Abstract
Background and aims
A gonadotropin-releasing hormone (GnRH)-based therapeutic vaccine candidate against hormone-sensitive prostate cancer has demonstrated its safety and signs of efficacy in phase I/II trials. In this study, we characterized the isotype/subclass profiles of the anti-GnRH humoral response generated by the vaccination and analyzed its association with patients’ clinical outcomes.
Methods
The immunoglobulin isotypes and IgG subclasses of the antibody responses of 34 patients included in a randomized, open, prospective phase I/II clinical trial were characterized. Every patient included in the study had a diagnosis of locally advanced prostate adenocarcinoma at stages 3 and 4 and received immunization with the vaccine candidate. Additionally, serum testosterone and prostate specific antigen (PSA) concentrations, serving as indicators of tumor response, were determined. The type of anti-GnRH antibody response was correlated to the time elapsed until the first biochemical recurrence in patients and the outcome of the disease.
Results
All patients developed strong and prolonged anti-GnRH antibody responses, resulting in a short- to mid-term decrease in serum testosterone and PSA levels. Following immunizations, anti-GnRH antibodies of the IgM/IgG and IgG1/IgG3 subclasses were observed. Following radiotherapy, the humoral response switched to IgG (IgG1/IgG4). Patients who experienced a short-term biochemical relapse were characterized by significantly higher levels of anti-GnRH IgG titers, particularly IgG1 and IgG4 subclasses. These characteristics, along with a high response of specific IgM antibodies at the end of immunizations and the development of anti-GnRH IgA antibody responses following radiotherapy, were observed in patients whose disease progressed, compared to those with controlled disease.
Conclusion
The nature of the humoral response against anti-GnRH, induced by vaccination may play a key role in activating additional immunological mechanisms. Collectively, these mechanisms could contribute significantly to the regulation of tumor growth.
Keywords
Introduction
Prostate cancer is currently a significant healthcare concern affecting the male population. Epidemiological data for the year 2018 is alarming, as it reveals that there were 1 276 106 diagnosed cases of the disease, leading to 358 989 deaths among men.1,2 These statistics are attributed to factors such as population aging and lifestyle choices.
The average age at which prostate cancer is typically diagnosed is 66 years. 3 The disease follows a prolonged natural course when diagnosed at an early stage. In most cases, it follows an indolent course, often requiring minimal or no treatments. 4 However, in many patients it presents as a localized disease with intermediate or high risk of progression, for which radical surgery or radiotherapy (RT) with or without adjuvant hormonal ablation are the treatments of choice.5,6 Approximately one-third of patients progress to locally advanced, hormone-sensitive disease (castration-sensitive prostate cancer [CSPC]), where neoadjuvant or concurrent androgen deprivation therapy (ADT) with RT has demonstrated effectiveness.6,7
Hormonal ablation therapy (HAT) is the cornerstone of castration-sensitive prostate adenocarcinoma (CSPC) treatment of advanced and metastatic stages. 8 Desensitization of the gonadotropin-releasing hormone receptor (GnRHR) using hormone analogs or direct competence by the binding of antagonists to the GnRHRs, are the common ways to inhibit androgen production in clinical practice. 9 Evidence of the antitumor and antimetastatic effect of the combined use of HAT and RT to treat nonmetastatic advanced prostate cancer (T3/4, N+/−, and M0) 10 is strong.
Most patients with advanced disease present with biochemical recurrence and disease progression developing metastatic castration-sensitive prostate disease (mCSPC). In the last 5 years, the life expectancy of these patients has increased, with the introduction of second-generation antiandrogens, such as abiraterone and enzalutamide, combined with ADT, and with or without docetaxel in the treatment of mCSPC. 11
The main issue regarding therapies against CSPC is the development of tumor resistance to castration in a relatively short time, which limits treatment choices, life quality, and patient survival drastically. 12 These patients receive therapies aimed to inhibit androgen receptor signaling, diminish tumor burden, and provide palliative care. Consequently, the primary goal of any new therapeutic option against CSPC should be to prevent or delay the onset of these complications.
Among all mammal species, gonadotropin-releasing hormone (GnRH) is a highly conserved decapeptide 13 and therefore, it is not immunogenic. Accordingly, to develop GnRH-based vaccines, researchers have had to overcome this problem by introducing immunogenic epitopes in the hormone structure, including potent adjuvants in their formulations approved for veterinary use, such as Adjuvac. 14 Our vaccine candidate for the treatment of locally advanced and metastatic hormone-sensitive prostate cancer was designed following that strategy. It is made of a synthetic GnRH analog peptide containing a Th cell immunogenic epitope of tetanic toxoid, to which very small size proteoliposomes (VSSP) have been added as immunomodulators, both forming a water in oil emulsion, Montanide ISA 51 VG (Seppic, France). 15 Phase I and II clinical trials have demonstrated the vaccine's safety, immunogenicity, and efficacy in controlling tumor growth among treated patients. 16
The induction of specific neutralizing antibodies that sequester circulating GnRH is the main mechanism of action of the vaccine candidate. The immunomodulating effect of immunoglobulins has been well-documented. 17 The effector functions of different antibody isotypes and subclasses induce the activation of other cellular populations of the immune system, stimulate the production of immunological mediators, and trigger the action of effectors involved in antitumor response, such as antibody-dependent cellular cytotoxicity (ADCC), phagocytosis, and complement-dependent cytotoxicity (CDC). 17
Phase II clinical trial with the anti-GnRH vaccine candidate comprised 4-dose arms of the GnRH analog (GnRHm1-TT) and VSSP. The patients immunized produced high anti-GnRH antibody responses and normalized their serum prostate specific antigen (PSA) values, which indicated the presence of a clinical response to the treatment. To further characterize the humoral response induced by immunizations with the candidate vaccine, isotyping of anti-GnRH serum antibody classes and subclasses was performed during the Phase II clinical trial. This paper explores the dynamic changes observed in the GnRH antibody isotypes and subclasses developed by the experimental group during the course of the treatment regimen. Besides, this study establishes a possible relation between the evolution of the anti-GnRH antibody response and the short- and long-term disease control in immunized patients. This serves as a way to further study the immunological mechanisms underlying the vaccine's biological action.
Materials and Methods
Ethical and Methodological Aspects
This study was conducted as part of the clinical trial protocol IG/GNRH1/NT/1002. The clinical trial was conducted in accordance with the principles outlined in the Declaration of Helsinki concerning clinical research involving human subjects. It was approved by the Research Ethics and Review Committee at Marie Curie Provincial Oncological Hospital, in Camagüey (clinical site participating in the study), Cuba, approval number Reg/11/002/GR/MEC/01, dated April 5, 2011. This committee comprised qualified medical specialists and community members who were not affiliated with the study in any way. Finally, the National Regulatory Authority of Cuba (CECMED) granted approval to conduct the study (approval number 796/06.017.11. B, dated June 10, 2011), following a thorough review of all scientific, methodological, and ethical considerations. Also, this study was conducted and reported in accordance with the guidelines outlined in The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 18 All patients signed a written informed consent document before the study began. The vaccine candidate was named Heberprovac for the purpose of facilitating its identification during the development of clinical trials or for publication. Studied serum samples were identified by a code containing numbers and letters.
Patients
A randomized, open, prospective phase I/II clinical trial was performed between September 2011 and April 2013, at “Marie Curie” Oncological Hospital, in Camagüey, Cuba. At admission, patients were diagnosed and staged considering clinical (PSA and digital rectal exam), histopathological (biopsy), and imaging tests (trans-rectal ultrasonography, computed tomography, and bone scan studies criteria). 19 A total of 47 patients, stage III/IV suffering from hormone-sensitive advanced prostate adenocarcinoma, without hormonal treatment, were included. However, the isotype assay of the anti-GnRH serum antibody response and its subsequent correlation with clinical outcome was only performed in 34 patients (Figure 1). Due to different causes, the humoral responses of 13 patients could not be characterized. In 4 of them, the trial was interrupted due to disease progression or poor response to vaccination, 1 was interrupted after adverse events, and 2 abandoned treatments by their own free will. In addition, 3 patients died before completing the treatment for causes not directly related to prostatic adenocarcinoma. In the other 3 cases, the serum samples were insufficient for the antibody isotyping assays (Figure 1).

Flowchart depicting the characterization study of the humoral response to anti-gonadotropin-releasing hormone (anti-GnRH) in patients immunized with Heberprovac.
Immunization With Heberprovac
The composition and preparation of the vaccine was described. 16 The patients were distributed at random, in 4 groups of doses: Group 1 (2.4 mg GnRH/245 µg VSSP) (n = 12); Group 2 (2.4 mg GnRH/125 µg VSSP) (n = 12); Group 3 (1.2 mg GnRH/245 µg VSSP) (n = 12); and Group 4 (1.2 mg GnRH/245 µg VSSP) (n = 11) adjuvated with Montanide ISA 51VG (Seppic, France). All patients received 7 doses of the vaccine candidate by deep intramuscular injections, the first 4 immunizations fortnightly, and the others with monthly frequency. A month after the last immunization, the patients received 30 sessions of external RT with a Co-60 accelerator, in fractionated doses, to a total of 60Gy (Figure 2). Vein blood samples were collected before the first immunization (Time 0), at the end of the 7th immunization (135 days), upon finalizing RT (270 days), and 435 days after the beginning of immunizations (Figure 2). The sera were stored at −80 °C, until use.

Flowchart depicting the treatment regimen during the trial. Circles represent immunizations, the diamond represents the 30 radiotherapy (RT) sessions that concluded on the 270th day after the beginning of the immunizations. The stars represent the times of serum sample collections to characterize the humoral response developed by patients.
Indirect Enzyme-Linked Immunosorbent Assays to Characterize Anti-GnRH Humoral Response
Seroconversion and anti-GnRH antibody titers developed by immunized patients were assessed through enzyme-linked immunosorbent assays according to Aguilar et al. 15 In both cases, plates were coated with synthetic native GnRH at a concentration of 10 μg/mL. Triplicates of each sample were analyzed. In the seroconversion assay, the sera were applied in a 1/50 dilution, in sampling buffer (phosphate-buffered saline 1×/1% bovine serum albumin/0.01% Tween- 20). Serial serum dilutions (1/50) were made to determine titers of the anti-GnRH humoral immune response in patients. The maximum sample dilution in which the average absorbance value was higher than the cutoff value (0.2) of the assay was considered anti-GnRH titer. The class and isotypes of the anti-GnRH responses were determined with the same procedure. However, to achieve this, we used horseradish peroxidase-conjugated mouse anti-human immunoglobulin antibodies (Southern Biotech, USA) specific to various antibody isotypes: anti-IgA (clone B3500B4); anti-IgE (clone HP6029); anti-IgG1 (Clone 4E3); anti-IgM (clone SADA4); anti-IgG4 (clone 6025); anti-IgG2 (clone 6002); anti-IgG3 (HP6050), according to the dilutions recommended by the manufacturer. In all the cases, the established criterion for positivity required sample absorbance values to exceed the assay's predefined cutoff value.
Endocrine and Biochemical Evaluation of Patients
The concentrations of serum testosterone and PSA were evaluated in the same time intervals as the GnRH-specific antibody response. The concentration of testosterone was determined by radioimmunoassay using TESTO CT2 (CIS Bio International, France) commercial kit, whereas PSA was done by using a ultramicroanalytic enzyme-linked immunosorbent assay-PSA kit (Immunoassay Center, Havana, Cuba). Both assays were performed according to the manufacturer’s instructions.
Influence of Class and Isotype of the Anti-GnRH Humoral Response Developed by Patients on the Short- and Mid-Term Control of the Disease
After the conclusion of a clinical trial, an 8-year follow-up study was conducted, during which the evolution of prostate cancer in immunized patients was evaluated on a quarterly basis. The follow-up included a range of clinical, biochemical, and imaging examinations. For this study, we specifically assessed the possible correlations between the class and isotype of anti-GnRH serum response developed by patients and their short-term serum testosterone concentration. Patients were considered to have achieved castration when their serum hormone concentration reached 1.7 nmol/L. A similar analysis was performed to explore the relationship between anti-GnRH humoral response and the duration of days required for patients to normalize their serum PSA values (≥4 ng/mL). 20
Besides, we evaluated the potential correlation between the type of anti-GnRH response and the timing of the initial biochemical relapse (increase in PSA serum concentration = nadir value + 2 ng/mL). 21 This analysis was conducted separately for each experimental group. Moreover, we examined whether the type of anti-GnRH antibody response elicited by the immunizations had any potential impact on the medium-term progression of the disease. This progression encompassed new diagnoses of bone, lymphatic, or distal-organ metastasis, the development of castration-resistant tumors, or cases where the disease directly led to patient mortality.
Statistical Analysis
The data were organized in tables and graphs. Initially, they were analyzed using descriptive statistics, and tests for normal distribution were made. Continuous data were presented as mean and median with a range or percentage in relation to the entire dataset. Given the limited sample size, we used the Mann–Whitney U test to compare variables evaluated within groups (p < .05), as well as the Wilcoxon rank-sum test (α = 0.05) to compare the behavior of intergroup variables. The correlations were established according to the Spearman test and the correlation matrix design. The confidence interval was 95% in all the statistical tests, and the P ≤ .05 values were considered statistically significant. The analyses were performed using GraphPad Prism Windows 6.07 (GraphPad Software Inc., USA).
Results
This study examines the temporal evolution of serum anti-GnRH antibody isotypes in 34 patients. Table 1 provides demographic information and disease characteristics of the patients, stratified by experimental group, prior to immunizations (Time 0).
Characteristics of patients included in the study prior to immunizations.

Abbreviations: CSPC, castration-sensitive prostate cancer; mCSPC, metastatic CSPC; PSA, prostate specific antigen.
Patients, disease stages IIIB and IIIC, GG1, T3a, PSA > 20 ng/mL, with locally advanced disease, were predominant.
Evolution of Anti-GnRH IgG Seroconversion and Titers Among Immunized Patients
Immunization with Heberprovac produced high anti-GnRH IgG titers in all the dose groups (Figure 3), which were more inclined to reach peaks after RT. At that moment, the average anti-GnRH titers (1 out of 525) observed by patients in Group 1 were significantly lower than the titers shown by patients in Group 3 (1 out of 1040) (p = .0263), representing the highest average among the 4 experimental groups. Patients who received the lowest doses of the peptide (Groups 3 and 4) tended to develop higher anti-GnRH titers. Furthermore, only in Group 3 (low peptide concentration/high VSSP concentration), the anti-GnRH titers observed after RT were significantly higher than the titers reached at the end of immunizations (p = .0075) (Figure 3).

Serum anti-GnRH titers developed by patients immunized with Heberprovac, by experimental group. Each dot corresponds to the average anti-GnRH antibody titers shown by patients in each group. The asterisk represents the difference between Group 3 and Group 1 titers at the end of RT.
Five patients (3 in Group 1, and 1 in Groups 2 and 3, respectively) showed little response to Heberprovac immunizations, corroborated by the low anti-GnRH titers developed.
Profile of Anti-GnRH Immunoglobulin Classes Developed by Immunized Patients in the Different Dose Groups
After the 7th immunization with Heberprovac (130 days), the anti-GnRH humoral response in all the groups studied was mainly of the IgM/IgG isotype. The highest IgM response occurred among individuals in Group 4, with the lowest peptide and VSSP dose. In Groups 3 and 4, the anti-GnRH IgM response started to decline significantly after RT. This phenomenon was not observed among patients who received the highest dose of the peptide (Groups 1 and 2). In these groups, the anti-GnRH IgM levels dropped 435 days following the beginning of immunizations, when the antibodies of this isotype became undetectable in all the dose groups (Figure 4).

Isotype profile of anti-gonadotropin-releasing hormone (anti-GnRH) humoral response induced by immunization with vaccine candidate Heberprovac. All the asterisks represent significant differences between the ranges of intra-group responses, comparing times 0–130 days; 130–270 days; and 270–435 days, according to the Mann–Whitney test (p < .05). The letters a and b, denote the statistical differences between the ranges of intergroups responses considering median values of absorbance of samples taken at same time, determined by Wilcoxon rank-sum test (α = 0.05).
Immunizations with Heberprovac produced an elevated and prolonged IgG response to GnRH. Notably, this response remained high in Group 4, without significant fluctuations from the conclusion of the immunization and for 435 days. In the other groups, the anti-GnRH IgG antibody levels decreased at the conclusion of RT, particularly in Groups 1 and 3, where the highest VSSP doses were administered (Figure 4).
After RT, only 13 patients (comprising 38.2% of the total; Group 1: 3; Group 2: 4; Group 3: 5; Group 4: 1) showed modest anti-GnRH IgA responses (Figure 4). None of the dose groups produced significant anti-GnRH responses of the IgE isotype. There were 3 exceptions: 1 patient from Group 1, and 2 from Group 4, who showed a very mild anti-GnRH IgG response on day 130 (Figure 4).
Characterization of Anti-GnRH IgG Response of Patients in the Different Dose Groups
The temporal progression of IgG subclasses within the anti-GnRH antibody response in serum was similar in the 4 dose groups (Figure 5). At the end of immunizations, an anti-GnRH IgG/IgG1 response profile was predominant. Subsequently, the anti-GnRH IgG3 response was more pronounced in Group 2 (high peptide/low VSSP). Following RT, the anti-GnRH IgG3 response declined significantly in all the groups, until it became undetectable 435 days after the commencement of immunizations.

Subclass switch dynamics of anti-gonadotropin-releasing hormone (anti-GnRH) IgG immune response in the serum from patients immunized with different Heberprovac doses. Every dot represents the mean absorbance value (492 nm) of anti-GnRH serum antibodies of patients included in every study group. The asterisks represent significant differences between intragroup response ranges, comparing times from 0 to 130 days; 130–270 days, and 270–435 days, according to the Mann–Whitney test (p < .05). Different letters (a, b, c, or d) represent intergroup differences in relation to the anti-GnRH antibody averages from a particular subclass at the same time of assay, according to the Wilcoxon rank-sum test (α = 0.05).
The anti-GnRH IgG1 response was strong and lasted in all the dose groups (Figure 5). Upon reaching the highest peak at 130 days, it decreased gradually with time and persisted on day 435. At that time, the average anti-GnRH IgG1 response shown by patients in Group 2 (high peptide/low VSSP) was higher than in the rest of the groups.
Ten patients developed anti-GnRH IgG2 subclass responses. At the end of immunizations, it was detected in 5 patients: 2 patients in Group 4, and 1 patient in all the other groups. Upon concluding RT, this type of response was observed in 6 patients: 3 patients in Group 4, 2 patients in Group 2, and 1 patient in Group 1. On day 435, it was persistent in 2 patients in Groups 1 and 4, respectively.
At the end of RT sessions, the anti-GnRH IgG humoral response profile changed, with the emergence of IgG4 subclass antibodies that prevailed on day 435 day after the beginning of immunizations (Figure 5). On the 270th day, in Groups 2 and 4 receiving the immunogen with a lower VSSP dose, the anti-GnRH IgG4 response was higher than the one reached by the other 2 groups. However, on the 435th day, Groups 1 and 2 (highest doses of the peptide) tended to develop higher anti-GnRH IgG4 responses, which were significantly higher in Group 1.
Relation Between the Anti-GnRH Response and the Short-Term Biochemical Outcome of Patients
The neutralizing antibodies generated through immunizations with Heberprovac effectively suppressed the activity of GnRH hormone, leading to a significant reduction in serum testosterone concentrations in all patients by day 135. This effect was minimal at the end of RT, particularly in Group 1 (highest peptide/highest VSSP) (Figure 6A). In the intergroup analysis, no differences were observed in the testosterone-inhibiting activity of antibodies against GnRH at various assay times (Figure 6A).

Effect of immunization with Heberprovac on (A) serum testosterone concentrations at different assay times and (B) the number of patients and times at which the testosterone concentration reached castration levels. The asterisks represent intragroup difference significance of testosterone values at a point in time, with respect to the previous point.
The correlation analysis demonstrated that the inhibition of testosterone production, and the neutralizing activity of anti-GnRH antibodies had inverse, mean, and significant correlations as anti-GnRH serum titers rose, as well as in the specific IgG1 response magnitude developed by patients (anti-GnRH titer, Spearman = −0.770; p = .0007, and anti-GnRH IgG1 titer, Spearman r = −.759; p = .001).
The serum concentration of testosterone began to increase 435 days after the beginning of immunizations, coinciding with a decrease of anti-GnRH titers in all the dose groups (Figure 6B). In 12 patients (50% of patients in Group 1; 37.5% of patients in Groups 2 and 4; and 20% of patients in Group 3), the decline in the concentration of serum testosterone reached castration levels >1.7 nmol/L. The gradual testosterone drop is a particular effect of hormonal ablation induced by Heberprovac. Hence, on average, castration occurred 452 days after the first immunization (Figure 6B).
The decline in serum PSA concentrations occurred similarly, and gradually, in all the experimental groups, reaching physiological values (<4 ng/mL) after RT, which remained practically unchanged on the 435th day (Figure 7). However, in Group 1, patients reached the PSA nadir practically at the same time (absolute values) faster than the individuals immunized with other dose combinations (Figure 7)

Temporal changes in serum prostate specific antigen (PSA) levels among immunized patients in each dose group. On the right, the chart depicts the timeframe within which patients in each experimental group reached their PSA nadir.
Spearman correlation demonstrated the existence of a strong inverse and significant correlation between PSA concentrations and the anti-GnRH IgG4 response in time, in all the dose groups (Spearman correlation coefficient r = −.859, p = .0005).
The first biochemical regression (BR) in immunized patients took place in the mean time of 706.3 days (1.9 years), after completing the immunization scheme, with no significant differences between groups. However, the mean time to the first BR tended to be longer in patients who received the lowest peptide dose: Group 1 = 904.7 days (2.5 years); Group 3 = 1129 days (3.1 years), and Group 4 = 1092 days (3 years) (data not shown).
Nevertheless, before day 706 (438.9 days on average), some patients in all the experimental groups underwent early BR: 3 patients in Group 1, and 2 patients in Groups 2, 3, and 4, respectively (Figure 8). On the contrary, 3 patients underwent the first BR after day 1 850 (1990.5 days on average), and the other 4 patients (1 patient in Groups 1 and 2, and 2 patients in Group 3) have shown no BR for 8 years, without any additional intervention, apart from the vaccine. Considering these differences, the humoral response profile in both groups of patients was analyzed (Figure 8). Surprisingly, the anti-GnRH IgG responses at the end of immunizations (p = .0115), and the IgG isotype response following RT (p = .0311) were significantly higher in patients with an early BR, compared to others with a late BR or no relapse (Figure 8).
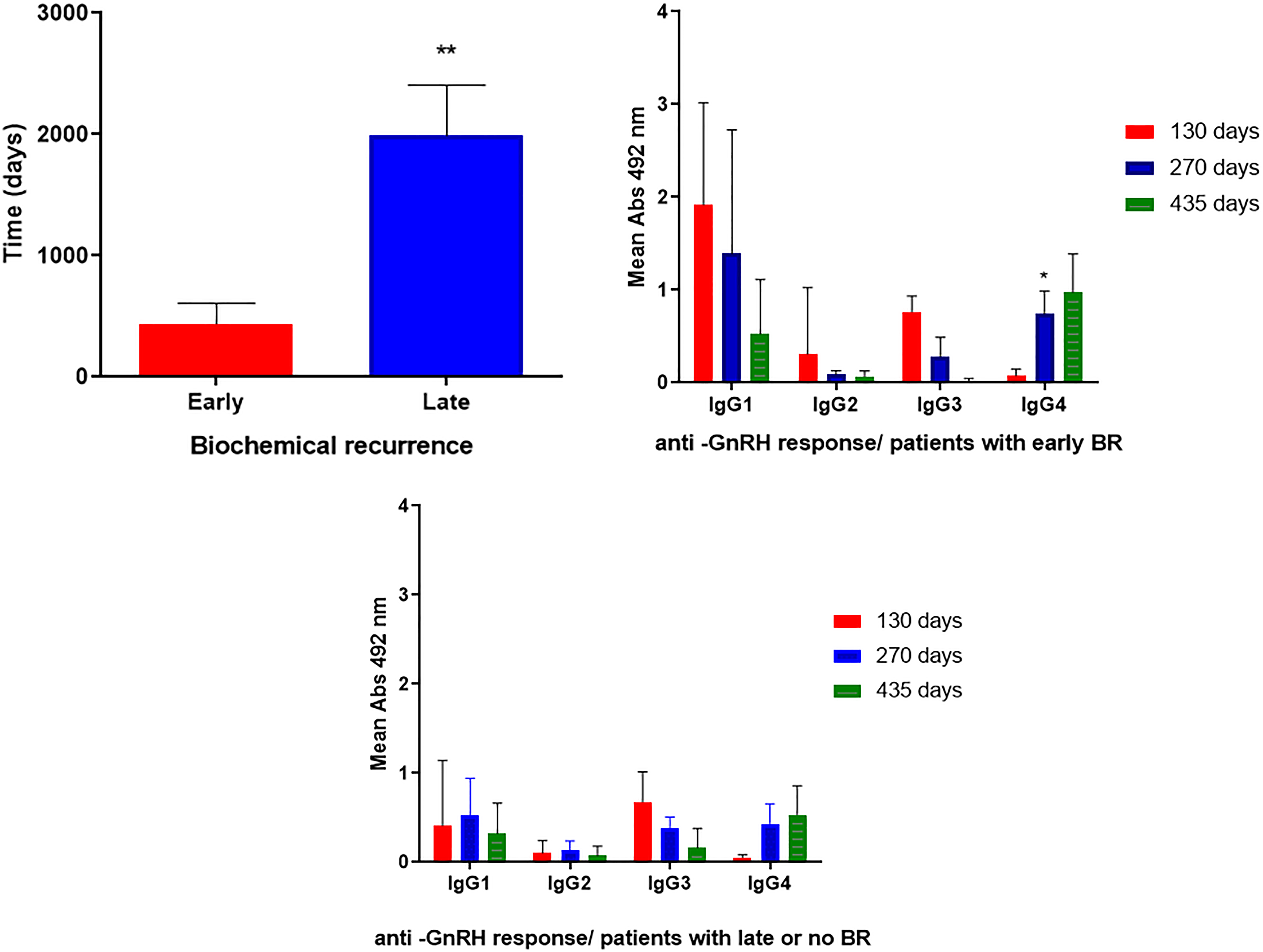
Differences in time of occurrence of BR (above), and the anti-GnRH IgG response (below) in patients immunized with Heberprovac that developed early (n = 9), and late (n = 7) BR. In the graphic above, the asterisks represent a statistical difference between time in days of BR occurrence (p = .0028), and in the graphics below, the asterisks represent the difference in the intensity range of anti-GnRH antibody responses of certain IgG subclass in patients with early and late BR.
Effect of Humoral Response to Anti-GnRH IgG Isotype and Subclasses on the Clinical Outcome of Patients
On average, at approximately 68.4 months (5.7 years) upon concluding the clinical trial, a disease progression was generally observed in 11 out of the 34 patients whose anti-GnRH immunoglobulin expression profiles were characterized (Figure 9, upper table). This accounted for 25.0% of patients in Groups 1 and 4; 30.0% in Group 3; and 50.0% in Group 2. The remaining patients have maintained control over their castration-sensitive disease, receiving conventional therapies as needed.

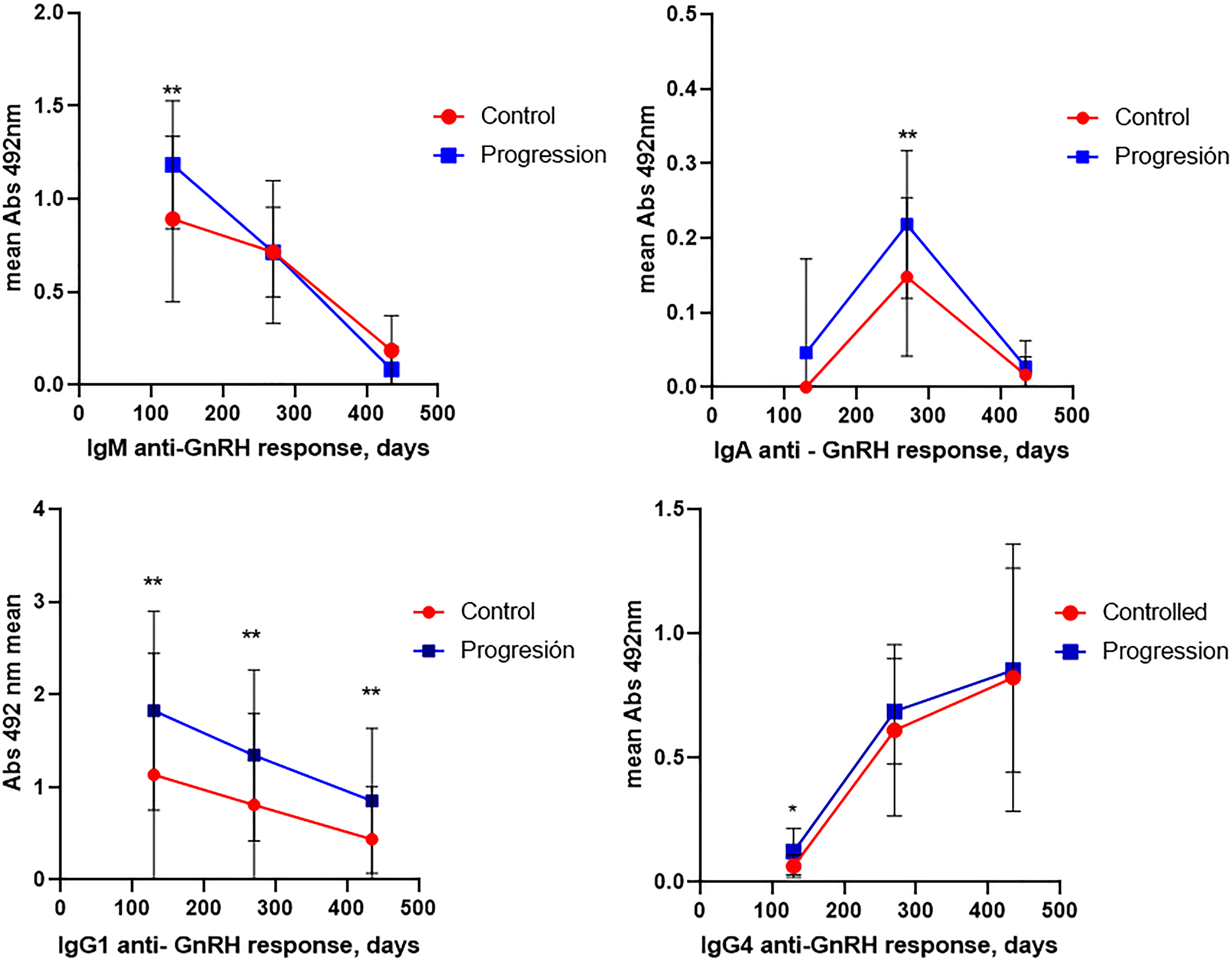
Outcome of patients in month 72 (6 years) after the clinical trial (upper table). The graphics below show the differences between the isotype and immunoglobulin subclasses in the anti-gonadotropin-releasing hormone (anti-GnRH) response developed by patients immunized with Heberprovac with a controlled disease, compared to patients with a progression of the disease.
In this study, an analysis was performed to determine the potential disparities in the anti-GnRH humoral response between patients who experienced disease control and patients who exhibited disease progression (Figure 9). Our findings indicate that several factors may be linked to disease progression in immunized patients. This includes the generation of a very high anti-GnRH IgM response following immunizations; the development of specific responses of the IgA isotype after RT; the appearance of early anti-GnRH responses of the IgG4 subclass; and the sustained presence of a high humoral IgG response against GnRH.
Discussion
The effectiveness of vaccines against cancer relies on the capacity to generate a strong and persistent immune response directed to the tumor cells, which can modify the tumor immunosuppressive microenvironment. Therefore, in order to control tumor growth, immunizations should induce the generation of specific B and T lymphocytes characterized by high effector and memory functions, leading to their recruitment to the tumor site.
Research has shown that immunizations based on GnRH peptides effectively inhibit the hormone cascade induced by GnRH in vivo. This inhibition is achieved through the action of specific neutralizing antibodies, resulting in the atrophy of the reproductive system and inducing castration in mammals.22–24 However, none of the GnRH-based vaccine candidates developed for prostate tumor therapy have managed to induce a specific homogeneous and lasting response in vaccinated individuals. This remains the case even when these vaccines have included immunostimulating molecules such as diphtheria toxin and oily adjuvants in their formulations. 25
In contrast to previous studies, most patients immunized with Heberprovac developed strong and prolonged anti-GnRH antibody responses in all dosage groups. In a few cases, low anti-GnRH titers were observed after vaccination. One such case was an 80-year-old patient with mCSPC, while the reasons behind the poor anti-GnRH response in the remaining cases remain undetermined. It is plausible that this reduced response could be due to the immunological senescence or secondary immunodeficiencies associated with advanced disease.
This underscores the participation of long-life circulating plasmatic cells that secrete anti-GnRH neutralizing antibodies, 26 along with memory B cells, in the construction of humoral response. Furthermore, memory B cells expressing membrane immunoglobulins possess the capability of capturing and presenting GnRH to CD4+ T cells through the major histocompatibility (MHC) class II complex. 27 Once activated, memory B cells proliferate and differentiate, generating new plasmoblasts and new memory B cells with a broader range of specificities than plasmatic cells. 28 Together, these mechanisms related to the B cell response might prolong the inhibition of GnRH action over time.
Immunizations with Heberprovac produced high anti-GnRH IgM and IgG responses. As expected, the IgM response declined as the immune response matured and became more specific over time. On the contrary, on day 435, the IgG response remained notably high, serving as an indicator of the efficacy of GnRHm1-TT peptide in activating CD4+ Th cells. In both instances, the lowest peptide dosage used for immunization produced higher anti-GnRH titers, especially after RT. Thus, it may be possible that high doses of the peptide favor an increase in the production of antibody-secreting plasma cells at the expense of memory B cell induction, as in the case of conventional antigens.29,30
RT is known to induce cell death and the release of proinflammatory mediators, such as reactive oxygen species, cytokines, and growth factors such as the transforming growth factor β1, and the tumor necrosis factor α (TNF-α), IL-6 and IL-10. 31 They include a change of immunoglobulin class into IgA, in a CD4+ T and CD40L cell-depending process. 32 The reason why this type of response was not observed in all patients is that the clinical trial requires further studies. However, 46.2% of patients with anti-GnRH IgA antibodies showed a progression of the disease, most of them diagnosed with locally advanced disease and 1 patient with mCSPC. Probably, in these cases, the advanced disease causes very high expression of inflammatory mediators, the production of which was exacerbated by RT. This, together with the high levels of Breg and Treg cells in the tumor microenvironment favored the immunosuppressive conditions necessary for tumor growth.
The prevalence of an anti-GnRH IgG3/IgG1 response after immunizations provides compelling evidence that the GnRHm1-TT peptide induces a Th1 response. This profile is more prominent when the lowest VSSP dose is administered to patients. Both subclasses have the capacity to opsonize, activate the complement system, and induce ADCC, natural killer-mediated ADCC, and complement-dependent cellular cytotoxicity. 17 However, since it is a soluble antigen, the anti-tumor effect of anti-GnRH antibodies essentially stems from the elimination of circulating GnRH. Additionally, the heightened potential of immunoglobulins IgG1 and IgG3 to activate leukocytes and stimulate the release of elevated levels of inflammatory cytokines through FcγRIIa is noteworthy.
Moreover, anti-GnRH IgG3 antibodies have a high affinity for activating FcγRs, thereby triggering important effector functions, including activation and maturation of dendritic cells (DCs) that enable efficient introduction of the antigen to CD4+ T cells through MHC class II complex, also triggering Th2 response. IgG3 antibodies also have effector functions through the Fcγ receptors in the antigen-presenting cells, where the antigen contained in the immune complex is endocytosed and cross-presented to CD8+ T lymphocytes through the MHC class I molecules, thus inducing a potent long-lasting anti-humoral response based on the generation of memory T cells. 33 These mechanisms might contribute to tumor growth control, amplifying the effects of hormonal ablation.
It was significant that the neutralizing effect of circulating GnRH and the drop in serum testosterone levels were inversely correlated to the anti-GnRH IgG1 response instead of the IgG3 subclass. It could be linked to the maturation of the immune response and higher avidity and affinity of anti-GnRH IgG1 antibodies toward the GnRH antigen.
The decline in serum testosterone levels led to a normalization of serum PSA concentrations in 95.3% of immunized patients, regardless of the dose group. An important finding of this study was that the normalization of PSA values also occurred in patients whose serum testosterone was above the castration levels. This effect remained on day 435, when the levels of serum testosterone were rising. It evidenced the existence of other immunological mechanisms of the neutralizing action of antibodies that control tumor growth.
An important limitation of this study is the absence of investigations to characterize cellular immunity resulting from immunizations with Heberprovac. However, we postulate that this vaccine candidate is likely to induce a memory-specific immune cellular response against GnRH. It is also possible that RT has exposed tumor-associated neoantigens to the immunological system. Both processes have the potential to trigger mechanisms of cellular cytotoxicity and immune spreading, leading to the infiltration of cytotoxic T lymphocytes, neutrophils, and other immunocompetent cell populations into the tumor microenvironment. This, in turn, could counteract the suppressive effects of regulatory T and B cell populations that typically reside within the tumor microenvironment. Altogether, these mechanisms may contribute to the long-term control of tumor growth, even in the presence of serum testosterone and in the absence of circulating anti-GnRH antibodies. This hypothesis requires validation through studies focused on cellular immunity.
From our perspective, there is no causal relationship between the decline in serum PSA levels and the rise in the anti-GnRH IgG4 response. Instead, these 2 phenomena seem to occur concurrently. The prolonged antigenic exposure resulting from repeated vaccinations and RT is the underlying cause of the shift in the immune response toward the IgG4 isotype, leading to a transition to a Th2 immune profile.
In this study, patients who were immunized with Heberprovac and managed to control tumor growth developed elevated and long-lasting anti-GnRH IgG1 responses. In contrast, 2 individuals with high initial responses (one with metastatic disease and the other with localized high-risk disease), with elevated, short anti-GnRH IgG1 responses experienced a shift to a Th2 response profile, a rapid biochemical relapse, and a short- or medium-term disease progression after RT. The shift toward the IgG4 isotype has been associated with the production of Th cytokines (IL-4, IL-13, and IL-10) by M2 macrophages, and regulatory T cells. 34 Previous research has reported a positive correlation between IgG4 levels and the number of Tregs, as well as a negative correlation with cytotoxic T lymphocytes, supporting their involvement in immune tolerance mechanisms in cancer. 35 The immunosuppressive effects of antibodies from this subclass mediate a tolerogenic microenvironment. This is achieved through macrophage polarization into M2b immunoregulatory cells which are believed to recruit Treg cells via the CCL1–CCR8 interaction. 35 Additionally, IgG4 exhibits lower affinity for FcγRIIa and no affinity for Fcγ. 36 Hence, it is less potent than IgG1 to mediate ADCC and antibody-dependent cellular phagocytosis. 37
Various authors have suggested additional mechanisms through which specific IgG4 subclass antibodies may foster tumor development. One such mechanism involves the ability of IgG4 to act as an antagonist to other IgG antibodies. This is due to its capacity to bind to the FcγRIIb inhibitor with a higher affinity than any other IgG subclass. 36 This receptor modulates both innate immunity (activation of macrophages, mastocytes, and basophils) and adaptative immunity (DCs activation and cross-presentation of antigens), and it has inhibitory effects on the actions of other FcγR when they are occupied by specific antibodies, particularly IgG1 subclass. 35 Reducing the intensity of IgG4 anti-GnRH humoral response could be a goal to be considered when planning the therapeutic scheme of Heberprovac in future clinical trials. Consequently, immunizations could be scheduled after the initial biochemical relapse following the first cycle of hormonal ablation and RT. Another potential approach might involve delaying RT for patients with heightened anti-GnRH IgG1 responses, thus mitigating the risk of antigenic overstimulation.
The development of significantly higher levels of anti-GnRH antibodies of the IgM isotype is another characteristic of the humoral response induced by Heberprovac. This distinguishes the 80% of patients who experienced disease progression from those who successfully maintained control over the disease, irrespective of the initial disease severity. In studies involving chronic lymphocytic leukemia in mice, secretory IgM was demonstrated to have immunosuppressive activity by recruiting and activating myeloid-derived suppressor cell (MDSC) effector functions. 38 However, it is still unknown if this action is directly induced by IgM binding to specific MDSC receptors, or if it takes place through IgM-antigen immune complexes binding to receptors of the complement via activated complement molecules and their receptors, such as C5a/C5aR, iC3b, and C3d. These complexes can promote tumor progression by recruiting and activating MDSCs. 38 It is also possible that IgM may stimulate other immune cell populations through small specific receptors different from MDSC. This could lead to an increased production of cytokines or chemokines that contribute to the expansion and regulation of immunosuppressive functions of MDSCs within tumors. 38 The role of these mechanisms in the progression of prostate adenocarcinomas requires further investigation.
This study included samples from patients with advanced disease and metastatic disease (distal n = 4 and nodal n = 2), which constitutes an additional limitation, analyzing the results and its conclusions more complex. It is a heterogeneous disease where, in addition to the tumor burden, the degree of differentiation of the tumor plays an important role in its outcome. However, in this study, we did not find significant differences regarding the intensity and type of anti-GnRH humoral response developed by patients with advanced and metastatic CSPC. As many as 67% of patients with mCSPC managed to maintain disease control throughout the study.
Factors such as peptide structure, formulation of the vaccine candidate, repeated immunizations, and the inclusion of RT in the therapeutic scheme have a decisive influence on the immune response observed. The rigidity of GnRHm1-TT peptide with respect to native GnRH due to substitution of L-glycine amino acid in position 6 by L-proline, its fusion to a 17-amino acid Th cell epitope of tetanic toxoid 39 and the use of Montanide ISA 51 VG as adjuvant (Seppic, France) are factors contributing to the induction of a highly specific IgG antibody response and potentially able to activate the Th immune response.
Furthermore, the inclusion of VSSP as an immune enhancer contributes to this response by activating human and murine DCs both in vitro and in vivo. This activation leads to the secretion of key cytokines such as IL-12p40/p70, TNF-α, and IL-6. Additionally, VSSP conditions the differentiation of virgin T cells toward the Th1 phenotype and triggers a robust cytotoxic T lymphocyte response to soluble peptides, 40 including GnRH.
Particularly, RT treatment could amplify the capacity of Heberprovac to control tumor growth. Kalina et al 41 reported that RT increases the expression of MHC class I molecules in tumor cells, therefore such a mechanism might bring about a more effective presentation of the GnRHm1TT peptide to the immune system of patients. Additionally, cellular necrosis and lysis induced by RT could trigger the release of damage signals and expose autologous neoantigens associated with the prostate tumor. This, in turn, activates the innate immune system and contributes to the expansion and improvement of adaptive immune response and the effector capacity of T cells. 41
Conclusions
In summary, this study demonstrates that immunization with Heberprovac induced potent and prolonged anti-GnRH serum antibody responses in patients with advanced and metastatic CSPC. The humoral response developed by the patients was characterized by the partial change of the IgM to IgG isotype, and the secretion of immunoglobulins IgG3 and IgG1 that define a Th1 response profile capable of contributing to tumor control through different effector mechanisms, including CDC and ADCC. Further studies are necessary to clarify mechanisms of the humoral and cellular immunity involved in the observed tumor growth control among the patients included in this study, and the contribution of each component involved in the therapeutic regimen to the clinical benefits observed in patients.
Footnotes
Abbreviations
Author’s Note
Ana Claudia Bover-Campal is also affiliated at Vaccine Research Group, Research Department, Center of Genetic Engineering and Biotechnology, Camagüey, Cuba.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ACCE, JAJB, FFA, LCA, EEBF, and HEGP are employees of the Centre for Genetic Engineering and Biotechnology, where this GnRH-based vaccine candidate is developed. The remaining authors have no conflict of interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
