Abstract
Objective:
To investigate role of microRNA-1/Golgi phosphoprotein 3/Foxo1 axis in bladder cancer.
Methods:
The expression of Golgi phosphoprotein 3 was determined in both bladder cancer tissues and cell lines using quantitative real-time polymerase chain reaction and Western blotting, respectively. Golgi phosphoprotein 3 was knocked down by small hairpin RNA. MicroRNA-1 was overexpressed or inhibited by microRNA-1 mimic or inhibitor. Cell viability and proliferation were determined by 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) and colony-formation assay. Cell apoptosis and cycle was detected using flow cytometer. The expression of microRNA-1 and Golgi phosphoprotein 3 was determined using quantitative real-time polymerase chain reaction and Western blotting was used to test the expression of Golgi phosphoprotein 3, Foxo1, p-Foxo1, AKT, p-AKT, p27, and CyclinD1. Binding between microRNA-1 and Golgi phosphoprotein 3 was confirmed by Dual-Luciferase Reporter Assay.
Results:
MicroRNA-1 was downregulated in bladder cancer tissues, while Golgi phosphoprotein 3 was overexpressed in bladder cancer cells and tissues. In both bladder cancer 5637 and T24 cell lines, the cell viability and proliferation were dramatically reduced when Golgi phosphoprotein 3 was knocked down. The inhibition of Golgi phosphoprotein 3 remarkably promoted cell apoptosis and induced cell-cycle arrest, as well as decreased the expression of p-Foxo1, p-AKT, and CyclinD1 and increased the expression of p27. The overexpression of microRNA-1 significantly inhibited cell viability and proliferation, induced G-S cell-cycle arrest, and decreased the expression of Golgi phosphoprotein 3, p-Foxo1, and CyclinD1 and upregulated p27, while inhibition of microRNA-1 led to opposite results. Golgi phosphoprotein 3 was a direct target for microRNA-1.
Conclusion:
Overexpression of microRNA-1 inhibited cell proliferation and induced cell-cycle arrest of bladder cancer cells through targeting Golgi phosphoprotein 3 and regulation of Foxo1.
Keywords
Introduction
Bladder cancer (BC), the fourth most common cancer in males and ninth most common in females, accounts for more than 350 000 new cases of cancer every year worldwide. 1,2 Despite development of surgical techniques, the prognosis of BC is still poor due to the metastasis of high occurrence. 3 Thus, early diagnosis and better understanding for BC are still urgent.
Golgi phosphoprotein 3 (GOLPH3) is a newly identified membrane protein in the trans-Golgi matrix, which is involved in progression of anterograde and retrograde Golgi trafficking. 4,5 Several studies have demonstrated that GOLPH3 is involved in development of many cancers, such as gastric cancer, 6 pancreatic ductal adenocarcinoma, 7 and colorectal cancer. 8 It was found that GOLPH3 could be used as a potential biomarker for prognosis of cancers, including BC. 9,10 However, deeper insights for how GOLPH3 affects BC development are still unclear.
MicroRNAs (miRNAs) are noncoding RNA with about 22 nucleotides in length. Studies show miRNAs play important roles in bioprocesses and are involved in cancer development. 11 MicroRNA-1, a kind of miRNA, is considered to act as a tumor suppressor in several cancers such as prostate cancer 12 and glioblastoma. 13 Studies also found that miR-1 could suppress tumor development of BCs. 14 -16 However, no study reported relationship between miR-1 and GOLPH3 in cancer development.
Recently, a study showed GOLPH3 could significantly promote cell proliferation and tumorigenicity of breast cancer cells by regulation of Foxo1. 17 However, to our best knowledge, no study focused on role of miR-1/GOLPH3/Foxo1 axis in BC development. In the present study, we for the first time demonstrated that inhibition of GOLPH3 could inhibit cell proliferation and induce cell-cycle arrest of BC cells through regulation of Foxo1, and overexpression of miR-1 could reverse the above effects by directly targeting GOLPH3. These results might give us deeper understanding for miR-1/GOLPH3/Foxo1 axis in cancer development.
Materials and Methods
Tissue Samples
A total of 30 tissue samples of BC and the adjacent tissue samples were obtained from patients at Affiliated Hospital of Hebei University. The samples were collected and frozen immediately after resected and were stored at −80oC before used. All patients involved in the study have signed the written informed consent for the biological studies, and the study was approved by the Ethics Committee of Affiliated Hospital of Hebei University.
Cell Culture and Transfection
Bladder cancer cell lines such as BC 5637, BIU-87, BT-B, and T24 cells, as well as normal immortalized human BC cell line SV-HUC-1 cells were all were purchased from American Type Culture Collection (Manassas, Virginia). Briefly, cells were cultured in RPMI-1640 (Thermo Fisher Scientific, Inc, Waltham, Massachusetts) containing 10% Gibco fetal bovine serum and 100 μg/mL penicillin-streptomycin (Sigma-Aldrich Co, St. Louis, MO) under 37oC and 5% CO2. Cells were then transfected with small hairpin RNA (shRNA) for GOLPH3 (sh-GOLPH3), miR-1 mimic, inhibitor (5 nM, all purchased from GeneChem Corp, Shanghai, China) or negative control (NC) using the Lipofectamine 3000 (Invitrogen, Waltham, Massachusetts) in serum-free Opti-MEM medium, according to the manufacturer’s instruction.
MTT Assay
To measure cell viabilities, 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) assay was conducted for different groups of cells at 1, 2, 3, and 4 days after transfection, respectively. Briefly, cells (4 × 103) were seeded in 96-well plates, cultured for 48 hours under 37oC and 5% CO2, and added with 10 μL MTT solution (5 mg/mL). Cells were then further cultured for 4 hours and the MTT was removed. Then, 100 μL dimethylsulfoxide was added and the value of optical density was evaluated 490 nm.
Colony Formation Assay
Clonogenic formation assay was performed 48 hours after transfection to further evaluate cell proliferation, as described elsewhere. 1 Briefly, cells at density of 5 × 103 were seeded into 6-well dishes. After culturing for 14 days, cells were fixed with glutaraldehyde and stained with crystal violet. The positive colony formation was manually counted. All experiments were performed in triplicate.
Measurement of Cell Apoptosis
For measurement of cell apoptosis, briefly, cells were harvested 48 hours after transfection, washed with phosphate-buffered saline (PBS), and fixed with 70% ethanol overnight at 4°C. The cells were stained Annexin V/propidium iodide double-staining kit (BD Biosciences, Waltham, Massachusetts) strictly according to the manufacturer’s instruction and a FSCAN flow cytometer (FCM; BD Biosciences) was used to measure the cell apoptosis.
Measurement for Cell Cycles
Briefly, cells were harvested with trypsinization, washed, and fixed. Then, cells were resuspended in 100 μL of PBS containing 50 μg/mL RNase for 30 minutes at room temperature. Propidium iodide (20 μg/mL) was used to stain the cells for 20 minutes using an FITC Annexin V kit (BD Biosciences). A FSCAN FCM (BD Biosciences) was used to measure the cell cycle.
Dual-Luciferase Reporter Assay
Dual-Luciferase Reporter Assay was performed to confirm the GOLPH3 3′-UTR as a target of miR-1. Briefly, the wild-type (WT) GOLPH3 3′-UTR or mutant (MUT) was amplified and subcloned into a pGL4.10 Luciferase Reporter Vector. Then, cells were transfected with either the vectors and the miR-1 mimics or NC control using Lipofectamine 3000. After 48 hours of transfection, Luciferase assays were performed using a Bright-Glo Luciferase Assay System (Promega, Madison, WI). The luciferase activity was normalized to values of Renilla Luciferase activity.
Quantitative Real-Time Polymerase Chain Reaction
The expression of miR-1 and GOLPH3 was determined using quantitative real-time polymerase chain reaction (qRT-PCR). Briefly, total RNA was extracted from the BC tissues or cells using the Trizol reagent (Tiangen Biotech, Beijing, China). Then, RNA was converted into complementary DNA using a Prime-Script one-step qRT-PCR kit (TAKARA, Dalian, China). The PCR reactions were conducted in an ABI 7500 Fast RealTime PCR System (Life Technologies, Carlsbad, CA). Primers used in PCR were listed below: miR-1, F 5′-GCCCGCTGGAATGTAAAGAAGT-3′, R 5′-GTGCAGGGTCCGAGGT-3′; GOLPH3, F 5′-GGGCGACTCCAAGGAAAC-3′, R 5′-CAGCCACGTAATCCAGATGAT-3′; U6, F 5′-CCTATTTCCCATGATTCCTTCATA-3′, R 5′-GTAATACGGTTATCCACGCG-3′; and GAPDH, F 5′-ACAGCAACAGGGTGGTGGAC-3′, R 5′-TTTGAGGGTGCAGCGAACTT-3′.
Relative RNA levels were calculated by the 2−ΔΔCt method. Both U6 and GAPDH were used as internal controls.
Western Blotting
Western blotting was used to test the expression of GOLPH3, Foxo1, p-Foxo1, AKT, p-AKT, p27, and CyclinD1. β-Actin was used as a control. Briefly, samples were extracted, loaded on sodium dodecyl sulfate polyacrylamide gel electrophoresis, and transferred to polyvinylidene difluoride membranes. Then, samples were incubated with primary antibodies (all purchased from Abcam, Cambridge, Massachusetts) of anti-GOLPH3 antibody (ab91492, 1/500), anti-Foxo1 antibody (ab70382, 1/3000), anti-p-Foxo1 antibody (ab131339, 1/1000), anti-AKT antibody (ab8805, 1/500), anti-p-AKT antibody (ab8933, 1/500), anti-p27 antibody (ab193379, 1/500), anti-CyclinD1 antibody (ab16663, 1/200), anti-AKT antibody (ab179463, 1/10 000), and anti-p-AKT antibody (ab38449, 1/500), following with a conjugated secondary antibody (the corresponding secondary horseradish peroxidase-conjugated anti-rabbit [ab6721] or anti-mouse [ab6785] immunoglobulin G antibodies). The Pierce ECL Western Blotting Substrate (Pierce, Shanghai, China) was used to scan the protein bands, according to the manufacture’s instruction.
Statistical Analysis
The measurement data were expressed by mean ± standard deviation. Comparison between 2 groups was performed using the Student t test. Comparison among 3 or more groups was conducted using 1-way analysis of variance. It was considered to be statistically significant when P value was less than .05. All calculations were made using SPSS version 22.0.
Results
Golgi Phosphoprotein 3 was Overexpressed in BC Tissues and Cells
First, we evaluated the expression of miR-1 and GOLPH3 in both BC tissues and cell lines. As shown in Figure 1A and B, the expression of miR-1 was significantly downregulated, while the expression of GOLPH3 was dramatically upregulated in BC tissues (P < .05). Further in vitro experiments demonstrated the expression of GOLPH3 was overexpressed in all BC cell lines compared with normal SV-HUC-1 cells for both messenger RNA and protein levels (P < .05, Figure 1C and D), indicating that GOLPH3 was overexpressed in BC. Since expression of GOLPH3 was higher in BC 5637 and T24 cell lines, these 2 cells were used for further experiments.
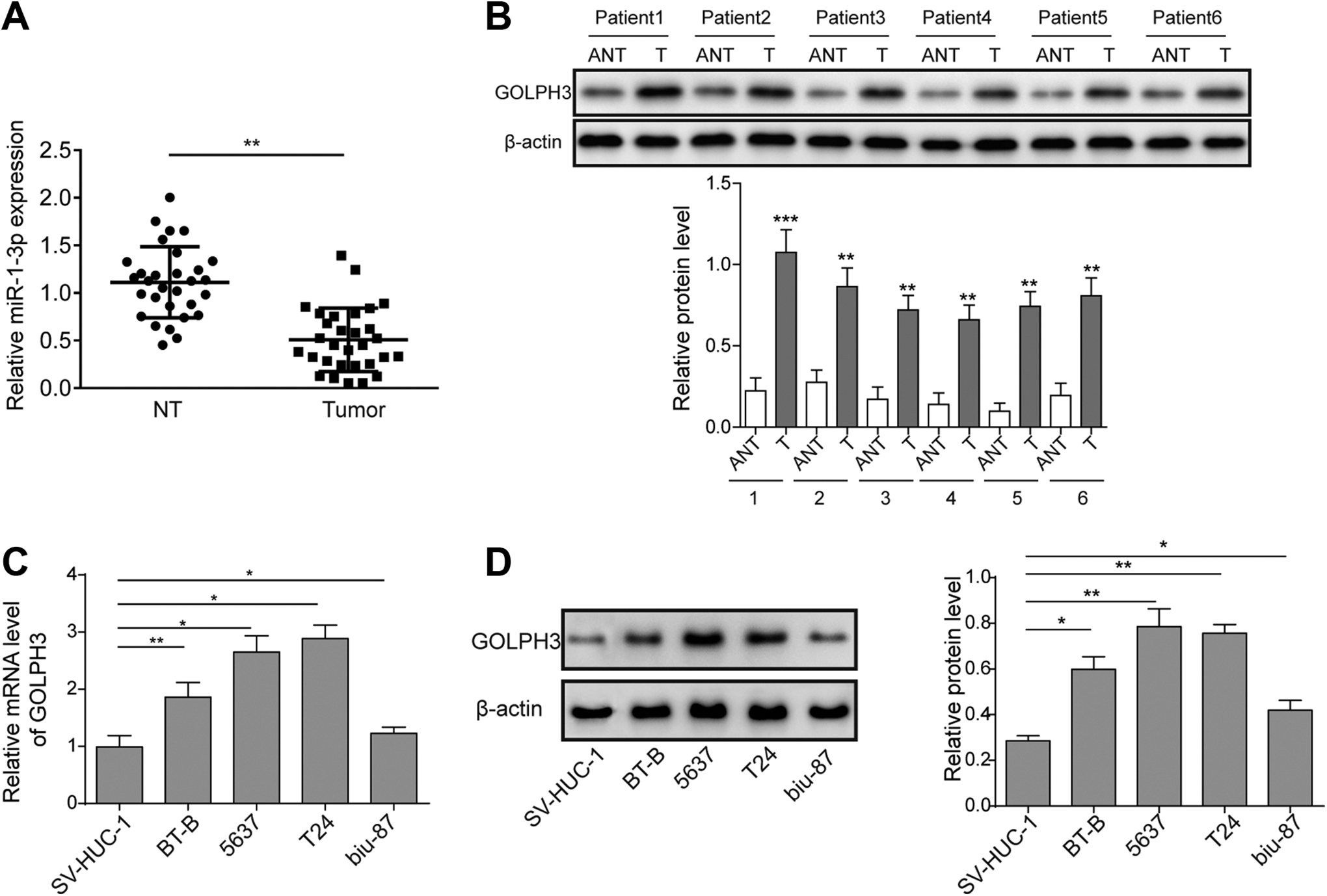
Golgi phosphoprotein 3 was overexpressed in BC cells. A, Expression of miR-1 in BC tissues and normal tissues by RT-qPCR. B, Expression of GOLPH3 in BC tissues and normal tissues by Western blotting. C, Expression of GOLPH3 in different BC cell lines by RT-qPCR. D, Expression of GOLPH3 in different BC cell lines by Western blotting. The result was a representative of 3 independent experiments. Error bars represented mean ± standard deviation. *P < .05, **P < .01. BC indicates bladder cancer; GOLPH3, Golgi phosphoprotein 3; qRT-PCR, quantitative real-time polymerase chain reaction.
Knockdown of GOLPH3 Inhibited Proliferation, Promoted Cell Apoptosis, and Induced Cell-Cycle Arrest of BC Cells
To further investigate role of GOLPH3 in BC development, we used shRNA to knockdown GOLPH3 in both BC 5637 and T24 cell lines. The morphology of both BC 5637 and T24 cell lines was shown in Figure 2A after inhibition of GOLPH3. Results showed in both 2 cell lines, GOLPH3 was significantly decreased when cells were transfected with sh-GOLPH3 compared with the NC (P < .05, Figure 2B), suggesting successful establishment of GOLPH3 knockdown model. Moreover, when transfected with sh-GOLPH3, the cell viability and proliferation were dramatically reduced in both BC 5637 and T24 cell lines by MTT assay and colony formation assay (P < .05, Figure 2C and D). Further FCM analysis showed knockdown of GOLPH3 remarkably increased cell apoptosis compared with the NC cells (P < .05, Figure 2E). What’s more, GOLPH3 knockdown also significantly induced cell-cycle arrest of G1-S phase (P < .05, Figure 2F). All these results suggested inhibition of GOLPH3 could depress cell proliferation, promote cell apoptosis, and induce cell-cycle arrest of BC cells.

Knockdown of GOLPH3 inhibited proliferation, promoted cell apoptosis, and induced cell-cycle arrest of BC cells. A, Morphology of BC 5637 and T24 cells at 1 and 4 days after inhibition of GOLPH3. B, Expression of GOLPH3 in BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by Western blotting. C, Cell viability of BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by MTT assay. D, Cell proliferation of BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by colony formation assay. E, Cell apoptosis of BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by FCM. F Cell cycle of BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by FCM. The result was a representative of 3 independent experiments. Error bars represented mean ± standard deviation. *P < .05, **P < .01. BC indicates bladder cancer; FCM, flow cytometry; GOLPH3, Golgi phosphoprotein 3; sh-GOLPH3, small hairpin RNA for GOLPH3.
Knockdown of GOLPH3 Downregulated p-Foxo1 and p-AKT
The expression of GOLPH3, Foxo1, p-Foxo1, AKT, p-AKT, p27, and CyclinD1 was then determined using Western blotting to further study the effect of GOLPH3 on related signaling and proteins. As shown in Figure 3A, when GOLPH3 was inhibited in both BC 5637 and T24 cell lines, the expression of both p-Foxo1 and p-AKT was dramatically decreased compared with the control (P < .05). Meanwhile, the expression of p27 was significantly upregulated while CyclinD1 was significantly downregulated when GOLPH3 was knocked down (P < .05), indicating that knockdown of GOLPH3 could inhibit phosphorylation of Foxo1 and AKT, as well as affect expression of cell-cycle–related protein p27 and CyclinD1.

Knockdown of GOLPH3 could downregulated phosphorylation of Foxo1 and AKT and regulate the expression of p27 and CyclinD1. A, Expression of GOLPH3, Foxo1, p-Foxo1, AKT, p-AKT, p27, and CyclinD1 in BC 5637 and T24 cells transfected with sh-GOLPH3 or negative control by Western blotting. The result was a representative of 3 independent experiments. Error bars represented mean ± standard deviation. *P < .05. BC indicates bladder cancer; GOLPH3, Golgi phosphoprotein 3; sh-GOLPH3, small hairpin RNA for GOLPH3.
MicroRNA-1 Inhibited Cell Proliferation and Induced Cell-Cycle Arrest of BC Cells by Targeting GOLPH3
Finally, we investigated relationship between miR-1 and GOLPH3 in BC cells. As shown in Figure 4A, the predicted binding for GOLPH3 and miR-1 was obtained using bioinformatic prediction by software Targetscan 7.2. Dual-Luciferase Reporter Assay also showed the Luciferase activity significantly deceased in WT-GOLPH3 (P < .05) and no significant difference was found in MUT-GOLPH3, indicating miR-1 directly targeted on GOLPH3 (Figure 4B). When transfected with miR-1 mimic, the expression of GOLPH3 was significantly inhibited, while when transfected with miR-1 inhibitor, the expression of GOLPH3 was significantly increased compared with NC cells by qRT-PCR (P < .05, Figure 4C), suggesting that miR-1 might negatively regulate expression of GOLPH3. Meanwhile, MTT assay and colony formation assay showed overexpression of miR-1 significantly inhibited the cell viability and proliferation, while inhibition of miR-1 led to opposite results (P < .05, Figure 4D and E). The cell-cycle analysis also showed the G-S cell-cycle arrest was remarkably induced by the overexpression of miR-1 and was significantly inhibited by miR-1 inhibitor (Figure 4F). The overexpression of miR-1 significantly downregulated the expression of GOLPH3, p-Foxo1, and p-AKT, as well as increased p27 and downregulated CyclinD1 compared to the NC cells, while the inhibition of miR-1 led to opposite results (Figure 4G). All these results indicated miR-1 might regulate cell proliferation and affect cell cycle of BC cells through the GOLPH3/Foxo1 pathway.

Overexpression of miR-1 decreased GOLPH3, inhibited cell proliferation, and induced cell-cycle arrest of BC cells. A, The predicted binding for GOLPH3 and miR-1 was obtained using bioinformatic prediction by software Targetscan 7.2. B, Luciferase activity in WT-GOLPH3 and MUT-GOLPH3 by Dual-Luciferase Reporter Assay. C, Expression of miR-1 and GOLPH3 in cells transfected with miR-1 mimic, inhibitor or NC in BC 5637, and T24 cells by RT-qPCR. D, Cell viability of BC 5637 and T24 cells transfected with miR-1 mimic, inhibitor or NC in BC 5637, and T24 cells by MTT assay. E, Cell proliferation of BC 5637 and T24 cells transfected with miR-1 mimic, inhibitor or NC in BC 5637, and T24 cells by colony formation assay. F, Cell cycle of BC 5637 and T24 cells transfected with miR-1 mimic, inhibitor or NC in BC 5637, and T24 cells by FCM. G Expression of GOLPH3, Foxo1, p-Foxo1, AKT, p-AKT, p27, and CyclinD1 in BC 5637 and T24 cells transfected with miR-1 mimic, inhibitor or NC in BC 5637, and T24 cells by Western blotting. The result was a representative of 3 independent experiments. Error bars represented mean ± standard deviation. *P < .05, **P < .01, ***P < .001. BC indicates bladder cancer; FCM, flow cytometry; GOLPH3, Golgi phosphoprotein 3; MUT, mutant; qRT-PCR, quantitative real-time polymerase chain reaction; WT, wild type.
Discussion
Despite development of surgical techniques, the prognosis of BC is still poor due to metastasis of high occurrence and molecular mechanisms for BC development are still unclear. Though several studies demonstrated role of GOLPH3 in BC, few studies focused on relationship between GOLPH3 and Foxo1 and GOLPH3 and miR-1 in BC development. In the present study, we for the first time demonstrated knockdown of GOLPH3 could inhibit cell proliferation and induce cell-cycle arrest of BC cells through regulation of Foxo1, and overexpression of miR-1 could inhibit expression of GOLPH3 by direct targeting on it, which further led to depression of cell proliferation and induction of cell-cycle arrest.
Role of GOLPH3 in cancer development, including BC, has been reported in several studies. Zhang et al 10 demonstrated the overexpression of GOLPH3 had significant correlation with poorer survival for patients with BC. It was also found GOLPH3 was increased in gemcitabine and cisplatin chemoresistant BC cell lines. 9 Meanwhile, GOLPH3 was found to overexpress in breast cancer and overexpression of GOLPH3 could significantly promote cell proliferation and tumorigenicity of breast cancer cells by regulation of Foxo1. 17 A recent research showed inhibition of GOLPH3 arrested the cell cycle at the G2/M phase. 18 All these results were consistent with our findings. In the present study, we also found GOLPH3 was overexpressed in BC cells and tissues, and knockdown of GOLPH3 inhibited cell proliferation. We also first confirmed suppression of GOLPH3 also induced cell-cycle arrest of G1-S phase in BC cells, and these effects might be through inhibition of Foxo1 phosphorylation.
Many studies have demonstrated role of Foxo1 in cancer development. Hou et al 19 found miR-196a could promote cervical cancer proliferation through downregulation of Foxo1. Haflidadóttir et al 20 showed upregulation of miR-96 also enhanced cellular proliferation of prostate cancer cells by targeting and negative regulation of Foxo1. Zeng et al 17 demonstrated overexpression of GOLPH3 could promote tumorigenicity of breast cancer cells by regulation of Foxo1. However, few other studies focused on relationship between GOLPH3 and Foxo1 in cancer progress. In our work, we for the first time showed knockdown of GOLPH3 suppressed cell proliferation of BC cells, which might be through inhibition of Foxo1 phosphorylation.
It was considered miR-1 could suppress cancer development in many cancers. Singh et al 21 showed overexpression of miR-1 decreased in vivo tumor growth in mice. It was found miR-1 was downregulated in osteosarcoma cell lines and overexpression of miR-1 significantly reduced cell proliferation and induced cell-cycle arrest in osteosarcoma cells. 22 Recently, Wang et al 14 showed the overexpression of miR-1 inhibited BC cell growth, induced apoptosis, and decreased cell motility. However, to our best knowledge, no study focused on relationship between miR-1 and GOLPH3 before. In the present study, we for the first time showed miR-1 might inhibit cell proliferation and induce cell-cycle arrest through directly targeting and negative regulation of GOLPH3.
In conclusion, the present study aimed to investigate role of miR-1/GOLPH3/Foxo1 axis in BC. Results showed that GOLPH3 could inhibit cell proliferation and induce cell-cycle arrest of BC cells through regulation of Foxo1, and overexpression of miR-1 could lead to inhibition of cell proliferation and induction of cell-cycle arrest by targeting GOLPH3. These results might give us deeper understanding for miR-1/GOLPH3/Foxo1 axis in cancer development.
Supplemental Material
Supplemental Material, FigS1 - MiR-1/GOLPH3/Foxo1 Signaling Pathway Regulates Proliferation of Bladder Cancer
Supplemental Material, FigS1 for MiR-1/GOLPH3/Foxo1 Signaling Pathway Regulates Proliferation of Bladder Cancer by Ming-Kai Liu, Tao Ma, Yang Yu, Yong Suo, Kai Li, Shi-Chao Song and Wei Zhang in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
Wei Zhang is also affiliated to Hebei Key Laboratory of Chronic Kidney Diseases and Bone Metabolism, Baoding, China.
Acknowledgments
The authors give sincere gratitude to the reviewers for their constructive comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
