Abstract
Objectives
This work aimed to prevent stoma stenosis and achieve tubeless cutaneous ureterostomy in elderly and high-risk patients with our modified cutaneous ureterostomy.
Methods
We retrospectively analyzed 40 and 49 patients (176 renal units) who underwent Toyoda (group 1) and modified cutaneous ureterostomy (group 2) between 2012 and 2021. The average follow-up period was 44 months. The primary results of our study were the catheter-free rate and clinical outcomes, especially renal function and urinary diversion-related complications. Significant differences in catheter-free rate and urinary diversion-related complications were found between our modified method and the Toyoda technique.
Results
A total of 56 (71.8%) of 78 renal units in group 1 and 89 (90.8%) of 98 renal units in group 2 remained catheter free. Compared with group 1, group 2 had a higher catheter-free rate (P = .001). Multivariate analysis indicated that the surgical procedure (HR = 0.268; P = .001) and body mass index (HR = 3.127; P = .002) were the predictors independently associated with catheter insertion. During follow-up, renal deterioration was observed in 32 (36.0%) patients. Patients with catheter insertion were more likely to suffer from renal deterioration (P < .001), postoperative pyelonephritis (P < .001), and urolithiasis (P < .001) than their counterparts.
Conclusion
Our modified cutaneous ureterostomy method may provide an effective and simple approach to tubeless cutaneous ureterostomy in elderly and high-risk patients.
Introduction
Cutaneous ureterostomy is a simple, quick, safe, and minimally invasive urinary diversion procedure that eliminates the risk of bowel and metabolic complications because the intestinal segment is not involved. Instead, a route to the upper urinary tract is facilitated without ureterointestinal anastomosis. However, postoperative ostomy stenosis and subsequent urinary tract infections have limited its application. 1 Although stoma stenosis can be avoided to some extent by changing the catheter periodically, this solution may greatly influence a patient's quality of life and cause catheter-related complications. After Toyoda first introduced a new technique for normal-caliber ureters to achieve tubeless cutaneous ureterostomy in 1977, 2 several modified technologies based on the Toyoda method have emerged, achieving catheter-free rates from 82% to 90%.3–6 Therefore, cutaneous ureterostomy, as a permanent rather than a temporary urinary diversion, especially for elderly and high-risk patients, has attracted the interest of urologists. 7 Furthermore, Kim et al 3 suggested that if the procedure succeeds without tubes, the complications of cutaneous ureterostomy are reduced, and the procedure is comparable with ileal conduit, which is the gold standard for urinary diversion. 8
To date, no studies have focused on large groups of elderly and high-risk patients who have undergone tubeless cutaneous ureterostomy. Although various modifications of tubeless cutaneous ureterostomy have been proposed, few studies have reported long-term dynamic estimated glomerular filtration rate (eGFR) changes after cutaneous ureterostomy. Given that the long-term follow-up of postoperative renal function is critical for evaluating urological reconstructive procedures, we introduced a modified method, evaluated and compared it with the Toyoda technique described by Kim et al, 3 and tracked patient clinical outcomes, especially eGFR changes.
Methods
This study followed the ethical standards set out in the 1964 Declaration of Helsinki. All patients signed consent forms before the procedure, and data from the procedure may be used for scientific research, but no personal information about the patients will be exposed.
The reporting of this study conformed to STROBE guidelines. 9 We retrospectively analyzed patients who underwent radical cystectomy and cutaneous ureterostomy (modified or Toyoda) between 2012 and 2021 who were 70 years of age or older and had an American Society of Anesthesiologists (ASA) grade 3 or higher; patients with grade 3 and above were defined as high risk based on ASA scores.10,11 The Toyoda and modified surgical procedures were performed by the same group of urologists. We routinely created the stoma on the right side (the midpoint of the connection of the navel and anterior superior iliac spine) at the relatively weak point of the abdominal muscles. If the left ureter was not long enough, then stomas were created on the left side (at a location corresponding to the right side). In patients with solitary renal units, the stoma was created on the renal unit side. During ureteral mobilization out of the pelvis, we primarily used the method described by Straffon et al 12 However, the process was not identical because the parietal peritoneum was opened bilaterally, and bilateral ureters were exposed. We transected the ureters as close to the bladder as possible. We dissected the ureters up to the crossing of the ureters with the common iliac artery, about 6 to 8 cm away from the bladder. We did not mobilize the ureters all the way up to the uretero-pelvic junction as we found that generally there was a sufficient ureteric length to create our cutaneous ureterostomy. When creating the abdominal wall tunnel, we primarily utilized used the method described by Kim et al 3 The abdominal wall tunnel passes through several layers including the abdominal transverse fascia, abdominal internal oblique muscle, abdominal external oblique muscle, superficial fascia, and skin. For group 1, we formed the ostomy using the Toyoda 2 method, in which the distal end of the ureter is cut longitudinally to make a fish-mouth-like opening and each separated ureteral segment is sutured to the corresponding skin area. By contrast, in group 2, the modified cutaneous ureterostomy was performed. An approximately 3 to 4 cm slit was made at each ureteral end. The ureters were conjoined side to side using 3 interrupted sutures (at the bottom of slits and at each side of slits). Before the ureters were brought out of the abdominal wall, we reshaped and sutured the bilateral ureters together. The surgical procedure is depicted in detail in Figure 1. The color of the ureteral mucosa is usually observed to determine blood supply. When necessary, suture acupuncture was used to Pierce the exposed ureteral mucosa, observe the bleeding in the pinhole, and evaluate the blood supply at the distal ureter. Intraoperative frozen pathology excluded positive surgical margins, and ureteral stents were placed at the end of the operation to prevent residual tumors. The stents were removed within 3 months. If the hydronephrosis is significantly aggravated after the removal of the ureteral stent, lumbago or fever, the ureteral stent (urovision Single J tube, diameter 7F, length 35 cm) should be reinserted anterograde through nephrostomy. We used an ostomy care device to maintain the ostomy, changed the ostomy bag approximately twice a week, and had no difficulty in performing either method. Patients with upper urinary tract tumors or ureteral stricture before surgery were excluded, and patients followed for less than 12 months after surgery were excluded from the final analysis.

Surgical procedure of modified tubeless ureterocutaneostomy. (a) An approximately 3 to 4 cm slit was made at each ureteral end. The ureters were conjoined side to side using 3 interrupted sutures (4-0 Coated Vicryl Plus Antibacterial Suture, at the bottom of slits and at each side of slits). Approximately 2 to 3 cm was not sutured, forming a Y shape. (b) We not only sutured the abdominal wall tunnel between the anterior and posterior rectus sheaths but also fixed the ureters to the abdominal wall tunnel using 4 interrupted sutures (5-0 Coated Vicryl Plus Antibacterial Suture). (c) Conjoined ureters were everted and sutured to the skin (4-0 Coated Vicryl Plus Antibacterial Suture), forming an everted double-nipple stoma.
The primary outcomes assessed in our study were the catheter insertion rate and patient clinical outcomes, especially renal function. We retrospectively followed up patients in our outpatient clinic using the European Association of Urology Guidelines,13,14 and renal function was tested every 6 months after the operation. Serum creatinine measurements were used to calculate eGFR using the equation described by Liu et al 15 renal deterioration was defined as >20% decrease in eGFR relative to the presurgery value. 16 Body mass index (BMI) was defined as a patient's weight (kilogram) divided by the square of the patient's height (meters). 17 Acute pyelonephritis was defined as a positive urine culture (>105 colony-forming units) and either frank pain or tenderness with fever (>38 °C) was observed in the absence of another indication for fever. Hydronephrosis was evaluated by ultrasound and divided into 4 grades. 18 The tumor-node-metastasis classification system was used for staging, 19 the ASA physical status classification system was used to quantify the anesthetic risk of concomitant disease, 20 and a modified Clavien classification system was used to evaluate postoperative complications. 21
Statistical Analyses
Two-sided t-test, Wilcoxon rank sum test, and Fisher's exact test were used to evaluate baseline variables. Analysis of covariance was used to compare eGFR at last follow-up. The Kaplan–Meier curve and log-rank test were used to analyze the overall mortality and catheter-free rate. Multivariable Cox proportional hazards regression analysis was conducted to identify independent predictors. Data were analyzed using SPSS version 26.0 (Corp., Armonk, NY, USA). A P-value of <.05 was considered statistically significant.
Results
A total of 89 patients with bladder cancer (72 males and 17 females aged 70 years or older) and with an ASA classification of 3 (71.9%) or greater (28.0%) were enrolled in our study. Muscle-invasive bladder cancer was diagnosed in 86 of these patients, and recurrent non-muscle-invasive bladder cancer resistant to nonsurgical treatment (immunotherapy/chemotherapy) was diagnosed in 3. All patients met the main indications for radical cystectomy.13,14 The 89 identified patients were divided into 2 groups: Toyoda cutaneous ureterostomy (group 1; 78 renal units from 40 patients including 2 solitary kidneys) and modified cutaneous ureterostomy (group 2; 98 renal units from 49 patients). Groups 1 and 2 had average ages of 77.2 ± 4.5 and 77.3 ± 4.1 years, respectively. Major clinical characteristics, patient demographics, and most common comorbidities are illustrated in Table 1. Two patients each had a solitary kidney, 3 had an average eGFR of 34.76 mL/min/1.73 m2 and disease-related acute renal insufficiency and bilateral upper urinary hydrocele, and 4 had unilateral upper urinary hydrocele. Overall, 10 renal units suffered from preoperative hydronephrosis (eight grade 2 and two grade 3). Bilateral pelvic lymphadenectomy was performed in 78 of 89 patients. The median DJ tube removal time was 85.5 days (range 82-90 days).
Baseline Demographics and Clinical Characteristics Based on Study Group.

Abbreviations: BMI, body mass index; ASA, American Society of Anesthesiologists; eGFR, estimated glomerular filtration rate.
Two-side t-test.
Fisher's exact test (2-sided).
Wilcoxon rank sum test. Results are number of patients and percentage in parentheses.
Bilateral ureters were used to construct unilateral stomas in 87 patients. Two patients create the stoma on the left side because the left ureter was short (positive surgical margin), but the right ureter was long enough, in 2 patients with solitary renal units, the stoma was created on the renal unit side (one right and one left). For the rest of the patients, we routinely create an ostomy on the right side.
As of June 2021, the mean follow-up was 44.1 ± 16.7 months, 94% (84) of the patients were followed for more than 24 months, and a total of 30 (33.7%) patients were alive and disease-free, whereas 52 (58.4%) patients had died. Tumor recurrence was the cause of death of 39 patients. There was no significant difference in survival between the 2 groups (Table 2; P = .525). Several major demographics and clinical characteristics were examined (Table 1), and no significant differences were found between the groups. Postoperative complications occurred in 43 (48.3%) patients, primarily those with Clavien grades I (78.0%) and II (15.0%). No significant differences were found (P = .92) between the 2 groups. Pneumonia, lymphatic fistula, hypotension (systolic blood pressure <90 mm Hg), decreased oxygen saturation (<90%), and cardiac arrhythmia were the most common postoperative complications. Although no postoperative deaths occurred, reoperation was necessary in 3 (7.0%) patients due to rectal injury, iliac vein contusion, and unknown bleeding.
Perioperative and Postoperative Complications and Pathological Results Based on Study Group.

Two-side t-test.
Wilcoxon rank sum test. Results are medians and range or number of patients and percentage in parentheses.
Analysis of covariance (Covariate: preoperative eGFR).
Fisher's exact test (2-sided).
Log-rank test.
* Statistically significant at P < .05.
Urinary diversion-related complications occurred in 49 (55.1%) patients. The modified group had a lower number of diversion-related complications than the Toyoda group, but P = .05, so we could not easily determine whether the difference was statistically significant or not. The most common complication was hydronephrosis (33.7%), but these hydronephrosis were mild and did not require intervention. Acute pyelonephritis was the second most common complication (21.4%; 11 cases were related to catheter insertion), followed by urolithiasis (11.2%) and stoma retraction or stricture (12.4%). Except hydronephrosis, the Toyoda group patients were more likely to experience stoma retraction or stricture (P = .01), urolithiasis (P = .005), and acute pyelonephritis than the patients in the modified group (P = .036; Table 2). Table 2 shows that the perioperative data between the 2 groups were not different.
The study included 89 patients, representing 176 renal units (right: 88; left: 88). A total of 56 (71.8%) of 78 renal units in group 1 and 89 (90.8%) of 98 renal units in group 2 remained catheter free. Ureteral stents insertion was required in 31 renal units, representing 19 patients (right ureter: 17; left ureter: 14). The indications for catheter insertion included stoma retraction or stenosis, ureteral stricture, unilateral or bilateral flank pain, recurrent pyelonephritis, extraluminal obstruction, and any other factor that would lead to or indicate ureter obstruction. The ureteral stent was replaced every 8 to 10 months. Table 3 shows that no significant differences were found between the ipsilateral and contralateral sides in relation to the catheter-free rate (P = .61). Compared with group 1, group 2 had a higher catheter-free rate (P = .001; Table 3). Kaplan–Meier curve and log-rank analysis indicated that group 1 had a higher catheter insertion rate than group 2 (P = .001; Figure 2). Furthermore, stepwise multivariate Cox proportional hazard regression analysis indicated that the surgical procedure (HR = 0.268; P = .001) and BMI (HR = 3.127; P = .002) were the predictors independently associated with the catheter-insertion rate (Table 4).

Relationship between catheter-free rate and catheter insertion time. Kaplan–Meier curve of catheter-free rate classified by Toyoda or modified renal units in univariate log-rank analysis (P = .001). In the Kaplan–Meier curve, catheter insertion was defined as a terminal event, and the period between the operation and ureteral stent reinsertion was defined as survival time.
Several Parameters Stratified by Catheter-Free or not.

Fisher's exact test (2-sided).
Log-rank test.
Analysis of covariance (covariate: preoperative eGFR), with equal variances assumed under Leven's test. Results are means ± SD.
* Statistically significant at P < .05.
Univariate and Multivariate Cox Proportion Hazards Regression Analyses of Clinicopathological Features Associated With Catheter Insertion.

Cox proportional hazards regression analysis using stepwise regression methods. Abbreviations: TNM, tumor-node-metastasis; BMI, body mass index.
The mean eGFR of the 89 patients decreased from 61.9 to 54.3 mL/min/1.73 m2 at the last follow-up, and renal deterioration was observed in 32 (36.0%) patients. No significant difference was found between the 2 groups (P = .125; Table 2). However, patients with catheter insertion were at higher risk of suffering from renal deterioration (P = .001), postoperative pyelonephritis (P < .001), and urolithiasis (P = .003; Table 3) than their counterparts. Figure 3 shows that the eGFR decreased dramatically in patients with catheter insertion.
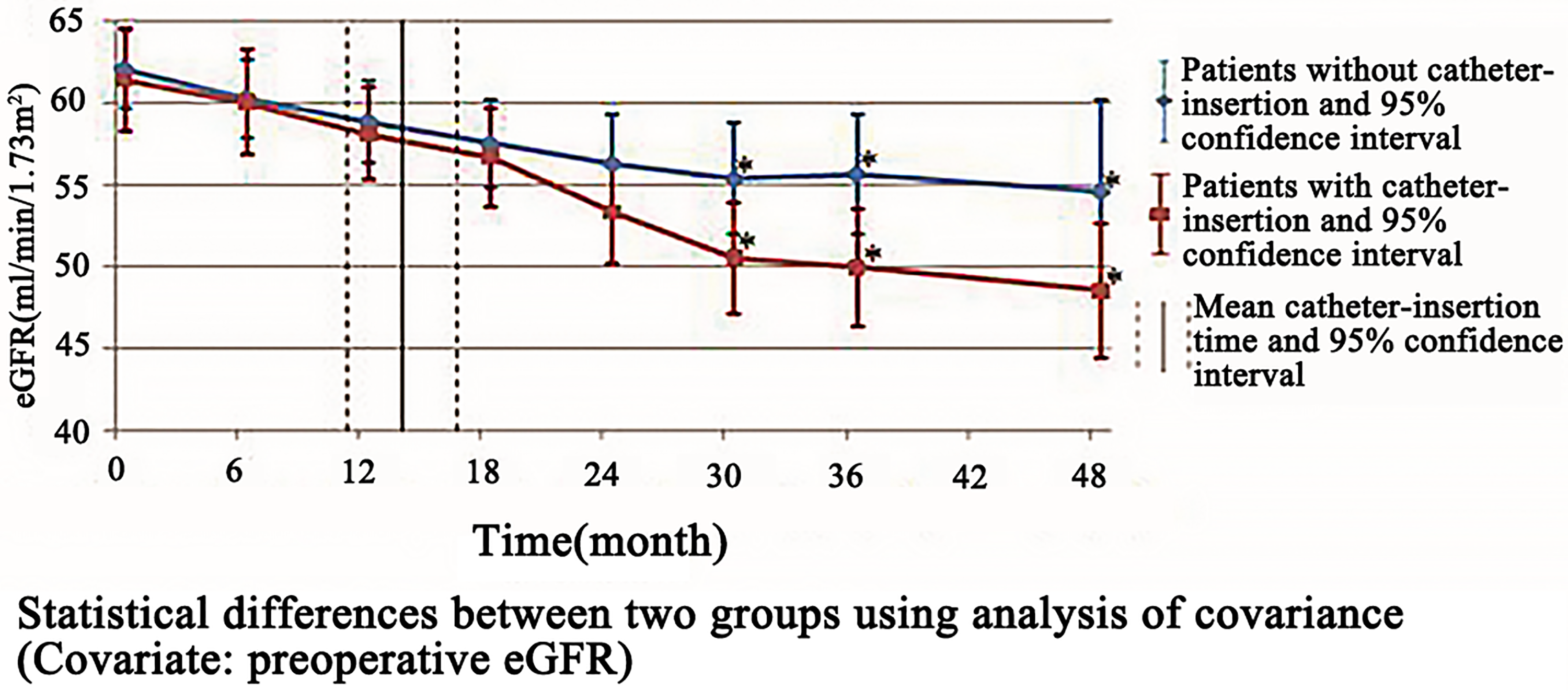
Estimated glomerular filtration rate (eGFR) changes in a time-dependent manner between patients with and without catheter insertion. After catheter insertion (mean time at 13.8 months), the eGFR decreased dramatically in patients with catheter insertion.
Discussion
For patients with cystectomy and urinary diversion, preserving renal function and improving patients’ quality of life are the primary goals of treatment. Compared with orthotopic neobladder and ileal conduit, cutaneous ureterostomy provides a simplified and safe alternative for urinary diversion in high-risk patients. The procedure can be performed quickly and has few severe postoperative complications.10,22 If stoma stenosis can be reduced without catheter insertion, cutaneous ureterostomy can be applied not only to unfit patients but also to patients with a good prognosis. 3
The baseline backgrounds of the patients in the 2 groups were similar (Table 1). In addition, no significant differences in perioperative data, postoperative complications graded by the Clavien grade system, and mortality were found, indicating that our modified surgical procedure did not complicate the surgical procedure, cause perioperative complications, or influence disease progress. In a retrospective study of 445 patients undergoing ileal conduit by Demaegd et al, 23 the incidence of postoperative complications was 49.4% for grade 1 to 2 and 13.9% for grade 3 to 5, which was higher than our results. It is important to note that the patients we included were clearly in worse physical condition. When performed in our institute, the original Toyoda method achieved a 71.8% catheter-free rate, which was relatively low compared with the 89.8% rate reported by Kim. However, Kim did not clearly describe the indications for catheter insertion, and patients in his study may have had a better baseline condition than those in ours, which would contribute to the difference in the catheter-free rate. Under similar conditions, a high catheter-free rate (91.8%) was achieved by the modified method.
Moreover, the modified method achieved lower urinary diversion-related complications than the Toyoda method. We observed that not only urolithiasis and subsequent urinary tract infection but also renal function deterioration were closely related to catheter insertion (Table 3). Therefore, tubeless ureterocutaneostomy is important to reduce diversion-related complications and preserve renal function for early high-risk patients.
Stoma stenosis can be explained by several mechanisms. First, the blood supply to the distal ureter is poor. Wada et al 24 reported that the stoma stenosis rate is reduced from 74% to approximately 10% when the fascia between the spermatic cord and ureter is preserved. In addition, proximal blood supplies from gonadal and renal vessels are not only important but also sufficiently protect the distal ureter from ischemia 25 ; thus, protecting these vessels is extremely important. We did not dissect the ureters to a high level to ensure that the proximal ureter remained untouched and to preserve essential vessels. Moreover, the distal parts of the ureters were sutured side to side, and the blood vessels of both ureters will spread and grow to the opposite side. Therefore, even when ischemia occurred on one side of the distal ureter, blood was supplied from the other side.
Second, Namiki and Yanagi 26 assumed that the main cause of stoma stenosis is increased tension in the distal ureters when blood supply decreases. Increased tension leads to mucosal and submucosal edema, resulting in ureteral degeneration and stenosis. This finding was supported by the large study (310 patients) of Rodriguez et al, 27 who showed that contralateral ureters, which have a longer trajectory and are more extensively mobilized with more tension, are more likely to exhibit obstruction than ipsilateral ureters. However, we did not face much difficulty when we mobilized the bilateral ureters to the stoma point partly because tension was reduced by suturing and anchoring the contralateral ureter to the ipsilateral ureter and fixing both to the abdominal wall before the contralateral ureter was pulled out of the abdominal wall. In addition, in all patients (except 2 positive surgical margin patients), we transected the ureters as close to the bladder as possible. This procedure ensured sufficient length for the contralateral ureter. Interestingly, Dreikorn 28 and Davari et al 29 preferred to preserve a short ureter in renal transplantation surgery, contradictory to our findings. As noted above, when we sufficiently reduced the tension of the ureter during surgery, although the blood supply to the ureter was affected during transplantation, it was not enough to cause distal ischemia; therefore, the risk of developing ostomy stenosis was also reduced. In this study, no distal ischemia or contralateral obstruction was observed (Table 3), which strongly supported this view.
Another important mechanism regarding stoma stenosis is the compression of ureters when crossing the abdominal wall tunnel. Burkhard and Wuethrich 30 regarded this factor as the main reason for postoperative stenosis because compression in the abdominal wall tunnel not only interferes with urine flow but also decreases the blood supply to the distal ureters. In a retrospective review, Terai et al 6 found that the abdominal wall tunnel is sufficiently enlarged in the majority of stoma stenosis cases. Thus, we selected the relatively weak points of the abdominal muscles as the stoma point and reshaped ureters and the abdominal wall tunnel (Figures 1a and 1b). This simple strategy not only shortens the distance of the abdominal wall tunnel by suturing the anterior and posterior rectus sheaths but also stabilizes the ureters by suturing 2 ureters side to side and affixing them to the abdominal wall tunnel.
Obesity was thought to have a negative effect on cutaneous ureterostomy because the ureter is subject to tension and compression when it reaches the skin due to abdominal wall thickness, 27 so patients with a large BMI who underwent cutaneous ureterostomy are more likely to rely on catheter insertion to avoid ureter obstruction compared with patients with a low BMI.
In our modified method, before being sutured to the skin, bilateral ureters were sutured together and formed a Y shape. Each side of the Y shape was a ureter, thereby having larger areas of mucosal ureter to be everted than 2 ureters everted separately and forming a larger jointed everted double-nipple stoma. We believe this is also one of the important reasons why our modified methods achieved a higher catheter-free rate than the Toyoda technique.
We did encounter 2 cases with insufficient ureter length because of oncological margins. Hence, we created a stoma on the left side at a corresponding location without difficulty. Both cases achieved a catheter-free status. However, for patients with insufficient length bilaterally or with a solitary kidney, our technique cannot be used. Moreover, many other methods can be used for short ureters.26,31 Given the nursing requirements and quality of life, bilateral stomas should be avoided and only used as a last resort.
The small sample size and single center diminished the significance of this retrospective study. In addition, this modified technique is not completely tubeless in the true sense. Ureteral stents still need to be retained for no more than 3 months after surgery, and the time of tube extraction for patients has individual differences. Whether the difference in the tube extraction time will affect postoperative complications of patients is unknown. Despite these limitations, our results are encouraging, and our modified technique can serve as a reference for others to explore urinary rerouting in high-risk patients.
Conclusions
Our results showed that modified cutaneous ureterostomy has a high catheter-free rate, and patients with catheter insertion are more likely to experience renal deterioration, postoperative pyelonephritis, and urolithiasis than their counterparts. Our modified cutaneous ureterostomy method may provide an effective and simple approach to tubeless cutaneous ureterostomy in elderly and high-risk patients.
Footnotes
Abbreviations
Acknowledgments
The authors are grateful to all staff of the Urology Department for providing excellent collaboration during the patient recruitment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study followed the ethical standards set out in the 1964 Declaration of Helsinki and was approved by the Ethics Committee of the Xiangya Hospital of Central South University (No. 202109187).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Hunan Province (2020JJ4542), the Clinical Research Project of University of South China (USCKF201902K01), and the Hunan Provincial Clinical Medical Technology Innovation Guiding Project (2020SK51801). Funders only provide financial support and do not interfere with the research process.
Informed Consent
All patients in the study signed an informed consent form. All patients agreed to publish the study without revealing their personal information.
