Abstract
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors, with more than 750,000 new cases diagnosed each year globally, approximately half of which are in China. 1 Hepatitis B virus (HBV) is the most important factor leading to HCC, and >85% of HCC in China is associated with HBV infection or carriage. 2 HCC has an insidious onset, and 70%–80% of the patients are already in the middle or advanced stage at the time of diagnosis, which limits the treatment options. The 5-year survival rate of HCC is <20%. 3 The emergence of immunotherapy represented by anti-programmed death 1 (PD-1)/programmed cell death-ligand 1 (PD-L1) therapy has broken the deadlock in the treatment of intermediate and advanced HCC. Especially, the combination of immuno–targeted therapy has shown significant efficacy. 4 Camrelizumab is a humanized PD-1 monoclonal antibody drug. Based on the satisfactory results of a national multicenter phase II clinical trial, this drug was officially approved by the Chinese Drug Administration in 2020 for the second-line treatment of advanced HCC, making it the first PD-1 inhibitor approved for HCC therapy in China. 5 Lenvatinib is an oral small-molecule multikinase inhibitor. The REFLECT phase III clinical trial reported that lenvatinib has a better overall response rate and time to progression than sorafenib and hence has been approved by the Food and Drug Administration as the first-line treatment of HCC. 6 This drug has been shown to inhibit angiogenesis and immunosuppression in the tumor microenvironment (TME), thereby acting synergistically with PD-1 inhibitors. 7 According to the findings of the KEYNOTE-524 study, the disease control rate of lenvatinib combined with pablizumab reached 85% in advanced HCC. 8 Camrelizumab is independently developed in China and has a relatively low price. Hence, the camrelizumab combined with lenvatinib (C + L) regimen has been widely adopted in Chinese patients with advanced HCC. Nonetheless, the efficacy of immunotherapy varies to a great extent in HCC. A study has shown that patients with HBV infection are more likely to benefit from immunotherapy than those without viral infection. 9 Identifying biomarkers that predict the efficacy of immunotherapy in patients with hepatitis B virus-associated hepatocellular carcinoma (HBV-HCC) is particularly important to maximize the treatment efficacy.
The eosinophil (EOS) count is a commonly used indicator in routine blood tests as it is easy to obtain and is inexpensive. EOSs can influence the TME and can recruit lymphoid T cells, thereby increasing the number of T cells in the TME.10–12 Certain studies have suggested that EOS counts and their extent of change may be correlated with the efficacy of immunotherapy in lung and kidney cancers; however, there is a lack of such studies in liver cancer.13,14 Both PLR and NLR serve as important proxies for inflammatory factors. These parameters are effective in assessing the efficacy and prognosis of immunotherapy for various solid tumors, such as gastric, esophageal, and pancreatic cancers. A study has reported NLR and PLR to be prognostic markers for HCC. 15
Therefore, two classical and commonly used indicators, PLR and NLR, were used as references in this study to explore the relationship between the levels of peripheral blood EOS and the prognosis of patients with HBV-HCC before C + L treatment and after three cycles of treatment.
Patients and Methods
Study Participants
A total of 200 intermediate and advanced HBV-HCC patients treated with carelizumab combined with lunvalvatinib (C + L) were admitted to The Affiliated Hospital of Xuzhou Medical University and The Second Affiliated Hospital of Xuzhou Medical University between January 2018 and August 2023 were selected for retrospective analysis. This retrospective study was approved by the Ethics Review Committee of the Affiliated Hospital of Xuzhou Medical University and The Second Affiliated Hospital of Xuzhou Medical University (XYFY2022-KL207-01) and was conducted in accordance with the principles of the Declaration of Helsinki. Due to the retrospective nature of this study, the requirement for written informed consent was waived.
Inclusion criteria: (1) histopathological or imaging-confirmed diagnosis of HCC; (2) serum HBV surface antigen-positive and serum HBV DNA-positive status; (3) completion of ≥3 cycles of C + L treatment; (4) staging of HCC assessed using mature imaging, including computer tomography (CT) and magnetic resonance imaging (MRI), with at least one target lesion measurable via imaging; (5) Barcelona clinic liver cancer, (BCLC) stage B and C; (6) Child–Pugh classification of A–B; (7) Eastern Cooperative Oncology Group (ECOG) score of ≤2.
Exclusion criteria: (1) HCC combined with hepatitis C or other viral infections; (2) HCC combined with other malignant tumors; (3) cholangiocellular carcinoma and mixed hepatocellular carcinoma; (4) patients with serious adverse reactions and adverse reaction-related deaths; (5) HCC combined with severe cardiac, cerebral, hepatic, or renal insufficiency or severe coagulation dysfunction; (6) HCC combined with other infectious, hematological, autoimmune, or other diseases that may affect inflammatory indexes or organ transplantation; (7) having recently received blood transfusion therapy.
Treatment Methods
Camrelizumab 200 mg IV every 3 weeks; lenvatinib (weight >60 kg 12 mg, weight <60 kg 8 mg) orally once a day.
Collection of Clinical Data
General information on the patients, including age, gender, and tumor stage, were collected. Furthermore, evaluation data on the therapeutic efficacy after three cycles of C + L, imaging data during the treatment period, and EOS, NLR, and PLR parameters in routine blood tests were obtained.
Evaluation Criteria
Efficacy evaluation indexes included short-term and long-term efficacy, and the chest and abdomen CT or MRI were reviewed after three cycles of C + L treatment. The short-term efficacy was classified into complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) in accordance with the modified Revised Evaluation Criteria for the Evaluation of the Efficacy of Solid Tumors. The long-term efficacy was evaluated in terms of PFS and overall survival (OS).
Variables Included in the Study and Follow-up
EOS, NLR, PLR, and tumor response were recorded before and after three cycles of C + L. PFS and OS served as the endpoint events. The cutoff date for follow-up was September 30, 2023, via regular visits to the hospital or via telephone.
Ethical Statement
This study was approved by The Ethics Committee of the Affiliated Hospital of Xuzhou Medical University and The Ethics Committee of the Second Affiliated Hospital of Xuzhou Medical University on June 23, 2022 (Approval Number: XYFY2022-KL 207-01), and all patients signed written informed consent prior to treatment with camrelizumab combined with lenvatinib.
Statistical Methods
The R software was used for data analysis. Data were presented in the form of median (interquartile spacing) or frequency (percentage). The Wilcoxon rank–sum test was used to compare the medians between groups, and the chi-square test or Fisher's exact test was employed to compare the rates. The diagnostic value of EOS was assessed by plotting the receiver operating characteristic (ROC) curve and determining the area under the curve (AUC). The Kaplan–Meier method was applied for survival curves, and the log-rank test was used to test the difference in survival rates between the two groups. Furthermore, univariate and multivariate analyses were performed using the Cox equiproportional risk model, and variables with P < 0.05 in the univariate Cox analysis were included in the multivariate regression analysis. The statistical test was a two-sided probability test, and the difference was considered statistically significant at P < 0.05.
Results
General Clinical Characteristics of the Patients
A total of 200 patients were enrolled in this study, of which 82 (41.0%) were treated with combined TACE. Of these, 160 (80.0%) were men and 31 (15.5%) patients were ≥70 years of age. Of the 200 patients with HCC, 156 (78.0%) were in BCLC stage C and 44 (22.0%) were in stage B. With regard to Child–Pugh grading, 126 (63.0%) patients were in grade A and 74 (37.0%) were in grade B. Mortality outcome occurred at the end of follow-up in 105 (52.5%) patients (Table 1).
General Situation of 200 Patients with Hepatocellular Carcinoma

Relationship among peripheral blood EOS and clinical data, NLR, and PLR in patients with HCC, EOS, NLR, and PLR before and after three cycles of C + L treatment were recorded as EOS0, NLR0, PLR0 and EOS3, NLR3, PLR3, respectively. EOS0 was 0.11 ± 0.14 and EOS3 was 0.14 ± 0.16; EOS3 was significantly increased compared with EOS0 (P = 0.012). Furthermore, EOS0 was correlated (P < 0.05) with portal vein invasion, tumor diameter, ALT, PLT, NLR3, and OS (P < 0.05). EOS3 was correlated with the patient's sex and PFS (P < 0.05). However, EOS0 and EOS3 were not correlated with NLR0, NLR3, PLR0, age, cirrhosis, extrahepatic metastasis, BCLC staging, Child grading, or efficacy evaluation after three cycles (P > 0.05).
Alterations in EOS, NLR, and PLR of Patients with HCC in Different Efficacy Groups Before and After Treatment
According to the tumor response, the 200 patients were categorized into two groups: progressed and nonprogressed. Patients whose treatment efficacy was evaluated as PD were included in the progressed group, whereas those whose efficacy was assessed as PR or SD were included in the nonprogressed group. Before C + L treatment, patients in the nonprogressed group showed a significantly higher EOS0 (0.14 ± 0.22) than those in the progressing group (0.10 ± 0.08) (P = 0.02); nonetheless, there was no statistically significant difference in NLR0 or PLR0 levels (P > 0.05). After three cycles of C + L treatment, patients in the nonprogressed group exhibited significantly higher EOS3 (0.18 ± 0.26) than those in the progressing group (0.13 ± 0.09) (P = 0.04). NLR3 was significantly lower in the nonprogressed group (3.50 ± 2.13) than in the progressed group (4.10 ± 3.21) (p = 0.026), and PLR3 was also significantly lower in the nonprogressed group (133.06 ± 63.70) than in the progressing group (144.13 ± 66.74) (P = 0.017). Moreover, the differences were statistically significant (P < 0.05).
Correlation among EOS, NLR, and PLR and Outcome in Patients with HCC
Figure 1 depicts the ROC curves based on the EOS3, NLR3, and PLR3 of patients after completing three cycles of C + L treatment. The results indicated that the AUC of EOS3 for the predictive ability of patient survival was 0.61 (95% CI: 0.52-0.71, P = 0.016). The AUCs were 0.60 (95% CI: 0.52-0.69, P = 0.027) for NLR3 and 0.60 (95% CI: 0.51-0.68, P = 0.034) for PLR3. These findings suggest that the predictive value of EOS3 for the long-term efficacy of C + L treatment in patients with intermediate and advanced HCC was better than that of NLR3 and PLR3. The specific results are presented in Table 2. In addition, Figure 2 illustrates the predictive ability of EOS for patient survival. The AUC of EOS0 was 0.63 (95% CI: 0.51-0.76, P = 0.04), with the best cutoff value of 0.045, and its sensitivity and specificity were 30.04% and 86.3%, respectively. The AUC of EOS3 was 0.61 (95% CI: 0.52– 0.71, P = 0.016), with the best cutoff value of 0.105, with a predictive sensitivity of 57.4% and a specificity of 67.8%.

Correlation of EOS3, NLR3, and PLR3 with the efficacy of C + L treatment in 200 HCC patients ROC curve.

EOS predicts ROC curves in 200 patients with HCC.
Correlation of EOS3, NLR3 and PLR3 with C + L Treatment Efficacy

Association Between EOS0 and EOS3 Levels and Clinical Characteristics of the Patients
The C + L pre-treatment group (EOS0) and the three-cycle C + L post-treatment group (EOS3) were categorized as high (EOS0 ≥ 0.045) and low (EOS0 < 0.045) EOS0 and high (EOS3 ≥ 0.105) and low (EOS3 < 0.105) EOS3, respectively, based on the cutoff values of the ROC curves. Different levels of EOS0 were correlated with Child grading and survival status (P = 0.03). Moreover, different levels of EOS3 and sex (P = 0.02) were correlated. Statistically significant differences were observed between different levels of EOS0 and EOS3 groups and efficacy evaluation after 3 weeks (both P < 0.05) (Table 3).
General Conditions of 200 HCC Patients Grouped According to High and Low EOS0 and EOS3 Conditions

Relationship of Peripheral Blood EOS with PFS and OS in Patients with HCC
Kaplan–Meier survival analysis indicated the following: (1) the median PFS of 9 months (95% CI: 1.1-2.9) in the high EOS0 group was higher than that of 5 months (95% CI: 0.3-0.9) in the low EOS0 group (P = 0.027), as depicted in Figure 3A; (2) The median PFS of 9 months (95% CI: 0.6-1.3) in the high EOS3 group was higher than that of 8 months (95% CI: 0.8-1.6) in the low EOS3 group (P = 0.018), as portrayed in Figure 3B; (3) The median OS of 10 months (95% CI: 0.4-1.2) in the high EOS0 group was higher than that of 7 months (95% CI: 0.8-2.3) in the low EOS0 group (P = 0.032), as presented in Figure 3C; (4) The median OS of 15 months (95% CI: 0.4-0.9) in the high EOS3 group was higher than that of 8 months (95% CI: 1.1-2.5) in the low EOS3 group (P < 0.0001). See Figure 3D.
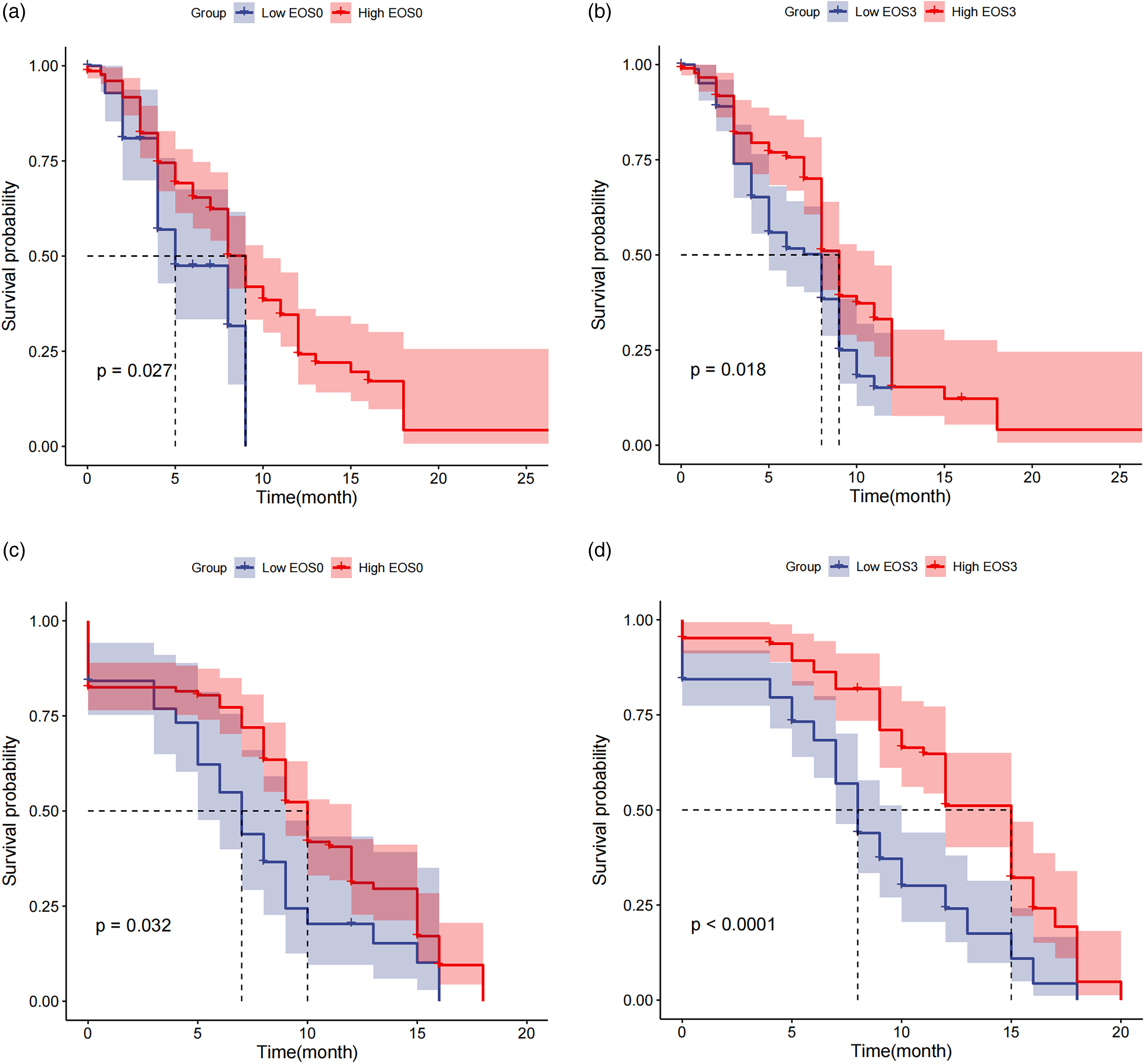
Kaplan–Meier survival curve analysis.
Analysis of Factors Affecting C + L Treatment in Patients with HCC
After calibrating EOS0, EOS3, NLR0, NLR3, PLR0, PLR3, BCLC staging, Child grading, ECOG scores, and other factors of the patients, Cox multifactor analysis revealed the following: (1) EOS3, age, cirrhosis, portal vein invasion, and efficacy evaluation after three cycles were independent predictors of PFS (all P < 0.05). On the contrary, E0S0, NLR0, NLR3, PLR0, and PLR3 did not have a significant predictive value (P > 0.05), as shown in Table 4. (2) EOS0, NLR0, PLR3, age, ECOG score, and efficacy evaluation after three cycles were independent predictors of OS (all P < 0.05). However, NLR3, PLR0, cirrhosis, portal vein invasion, and BCLC stage showed no significant predictive value for OS (P > 0.05), as presented in Table 5.
Multivariate Analysis of PFS Affecting 200 HCC Patients

Multivariate Analysis Affecting the OS of 200 HCC Patients

Discussion
The most common cause of liver cancer in East Asia is chronic HBV infection. 16 HBV is a partial-duplex DNA virus comprising a core protein and a DNA polymerase envelope wrapped by an envelope protein (HBsAg). The virus enters hepatocytes via receptor-mediated endocytosis and subsequently gains entry into the nucleus, where the relaxed circular DNA is converted to covalently closed circular DNA, which is often stochastically integrated into the host genome and plays a pertinent role in the oncogenic transformation of the infected cell. On one hand, the integration of HBV DNA into the host chromosome affects cellular gene transcription, thus inducing chronic inflammation with hepatocyte death, regeneration, and fibrosis. On the other hand, it activates protooncogenes, growth factors, receptors, as well as inactivates antioncogenes. Chronic inflammation and repeated necrosis and regeneration of hepatocytes are the major factors in the occurrence of cirrhosis and liver cancer.17,18
Camrelizumab, a monoclonal antibody against human immunoglobulin G4, can bind to programmed death receptor 1 and block the immunosuppressive response mediated by this pathway, thereby enhancing the ability of immune cells to attack cancer cells. 19 Lenvatinib is a tyrosine kinase receptor inhibitor capable of inhibiting the kinase activity of vascular endothelial growth factor receptors, which reduces tumor angiogenesis and controls tumor growth. 6 While monotherapy with camrelizumab or lenvatinib is prone to drug resistance, the combination of the two can enhance the therapeutic effect by augmenting the infiltration and activity of immune cells, alleviating the production of immunosuppressive factors in the tumor, and other mechanisms. 20 A study observed that the overall remission rate of camrelizumab in combination with lenvatinib in treating intermediate and advanced HCC was 52.0%. 21 Moreover, subgroup analyses of international multicenter phase III trials have reported lower response rates in combined immuno–targeted therapies in nonviral-associated HCC than in viral HCC.22–24 However, reliable biomarkers are still lacking to predict the efficacy of combined immuno–targeted therapy for HBV-HCC. The presence of a large number of immune cells, especially cytotoxic CD8+ T cells, in the TME is closely associated with disease progression.25,26 Eosinophils are bone marrow-derived granulocytes that are evolutionarily conserved pleiotropic cells that display key effector functions in allergic diseases (eg, asthma). Moreover, eosinophils penetrate into multiple tumors and regulate tumor progression,27,28 either by directly interacting with tumor cells or indirectly by shaping the TME. Once in the TME, eosinophils release many cytotoxic molecules, including the main essential protein, 29 tumor necrosis factor-α, as well as granzyme A as well as B, 30 which directly causes cell lysis. In addition, eosinophils may play a role in stabilizing the TME by promoting the differentiation of M2 macrophages into M1 phenotypes, and the M1 phenotype macrophage environment tends to be associated with reduced release of vascular-derived growth factors (including vascular endothelial growth factor and HIF-1α), thus attenuating tumor-mediated angiogenic. 31 At the same time, eosinophils recruit tumor-reactive CD8+ T cells by secreting chemokines, including CCL 5, CXCL9, and CXCL10, which increases the number of T cells in the cancer environment, thus fighting tumors and slowing and inhibiting the development of cancer. 32 Overall, eosinophils can enhance anti-tumor activity and prolong the survival of tumor patients by attenuating tumor angiogenesis, releasing toxic molecules, and recruiting CD8+ T cells.29,30 Increasing the number of T cells as well as their power is one of the main targets of immunotherapy for cancer patients today. Considering the correlation of eosinophil numbers with T cell numbers, changes in the number of peripheral eosinophils in peripheral blood may affect the efficacy of immunotherapy.
In recent years, several studies have shown that NLR as well as PLR are important prognostic markers33,34 associated with cancer immunotherapy. Neutrophils play an important role in the development of tumor inflammation by producing chemokines that inhibit the lymphocyte immune activity as well as cytokines.35–39 Therefore, if large numbers of neutrophils are present in the tumor environment, an inflammatory response will lead to cancer cell proliferation as well as metastasis. 40 Platelets are also involved in the tumor metastasis process, The specific mechanisms may be as follows: 1. Platelets promote epithelial—mesenchymal transition (EMT), cause tumor cells to lose apical-basal polarity, overcome apoptosis and exhibit migration as well as an invasive phenotype; on the other hand, EMT promotes the degradation of the basement membrane as well as the underlying extracellular matrix, which further promotes the migration of tumor cells 41 ; 2. Platelets release various cytokines, such as the platelet-derived growth factor as well as the transforming growth factor-β, and transport it to specific sites, indicating involvement in angiogenesis and tissue repair process. 42 EOS is also a routine test item for cancer patients during the first diagnosis and treatment, which offers the characteristics of low price, safety, speed, and high repetition rate. Recently, eosinophils have been highlighted as potential cellular biomarkers, even end-stage effector cells 43 for cancer therapy, and the relationship between EOS and its dynamic changes and immunotherapy efficacy and prognosis has become the current research hotspot. 44 The association of EOS and its dynamic alterations with the efficacy and prognosis of immunotherapy has become a hot research topic.
In this study, EOS was significantly higher after C + L treatment, that is, EOS3 was higher than EOS0 (0.15 vs 0.10, P = 0.005). Before C + L treatment, patients in the nonprogressed group showed a significantly higher EOS0 (0.14 ± 0.22) than those in the progressed group (0.10 ± 0.08) (P = 0.02). Nevertheless, there was no statistically significant difference in NLR0 and PLR0 levels (P > 0.05). After three cycles of C + L treatment, patients in the nonprogressed group had significantly higher EOS3 (0.18 ± 0.26) than those in the progressed group (0.13 ± 0.09) (P = 0.04). In addition, patients in the nonprogressed group had significantly lower NLR3 (3.50 ± 2.13) than those in the progressed group (4.10 ± 3.21) (P = 0.026), while those in the nonprogressed group had significantly lower PLR3 (133.06 ± 63.70) than those in the progressed group (144.13 ± 66.74) (P = 0.017). All differences were statistically significant (P < 0.05). The results of ROC curves based on EOS3, NLR3, and PLR3 of patients after completing three cycles of C + L therapy indicated the following: the AUC for the predictive value of EOS3 for patient survival was 0.61 (95% CI: 0.52-0.71, P = 0.016), that of NLR3 was 0.60 (95% CI: 0.52-0.69, P = 0.027), and that of PLR3 was 0.60 (95% CI: 0.51-0.68, P = 0.034). Upon calibrating patients’ EOS0, EOS3, NLR0, NLR3, PLR0, PLR3, age, gender, ECOG score, and BCLC stage, cox multifactorial analysis revealed that EOS3, age, cirrhosis, portal vein invasion, and efficacy evaluation after three cycles were independent predictors of PFS (all P < 0.05) and that EOS0, NLR0, PLR3, age, ECOG score, and efficacy evaluation after three cycles were independent predictors of OS (P > 0.05). These findings signify that EOS has a certain predictive value for the efficacy of C + L therapy in patients with intermediate and advanced HCC and may be superior to NLR and PLR. The possible reasons for this are as follows: 1. EOS can secrete signal substances to call lymphatic T cell aggregation and increase the number of T cells in the cancer environment, and the number and strength of T cells are closely related to the efficacy of immunotherapy. 2. NLR and PLR are both ratios, bivariate, with more affected factors, and the uncertainty may be higher. Relatively, EOS is a single variable, and only a few factors are affected; therefore, the prediction accuracy may be higher. Different levels of EOS0 were correlated with child grading and survival status (P = 0.03), and different levels of EOS3 were correlated with sex (P = 0.02). There were statistically significant differences between groups with different levels of EOS0 and EOS3 and evaluation efficacy after 3 weeks (both P < 0.05). Median PFS was higher in the high EOS0 group than in the low EOS0 group (9 months vs 5 months, P = 0.027); median PFS was higher in the high EOS3 group than in the low EOS3 group (9 months vs 8 months, P = 0.018); median OS was higher in the high EOS0 group than in the low EOS0 group (10 months vs 7 months, P = 0.032); median OS was higher in the high EOS3 group than in the low EOS3 group (15 months vs 8 months, P < 0.0001). Thus, it was evident that median PFS and OS were significantly higher in the high EOS0 and EOS3 groups than in the low EOS0 and EOS3 groups. A past retrospective study on patients with metastatic renal cell carcinoma treated with navuliumab 13 showed that an increase in EOS and relative EOS change at 6 weeks of treatment was associated with a favorable response to immunotherapy (P = 0.012 and P = 0.024, respectively). In the group of patients with a 10% decrease in the relative change, PFS was significantly lower than that in the other groups (P = 0.0044 for the 10% increase group and P = 0.03 for the unchanged group). Another retrospective study performed on patients with advanced non-small cell lung cancer receiving immune checkpoint inhibitors 14 reported higher baseline EOS (compared with lower baseline EOS) in association with a higher objective remission rate (40.9% vs 28.8%, P = 0.029) and longer PFS (8.93 months vs 5.87 months, P = 0.038). The results of these two studies are similar to those of the present study. The uniqueness of our study is that it reports, for the first time, the predictive role of EOS in C + L treatment in patients with HCC, with longer PFS and OS in patients with high EOS0 and EOS3. However, there are some limitations in this study. First, China is a major country with hepatitis B, and all the patients in this study are from China, which may have led to a selection bias. Second, the patients in this study were treated with only one PD-1 inhibitor (camrelizumab); therefore, future studies will need to explore whether other PD-1 inhibitors have similar effects. Third, in our study, the significant p-value of EOS3 as a possible prognostic marker for treatment efficacy the sensitivity was low (<80%), which could affect EOS3 usability as a prognostic marker for HCC treatment. Finally, our study was a retrospective study conducted in dual centers with a small sample size. Therefore, future randomized controlled trials with large sample sizes in multiple centers are needed to confirm the present findings.
Conclusion
EOS may be an ideal indicator for predicting the treatment efficacy and prognosis of C + L in patients with intermediate and advanced HBV-HCC. However, this research is a two-center retrospective study with a small sample size. Therefore, future validation with large sample sizes and multicenter randomized controlled trials is necessary to validate the findings.
Footnotes
Authors’ Contribution
PFQ designed the study. HNL, PFQ, and DP collected the clinical data. ZYY, and DP analyzed the data. XXC wrote the paper. PFQ revised the paper. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Affiliated Hospital of Xuzhou Medical University (XYFY2022-KL207-01).Due to the retrospective nature of this study, we waived the requirement for written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
