Abstract
Introduction
It is possible to predict immune-related adverse events (irAEs) in the treatment of immune checkpoint inhibitors (ICIs) based on clinical and hematological parameters. Nevertheless, the specific parameters which can predict irAEs are still in the exploration. The purpose of this retrospective study was to develop a predictive model for irAEs in non-small cell lung cancer (NSCLC) patients in the treatment of ICIs.
Methods
Researchers enrolled NSCLC patients treated with at least 1 type of ICIs at Harbin Medical University Cancer Hospital between January 30, 2019 and December 31, 2021. Baseline parameters including demographic, clinicopathology, treatment information, and peripheral blood markers were selected retrospectively. Type, onset time, grade, and treatment of irAEs were also assessed. By analyzing the risk factors for irAEs, an irAEs prediction model was established using univariate and multivariate logistic regression.
Results
In a total of 484 patients, 81 patients experienced 112 irAEs in which thyroid dysfunction was the most common irAE (n = 38, 33.9%) and ICI pneumonitis was the most serious irAE (n = 6, 33.3%). Finally, a prediction model based on lines and combination therapy of ICIs, ECOG performance status, neutrophils/lymphocytes ratio (NLR), platelet (PLT), and lymphocyte (LYM) was established. Multivariate logistic regression analysis showed that 2 or ≥3 lines of immunotherapy, ICIs combination therapy, and ECOG PS 1–2 were independent risk factors for irAEs. Baseline LYM was positively associated with irAEs (OR = 2.599, P = 0.048) while baseline NLR and PLT were negatively associated with irAEs (OR = 0.392, P = 0.047; OR = 0.992, P = 0.035, respectively). The model showed great prediction performance with the AUC value of 0.851 and 0.779 in the training cohort and validation cohort, respectively.
Conclusion
Our study identified the risk factors related to irAEs occurrence and constructed and assessed the predictive model of irAEs in patients with NSCLC treated by ICIs using clinical and hematological parameters, thus guiding clinicians to select precisely the population receiving immunotherapy and develop individualized treatment therapy.
Introduction
Lung cancer has been characterized by the highest incidence and mortality worldwide, with an estimated 2.2 million newly diagnosed cases and 1.8 million deaths worldwide. 1 As the most frequent type of lung cancer, non-small cell lung cancer (NSCLC) can be treated with a series of therapeutic modalities such as radiation therapy, chemotherapy, targeted therapy, and surgery.2,3 Nevertheless, the probability of treatment failure is still high. 4 Recently, immune checkpoint inhibitors (ICIs) have greatly increased therapeutic efficacy and changed treatment paradigms in the management of NSCLC. 5
Over the past few years, a series of phase III clinical trials have observed the durable therapy responses and great improvement in OS of patients with ICIs or combined platinum-based chemotherapy compared with chemotherapy alone, thus ICIs have become the first-line treatment options for patients with advanced NSCLC without sensitizing mutation site.6–8 The ICIs in clinical application are mainly antibodies that block the inhibitory signaling molecules expressed on immune cells or tumor cells such as PD-1, CTLA-4, or PD-L1. 9 Currently, approved ICIs in clinical application mainly include PD-1 inhibitors such as pembrolizumab and nivolumab, PD-L1 inhibitors such as atezolizumab and durvalumab, and the CTLA-4 inhibitor such as ipilimumab.6–8,10
The use of ICIs may cause hyperactivation of the immune system against normal tissues and immune-mediated toxicity, which are also referred as immune-related adverse events (irAEs). 11 Specifically, these agents can enhance T-cell activation and proliferation, weaken Treg cell functions, and increase autoimmune response, thus resulting in multiple irAEs. 12 Based on the diverse mechanisms from above, the affected organs, onset time, and incidence of irAEs are highly heterogeneous. The organs including the skin, lung, colon, endocrine system, and liver are the most frequently affected by irAEs. 11 In addition, majority of irAEs initiate from weeks to years after ICIs administration, while others may occur within several years even after the whole treatment. 13 The incidence of irAEs depends on the ICIs type and combination therapy, with a lower incidence treated with anti-PD-1/PD-L1 antibodies compared to anti-CTLA-4 antibodies and the highest incidence treated with anti-PD-1 + anti-CTLA-4 treatment. 14 Studies have suggested that many factors can be regarded as biomarkers to assess the risk of irAEs, such as host parameters, ICIs type and dose, tumor immune environment, and gut microbiome. 15 Since conducting a lesion biopsy to acquire immunomic-related data is a great challenge, most studies use more accessible data including clinical and hematological factors to predict the occurrence of irAEs. For example, studies have shown that NSCLC patients with neutrophils/lymphocytes ratio (NLR) < 3 and platelets/lymphocytes ratio (PLR) < 180 at the baseline of immunotherapy were more likely to develop irAEs. 16 Another study by Okada et al 17 also found that ECOG PS ≥2 could be used as an independent predictive factor for interstitial lung disease, a serious type of irAEs.
With the extensive use of ICIs in NSCLC therapy, it is crucial to establish biomarkers of immunotherapy response to optimize patient benefit and minimize toxicities risk. As the biomarker with the greatest focus, high PD-L1 expression on tumor cells tends to indicate better clinical benefit, but PD-L1 testing alone is not sufficient. Recently, emerging studies have demonstrated the utility of tumor mutational burden (TMB) in predicting ICI benefits, regardless of PD-L1 level. 18 Immune gene signatures, T-cell receptor clonality, oncogenic driver mutation, and tumor-infiltrating lymphocytes (TILs) within the tumor microenvironment are also being investigated as they are more likely to affect immunotherapy response.19,20 However, the application of these biomarkers is still limited by many problems, such as the difficulty in obtaining optimal cutoff value, high technical difficulty, and the complexity of the tumor immune microenvironment, which needs to be deeply investigated and validated. A healthy positive correlation between onset of irAEs and response to immunotherapy has been identified in multiple of studies. In a single-center retrospective study of 155 patients receiving monotherapy with a PD-1 inhibitor for advanced NSCIC, dermal toxicity was associated with higher objective response rate (ORR) and disease control rate (DCR; ORR:57% vs 19%, P < 0. 001; DCR: 94% vs 55%, P < 0.001). 21 Similarly, a retrospective study analyzing data from 48 patients with advanced NSCLC in KEYNOTE-001 found that thyroid function abnormality was significantly associated with the prolongation of OS (40 months vs 14 months, HR = 0.29, 95%CI: 0.09-0.94, P = 0.04). 22 Moreover, there is better tolerance with PD-1 and PD-L1 inhibitors than CTLA-4 inhibitors, and CTLA-4 inhibitors were more likely to cause grade 3 or 4 irAEs reported by a systematic review.23,24 Therefore, it is urgent for us to develop a better understanding of irAE onset and explore potential biomarkers of irAE development.
In our retrospective study, we developed and validated successfully a predictive model to evaluate the incidence of irAEs collecting clinical and hematological parameters from real-world evidence and further explored the type, onset time, grade, and treatment of irAEs. Besides, we also investigated the correlation of irAEs with efficacy in ICI treatment.
Materials and Methods
Patients Enrollment
A total of 522 NSCLC patients were enrolled in our study. The inclusion criteria for NSCLC patients were as follows: pathologically confirmed NSCLC between January 30, 2019 and December 31, 2021, age ≥18 years, treated with anti-PD-1 antibody (nivolumab, pembrolizumab, camrelizumab, sintilimab, tislelizumab, toripalimab, etc) or anti-PD-L1 antibody (atezolizumab, durvalumab) combined or not with anti-CTLA-4 antibody (ipilbuzumab) at least 1 cycle until disease progression, toxicity intolerance, withdrawal, or death from the registry data in Department of Medical Oncology, Harbin Medical University Cancer Hospital. The exclusion criteria included the following: incomplete baseline demographic, clinicopathology and therapeutic information, autoimmune disease history, liver and kidney dysfunction, and acute infections. Written informed consent from patients that medical data during the treatment process could be used for scientific research was confirmed before ICI treatment. The flow diagram of the patients’ enrollment is presented in Figure 1. The protocol in this study was approved by the ethics committee of Harbin Medical University Cancer Hospital on October 28, 2020 with the approval number 2020-140-IIT. The reporting of this study conforms to the TRIPOD guidelines. 25

Flow diagram of patients’ enrollment.
Data Collection
We collected retrospectively baseline demographic (sex, age, BMI, smoking history), clinicopathology and treatment (stage, performance status, single or combination agent therapy, type and line of ICI treatment) information in our research. Baseline peripheral blood biomarkers comprised white blood cell (WC), lymphocytes (LYM), eosinophil (EOS), monocytes (MONO), neutrophils (NEU), basophils (BASO), platelet (PLT) counts, serum albumin level (ALB), and lactate dehydrogenase (LDH) as well as NLR, PLR, monocytes-to-lymphocyte ratio (MLR). All baseline parameters for enrolled NSCLC patients were selected within 1 week before ICI therapy. We also assessed optimal efficacy including complete response (CR), partial response (PR), stable diseases (SD), and progressive diseases (PD) in patients during ICI cycle. In addition, the overall survival of patients receiving ICIs was identified by the researchers. Besides, we used the median to perform simple interpolation for missing values.
Outcome Assessment
IrAEs were defined as adverse events causing immunological dysregulation in patients treated with ICIs such as thyroid dysfunction, ICI pneumonitis, rash, and myocarditis. Researchers who did not know about the relevant information in advance evaluated irAEs using the Common Toxicity Criteria for Adverse Events (CTCAE 4.0) of the National Cancer Institute during or after the treatment of ICIs. Data about irAEs including type, onset time, grade, and treatment of irAEs were obtained.
Statistical Analyses
Based on random seed, the NSCLC immunotherapy data was divided into the training cohort and validation cohort randomly to establish and verify the predictive model. We applied receiver operating characteristic curve (ROC) to identify the optimal cutoff value of NLR, PLR, and MLR and classified patients into 2 cohorts based on the cutoff value (NLR <3.2 and ≥ 3.2, respectively; PLR <143 and ≥ 143, respectively; MLR < 0.3 and ≥ 0.3, respectively). We also performed student's t-test and chi-square test to compare baseline characteristics between patients with irAEs and those without irAEs. Univariate and multivariate logistic regression analyses were used to confirm factors tightly linked to the occurrence of irAEs and included in the establishment of predictive model using nomogram through “rms” and “Dynnom” R package. ROC, calibration curves, and decision curve analyses (DCA) were applied to verify the predictive performance of model through “survivalROC” and “ggplot2” package. The Kaplan-Meier survival curve was performed to compare the OS between the irAEs and non-irAEs group using R packages “survival” and “survminer.” All statistical tests were disposed by R software (version 4.0.2), and P < 0.05 was considered statistically significant.
Results
Patient Characteristics
A total of 484 patients who were treated with at least 1 cycle ICIs at anonymized issue Cancer Hospital have been included. Most of the patients (n = 441, 91.12%) received anti-PD-1 antibody, 27 patients were administered anti-PD-L1 antibody, 5 patients with anti-PD-L1 + anti-CTLA-4 antibody, and 11 patients with anti-PD-1 + anti-CTLA-4 antibody. In total, 53.3% of patients were treated with ICIs monotherapy whereas other patients received ICIs combined therapy including chemotherapy, antiangiogenic therapy, and radiotherapy. Majority of the patients were given the first-line treatment of ICIs (62.4%) and had 0 to 1 ECOG performance status (92.15%). Patients with IV stage (75.41%), male (71.69%), age <65 (64.88%), BMI ≤23.9 (59.92%), and smoking history (50.41%) also had a larger proportion of the total population. In addition, the NSCLC immunotherapy data was divided into the training cohort (n = 339) and validation cohort (n = 145) randomly to establish and verify the predictive model according to random seed. The baseline characteristics of enrolled patients were compared between the training and validation cohort, and no statistical significance was found (P > 0.05; Table 1).
Baseline Characteristics of Enrolled Patients in Overall, Training, and Validation Cohort.
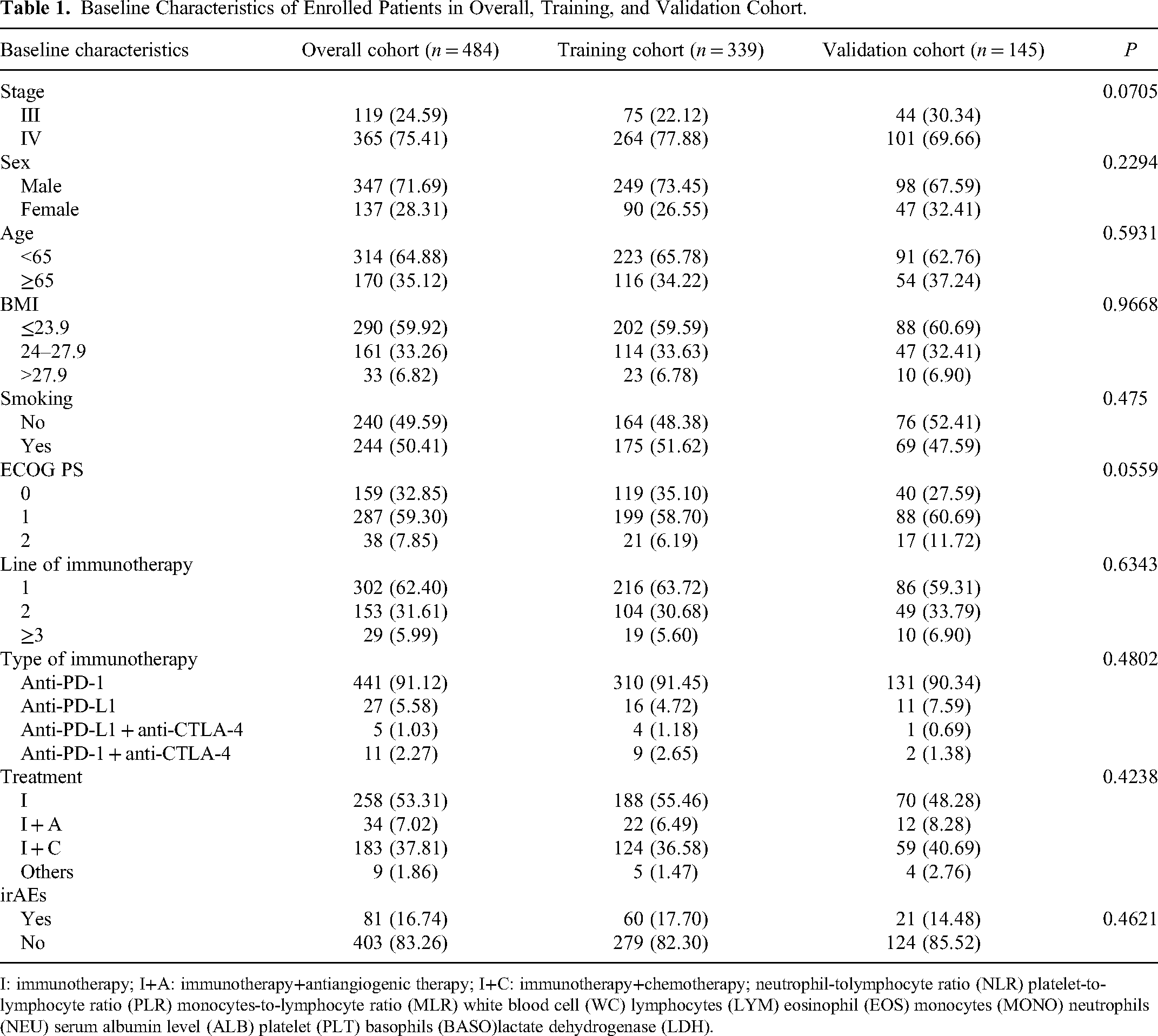
I: immunotherapy; I+A: immunotherapy+antiangiogenic therapy; I+C: immunotherapy+chemotherapy; neutrophil-tolymphocyte ratio (NLR) platelet-to-lymphocyte ratio (PLR) monocytes-to-lymphocyte ratio (MLR) white blood cell (WC) lymphocytes (LYM) eosinophil (EOS) monocytes (MONO) neutrophils (NEU) serum albumin level (ALB) platelet (PLT) basophils (BASO)lactate dehydrogenase (LDH).
Immune-Related Adverse Events
As shown in Table 2, a total of 81 patients experienced 112 irAEs of any grade and 18 irAEs of grade 3 or higher. Thyroid dysfunction was the most common irAEs with the incidence rate of 33.9%, followed by ICI pneumonitis (21.4%), rash (17.9%), and myocarditis (16.7%; Figure 2). Notably, ICI pneumonitis was the most serious adverse event with the proportion of 33% in all grade ≥3 irAEs. A total of 34.8% of patients who developed irAEs received corticosteroid treatment, while the remaining patients were treated with symptomatic treatment and immunosuppressants as recommended by CSCO, ASCO, and NCCN guidelines for irAEs management. Most patients with irAEs showed symptom relief after irAEs treatment, and 61.6% (n = 69) of them underwent immunotherapy retreatment (Table 2). We also investigated the onset time of various irAEs and found that the average time of irAEs occurrence was 0.3 to 122 weeks, which showed high variability among them (Figure 2). Furthermore, the relationship of irAEs with efficacy was explored. In total, 59.3% and 52.1% of irAEs and non-irAEs patients showed CR or PR, respectively, although there was no significance found between the 2 groups (P > 0.05; Figure 3). Kaplan-Meier survival curve has revealed that compared with the non-irAE group, the overall survival of patients in the irAE group was longer (P < 0.05), tending to have a better prognosis (Figure 3).

(A) Distribution characteristics of irAEs. (B) Median time for the development of different irAEs.
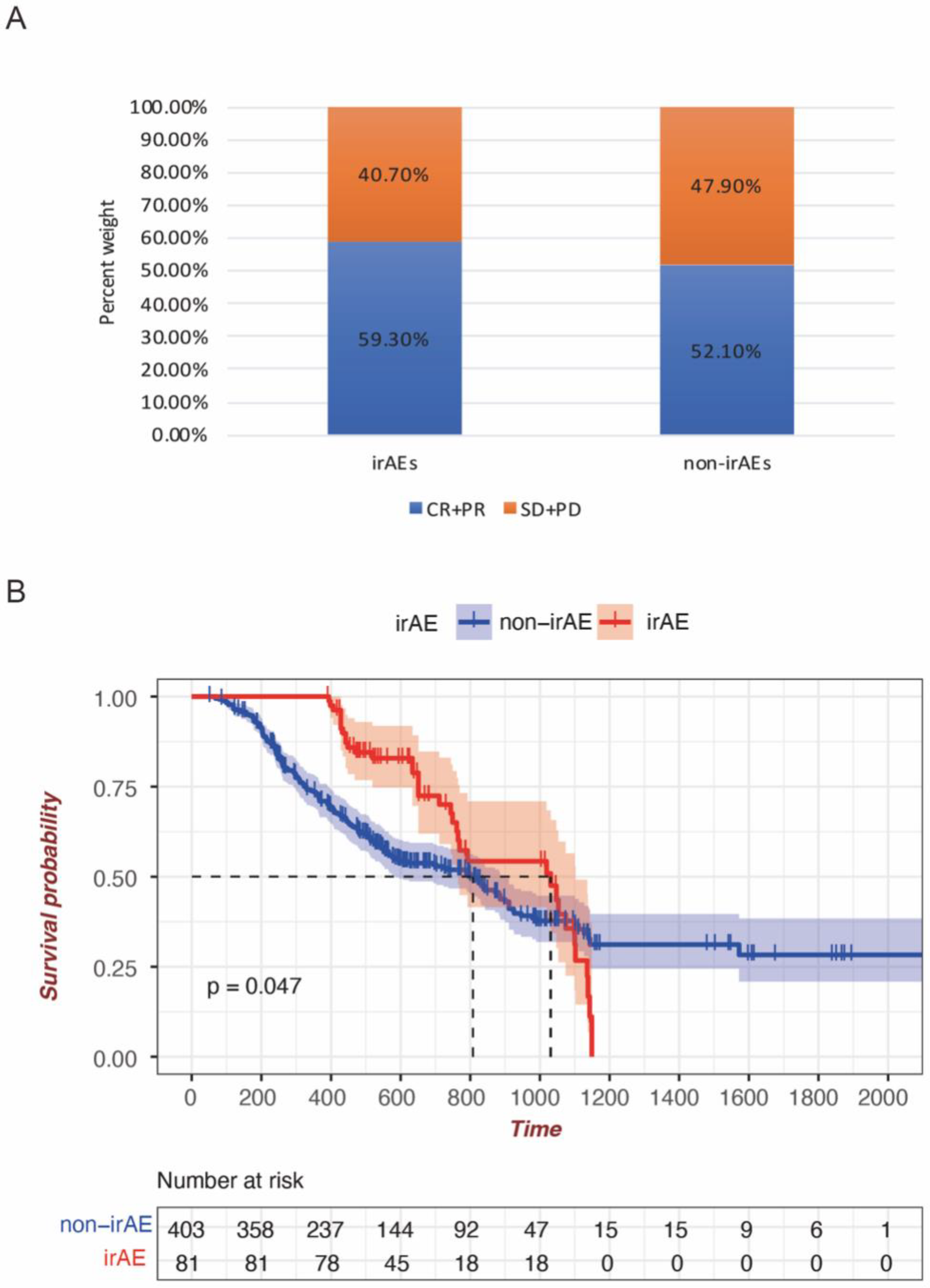
(A) Association of irAEs with efficacy of immunotherapy. (B) Kaplan-Meier survival curve of NSCLC with the irAE and non-irAE group.
Immune Checkpoint Inhibitor-Related Adverse Effects.

I: immunotherapy; I+A: immunotherapy+antiangiogenic therapy; I+C: immunotherapy+chemotherapy; neutrophil-tolymphocyte ratio (NLR) platelet-to-lymphocyte ratio (PLR) monocytes-to-lymphocyte ratio (MLR) white blood cell (WC) lymphocytes (LYM) eosinophil (EOS) monocytes (MONO) neutrophils (NEU) serum albumin level (ALB) platelet (PLT) basophils (BASO)lactate dehydrogenase (LDH).
Construction of a Predictive Model for IrAEs
The risk factors for irAEs were assessed by univariate binary logistic regression analysis in the training cohort. Then, variables including BMI, ECOG PS, line of immunotherapy, type of immunotherapy, treatment, NLR, PLR, MLR, LYM, and PLT were selected for inclusion in the multivariate logistic regression analysis based on statistical significance in univariate analysis (P < 0.05; Table 3). A final irAEs prediction model was constructed using the significant variables (P < 0.05) including ECOG PS, line of immunotherapy, treatment, NLR, LYM, and PLT in multivariate logistic regression analysis (Table 4). We can conclude from multivariate logistic regression analysis that patients with ECOG PS 1 to 2, 2 or ≥3 line of immunotherapy, and ICIs combined therapy are independent risk factors for irAEs. Baseline LYM are positively associated with irAEs (OR = 2.599, P = 0.048) while baseline NLR and PLT are negatively associated with irAEs (OR = 0.392, P = 0.047; OR = 0.992, P = 0.035, respectively; Table 4). A nomogram was established as a quantitative tool to predict the incidence of irAEs in NSCLC patients more precisely (Figure 4). Furthermore, the web-based nomogram (https://weitonggao.shinyapps.io/DynNomapp/) was also established by which you can select the corresponding variable options, and then click Predict on the page to get the probability of irAEs in NSCLC patients and its 95% confidence interval.

Nomogram of predictive model in the training cohort.
Univariate Binary Logistic Regression Analysis.

I: immunotherapy; I+A: immunotherapy+antiangiogenic therapy; I+C: immunotherapy+chemotherapy; neutrophil-tolymphocyte ratio (NLR) platelet-to-lymphocyte ratio (PLR) monocytes-to-lymphocyte ratio (MLR) white blood cell (WC) lymphocytes (LYM) eosinophil (EOS) monocytes (MONO) neutrophils (NEU) serum albumin level (ALB) platelet (PLT) basophils (BASO)lactate dehydrogenase (LDH).
Multivariate Binary Logistic Regression Analysis.

I: immunotherapy; I+A: immunotherapy+antiangiogenic therapy; I+C: immunotherapy+chemotherapy; neutrophil-tolymphocyte ratio (NLR) platelet-to-lymphocyte ratio (PLR) monocytes-to-lymphocyte ratio (MLR) white blood cell (WC) lymphocytes (LYM) eosinophil (EOS) monocytes (MONO) neutrophils (NEU) serum albumin level (ALB) platelet (PLT) basophils (BASO)lactate dehydrogenase (LDH).
Evaluation and Validation of a Predictive Model for irAEs
Figure 5A exhibits the ROC curve, and the area under the curve (AUC) was 0.851 (95% confidence interval: 0.804-0.899). The corresponding sensitivity and specificity were 86.7% and 69.2%, respectively. As depicted in the calibration plots, there was excellent agreement between the observed and predicted probabilities, demonstrating the superiority of the nomograms (Figure 6A). In decision curve analysis (DCA), the net benefit of model was significantly higher than all positive and negative curves, showing high reliability (Figure 7A). In order to validate the prediction model, the above parameters were enrolled into the validation cohort, and ROC curves were drawn with the AUC of 0.779 (95% CI: 0.690-0.867). The corresponding sensitivity and specificity were 95.2% and 54.8%, respectively (Figure 5B). Furthermore, the calibration and DCA plots also exhibited good predictive efficiency in the validation cohort, which was consistent with that in the training cohort (Figures 6B and 7B).

ROC curve analysis of the predictive model. (A) Training cohort: AUC = 0.851, sensitivity 86.7%, specificity 69.2%. (B) Validation cohort: AUC = 0.779, sensitivity 95.2%, specificity 54.8%.

Calibration plots showed the agreement between predicted and observed probabilities of the predictive model in the training cohort (A) and validation cohort (B).

DCA curve showed the net benefit of model was significantly higher than all positive and negative curves in training cohort (A) and validation cohort (B).
Discussion
Over the past few decades, immunotherapy dominated by ICIs including anti-PD-1, anti-PD-L1, and CTLA-4 antibody has dramatically altered the landscape of NSCLC treatment. 26 However, with increased application of ICIs, many irAEs have emerged that impose adverse effects on patients’ quality of life and long-term outcomes. 27 Thus, it is important for clinicians to early identify patients with high risk of irAEs to reduce or prevent the occurrence of severe irAEs. Emerging studies confirmed irAEs-related biomarkers in multiomics data, such as genomics, epigenomics, and transcriptomics, from tumor samples that was not easily accessible.28,29 In contrast, baseline demographic, clinicopathology, and treatment information in combination with laboratory test results are the most convenient and available predictive factors. Recently, Zhao et al 30 established successfully a nomogram including interleukin (IL)-2, IL-8, hepatic metastases, and duration of time to predict the occurrence of irAEs in cancer patients receiving ICIs. As a result, based on clinical and hematological parameters from real-world evidence, a predictive model was established to early identify and reduce the onset of irAEs in our study.
In our research, 81 patients experienced 112 irAEs treated with ICIs in which thyroid dysfunction was the most common irAEs (n = 38, 33.9%), followed by ICI pneumonitis (n = 24, 21.4%), and rash (n = 20, 17.9%). Similar with above, data from a systematic review showed that pneumonitis, hypothyroidism, arthralgia, and vitiligo were more common with patients treated by PD-1 inhibitors. 24 One meta-analysis also found that arthralgia and pneumonitis are more likely to develop with pembrolizumab, endocrine toxicities with nivolumab, and hypothyroidism with atezolizumab. 31 Besides, as the most commonly fatal irAE treated with anti-PD-1/PD-L1 antibodies, 32 33% of ICI pneumonitis was graded ≥3 by our researchers. It has been demonstrated that most of irAEs can occur within 2 to 16 weeks after the initial ICI therapy. 33 Nevertheless, some irAEs are likely to occur within a few days at the beginning of ICIs, and some even occur up to 1 year after the end of therapy.34,35 Similarly, the onset time of ICIs in our study was highly heterogeneous with the range of 0.3 to 122 weeks. As glucocorticoids are regarded as the mainstay of ICIs treatment with the exception of endocrine irAEs, 36 39 out of 112 (34.8%) patients received glucocorticoid therapy in our study.
To better predict the immunotherapy response of ICIs, we investigated the correlation of irAEs occurrence with therapeutic efficacy. We found that compared with the non-irAEs patients, a larger percentage of the irAEs patients showed a CR or PR. It was also revealed that the overall survival of patients in the irAEs group was significantly longer than that of the non-irAEs patients, tending to have a better prognosis. This may owe to the mechanism of irAEs onset that ICIs can activate several key cellular events, such as activation and proliferation of T cells, inhibitory function of Treg cells, the induction of cross-reactivity between antigens on normal tissues and antitumor T cells, the production of cytokines as well as antibody. 12 Therefore, it is reasonable for us to infer that once the antitumor immune response was stimulated by receiving ICIs, the activation of the immune system could also lead to the development of irAEs. In the future, we will deeply explore the influence of irAE occurrence time and type as well as induced by different types of ICIs on treatment efficacy.
Next, we evaluated the demographics, clinical characteristics, and peripheral blood biomarkers in NSCLC patients treated with ICIs and identified the risk factors related to irAEs. Ultimately, 6 parameters including lines and combined therapy of ICIs, ECOG PS, baseline NLR, PLT, and LYM were integrated into the construction of predictive model for irAEs using univariate and multivariate logistic regression analysis. The AUC of ROC curve in the training cohort was 0.851 (95% confidence interval: 0.804-0.899). The corresponding sensitivity and specificity were 86.7% and 69.2%, respectively. In the validation cohort, ROC curve was drawn with the AUC of 0.779 (95% CI: 0.690-0.867). The corresponding sensitivity and specificity were 95.2% and 54.8%, respectively, showing a good predictive ability.
An increasing use of combination therapeutic regimens (ICIs combined with traditional cancer therapy, such as chemotherapy, antiangiogenic therapy, and radiotherapy) might improve the effectiveness of the treatment but amplify irAEs, which was consistent with our finding. 37 Several preclinical studies have demonstrated that most chemotherapeutic drugs stimulate the immune system by reducing the density of immunosuppressive cells and increasing the infiltration of T cells, thus enhancing immunogenicity. 38 For example, neoadjuvant FOLFIRINOX can increase CD8 + T-cells amount and decrease M2 macrophage and regulatory T-cells amount to reactivate the immune system in pancreatic ductal adenocarcinoma. 39 Similarly, antiangiogenic agents and immunotherapy exhibit synergistic effects in preclinical models that may be explained by the immune status regulation. 40 Radiotherapy can also induce immunostimulatory mechanisms that trigger the immune response especially when combined with immunotherapy. 41 In our study, ICIs combined with chemotherapy and antiangiogenic agents (I + C + A) or ICIs combined with radiotherapy (I + R) were both included in other groups, which predicted high incidence of irAEs. Moreover, our multivariate regression analysis found that the incidence of irAEs was higher in ≥3 lines of therapy, reflecting high tumor burden. 42 A retrospective study conducted by Sakata et al 43 showed that severe irAEs were correlated with a high tumor load among advanced NSCLC patients treated with ICIs. It could be explained by the mechanisms that tumor size can influence the intensity of the drug-induced T-cell response. 44 Besides, in agreement with our findings, a study by Okada et al 17 identified the relationship between ECOG PS and lung irAEs in lung cancer patients. However, patients with multiple lines of treatments tend to have poor physical activity, showing a potential interaction between therapy lines and ECOG PS.
We also observed that baseline NLR (OR = 0.392, P = 0.047), LYM (OR = 2.599, P = 0.048), and PLT (OR = 0.992, P = 0.035) were associated with the risk of irAEs. The NLR and LYM are common biomarkers of systemic inflammation, which can be readily detected by peripheral blood analysis. 45 Inflammation can trigger immunosuppression and compromise anticancer immune response, thus reflecting a dysfunctional host immune status. 46 Liu et al 47 also found an increased risk of severe irAEs (grade 3/4) was linked to decreased baseline NLR and PLR among NSCLC patients receiving anti-PD-1 treatment. LYM was regarded as a risk factor for the occurrence of irAEs in patients with NSCLC treated by PD-1 inhibitors in a study by Isono et al. 48 As another component of peripheral blood, PLT have been recognized as immune cell to facilitate inflammation and immune response. 49 A recent study demonstrated that PLT may have immune suppressive role in tumor microenvironment by inhibiting the tumor lysis mediated by natural killing cells. 50 Consistent with our results, a decreased amount of PLT was found significantly in patients with thyroid dysfunctions under the immunotherapy. 51
However, there are still some limitations in our study. Firstly, this was a retrospective study in which potential confounders could not be excluded. Secondly, emerging irAEs-related biomarkers such as chemokines and cytokines were not included in the establishment of our prediction model, which needs to be further explored.30,52 Finally, the internal validation we applied in our study may not be sufficient compared to the external validation.
Conclusion
In conclusion, our study identified the risk factors related to irAEs occurrence and constructed and assessed successfully the predictive model of irAEs in patients with NSCLC treated by ICIs using clinical and hematological parameters from real-world evidence. Although it needs further investigation, the predictive model may be possibly incorporated into clinician decision-making and minimize adverse effects while optimizing the quality of life of the cancer patients receiving immunotherapy.
Footnotes
Abbreviations
Authors’ Note
W.G. and Q.L. contributed equally to this work. Y.Y. conceived the manuscript. W.G. wrote the initial draft of the manuscript and processed the data. Q.Q. collected and processed data. Y.Z. and M.Y. participated in the revision of the manuscript. All authors have read and approved the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
