%, while the pooled resection rate for the RCTs was 82% (Supplemental Figure 1(Figure S1)).
Pathological reaction
The single-arm studies and RCTs with MPR and pCR data showed an overall MPR of 44% (95% CI, 0.37-0.52) (Figure 3B) and a pCR of 26% (95% CI, 0.21-0.31) (Figure 3C).
Subgroup Analysis From the RCTs
Surgical resection rate
The analysis of 5 studies found that patients receiving neoadjuvant immunotherapy had a higher resection rate than the control group (OR = 1.32; 95% CI, 1.10-1.59; P = .003; I2 = 11.8%) (Figure S1).
Pathological reaction
In the 7 studies reporting pCR data, patients undergoing neoadjuvant immunotherapy achieved a higher rate of pCR than the placebo group (OR = 7.83; 95% CI, 5.95-10.31; P < .001; I2 = 48.1%) (Figure 4A).

Efficacy analysis of neoadjuvant therapy. Forest plot of pCR (A), MPR (B), HR of EFS (C), and HR of OS (D) in RCTs.
In the 6 studies in which MPR data were available, the ICI-based neoadjuvant therapy group had a higher incidence of achieving MPR than the neoadjuvant chemotherapy alone group (OR = 5.13; 95% CI, 3.56-7.40; P < .001; I2 = 69.0%). Since the level of heterogeneity was high, a random-effects model was used to account for the high heterogeneity (Figure 4B).
Survival outcomes
The 5 studies reporting EFS data showed that the group receiving neoadjuvant immunotherapy had improved EFS compared to the placebo group (HR = 0.59; 95% CI, 0.52-0.67; P < .001; I2 = 34.9%) (Figure 4C).
Analysis of the OS data from the 4 studies showed that neoadjuvant treatment improved patient OS significantly (HR = 0.65; 95% CI, 0.52-0.82; P < .001; I2 = 0.0%) (Figure 4D).
Single-arm Trials
Surgical resection rate
Among the 9 single-arm trials, 7 data points were included in the analysis. The combined weighted resection rate with neoadjuvant immunotherapy was 90.0% (95% CI, 0.86-0.94) (Figure S1).
Pathological reaction
Analysis of 9 single-arm trials showed that the combined weighted proportions of patients achieving partial response and complete response during neoadjuvant immunotherapy were 57.0% (95% CI, 0.35-0.78) (Figure 5A) and 5.0% (95% CI, 0.01-0.08) (Figure 5B). Notably, there was significant heterogeneity in the partial response rate, which led to the use of a random-effects model.

Efficacy analysis of neoadjuvant therapies. Forest plot of partial response (A), complete response (B), pCR (C), and MPR (D) in single-arm trials. pCR, pathological complete response; MPR, major pathological response.
In the single-arm trials analysis for pCR, 7 of the 9 data points were included. The combined weighted proportion of patients achieving pCR during neoadjuvant immunotherapy was 26.0% (95% CI, 0.14-0.38). However, significant heterogeneity was observed, necessitating the use of a random-effects model. When conducting a subgroup analysis of neoadjuvant immunotherapy with or without chemotherapy in the treatment regimen, the results indicated that this factor did not have a heterogeneous source. Furthermore, it was observed that in the single-arm trials, neoadjuvant immunotherapy combined with chemotherapy had an ES of 31%, which was superior to monotherapy (ES = 22%) (Figure 5C).
In 8 of the 9 single-arm trials, the combined weighted proportion of patients achieving MPR during neoadjuvant immunotherapy was 46.0% (95% CI, 0.32-0.60). However, there was significant heterogeneity, which warranted the use of a random-effects model. A similar subgroup analysis showed that neoadjuvant immunotherapy combined with chemotherapy had a superior effect size (ES = 53%). These results suggest that neoadjuvant immunotherapy combined with chemotherapy may be more effective in this subgroup (Figure 5D).
Safety and Toxicity
Adjuvant Immunotherapy
In the 2 included studies that utilized adjuvant immunotherapy, the incidence of ≥TRAE was higher in the adjuvant immunotherapy group than the placebo group (OR = 1.75; 95% CI, 1.20-2.56; P = .003; I2 = 68.7%) (Figure 6A).
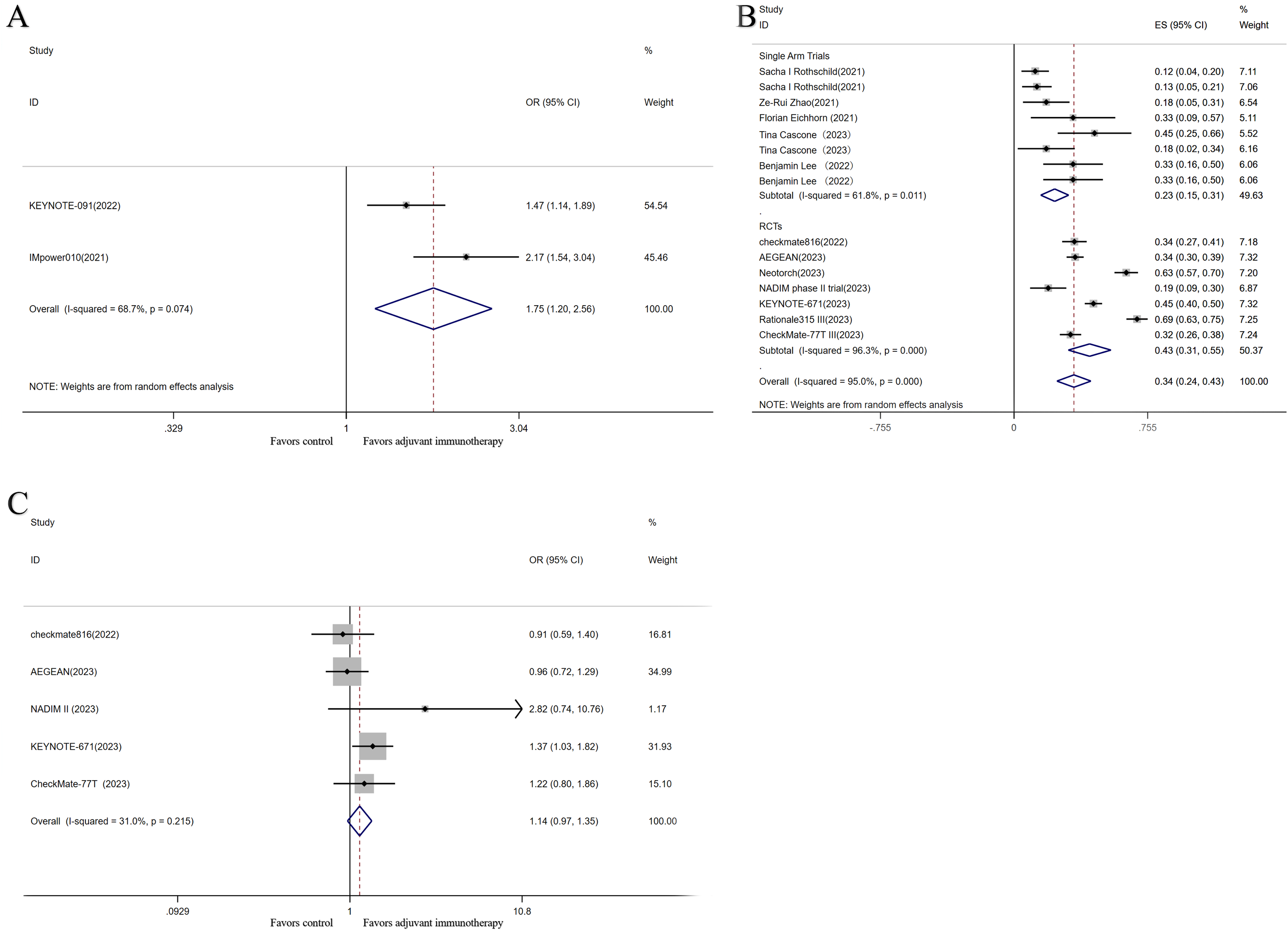
Safety and toxicity analysis of adjuvant and neoadjuvant immunotherapy. Forest plots of TRAEs for adjuvant immunotherapy (A); forest plots of pooled TRAEs for neoadjuvant immunotherapy RCTs and single-arm trials (B); forest plots of TRAEs (C) for RCTs.
Neoadjuvant Immunotherapy
Overall Data
A summary of the studies on TRAEs showed an overall TRAE incidence rate of 34% (95% CI, 0.24-0.43) (Figure 6B). Among these, the incidence rate in the neoadjuvant RCT group reached 43%.
TRAEs Between the 2 Groups
Five RCTs compared the incidence of TRAEs above grade III between 2 ICI-based neoadjuvant and chemotherapy neoadjuvant groups. The pooled results showed that the rate was not statistically significant for neoadjuvant treatment with ICIs combined with chemotherapy compared with chemotherapy alone (OR = 1.14; 95% CI, 0.97-1.35; P = .115; I2 = 31.0%) (Figure 6C).
Postoperative Complications
An analysis of 8 single-arm trials reported that the combined weighted proportion of patients experiencing postoperative complications during neoadjuvant immunotherapy was 35% (95% CI, 0.21-0.49). However, there was significant heterogeneity, warranting the use of a random-effects model (Figure S2).
Surgical Delay Rate
The surgical delay rate was defined as the ratio of patients whose surgery was delayed due to adverse events caused by neoadjuvant immunotherapy compared to all patients who were expected to undergo surgery. This is a commonly used metric for evaluating the safety of neoadjuvant immunotherapy. The pooled data from the study showed that the neoadjuvant immunotherapy surgical delay rate was 7% (95% CI, 0.02-0.11) (Figure S2). It is noteworthy that in 3 of the studies, all surgeries were completed with surgical resections as planned, and no surgery was delayed.
Publication Bias and Sensitivity Analysis
Publication Bias
Adjuvant Immunotherapy
Research bias was evaluated using a funnel plot. The 2 studies were asymmetrically distributed along the longitudinal axis, the sample sizes of the 2 studies were similar, and one had a significant publication bias. The bias could be attributed to (1) the different blinding methods chosen for the trials, with one study being a triple-blind trial and the other being an open-label trial; (2) the inconsistent proportion of the population with EGFR/ALK mutations. This sample showed high heterogeneity due to the above-mentioned factors (Figure S3).
Neoadjuvant Immunotherapy
RCTs
In addition, bias analysis for the use of neoadjuvant immunotherapy methods indicated the presence of bias in the data. Factors that may have caused bias due to differences in baseline and clinical characteristics included: (1) differences in patient ECOG scores; (2) variations in the proportion of patients with adenocarcinoma or squamous cell carcinoma among the different groups; (3) differences in smoking status; (4) differences in the histopathological diagnostic stage with 2 of the 5 trials including only stage III patients and the other 3 including patients at various stages from I to II (Figure S3).
Single-arm test
A bias analysis of the single-arm trials showed, from the distribution, the presence of biased data (Figure S3). Possible reasons included: (1) age differences, with 1 of the 6 datasets having an average age of 69 years while the others ranged from 62 to 64; (2) variations in the proportion of adenocarcinoma and squamous cell carcinoma across different studies; (3) differences in the histopathological diagnostic stage with 7 trials including patients with stage I-II.
Sensitivity Analysis
Adjuvant Immunotherapy
The results from the sensitivity analysis did not have statistical significance. This could have been caused by the inclusion of too few studies in the analysis (Figure S4).
Neoadjuvant Immunotherapy
RCTs
Even after excluding most of the literature, the sensitivity analysis results retained statistical significance (OR = 5.13; 95% CI, 3.19-7.40) (Figure S4).
The sensitivity analysis revealed that even after excluding most of the literature, the results remained statistically significant (OR = 0.26; 95% CI, 0.14-0.38), suggesting the robustness of the findings (Figure S4).
Discussion
The present meta-analysis summarized the evidence from studies on neoadjuvant immunotherapy and adjuvant immunotherapy in patients with NSCLC. In total, 5850 patients were included, with 2020 patients receiving neoadjuvant ICI combined with chemotherapy and 1097 patients receiving adjuvant ICI combined with chemotherapy. The present analysis showed that perioperative immunotherapy combined with chemotherapy compared with chemotherapy alone could increase the pCRs and improve survival without increasing toxicities in patients with resectable NSCLC. A baseline analysis of the population reveals that the male population is approximately twice as large as the female population, which may be related to the higher number of male smokers, although detailed statistics are not available. In terms of the smoking population, there were 6 times as many current or former smokers as non-smokers. According to available statistics, squamous carcinoma was 1.4 times more common than adenocarcinoma in the population included in the study, and there were only 23 cases of other types of cancer (eg, lymphoepithelioma-like carcinoma, large cell carcinoma). Lymph node staging, ECOG, and diagnostic grading are shown in Table 2, and no significant differences were seen.
Recently, several meta-analyses have shown that neoadjuvant immunotherapy is safe and effective in advanced resectable NSCLC, and data from trials such as NADIM, NEOSTAR, and SAKK 16/14 have been analyzed.31–34 These meta-analyses included only RCTs or retrospective studies up to 2021, and only pCR, MPR, resection rates, and complications were discussed. This meta-analysis analyzed data from the most recent trials through February 2024, including large trials such as AEGEAN, CheckMate816, CheckMate-77T, Rationale315, Neotorch, NADIM II, and KEYNOTE-671, which strengthens the evidence for the efficacy of neoadjuvant immunotherapies and complements the results of the meta-analyses described above. Most importantly, we have updated and supplemented the data on pCR, MPR, EFS, and OS for neoadjuvant immunotherapy. This can be considered as add-on value compared to those previous reports.
pCR is defined as the absence of residual viable tumor cells in the primary tumor or cleared lymph nodes. Studies have found that pCR rates with neoadjuvant chemotherapy for NSCLC are extremely low and may not be suitable as an alternative indicator. Consequently, in 2014, Professor Matthew Hellman introduced MPR, a novel metric. MPR is defined as the presence of residual tumor cells of less than 10% on postoperative pathology examination. Increased MPR and pCR may be associated with survival benefits in NSCLC patients treated with neoadjuvant immunotherapy. 23 The MPR may even serve as a surrogate endpoint of OS to evaluate neoadjuvant immunotherapy. 35 This study conducted a meta-analysis on the overall data for postoperative pathological pCR and MPR in RCTs and single-arm trial groups for neoadjuvant immunotherapy, revealing that MPR could reach 44% (95% CI, 0.37-0.52) while pCR reached 26% (95% CI, 0.21-0.31). Notably, nivolumab has achieved high levels of both MPR and pCR rates. It is worth noting that the NADIM study involving the combination of nivolumab, paclitaxel, and carboplatin achieved favorable clinical results. MPR reached 83%, pCR was 63%, and the 2-year OS was 89.9%. However, it is important to consider that the study reported a TRAE rate of 30%. When analyzing the data from the RCT group separately, it is evident that neoadjuvant immunotherapy had significantly higher rates of both pCR (OR = 7.83; 95% CI, 5.95-10.31; P < .001) and MPR (OR = 5.13; 95% CI, 3.56-7.40; P < .001) than chemotherapy alone. Previous studies found that neoadjuvant chemotherapy had a pCR rate of 7.7% and an MPR rate of less than 25%.36,37 Subgroup analysis of single-arm trials showed that the combination of neoadjuvant immunotherapy with chemotherapy resulted in an MPR rate of 59% and a pCR rate of 33%, both of which were higher than immunotherapy alone (MPR = 45%, pCR = 20%). In addition, the enhanced MPR and pCR rates resulted in survival benefits. In terms of long-term clinical benefits, data from the RCTs found that neoadjuvant immunotherapy reduced the risk of death by 35% (HR = 0.65; 95% CI, 0.52-0.82; P < .001) and improved EFS (HR = 0.59; 95% CI, 0.52-0.67; P < .001). These data suggest that adjuvant and neoadjuvant immunotherapies provide significant benefit for long-term prognosis. Meanwhile, analysis of short-term survival revealed that adjuvant immunotherapy reduced the risk of disease progression or death by 22% (HR = 0.78; 95% CI, 0.69-0.90; P < .001). However, the long-term survival data for adjuvant immunotherapy has not been reached.
When neoadjuvant ICI combined with chemotherapy was compared with neoadjuvant immunotherapy alone, patients who received neoadjuvant chemoimmunotherapy had remarkably improved pathological response (MPR rate: 53.3% vs 28.6%; pCR rate: 28.6% vs 9.9%) compared with those receiving neoadjuvant single-agent immunotherapy. 38 The present analysis supports better clinical outcomes with neoadjuvant ICI combined with chemotherapy than single ICI or single chemotherapy alone. This aligns with previous studies suggesting a greater antitumor effect of immunotherapy combined with chemotherapy than chemotherapy or immunotherapy alone in the setting of neoadjuvant in resectable NSCLC patients.
Regarding the benefit of immunotherapy in the current population, neoadjuvant chemotherapy-immunotherapy demonstrated higher MPR rates at 56% and pCR rates at 39% for stage III NSCLC in one study. However, it was associated with a higher summary rate of TRAEs at 65%. 31 Based on the inclusion criteria from previous chemotherapy trials and other research findings, it is clear that the majority of patients who benefited from perioperative ICI-based trials had at least stage IB disease (>4 cm) or more advanced. 39 In addition, despite the poor efficacy of anti-PD-1/PD-L1 treatment in most advanced EGFR-mutated NSCLC, specific subpopulations with advanced EGFR-mutated NSCLC could benefit from immunotherapy. 23 Notably, few populations with EGFR/ALK mutations were included in the KEYNOTE-671, AEGEN, Impower 010, and KEYNOTE-091 trials. Due to unavailable data, we did not conduct a meta-analysis in these patients to evaluate the efficacy of perioperative therapy with ICI combined with chemotherapy compared with chemotherapy or targeted EGFR/ALK treatment. However, the better EFS or DFS in the EGFR mutation subgroup in these trials indicated that perioperative immunotherapy combined with chemotherapy might be an alternative choice in these patients in the future.
On the need for postoperative adjuvant immunization, the current standard of care for patients with perioperatively resected stage II or III NSCLC remains adjuvant cisplatin-based chemotherapy. However, as more evidence finds that ICI monotherapy and ICI combination chemotherapy improve survival in advanced NSCLC, adjuvant immunotherapy will certainly become a standard of care.23,40 In terms of mechanistic insights, several trials have shown an association between PD-L1 expression and therapeutic benefit. It was found that cancer surgery induces changes in cytokine levels, which in turn causes bone marrow-derived suppressor cells. Cellular immunosuppression can lead to PD-1/CTLA-4 expression and cause T-cell and NK-cell dysfunction, leading to postoperative immunosuppression in patients. 41 In contrast, ICI can block the binding of PD-1 and PD-L1, upregulate the growth and proliferation of T cells, enhance the recognition of tumor cells by T cells, and activate their attacking and killing functions, thus realizing antitumor effects. In the most representative large adjuvant immunotherapy IMpower010 trial, atezolizumab improved DFS in stage II-III PD-L1+ (defined as PD-L1 expression in ≥1% of tumor cells) with an HR of 0.66 (P = .004) and in stage II-III with an HR of 0.79 (P = .02). 42 While discussions on the optimal duration of postoperative adjuvant immunotherapy remain controversial, the 2022 NCCN Clinical Practice Guidelines in Oncology recommend 1 year for atezolizumab monotherapy as adjuvant immunotherapy. 7 It has been reported that ctDNA monitoring during ICI treatment is able to accurately distinguish between pseudo-progression and true progression resistance in metastatic melanoma. 43 Several IO-based trials have suggested that ctDNA may be particularly important in detecting molecular residual disease and, thus, supports the early implementation of adjuvant or escalated therapy to prevent the development of metastatic disease. 44 Further research is needed to investigate the value of ctDNA in defining molecular residual disease, to direct the duration of adjuvant immunotherapy, and to conduct a personalized adjuvant treatment plan.
The safety of the treatment regimen is also a crucial concern. The occurrence rate of TRAEs (such as neutropenia, decreased neutrophil count, anemia, diarrhea, and rash) in adjuvant immunotherapy (OR = 1.75; 95% CI, 1.20-2.56; P = .003) was higher than in the chemotherapy-only group. No unreported adverse reactions were observed. The overall incidence rate for TRAEs in the neoadjuvant immunotherapy single-arm trial and RCT groups was 34% (95% CI, 0.24-0.43). Separate statistics for the RCT group showed no statistically significant incidence of TRAEs (OR = 1.14; 95% CI, 0.97-1.35; P = .115). This indicates no increase in toxicity in the RCT group of neoadjuvant chemotherapy combined with immunotherapy compared to the chemotherapy-alone group. In conclusion, it can be demonstrated that neoadjuvant immunotherapy is safer and less toxic than adjuvant immunotherapy.
On the downside, there have been few RCTs of adjuvant immunotherapy within the last decade, and only 2 studies are included in the current statistical analysis. This results in a high level of bias. In addition, most of the RCTs included in the present are ongoing clinical trials with data that is continuously being updated. However, the current data are suggestive of a better prognosis. Our study has some other limitations. First, an imbalance in the patients’ baseline characteristics between the different treatment groups may still exist. Although most studies provided a balance between intervention arms, subgroup analysis by nature might introduce bias in our analysis. Second, we conducted the present study at the trial level, and no clinicopathological characteristics were examined at the individual level. This might have reduced our ability to test for associations between variables in specific subgroups.
Due to the inclusion of some single-arm trials in the literature, this portion of the analysis performed only a single-rate meta-analysis on postoperative pathological indicators. Therefore, more and larger RCTs are needed to enrich and refine our results. Additionally, some studies presented at conferences (CheckMate816, AEGEAN, Neotorch, KEYNOTE-671) are still ongoing. With the disclosure of more results and the conduct of additional phase II and III clinical trials, it is believed that the effectiveness of neoadjuvant immunotherapy for NSCLC will be further demonstrated. This has the potential to benefit a larger number of patients.
Conclusion
In summary, the present study showed that perioperative immunotherapy combined with chemotherapy was superior to chemotherapy alone in the aspects of MPR, pCR, EFS, DFS, and OS. This conclusion was based on data analysis from existing ongoing and up to 10-year RCTs and single-arm studies. Both neoadjuvant and adjuvant immunotherapy combined with chemotherapy did not significantly increase the occurrence of TRAEs compared with chemotherapy alone.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241258164 - Supplemental material for The Value of Perioperative Immunotherapy for Non-Small Cell Lung Cancer: A Pool- and Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338241258164 for The Value of Perioperative Immunotherapy for Non-Small Cell Lung Cancer: A Pool- and Meta-Analysis by Yanmeng Wu, Lin Hu, Shuling Zhang and Hui Zhang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the 345 Talent Project of Shengjing Hospital and National Natural Science Foundation of China (grant No. 82103338).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
