Abstract
Background
The incidence and mortality of hepatocellular carcinoma (HCC) had increased globally over the past decades. Previous studies found that transarterial chemoembolization (TACE) combined with lenvatinib had also shown efficacy in the unresectable HCC. We aimed to evaluate the safety and efficacy of TACE combined with lenvatinib and camrelizumab to treat unresectable multiple nodular and large HCC (>5 cm).
Materials and methods
Between November 2018 and June 2021, we retrospectively recruited 82 patients with unresectable multiple nodular and large HCC (BCLC stage B or C with a single nodular diameter of >5 cm). Of the patients who had not previously been treated, 33 patients received TACE + lenvatinib + camrelizumab (group A, TLC), and 49 patients treated with TACE + lenvatinib (group B, TLB) as the initial treatment. Related efficacy and safety results were recorded and assessed.
Results
The median follow-up periods of groups A and B were 14.5 ± 7.9 months (range, 3-36) and 12.5 ± 8.2 months (range, 3-32), respectively (P = 0.799). The progression-free survival (PFS) of groups A and B was 9.4 months and 5.9 months (P < 0.01), respectively, and overall survival (OS) was significantly longer in group A (16.4 months vs 11.0 months, P < 0.01). In group A, the local response rate (LRR) and disease control rate (DCR) were 51.5% and 81.8%, respectively, which was higher than the corresponding 46.9% and 77.6% observed in group B (P = 0.233; 0.429). Patients with BCLC B stage had better PFS and OS (P < 0.05). The BCLC stage was an independent factor that affected PFS and OS. There were no massive bleeding or treatment-related deaths.
Conclusions
In patients with unresectable multiple nodular and large HCC (single nodular diameter of >5 cm), TACE combined with target therapy and immunotherapy is safe and effective.
Introduction
In recent years, survival in patients with unresectable large HCC has been prolonged due to systemic treatment. 1 Over the past 10 years, sorafenib has emerged as the only approved first-line drug for patients with unresectable HCC. 2 Since 2013, oxaliplatin-based interventional therapy (eg, hepatic artery infusion chemotherapy and HAIC) has been used as standard systemic therapy in China. 3 However, the objective response rate (ORR) was only 2–8.2%, and survival was 6.4–10.7 months in two treatments.
In 2018, the REFLECT study confirmed that patients treated with lenvatinib had longer PFS, time to progress (TTP), and better ORR. These results were similar to those of previous studies on sorafenib, and then lenvatinib was seen as a new treatment for advanced-stage HCC. 4 At the same time, for BCLC stage C, phase I and II clinical studies confirmed that lenvatinib, combined with embolization-based interventional therapy such as transarterial chemoembolization (TACE), could achieve higher ORR and longer survival without increasing side effects and reducing the quality of life.5,6 However, additional studies revealed that more frequent TACE would lead to poorer liver function and lower curative effect.7,8 Especially in massive (>5 cm) HCC, frequent administration of TACE could result in lower ORR, faster disease progression, and worse liver fibrosis. 9
In 2020, carrelizumab was approved for the second-line treatment of unresectable HCC. Phase 1 clinical trials showed that patients with solid tumors could tolerate the drug well, and carrelizumab exhibited antitumor activity.10,11 Additionally, the second phase of the study revealed that patients who had previously been treated with other therapies benefited from carrelizumab. Nevertheless, the ORR was 14.7% and the rate of OS in 6 months was 74.4%. Hence, single carrelizumab would be an alternative therapy in advanced-stage HCC. 12 However, in phase 3 clinical trials, the effective rate of a single-drug PD-1 inhibitor was only 15–20%, which neither improved efficacy nor prolonged the survival of patients. 13 Therefore, for unresectable multiple nodular and massive HCC (single nodular diameter of >5 cm, BCLC B or C stage), it is possible that TACE + lenvatinib + carrelizumab could be used as an initial treatment option upon disease diagnosis. However, there were no similar reports.
Thus, the purpose of the present study was to investigate the safety and efficacy of TLC in the treatment of unresectable multiple nodular and large HCC (single nodular diameter of >5 cm).
Materials and Methods
Patient Selection
Between November 2018 and June 2021, 82 patients with unresectable multiple nodular and large HCC (>5 cm) receiving systemic therapy from our center were retrospectively screened. Based on BCLC staging systems, these patients were identified as unresectable BCLC-B stage or BCLC-C stage. No previous TACE or other systemic therapies were administered for HCC, and there were measurable lesions upon imaging (CT/MRI). Patients were then divided into two groups: 33 patients received TACE + lenvatinib + camrelizumab (group A, TLC), and 49 patients treated with TACE + lenvatinib (group B, TLB) (Figure 1). The patient characteristics are summarized in Table 1. The reporting of this study conforms to STROBE guidelines. 14

A total of 202 patients received TACE or lenvatinib or camrelizumab as a first-line treatment for BCLC B or C-stage HCC. Thirty-five TLC-treated patients and 50 TLB-treated patients met the eligibility criteria. After follow-up treatment, safety and efficacy were compared between the two groups (TLC: 33; TLB: 49).
Patient Characteristics in This Study.
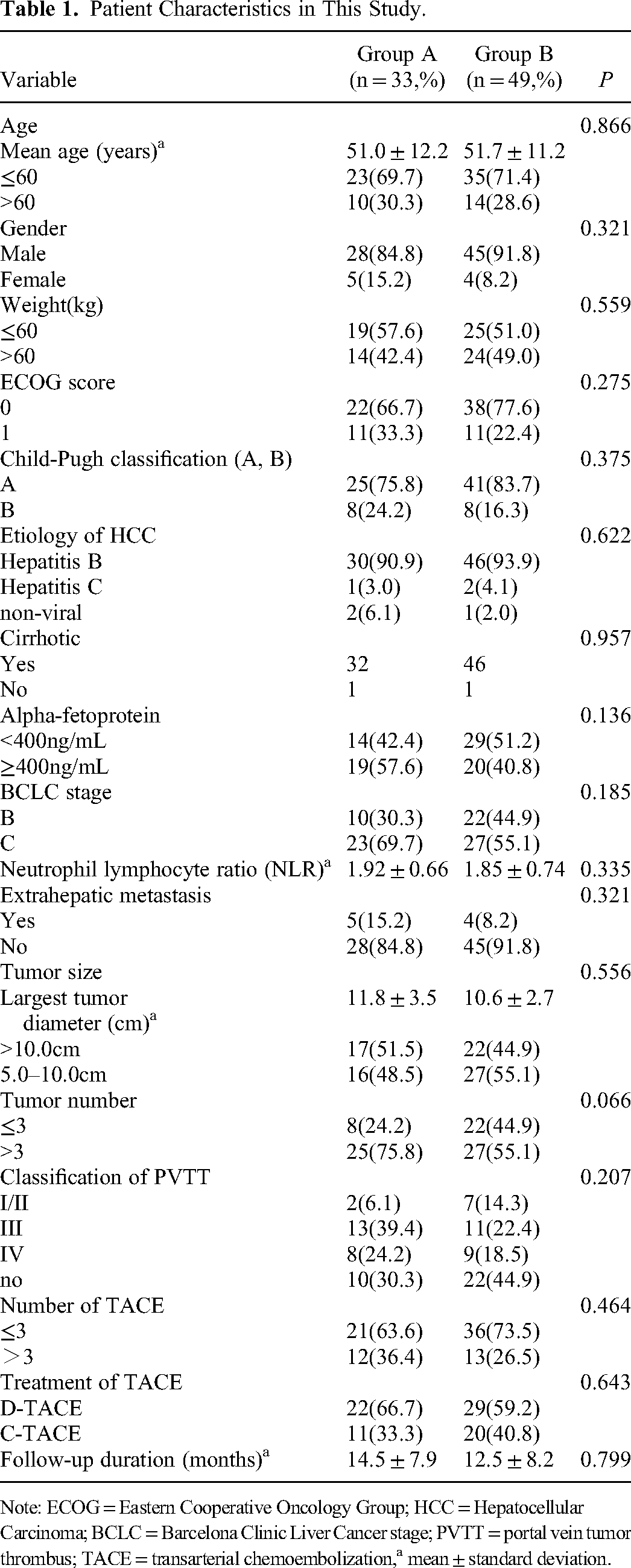
Note: ECOG = Eastern Cooperative Oncology Group; HCC = Hepatocellular Carcinoma; BCLC = Barcelona Clinic Liver Cancer stage; PVTT = portal vein tumor thrombus; TACE = transarterial chemoembolization,a mean ± standard deviation.
The inclusion criteria were as follows: a) confirmed by either imaging or histological evaluation according to the AASLD or EASL guidelines; b) patients with unresectable large HCC (>5 cm); c) BCLC-B or C stage; d) ECOG score of 0 or 1; e) Child–Pugh score of A5–B8; and f) longer than 3 months of life expectancy. The exclusion criteria were as follows: treatment with any systemic or loco-regional therapies; symptomatic ascites; less than 6 weeks of follow-up; gastrointestinal bleeding; comorbidity with other malignancies; and absence of baseline imaging information.
Treatment Procedures
All patients were treated after being reviewed by a multidisciplinary team (MDT) comprising oncologists, surgeons, interventional physicians, radiotherapists, and imaging specialists. Before treatment, all patients had undergone CT or MRI in our hospital.
Conventional TACE (C-TACE) and drug-eluting beads TACE (D-TACE) were confirmed. An 18-gauge Seldinger needle was applied to the femoral artery, and hepatic arteriography or navigation imaging was performed with a 5-F Yashiro catheter and a 1.9-Fr microcatheter (Shirabe®; Piolax, Yokohama, Japan). Tumor-feeding vessels were embolized either selectively or super-selectively. On-demand TACE procedures were scheduled at intervals of 4–8 weeks. Based on the review of CT/MRI, patients’ status, and liver function, the MDT decided if the next TACE was required.
During the C-TACE procedure, an emulsion of lipiodol (5-20 ml) and chemotherapeutic drugs was injected. The application of epirubicin hydrochloride (20-50 mg) was determined based on the tumor volume and the condition of each patient. Then the tumor arterial flow ensued, which decreased with embolization using polyvinyl alcohol foam (PVA) or absorbable gelatin sponge particles (300-500 μm or 500-700 μm; Alicon Medical Co., Hangzhou, China). CalliSpheres drug-eluting beads (Jiangsu Hengrui Medicine Co., Ltd, China) were applied to the D-TACE procedure. First, epirubicin hydrochlorides (60-80 mg) were dissolved to 20 mg/ml and mixed with dehydrated beads for approximately 30 min. Second, mixed dehydrated beads and appropriate contrast agents (ratio 9:1) were infused slowly into the hepatic arteries of the tumor (1 ml/min). The embolization was monitored until vascular stagnation and inconspicuous tumor staining on angiography.
Lenvatinib was applied once daily based on body weight (≥60 kg, 12 mg or <60 kg, 8 mg) for 1 week after the first application of TACE in the two groups. The dosage of lenvatinib was reduced or discontinued in case of grade ≥3 severe adverse events (AEs) or any unacceptable grade 2 drug-related AEs (National Cancer Institute Common Terminology Criteria for Adverse Events 4.0). If TACE was needed again, lenvatinib was suspended for 2–3 days during the operation. After liver function was rechecked and reached A level (Child–Pugh classification), the initial dosage of lenvatinib was continued.
In group A, patients were given 3 mg/kg camrelizumab (Jiangsu Hengrui Medicine Co., Ltd, China) intravenously over 30 min every 3 weeks. First, lenvatinib was first administered, followed by camrelizumab within 3 days. When ≥3 severe AEs occurred, treatment with camrelizumab was discontinued.
Follow-up
All patients underwent at least one follow-up. The first contrast agent–enhanced CT or MRI was performed within 6 weeks, and subsequently every 2–3 months during 12 months of treatment, and then once every 3 months (Figure 2A–D). If TACE was repeated, tumors were evaluated by CT or MRI every 4–6 weeks. The therapeutic efficacy of the combined treatment was evaluated by imaging examinations. All clinical symptoms and post-treatment laboratory tests were also recorded.

A. The patient was a 55-year-old male with advanced HCC, after one cycle TLC treatment was given, Enhanced MRI revealed a complete remission at one month; B–D tumor necrosis, and no recurrence or occurrence of intrahepatic metastasis upon imaging after 3, 6, and 12 months.
Evaluation Criteria
The PFS, OS, LRR, DCR, and AEs were reviewed in this study. PFS (time from initial TACE to initial radiological progression or death) and OS (time from initial TACE to death) were defined. Modified RECIST (mRECIST) criteria were used to evaluate LRR and DCR. LRR was defined as complete response combined with partial response, whereas DCR was complete response combined with partial response and stable disease. The severity of AEs or irAE was assessed by the NCI Common Terminology Criteria for Adverse Events (version 4.0).
Statistical Analysis
SPSS 20.0 was employed to analyze these data (International Business Machines Corporation, New York, USA). The level of significance was P < 0.05. Both PFS and OS were compared between the two groups using a stratified log-rank test, and the survival curves of the two groups were assessed by Kaplan–Meier analysis. A stratified cox proportional hazards regression model was used to assess univariate and multivariate analyses. Differences in patients’ characteristics between the two groups were compared using the Pearson χ2 test.
Results
Patient Characteristics
Patients’ characteristics in the two groups were summarized in Table 1. According to the study criteria, 82 patients who were diagnosed with unresectable multiple nodular and large HCC (>5 cm) and who were not subjected to other therapies were included in the research (groups A, N = 33; group B, N = 49). The median follow-up period of groups A and B was 14.5 ± 7.9 months (range, 3-36) and 12.5 ± 8.2 months (range, 3-32), respectively (P = 0.799). All patients successfully underwent TACE at least once, and no lenvatinib discontinuation patients were found due to AEs. In group A, 12 out of 33 patients (36.4%) received TACE more than three times, and this percentage was 26.5% (13/49) in group B. The largest tumor diameter was 11.8 ± 3.5 cm in group A, and 10.6 ± 2.7 cm in group B (P = 0.556). There were no significant differences in the characteristics of patients.
Treatment Response
Tumor responses are shown in Table 2. LRR and DCR in group A were 51.5% and 81.8%, respectively, compared to 46.9% and 77.6% in group B; however, there was no statistical significance (P = 0.233; 0.429). In the imaging evaluation of six patients with progressed disease in group A, three (50.0%) developed new extrahepatic metastases (two in the lung and one in the lumbar spine), two (33.3%) developed enlarged residual lesions, and one (16.7%) developed portal invasion. However, in group B, the results were different, and 10 out of 11 patients (90.9%) had new extrahepatic metastases, while the remaining one patient (9.1%) developed a new residual lesion.
Clinical Efficacy of Treatment Between Two Groups.

Note: CR = complete response, PR = partial response, SD = stable disease, PD = progressive disease, based on the mRECIST (modified Response Evaluation Criteria in Solid Tumors).
Progression-Free Survival
PFS was significantly longer in group A (9.4 months, 95% CI 7.2-11.6) than in group B (5.9 months, 95% CI 4.4-7.3; P < 0.01; Figure 3A). Moreover, the BCLC stage was an independent factor that affected PFS, and stage-B patients [P < 0.05, HR = 0.682, (95% CI 0.412-1.128), Figure 3B] had longer PFS. There was no association between PFS and age, gender, weight, ECOG performance status, Child–Pugh classification, etiology of HCC, cirrhotic, AFP, tumor size, tumor number, TACE number, and treatment with TACE.
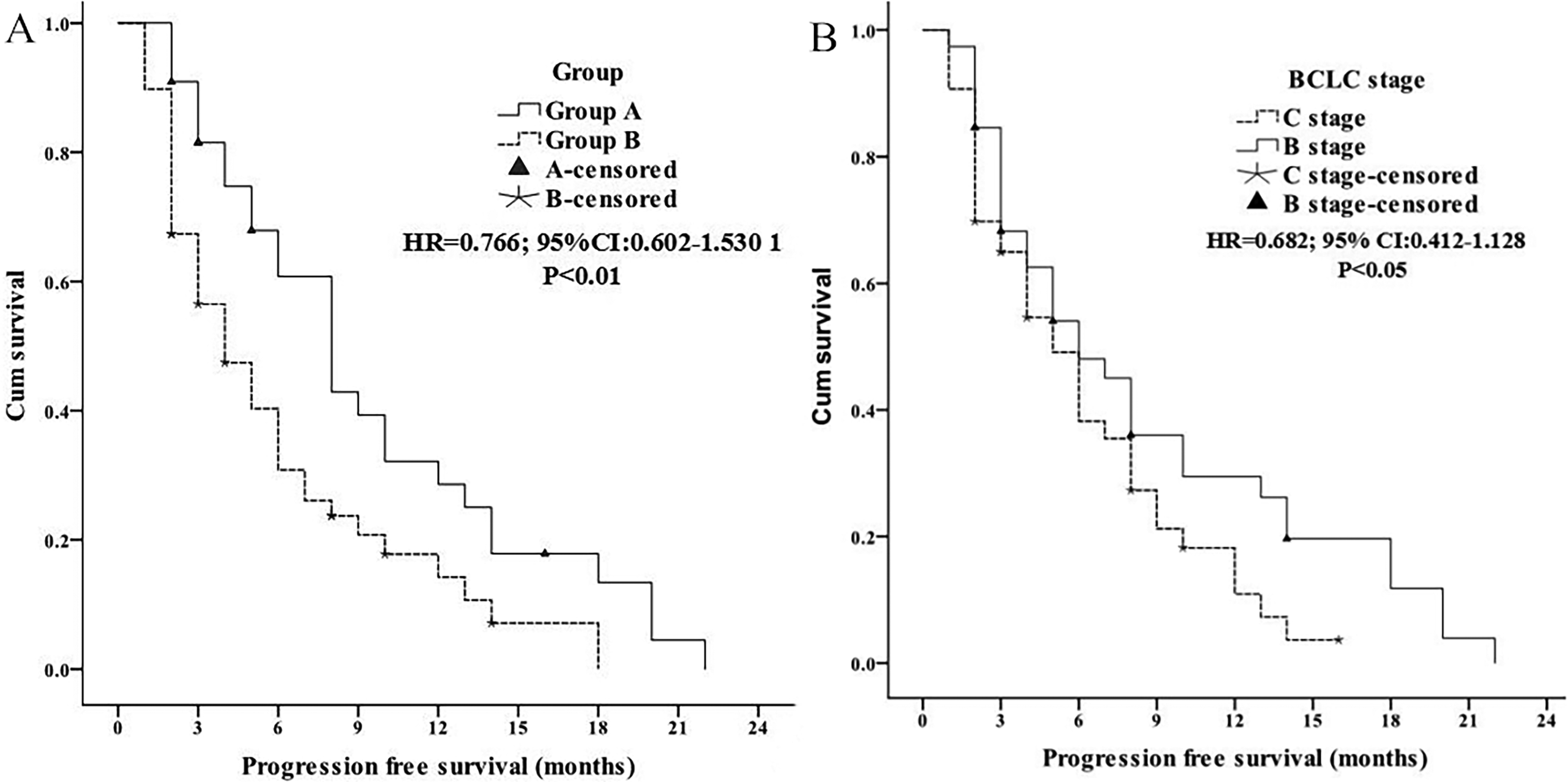
A. Comparison of PFS in two groups; B. Cox proportional hazards regression model explored the BCLC stage related to PFS.
Overall Survival
OS was significantly longer in group A (16.4 months, 95% CI 14.2-18.7) than in group B (11.0 months, 95% CI 9.3-12.7; P < 0.01; Figure 4A). The 6-, 12-, 18-, and 24-month OS was respectively 93.9%, 63.6%, 42.4%, and 6.1% in group A, and 77.6%, 40.8%, 18.3%, and 4.1% in group B. The BCLC stage was an independent factor that affected OS, and patients with the B stage (P < 0.05, Figure 4B) had longer OS. The study variables (age, gender, Child–Pugh classification, etiology of HCC, cirrhotic, AFP, tumor size, tumor number, TACE number, and treatment with TACE) had no association with OS.

A. Overall survival in two groups; B. Cox proportional hazards regression model explored the BCLC stage related to OS.
Safety of Treatment
AEs or irAE of the two groups are shown in Table 3. There was no significant difference in the occurrence rates of AEs (acute liver function impairment, acute renal insufficiency, abdominal pain, pyrexia, vomiting, hypertension, and fatigue). In Group A, no irAE (endocrine toxicity, immune pneumonia, autoimmune myocarditis) has been found. Although group B had no patients with grade 3 or 4 AEs during follow-up, four patients in group A developed grade 3 or 4 AEs (three experienced grade 3 proteinuria and one experienced grade 4 proteinuria) and gradually benefited from reduced medication or empirical treatment (P < 0.05). There were no treatment-related deaths.
Safety of Treatment Between Two Treatment Groups.

Discussion
Tyrosine kinase inhibitors (TKIs) were the first approved systemic treatments for the advanced stage of HCC and had been confirmed to be effective and safe. 15 However, thanks to characterization of the immunological features of the tumor microenvironment, more and more clinical research had found that TKIs combined with immune checkpoint (ICI) would be a promising method for prolonging survival with unresectable HCC. 16 In 2023, Bernardo Stefanini et al reviewed that efficacy and safety profiles of current and emerging ICI/TKI combination treatments, these results were consistent with previous research findings. 17 At the same time, previous studies had also found that TACE combined TKIs could achieve higher ORR and longer survival without increasing side effects. 18 Alessandro Granito et al reported that regorafenib combined with TACE could enhance the therapeutic potency of TACE for the treatment of HCC, and has promising clinical implications in future. 19 Therefore, it would be vital to find suitable populations to use combined therapies.
Based on several previous studies, these results showed that patients with unresectable BCLC-B were treated with combination therapies (TACE + targeted drugs), whereas BCLC-C patients were treated with systemic therapy.20–22 However, to the best of our knowledge, for patients with BCLC-B HCC (more than 7 criteria, single nodule >5 cm) or BCLC-C HCC (single nodule >5 cm), our study was the first to confirm the beneficial effects of TLC on the survival, whereupon we compared TLC results with those of TLB. The results revealed that the combination of the three methods was better than TACE combined with lenvatinib, thereby providing a central direction in the treatment of unresectable HCC (>5 cm), as well as an initial treatment for patients with more than seven criteria related to BCLC B- or BCLC C-stage HCC with regard to LRR, DCR, and survival. The combination of three methods was an initial treatment.
In the present study, the diagnosis of HCC depended on imaging (enhanced CT or MRI), and we retrospectively compared the clinical outcomes of these patients treated with TLC and TLB. This study showed that TLC could be effectively used for unresectable multiple nodular and large HCC (single nodular diameter of >5 cm). Patients in group A achieved higher LRR (51.5% vs 46.9%, P = 0.233) and DCR (81.8% vs 77.6%, P = 0.429) than patients in group B. Moreover, TLC resulted in better efficacy in patients than TLB did, which might be mainly attributed to a longer PFS (9.4 vs 5.9 months, P < 0.01) and OS (16.4 vs 11.0 months, P < 0.01). Similarly, many studies have also confirmed that the combination therapy for these patients should be used in advance in the clinical treatment plan.23,24 Allen et al reported that antiangiogenic therapy (TKI) can improve anti-PD-L1 treatment. Conversely, anti-PD-L1 therapy can prolong the efficacy of TKI and sensitize tumors to TKI. 25 Thus, despite no statistical significance in LRR and DCR, these data confirmed the use of TLC in the initial treatment of unresectable multiple nodular and large HCC (single nodular diameter of >5 cm).
The BCLC stage was an independent factor for OS and PFS in this study. Patients with BCLC B stage survived longer. Many previous studies also confirmed that targeted drugs and immunotherapy should be used early for unresectable HCC.26–28 As the primary tumor of the liver, alpha-fetoprotein (AFP) directly reflected the recurrent and metastatic ability of HCC29,30 However, AFP was not an independent factor in the two groups, which was inconsistent with other reports, and the inclusion criteria can be used to explain the reason for this (>5 cm unresectable B/C stage HCC in this study and not only a single stage).
Treatment-related AEs in HCC patients were significantly associated with longer overall survival as recently demonstrated. Francesco Tovoli et al found that experience in the management of TKI-related AEs prolonged treatment duration and survival. 31 In our study, another finding was that the rate of proteinuria was higher in group A than in group B (39.4% vs 10.2%, P < 0.05), and 27.3% proteinuria was considered mild or moderate, clinically controllable, and self-limiting in group A. Qin et al reported that patients were prescribed anti-PD-1 inhibitor camrelizumab, and the rate of proteinuria was approximately 23%. 12 Although anti-PD-1 inhibitor could lead to proteinuria, this study did not match the results of previous reports. Many studies have confirmed that anti-angiogenic drugs combined with the anti-PD-1 inhibitor could decrease the incidence of AEs.32,33In a previous study, Xu et al reported that the incidence of proteinuria with camrelizumab + apatinib (500 mg, a VEGFR2 inhibitor) was 20%, indicating that a reduced dosage of apatinib (250 mg) could lead to prolonged vessel normalization and decreased proteinuria. 34 However, the value of camrelizumab in combination therapies as a means to reduce the frequency of AEs has not been clearly shown in HCC (eg, proteinuria), and this needs to be further investigated in future research.
This study had some limitations. First, the control group was TACE combined with lenvatinib and not lenvatinib or sorafenib alone. Such a comparison and a prospective randomized controlled trial should be of value. Second, although 82 patients were enrolled in this study, only 33 were treated by TLC; more cases are required for future survival analysis. Third, AFP, tumor size, and TACE number in subgroups should have been analyzed. AFP, as an important factor, needs to be further studied.
To conclude, the current study showed that target therapy and immunotherapy in combination with TACE was effective and safe for unresectable multiple nodular and large HCC (with a single nodular diameter of >5 cm) and could be an alternative to first-line therapy.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank all colleagues involved in the study for their contributions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Third affiliated hospital of Sun Yat-sen University (address: 600 Tianhe Road, Guangzhou, China; approval number: [2020]-02-134-01; date: September 4, 2020). Informed consent was waived by the committee because of the retrospective nature of this study. All individual participants agreed the study and signed informed consent.
Funding
This study was funded by National Natural Science Foundation of China (grant No. 82072035 to Mingsheng Huang, 2020).
