Abstract
This study aimed to investigate the safety and effectiveness of irreversible electroporation ablation for unresectable large liver cancer. Fourteen patients were enrolled: 8 with large hepatocellular carcinoma (tumor diameter: 5.1-11.5 cm) and 6 with medium hepatocellular carcinoma (tumor diameter: 3.0-4.1 cm). All patients received percutaneous irreversible electroporation ablation. Ablation time and the incidence of complications were assessed by a
Introduction
Primary liver cancer is the most common malignant tumor, mainly caused by alcoholic cirrhosis, viral hepatitis, and fatty hepatitis. The incidence of primary liver cancer is 5 to 8 times higher in China than in the United States and Europe because of the increased prevalence of hepatitis B virus infection in China. Although surgery is the gold standard treatment for patients with primary liver cancer, it is suitable only for less than 20% of patients. 1 Patients with unresectable tumors are often treated with chemotherapy; however, there are some side effects and its efficacy is limited. Currently, radiofrequency ablation (RFA), microwave ablation (MWA), cryoablation, and other minimally invasive therapies and local treatments are being used increasingly to treat liver cancer. However, RFA and MWA are not suitable for tumors larger than 5 cm, and because of the “heat sink” effect, they cannot completely ablate tumors near the great vessels. 2 Using cryoablation, it is difficult to avoid the damage in large blood vessels and other important structures, and there are a series of complications caused by rupture puck. In recent years, irreversible electroporation (IRE) has been used increasingly in the clinic, providing a new choice for liver cancer. The IRE ablation uses a high-pressure pulse current that acts on the target cell membrane, resulting in unrecoverable nanoscale perforation. This leads to cell death and tumor ablation, without damaging adjacent tissue blood vessels, gall bladder, and other vital organs. 3 Thus, for large hepatocellular carcinoma (HCC) and/or HCC adjacent to large vessels, IRE ablation may be a more appropriate choice. This study focused on the safety of the clinical application of IRE ablation for patients with large liver HCC whose tumors had maximum diameter greater than 5 cm.
Materials and Methods
The clinical trial was registered with the US National Institutes of Health (ID.NCT02329106) and was approved by the regional ethics committee at Guangzhou Fuda Hospital. Each participant provided written informed consent in accordance with the Declaration of Helsinki.
We performed a prospective search for patients undergoing IRE for HCC from July 2015 to March 2016. The inclusion criteria were preoperative histopathological diagnosis of primary liver cancer, preoperative performance status score ≤2, and not eligible for surgical resection. The exclusion criteria were: could not tolerate anesthesia through the trachea, severe coagulopathy insufficiency, severe liver and kidney function insufficiency, and cardiac pacemaker or defibrillator.
According to the inclusion and exclusion criteria, 14 patients were enrolled. According to the preoperative tumor size, the 14 cases were divided into a large HCC group (preoperative imaging determine the maximum tumor diameter greater than 5 cm) 4 and a medium HCC group. The large HCC group had 8 patients (5 males and 3 females, age 24 to 65 years, mean age 50 ± 13 years): 6 cases of HCC and 2 cases of intrahepatic cholangiocarcinoma (tumor diameter 5.1-11.5 cm, mean diameter 7.2 ± 2.2 cm). The medium HCC group had 6 patients (all males, mean age 57 ± 11 years): 2 cases of HCC and 4 cases of intrahepatic cholangiocarcinoma (tumor diameter 3.0-4.1 cm, mean diameter 3.5 ± 0.5 cm). General information for all the patients is shown in Table 1.
General Information for 14 Cases of Primary Liver Hepatocellular.

Abbreviations: AFP, alpha fetoprotein; HCC, hepatocellular carcinoma; ICC, intrahepatic cholangiocellular carcinoma.
aAll of the patients were from China.
bCancer stage, according to American Joint Committee on Cancer (AJCC) staging system (2002).
The IRE was performed using an IRE ablation system (NanoKnife; AngioDynamics, Queensbury, New York). The main configuration included a high-voltage current generator (maximum power output of 3 kV, 50 A), an electrocardiogram (ECG) synchronization (an AccuSyneR synchronizer Device, AccuSync Medical Research Corporation, Milford, Connecticut), and two 15-cm monopolar probes.
The perioperative treatment plan was based on preoperative computed tomography (CT) imaging, which decided the number and spacing of the probes. Patients were given general anesthesia and muscle relaxants. Probes were placed under ultrasound (US; IU22; Philips Medical Systems, Bothell) and CT (CT SOMATOM Definition 64 AS; Siemens Medical Solutions, Forchheim, Germany) image-guidance. The probe spacing was 1.2 to 2.5 cm, and the effective exposure probe distance was 1.5 to 2.5 cm. 5 Energy was then applied at 800 to 2200 V/cm (pulse length was 90 μs, 6 pulse repetition was 70-90). Before delivering all therapeutic pulse, a test pulse was delivered. After the test pulse proceeded smoothly, a therapeutic pulse was delivered in less than 2 minutes. All pulses were delivered in the ventricular refractory period to avoid the occurrence of arrhythmias because electrocardiogram (ECG) synchronization monitored the patients’ R wave and sent the signal to the IRE generator which released the pulse energy after a 50-μs delay. Treatment was repeated to cover the entire target if the target treatment zone was greater. 7 After removal of the 2 needles, CT imaging was performed. The patients continued to be sedated with mechanical ventilation for 2 hours. Patients were then transferred to the intensive care unit for 24 hours. After their vital signs were stable, the patients were transferred to general wards and were provided with anti-infection treatment, drugs to protect the stomach and liver, and nutrition and other symptomatic and supportive treatments.
After the procedure, patients were followed up with contrast-enhanced CT scans at 1 to 3 months and at 3-month intervals. Complete ablation of the tumor was defined as an ablation region beyond the tumor, with clear boundaries and no evidence of arterial enhancement. Adverse events were recorded as per the unified standardized Society of Interventional Radiology grading system. 4 The definition of a major complication is an event that leads to substantial morbidity and disability that increases the hospital stay or the morbidity. All other complications were considered minor. Common procedural side effects such as pain, fever, and transient elevation of liver enzyme levels were excluded from the evaluation.
Data were analyzed using the SPSS 13 software for statistical analysis and were represented as
Results
Eight large liver tumors received a total of 136 cycles of IRE ablation. The procedure time was 70 to 125 min/case, with an average procedure time of 99 ± 23 min/case. The 6 medium liver tumors received a total of 33 cycles of IRE ablation. The procedure time was 45 to 75 min/case, with an average procedure time of 61 ± 10 min/case. The procedure time for medium liver tumors was significantly lower (
Irreversible Electroporation (IRE) Ablation Parameters of 14 Patients.

Complications
All patients tolerated IRE well. In the large HCC group, no treatment-related bleeding or other major adverse events occurred in the perioperative period. One patient had intraoperative blood pressure up to 200/83 mm Hg (1 mm Hg = 0.133 kPa), with a suspected IRE ablation zone near the adrenal gland, which returned to normal after treatment with intravenous nicardipine (0.5 mg). Minor adverse events comprised 3 cases of hypokalemia on postoperative day 1, which was improved by oral or intravenous potassium; 1 patient had low blood pressure, low white blood cells, and platelet function abnormalities postoperatively, which were improved by intravenous infusion of dopamine and phenylephrine or the same type of plasma. There were 4 cases of abdominal distension and 2 cases of limb edema, both were mild and improved after general symptomatic treatment.
In the medium HCC group, no major adverse events occurred in the perioperative period. One patient showed intraoperative heart rate acceleration up to 140 beats/min. Minor adverse events were 3 cases of hypokalemia, 3 cases of low serum albumin, 1 case of low blood pressure, and 1 case of stomach pain. The frequency of adverse events in the medium HCC group was not significantly different (
Clinical Follow-Up
In the large HCC group, the mean follow-up was 2.8 ± 2.1 months, with a median follow-up of 2.5 months. The rate of complete ablation was 25% (2/8), with a residual rate of 75%. One HCC patient, with stage II HCC, had a tumor mass of 5.3 × 4.4 cm on preoperative contrast-enhancement CT (Figure 1A and B). The intraoperative CT treatment planning system guided the 2 electrode needles into the liver tumor (Figure 1C and D). An immediate postoperative CT examination showed the multiple treatment areas of the liver tumor as shadows. A contrast-enhanced CT scan on postoperative day 7 showed that the tumor had bubble, the tumor size was 8.8 × 6.6 × 5.2 cm, the edge of this lesion was heterogeneously enhanced, and the central necrosis area showed no enhancement (Figure 1E). A postoperative contrast-enhanced CT scan at 2 months showed that the size of tumor was 4.9 × 4.3 × 4.3 cm, without evidence of enhancement, representing complete tumor ablation (Figure 1F).
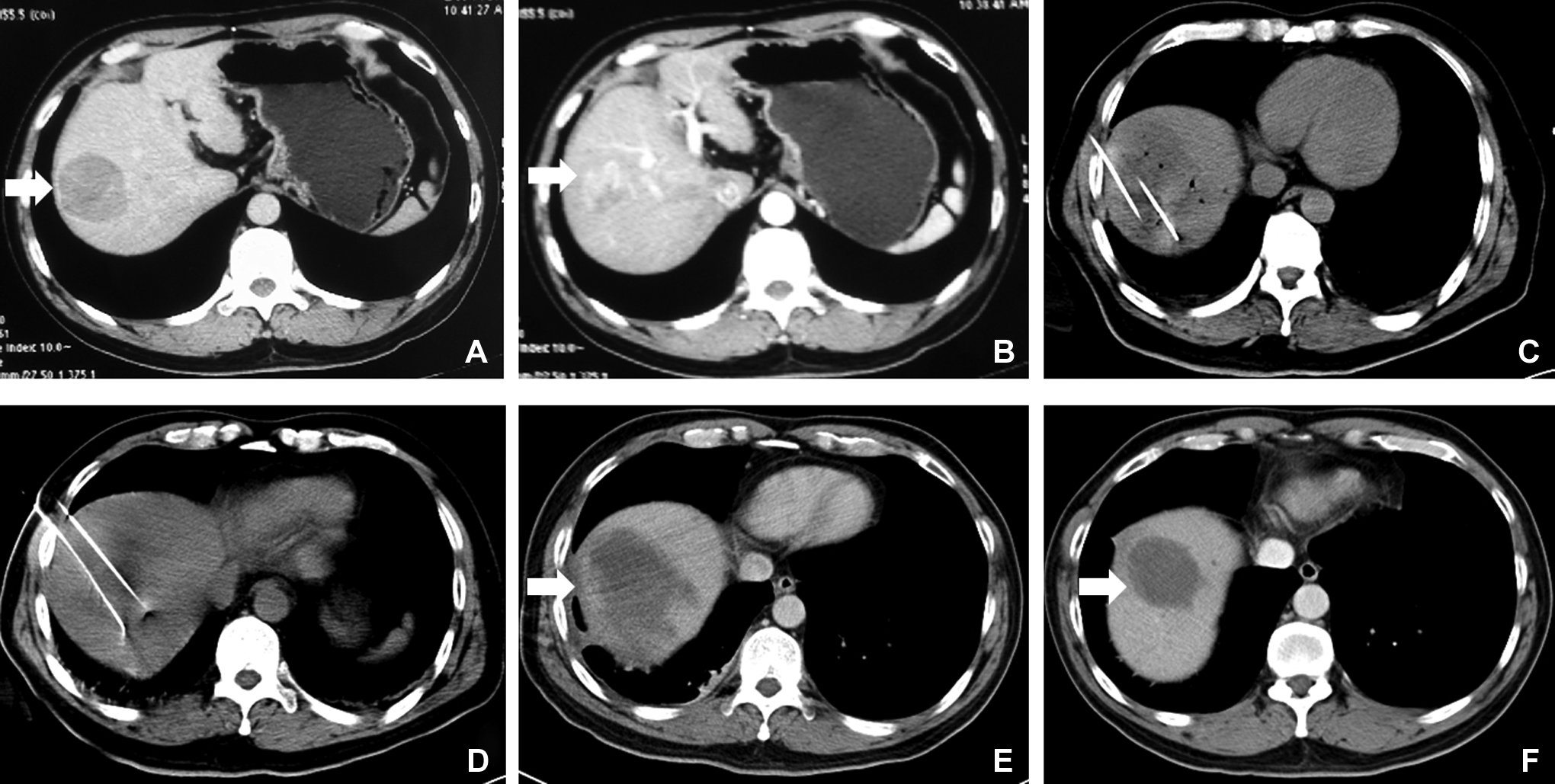
A 47-year-old male patient with HCC. A, B, Perioperative contrast-enhancement CT venous phase and arterial phase showing a tumor of approximately 5.3 × 4.4 cm. C, D, Two IRE probes inserted into the tumor under intraoperative CT guidance. Probe distance = 1.5 cm. E, Post-IRE 7 days contrast-enhancement CT showing that the tumor had a gas bubble, the size of the lesion was approximately 8.8 × 6.6 × 5.2 cm. F, Post-IRE 3 months contrast-enhancement CT showing that the size of the tumor was 4.9 × 4.3 × 4.3 cm with no obvious enhancement. CT indicates computed tomography; HCC, hepatocellular carcinoma; IRE, irreversible electroporation.
In the medium HCC group, the mean follow-up was 4.3 ± 3.2 months, with a median follow-up of 4.5 months. The rate of complete ablation was 66.6% (4/6), which was not significantly different compared with the large HCC group (
Effect of Tumor Size on Outcome of IRE Ablation.

Abbreviation: IRE, irreversible electroporation.
Discussion
Large HCC (>5 cm) is difficult to treat surgically because of the proximity of blood vessels, the biliary tract, and vital organs; thus, ablation is an alternative treatment. Traditional thermal ablation has certain limitations. First, thermal ablation is only useful for lesions smaller than 3 cm; for large HCC, the complete ablation rate is 10% to 25%. 2 Second, thermal ablation needs to reach 60°C in the target tissue to cause cell death; however, large tumors near blood vessels cannot be completely ablated and show a high recurrence rate because it is difficult to reach this temperature in the tumor due to the heat sink effect. 8 Finally, thermal ablation damages the adjacent normal structures of the ablation zone, causing a high incidence of complications. The IRE ablation is nonthermal, with no heat sink effect, and can protect the structure of blood vessels, bile ducts, and other vital organs. Thus, IRE can ablate the tumor near the inferior vena cava, the large hepatic artery, or the portal vein. 9,10
In this study, in the 8 patients with large HCCs, the IRE procedure was well tolerated: no deaths, major complications such as bleeding, or other treatment-related adverse events occurred. Minor complications in the large HCC group were not significantly different from those in the medium HCC group. Livraghi et al 11 reported that the major complication rate of 2320 patients with liver tumors who received multicenter RFA was 2.2%, and the mortality was 0.3%; the main complications were bleeding and intestinal perforation. In this study, the major adverse reaction rate was 0, indicating that IRE ablation of unresectable liver cancer is safe and feasible. Dollinger 12 reported that the major complication rate of 56 patients with 85 liver tumors who received IRE ablation was 7.1% (6/85), which showed that IRE ablation of unresectable liver tumors was safe and was consistent with our findings.
This study was the first to focus on patients with large liver tumors who received IRE ablation. In the large HCC group, the average follow-up time was 2.8 ± 2.1 months, and the complete ablation rate was 25% (2/8). In the medium HCC group, the mean follow-up time was 4.3 ± 3.2 months, and the complete ablation rate was 66.6% (4/6). The complete ablation rate in the large HCC was not significantly different (
When IRE probes were inserted percutaneously into the tumor tissue, it was difficult to determine accurately the ablation region because of the metal artifacts or angle limitation, which affected the imaging monitor, so there was a certain risk of accidental injury to other organs or blood vessels. In this study, percutaneous IRE ablation used 2 probes that could divide the tumor into multiple partitions and perform the ablation many times, which greatly reduced the risk of vascular puncture and improved the safety of IRE ablation. The disadvantage was the increased ablation time: the large liver cancer ablation time was significantly longer than that for patients with medium cancer in this study (
In conclusion, IRE ablation for large HCC is safe and highly effective compared with RFA and should be promoted in the clinic. However, this study focused on the safety of IRE ablation and only studied its short-term efficacy. The study had the following limitations: the small group of patients and the short follow-up time; therefore, the long-term efficacy of IRE should be studied by extending the group of patients and increasing the period of follow-up.
Footnotes
Abbreviations
Authors’ Note
Jianying Zeng and Guifeng Liu contributed equally to this work and share first authorship.
Acknowledgments
The authors thank all patients who took part in this clinical research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by International Scientific Fund of Fuda Cancer Hospital, Guangzhou (Y2015-ZD-001).
