Abstract
Keywords
Introduction
Primary liver cancer represents a common malignancy in China. 1 Due to the insidious onset of hepatocellular carcinoma, lack of typical clinical manifestations in the early stage, and high malignancy and rapid progress, most patients have already progressed to the middle and late stages at initial diagnosis, 2 and only a few patients can undergo surgical removal. 3 Primary liver cancer is a blood-rich tumor, with the majority of its blood supply coming from the hepatic artery and less from the portal vein. For patients with unresectable hepatocellular carcinoma, transcatheter arterial chemoembolization (TACE) is an important treatment option.4,5 Drug-carrying microspheres, a new embolic material, are characterized by high local drug concentration in tumors, low drug concentration in peripheral blood circulation, and fewer adverse effects. However, whether drug-eluting bead TACE (DEB-TACE) may more effective than conventional TACE (C-TACE) in intermediate to advanced tumors is controversial due to the lack of relevant clinical studies with large samples. This study compared the effectiveness and safety of C-TACE with DEB-TACE in patients with unresectable hepatocellular carcinoma.
Materials and Methods
Research Subject
We conducted a retrospective analysis of the data of patients with unresectable hepatocellular carcinoma who underwent hepatic artery chemoembolization at our hospital from July 2019 to April 2020. The reporting of this study conforms to STROBE guidelines. 6
Inclusion criteria: (1) conform to the diagnosis of hepatocellular liver cancer based on hepatocellular carcinoma7,8; (2) China Liver Cancer Staging (CNLC) stages IIb, IIIa, and IIIb; liver function Child–Pugh grade A or B; Eastern U.S. Tumor Collaborative Group physical status score 0 to 2; (3) who are candidates for surgical resection or ablation; local ablation treatment of CNLC stage I and IIa liver cancer, due to reasons such as advanced age and severe cirrhosis; (4) partial obstruction of the main trunk of the portal vein or presence of more compensatory collateral vessels in the hepatic portal even with complete obstruction of the main trunk of the portal vein.
Exclusion criteria: (1) hepatocellular carcinoma has metastasized extensively and cannot be treated with hepatic artery chemoembolization and (2) combination of other tumors and presence of multiorgan failure. This study was a retrospective cohort study that enrolled 282 patients, including 233 men and 49 women, with an average age of 55.9 ± 10.0 years. The patients were divided into the C-TACE group (
Observation Indicators
We collected the following information: age, sex, cirrhosis, tumor length, tumor number, tumor location, presence of large vessel invasion, distant metastases, CNLC clinical liver cancer stage, and alpha-fetoprotein (positive for alpha-fetoprotein > 7 ng/mL), etc. Adverse reactions after embolization include fever, nausea, vomiting, nausea, and abdominal pain.
Surgical Procedure
All procedures were performed in the digital subtraction angiography suite. Patients with hepatocellular carcinoma were evaluated for tumor status preoperatively using computed tomography (CT) or magnetic resonance imaging (MRI) of the upper abdomen. Patients underwent routine sterilization following anesthesia. The patients were routinely sterilized after anesthesia. The femoral artery was punctured using the Seldinger technique, then a 4-F vascular sheath was inserted, and the catheter was advanced to the abdominal trunk artery under x-ray fluoroscopy to identify the tumor's feeding artery and any collateral hepatic arteries before embolization. A microcatheter was then selected for the tumor's feeding artery. 9 The DEB-TACE group used 100 to 300 or 300 to 500-mm diameter DC® drug-loaded microspheres (Biocompatibles, UK) as carriers and embolic agents. The drug-carrying microspheres were loaded with pirarubicin hydrochloride (60 or 80 mg; Shenzhen Main Luck Pharmaceuticals Inc., Lot No.: GMP H10930105) and mixed with a high-concentration contrast agent in ratios of 1 : 1, 1 : 1.1, or 1 : 1.2. The chemotherapeutic drug in the C-TACE group contained pirarubicin hydrochloride (60 or 80 mg, Shenzhen Main Luck Pharmaceuticals Inc., Lot No.: State Pharmaceutical Code H10930105), iodine oil as the drug carrier, and gelatin sponge granules (Hangzhou Alicon Pharmaceutical Co., Ltd) as the embolic agent. Subsequently, a mixture of embolic and chemotherapeutic agents was pulsed through a microcatheter into the tumor's blood supply vessels. The endpoint of embolization is the complete disappearance of tumor staining on imaging. 10 The microcatheter was removed after the procedure ended, and the wound was compressed and dressed to stop the bleeding (Figure 1).

Angiography before and after hepatic artery chemoembolization in patients with primary liver cancer stage IIb in China. A. Transcatheter hepatic artery angiography before chemoembolization (tumor lesions indicated by arrows). B. Reangiography after transcatheter hepatic artery chemoembolization with drug-laden microspheres shows no tumor staining of the original lesion.
Efficacy Assessment and Follow-up
To assess the outcome (modified Response Evaluation Criteria in Solid Tumors [mRECIST]), all patients underwent enhanced CT or MRI of the abdomen for review at 1, 3, and 6 months postoperatively. (1) Complete remission: disappearance of enhanced contrast in the arterial phase of all target lesions; (2) Partial remission: a 30% reduction in the sum of the diameters of the target lesions (arterial phase enhancement visualization); (3) Disease stabilization: does not meet criteria for partial remission or disease progression; (4) Disease progression: a 20% increase in the sum of the diameters of the target lesions, visualized during the arterial phase enhancement or appearance of new lesions; (5) Progression-free survival: the time from randomization to disease progression or death from any cause.
The patients were followed up by phone or reviewing their outpatient charts to follow up on relapse and survival. Follow-up began 1 month post-TACE and conducted every 2 to 3 months on average, continuing until November 2021.
Statistical Processing
All data were processed using SPSS 20.0 software. Measurement data conforming to a normal distribution were expressed as mean
Results
Comparison of Preoperative Indicators Between the Two Groups
We compared sex, age, cirrhosis, tumor site, large blood vessel invasion, lymph node metastasis, and CNLC stage between the two groups. However, the differences were not statistically significant (all
Comparison of Preoperative Data Between the Two Groups of Patients With Liver Cancer.
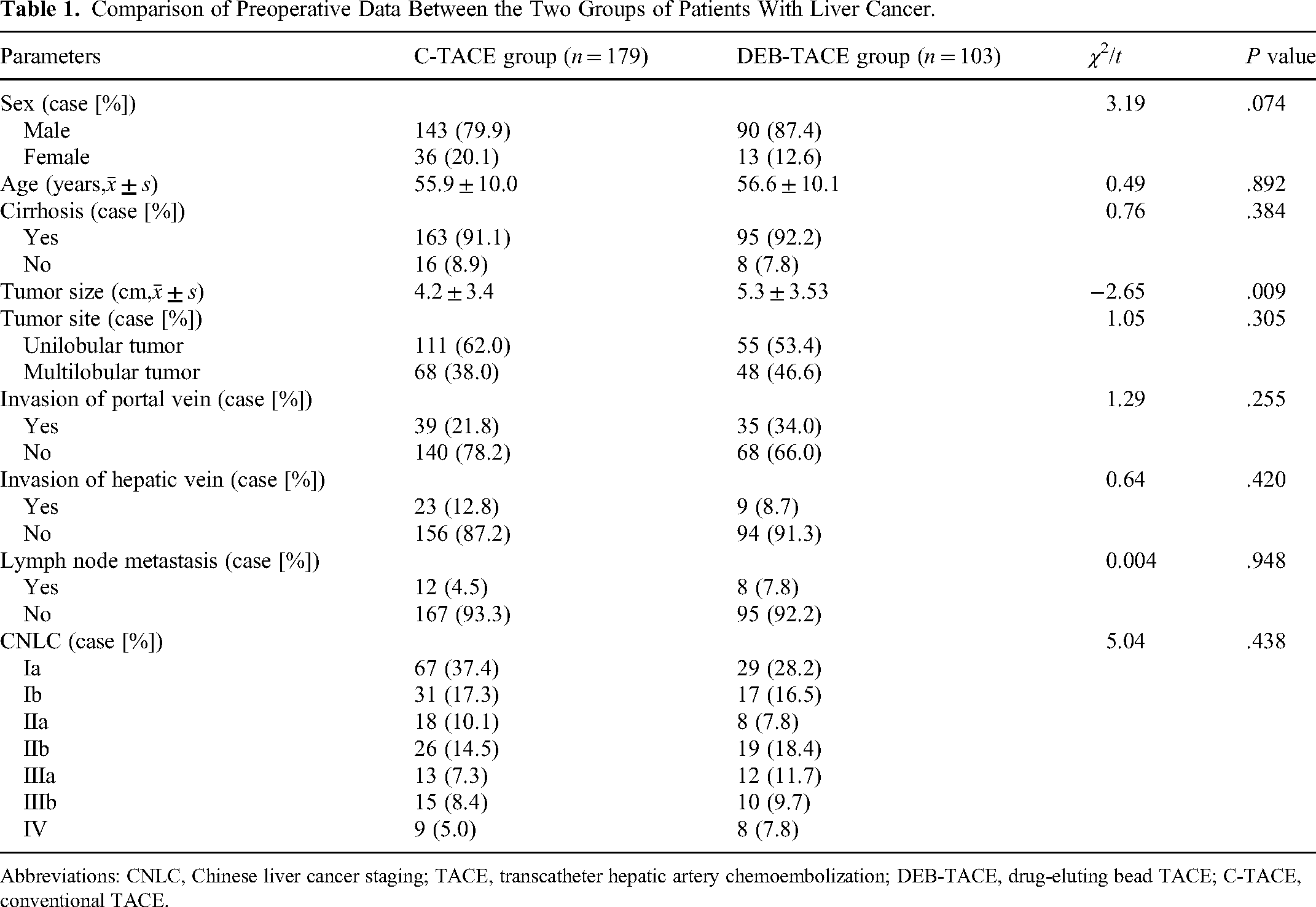
Abbreviations: CNLC, Chinese liver cancer staging; TACE, transcatheter hepatic artery chemoembolization; DEB-TACE, drug-eluting bead TACE; C-TACE, conventional TACE.
Evaluation of C-TACE and DEB-TACE Outcomes Using Liver Biochemical Parameters
Liver biochemical indexes after C-TACE and DEB-TACE were analyzed using the
Liver Biochemical Parameters of the Patients in C-TACE and DEB-TACE Groups.

Abbreviations: DEB-TACE, drug-eluting bead transcatheter arterial chemoembolization; C-TACE, conventional TACE.
Comparison of Adverse Reactions Between the Two Groups
The incidence of adverse reactions was 53.1% (95/179) in the C-TACE group and 34.0% (35/103) in the DEB-TACE group, respectively, with the difference being statistically significant (χ2 = 7.34,
Adverse Reactions of the Patients in the C-TACE and DEB-TACE Groups.

Abbreviations: DEB-TACE, drug-eluting bead transcatheter arterial chemoembolization; C-TACE, conventional TACE.
Comparison of the Efficacy of the Two Groups
After excluding patients who died or were lost to follow-up at 6 months postoperatively, 240 patients (148 in the C-TACE group and 92 in the DEB-TACE group) were included in the efficacy and survival analyses. Based on the mRECIST criteria to assess the efficacy at 1 month postoperatively, 75 patients (50.7%) had complete remission, 49 (33.1%) had partial remission, 9 (6.1%) had stable disease, and 15 (10.1%) had disease progression in the C-TACE group, and 46 (50.0%), 31 (33.7%), 8 (8.7%), and 7 (7.6%) in the DEB-TACE group, respectively, and the difference between the two groups was not statistically significant (χ2 = 0.84,
Efficacy of the C-TACE and DEB-TACE Groups.

Abbreviations: C-TACE, conventional transcatheter arterial chemoembolization; DEB-TACE, drug-eluting bead transcatheter arterial chemoembolization.
Survival Analysis
All patients were followed from 1 to 31 months (median, 13 months). The median survival time was 13 months for the C-TACE group and 14 months for the DEB-TACE group. The cumulative survival rates at 6 months/1 year/2 years postoperatively were 85.1%, 75.0%, and 66.7% in the DEB-TACE group and 82.8%, 71.6%, and 63.8% in the C-TACE group, respectively, and no statistically significant difference was observed (χ2 = 0.01,
Statistical Table for Analysis of Patient Survival Postoperatively.

Abbreviations: DEB-TACE, drug-eluting bead transcatheter arterial chemoembolization; C-TACE, conventional TACE.
Overall survival and progression-free survival were analyzed using the Kaplan–Meier method for patients after C-TACE and DEB-TACE treatments (Figure 2). The overall survival analysis results showed that the survival rate of patients undergoing DEB-TACE was not significantly different from that of patients undergoing C-TACE within 20 months postoperatively. Additionally, the chi-square test results for overall survival rates between the two treatments showed no significant difference (χ2 = 0.20,

Analysis of patient survival postoperatively.
Discussion
The ideal therapeutic effect of transcatheter hepatic artery embolization is to accurately embolize the supporting vessels of the tumor, so as to continuously maintain the maximum concentration of antitumor drugs in the local tumor lesion and the minimum systemic drug exposure. Two of the most commonly used clinical methods for unresectable hepatocellular carcinoma are C-TACE, which uses an iodine oil emulsion plus granular embolic agent, and DEB-TACE. Compared with C-TACE, embolic microspheres can be deposited in the peripheral vessels to maintain high local drug concentration for sustained embolic effect. We compared the liver indices of the two groups at 3 and 6 months postoperatively, with AST and bilirubin levels at 3 months postoperatively being significantly higher in the DEB-TACE group were than those in the C-TACE group (
In C-TACE, due to the lack of good stability of the mixture, chemotherapeutic drugs can easily separate from the target lesion and enter the blood circulation system,
13
and cancer cells may decompose and metabolize the iodinated oil in the lesion, which is gradually cleared by phagocytosis of the mononuclear–phagocytic system and blood flushing, whereas gelatin sponge particles are biodegradable embolic materials, which are generally absorbed by the organism after 2 weeks, and the blocked vessels can recanalize. Drug-loaded microspheres are a class of nondegradable embolic materials, which stably bind to adriamycin through ionic bonds and slowly release the drug in the bloodstream through an ion-exchange mechanism to permanently embolize tumor supply arteries while synergistically causing tumor tissue necrosis in combination with chemotherapeutic drugs.14,15 Zhang et al
16
reported that 1 mL of drug-loaded microspheres loaded with 40-mg pirarubicin can reach 200 µm from the microsphere and persist for at least 1 month, and Namur et al
17
showed in a nontumor animal model that although the half-life of adriamycin is only 35 to 48 h, 57% of its drug-loaded microspheres are retained after binding to the initial load, and 11% of the initial loading dose after 3 months is retained, leading to persistent local tissue necrosis and inflammatory response. At 6 months postoperatively, the DEB-TACE group had significantly decreased bilirubin levels compared with the C-TACE group, which was consistent with the above mechanism of action of DEB-TACE, and the long-term hepatoprotective properties may be more advantageous. The overall incidence of postoperative adverse effects was 53.1% in the C-TACE group, which was higher than that of the DEB-TACE group at 34.0% (
Although several studies have compared the efficacy of DEB-TACE and C-TACE for unresectable hepatocellular carcinoma, the results were quite different. Although Sacco et al 18 and Ferrer et al 19 found no statistically significant difference between the two groups, Burrel et al 20 showed complete, partial, and overall remission rates of 16.2%, 59.6%, and 75.8% after treatment with DEB-TACE, respectively. In this study, at 6 months postoperatively, the DEB-TACE group had complete, partial, and overall remission rates of 41.3%, 18.5%, and 59.8%, respectively, and 28.3% of patients had stable disease, and 12.0% had tumor progression. The DEB-TACE group was slightly more effective than the C-TACE group in terms of overall remission rate (59.8% vs 55.4%), proportion of patients with stable disease (28.3% vs 18.2%), and proportion of patients with tumor progression (12% vs 26.4%). However, the overall and progression-free survival of the two groups were not significantly different.
This study presents some limitations. First, selection bias and confounding factors may be present because this was a retrospective study. Second, longer postoperative survival rates and survival remain to be followed up further because of our short follow-up period and the lack of more accurate indicators for some patients who did not reach the survival endpoint. Finally, confounding factors, such as errors in judging tumor efficacy, may exist, and some patients may have died for reasons other than tumor progression alone. We will continue to perform subgroup analyses to address these situations.
Conclusion
In summary, based on our data, DEB-TACE may be slightly better than C-TACE in terms of safety and efficacy in the treatment of unresectable hepatocellular carcinoma. Our team will also continue to conduct subgroup analysis to gather more data to assist in guiding future clinical treatment.
Footnotes
Abbreviations
Authors’ Notes
Long Hai and Shuaiwei Liu contributed equally to this work.
Data Availability
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
This study was approved by the Medical Ethics Committee of the General Hospital of Ningxia Medical University (approval number: KYLL-2021-1069) and followed the principles of the Declaration of Helsinki. Written informed consent was obtained from each patient.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ningxia Natural Science Foundation, Ningxia Key Research and Development Program, and Ningxia Natural Science Foundation Key Project (Grant Nos. 2021AAC03364, 2022BEG02039, and 2022AAC02065).
