Abstract
Introduction
Lung cancer is the primary cause of cancer death globally, 1 and the main histological subtype of it is lung adenocarcinoma. 2 With the universal use of high-resolution computed tomography (CT) and the increased awareness of physical examination, a growing number of patients are being identified with multiple primary lung adenocarcinomas (MPLAs).
Multiple primary lung cancer (MPLC) is defined as 2 or more primary lung cancers occurring synchronously or sequentially in the same patient. 3 Surgery may be generally considered the primary treatment for MPLC patients, but the optimal surgical approach is still up for debate. There is no consensus on whether sublobar resection (segmentectomy or wedge resection) would result in a poorer prognosis for MPLC patients than conventional resection (anatomical lobectomy or pneumonectomy).4,5
In addition, several studies have proved that the survival of patients with MPLC is related to age, gender, laterality, differentiation, and lymphatic metastases.6–8 However, there is no definitive method for predicting the prognosis of MPLA based on CT characteristics. Of note, it has been reported that CT signs can indicate the prognosis of patients with solitary lung adenocarcinoma. Therefore, other independent prognostic factors, such as CT characteristics, are thought to contribute to individualized survival prediction in MPLA patients.
In the current study, we retrospectively analyzed a group of MPLA patients and explored the survival outcomes, hoping to draw valuable conclusions for prognosis evaluation that will contribute to future studies.
Materials and Methods
This single-center retrospective study was approved by the Ethics Committee of Wuhan Union Hospital (approval number: S377), and the requirement for informed consent was waived by the committee because of the retrospective nature of this study. The reporting of this study conforms to the REporting recommendations for tumor MARKer prognostic studies (REMARK) guidelines. 9 We have removed all patient details to ensure the confidentiality of patient information.
Study Population
As AAH and AIS were adjusted from preinvasive lesions to precursor glandular lesions in the 2021 WHO Classification of Lung Tumors, 10 we excluded AAH or AIS nodules. A total of consecutively 6052 patients who underwent resections of lung adenocarcinomas were screened from January 2013 to October 2021. 5632 patients were removed due to a single lung lesion (n = 5192), tumors spread from other body sites (n = 52), no thin-section CT before surgery (n = 369), or inadequate CT image quality (n = 19); 420 patients who underwent surgical resection of at least 2 separate lung cancer foci were collected. Among them, we found 283 patients with MPLA based on the comprehensive histological assessment of Girard et al 11 (Figure 1). Patients with resection had all their nodules removed, regardless of whether they were distributed unilaterally or bilaterally.

Flow diagram shows patient selection and exclusion criteria. CT, computed tomography; CHA, comprehensive histological assessment.
Clinical and demographic characteristics, including age, gender, smoking history, types of lung resection (lobectomy, segmentectomy, and wedge resection), and adjuvant therapy, were analyzed as variables. Follow-up was performed via telephone. According to the consensus agreement in Punt et al, 12 overall survival (OS) was defined as the time from the last surgery until death from any cause, with the patients who did not die during the study period censored at the date of the last available follow-up evaluation. Recurrence-free survival (RFS) was defined as the time from surgery until recurrence/metastasis or death from any cause. The patients were followed up until February 18, 2022.
Histopathological Examinations
Two expert pathologists reviewed histologic classifications according to the World Health Organization (WHO) classification of lung cancer. 10 According to the eighth edition of the tumor–node–metastasis classification, 13 MPLC was classified separately, each with a T, N, and M descriptor. The TNM stage of MPLC was considered the highest stage among different lesions.
Computed Tomography Image Acquisition
Computed tomography examinations were obtained by a multislice spiral CT system (SOMATOM Definition AS +, Siemens Healthineers). Imaging parameters were as follows: detector collimation width, 64 × 0.6 mm and 128 × 0.6 mm; tube voltage, 120 kV; reconstructed slice thickness and interval 1.5 mm/1.5 mm. An automatic exposure control system (CARE Dose 4D) was used for regulating the tube current. The reconstructed image was then transmitted to the workstation and picture archiving and communication systems for multiplanar reconstruction postprocessing (lung window setting: level, −600 HU and width, 1200 HU; mediastinal window setting: level, 50 HU and width, 350 HU).
Computed Tomography Image Interpretation
Two experienced radiologists (with 35 and 4 years of experience in interpreting CT images, respectively) evaluated imaging data independently. Computed tomography features, including lesion number, tumor density (pure ground-glass opacity [pGGO], mixed ground-glass opacity [mGGO], and solid), maximum tumor diameter on the lung window, homogeneity, margin (well-defined or ill-defined), pleural indentation, spiculation, calcification, vacuole sign, air bronchogram, vascular convergence, and cavitation were used as predictive variables. Any discrepancies between the radiologists were solved through discussion.
Computed Tomography Features of Lung Adenocarcinoma
On thin-section CT, pGGO tumors meant tumors with no solid component, mGGO tumors meant tumors with a combination of GGO and solid component, and solid tumors meant tumors with only consolidation. Homogeneity meant that the difference in CT values in the tumor was less than 20 HU. The margin was defined as well-defined or ill-defined. Pleural indentation referred to the retraction of the pleura toward the tumor. Spiculation was characterized by linear fibrotic strands radiating into the surrounding lung parenchyma. A vacuole sign referred to air attenuation spots within the tumor. Air bronchogram meant the presence of air-containing bronchi in the tumor. Vascular convergence was presented as the convergence of vessels surrounding the tumor. Cavitation was defined as air space within the tumor with a thick wall.
Grouping Criteria
According to the combination of tumor density, the patients were divided into 3 groups: multi-pure ground-glass nodules (multi-pGGN), at least one part-solid nodule without solid nodules (multi-GGN), and at least one solid nodule (solid-GGN). When a tumor had different types of lung resection, we chose a more extensive resection to represent it. In addition, we separated patients into 2 groups based on the number of tumors (ie, <3 and ≥3). Pathologically, patients were divided into 3 groups according to the WHO classification of lung cancer 10 : minimally invasive adenocarcinoma (MIA) group, mixed group, and invasive adenocarcinoma group. The MIA group was defined as all tumors were MIA. The mixed group consisted of tumors with both MIA and invasive adenocarcinoma. The invasive adenocarcinoma group meant all tumors were invasive adenocarcinoma. Categorization of continuous variables followed clinical relevance: tumor size cutoff was based on T-designation, 13 and age was dichotomized near the median age of patients. Furthermore, to investigate other features of MPLA, we recorded 0 when no tumor had the sign and 1 when at least one tumor had the sign.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics 26 (IBM Inc.). The patients were characterized by clinicopathological and radiological variables. Differences among the groups were evaluated using χ2 tests for categorical variables and 1-way analysis of variance tests for continuous variables. Overall survival and RFS were calculated using the Kaplan-Meier method. Subgroup comparisons of survival curves were determined by the log-rank test. Overall survival and RFS were performed by univariate and multivariate analyses using the Cox proportional hazard regression model. For variables with >2 levels (pathological, density, and TNM stage), we set invasive adenocarcinoma, Solid-GGN, and III as the references, respectively. The repeatability test of tumor diameter was analyzed using the intraclass correlation coefficient (ICC) with a 95% confidence interval (CI). For other CT signs, interobserver agreement was assessed with the k coefficient. A difference was considered statistically significant when the P-value was less than .05.
Results
During the study period, 6052 patients with lung adenocarcinomas underwent surgical resection. Multiple primary adenocarcinomas were present in 283 patients (4.7%) with 623 lesions. Women comprised 66.8% of the cohort. The median patient age in those with MPLA was 58 years (age range, 26-77 years).
Surgical methods included wedge resection, segmentectomy, and lobectomy, and the combination of the surgical type and tumor location are detailed in Table 1. The CT findings of all 623 lesions were analyzed (Table 2). Computed tomography primarily indicated pGGN (48.5%), ≤3 cm (93.9%), heterogeneity (53.9%), well-defined margin (50.5%), absence of spiculation sign (92.9%), pleural indentation (62.2%), calcification (99.3%), vacuole (82.9%), air bronchogram (84.1%), vascular convergence (88.6%), and cavity (97.8%).
Surgical Type and Tumor Location in 283 Cases of MPLA (623 Lesions).
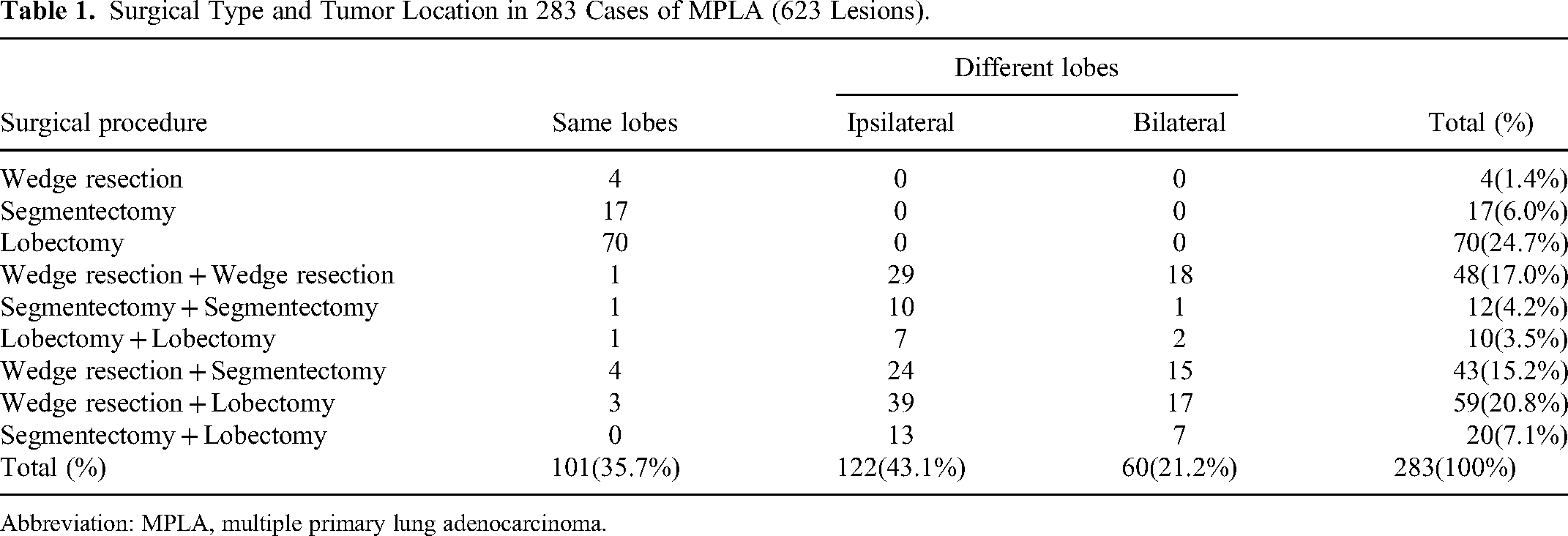
Abbreviation: MPLA, multiple primary lung adenocarcinoma.
Computed Tomography Findings in 283 Cases of MPLA (623 Lesions).

Abbreviations: MPLA, multiple primary lung adenocarcinoma; pGGN, pure ground-glass nodules.
The patient clinicopathological and radiological demographic characteristics based on the tumor density and lesion number are summarized in Tables 3 and 4, respectively. Representative cases are shown in Figure 2.
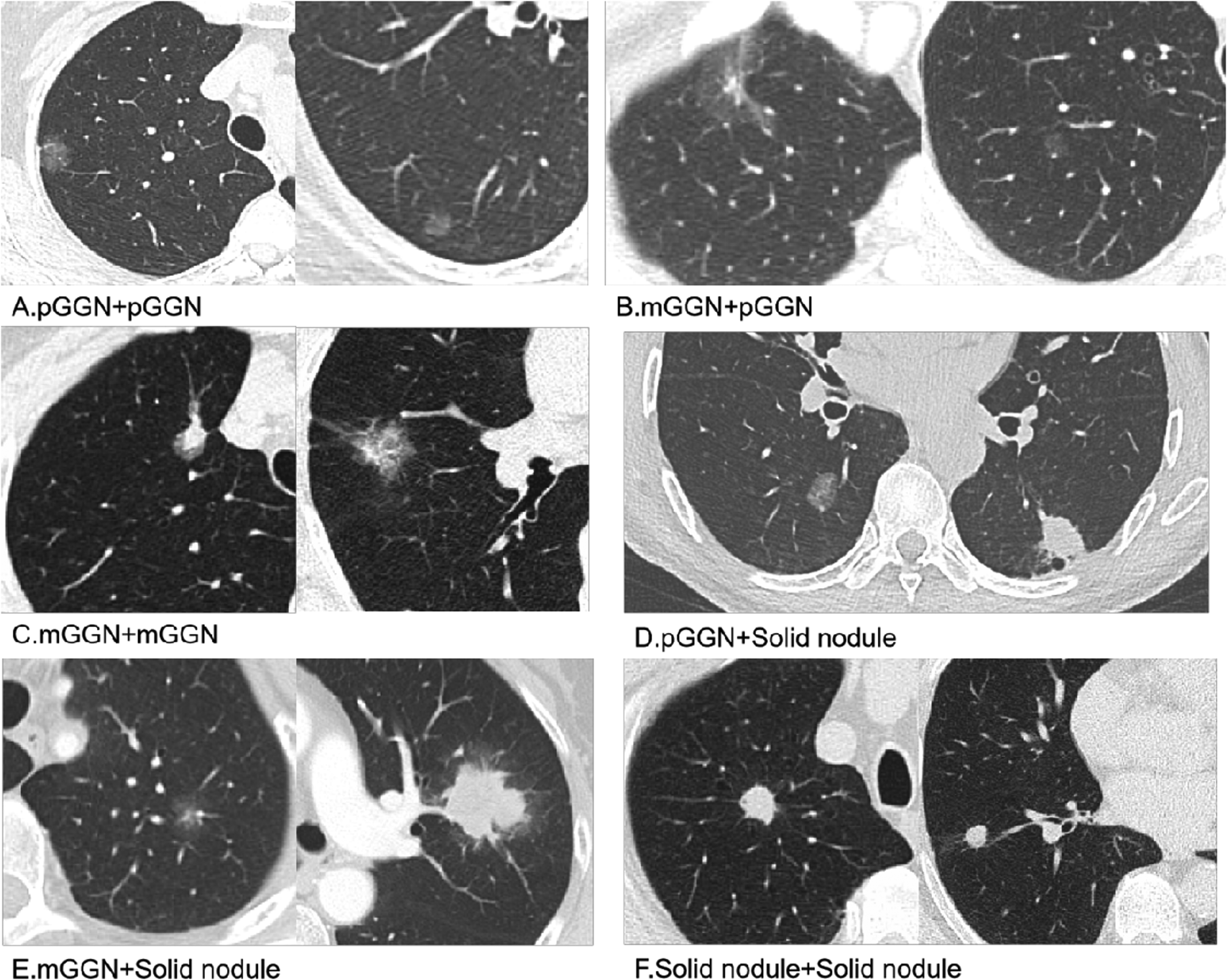
Radiological classification of multiple primary lung adenocarcinomas based on the tumor density on thin-section computed tomography (CT). It demonstrated typical radiological findings of multi-pGGN (A), multi-GGN (B and C), and solid-GGN (D-F). pGGN, pure ground-glass nodule; mGGN, mixed ground-glass nodule; multi-pGGN, multi-pure ground-glass nodule; multi-GGN, at least one part-solid nodule without solid nodule; solid-GGN, at least one solid nodule.
Clinicopathological and Radiological Characteristics of Patients With Multiple Primary Lung Adenocarcinoma, Classified by the Tumor Density.
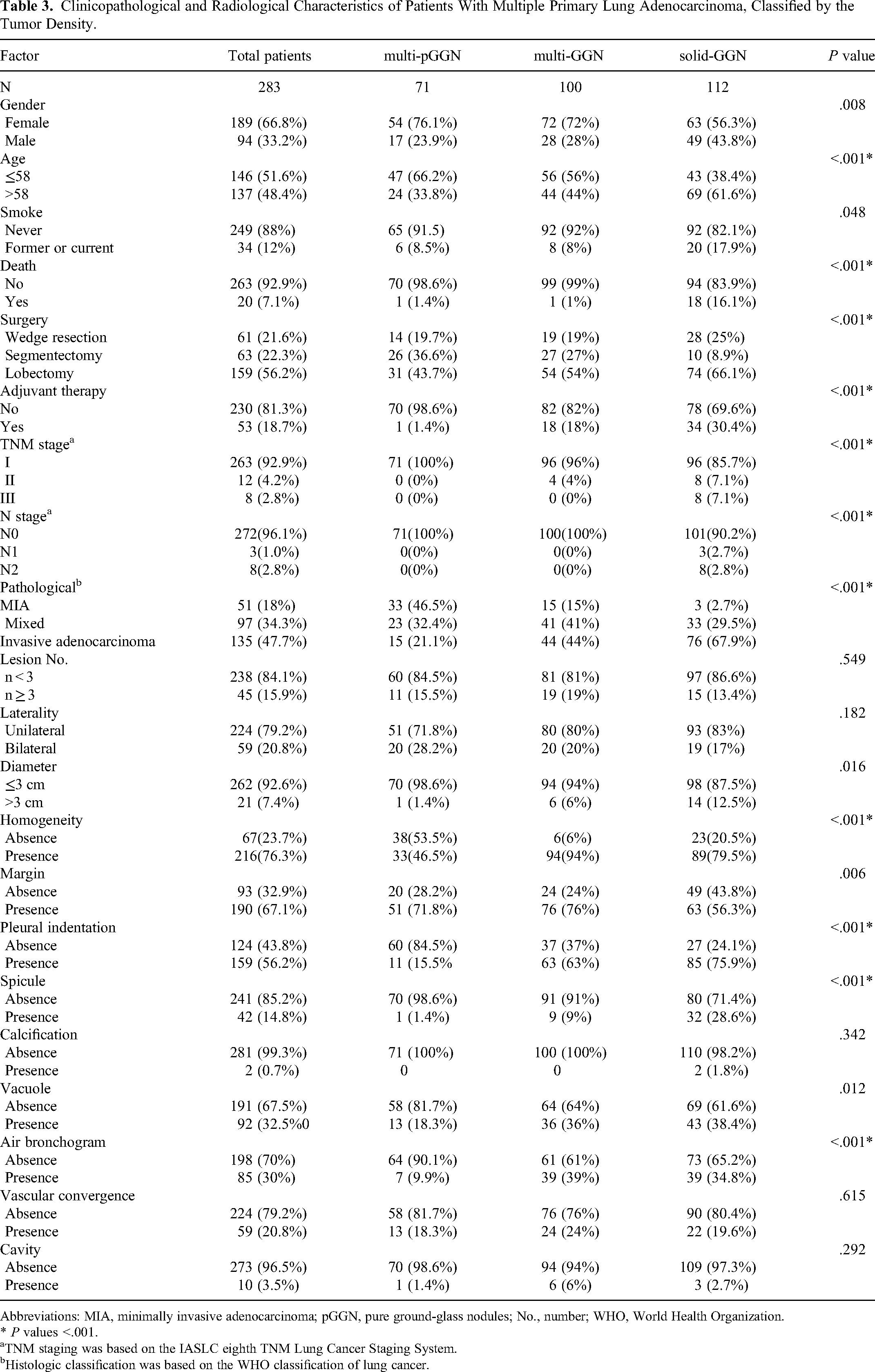
Abbreviations: MIA, minimally invasive adenocarcinoma; pGGN, pure ground-glass nodules; No., number; WHO, World Health Organization.
* P values <.001.
TNM staging was based on the IASLC eighth TNM Lung Cancer Staging System.
Histologic classification was based on the WHO classification of lung cancer.
Clinicopathological and Radiological Characteristics of Patients With Multiple Primary Lung Adenocarcinoma, Classified by the Number of Tumors.

Abbreviations: MIA, minimally invasive adenocarcinoma; pGGN, pure ground-glass nodules; WHO, World Health Organization.
* P values <.001.
TNM staging was based on the IASLC eighth TNM Lung Cancer Staging System.
Histologic classification was based on the WHO classification of lung cancer.
Clinicopathologic Demographics of MPLA Patients
Of 283 patients, 71 (25.1%) presented as multi-pGGN, 100 (35.3%) as multi-GGN, and 112 (39.6%) as solid-GGN. The solid-GGN group had a more significant number of males (P = .008). People over the age of 58 (P < .001), with a smoking history (P = .048), and people who underwent lobectomy (P < .001) could also be seen more in the solid-GGN group. Furthermore, 30.4% (34 of 112) of patients in the solid-GGN group underwent adjuvant therapy, and only one person was in the multi-pGGN group. Pathologically, the solid-GGN group showed higher rates of invasive adenocarcinoma (P < .001). In contrast, those of the multi-pGGN group varied, and nearly half of the patients had MIA. In the TNM stage, only the solid-GGN group contained patients in stage III, whereas all patients in the multi-pGGN group were in stage I (P < .001). In addition, patients with fewer than 3 tumors predominated (238/283, 84.1%), whereas 15.9% had 3 or more tumors. Bilateral distribution was more frequently observed (P = .029) among patients with 3 or more tumors.
Interobserver Agreement in CT Interpretation
The ICC for tumor diameter was 0.982 (95% CI: 0.973-0.987). Regarding other CT features, the concordance between the 2 observers was good, with k coefficients ranging between 0.485 and 0.848 (Table 5).
Analysis of Interreader Agreement Percent of Concordance and Kappa of Agreement.
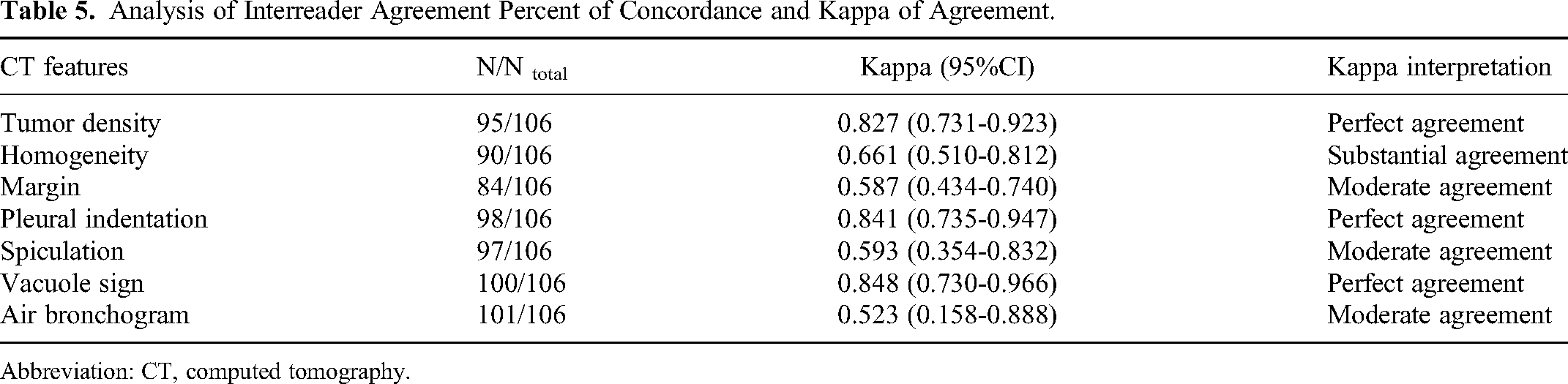
Abbreviation: CT, computed tomography.
Computed Tomography Features of MPLA
The number of resected tumors among the 3 groups based on tumor density did not differ statistically (P = .549). However, the size of the tumors varied. Tumors were larger in the solid-GGN group than in the other groups (P = .016). Homogeneity (6 of 100; 6%, P < .001) was uncommon in multi-GGN, and the margin (76 of 100; 76%, P = .006) of the group was mainly ill-defined. Pleural indentation (15.5% vs 63% vs 75.9%; P < .001), spicule (1.4% vs 9% vs 28.6%; P < .001), and vacuole sign (18.3% vs 36% vs 38.4%; P = .012) were more prevalent in the solid-GGN group, while air bronchogram(P < .001) could be seen more in the multi-GGN group.
Univariate and Multivariate Analyses
The median follow-up period for all 283 patients was 15 months (1-87), and the lost follow-up rate was 1.8% (5/283); 14 patients (4.9%) experienced recurrence or distant metastases, and 20 (7.1%) had died by the end of the follow-up period. Overall survival at 2 and 3 years was 93.0% and 76.8%, respectively, with a median cumulative OS time of 63 months. Recurrence-free survival at 2 and 3 years was 86.0%% and 65.1%, respectively. The median cumulative RFS time was 51 months.
We investigated the significant clinicopathological and radiological factors that affected OS in patients with MPLA. In the univariate analysis, OS was significantly related to adjuvant treatment (P = .047), stage III (P = .01), lesion number (P < .001), and solid-GGN (P = .016). No association was found between OS and other features. On multivariate analysis, no variable showed multicollinearity on VIF analysis; thus, no variable was removed from the multivariate analysis for that reason. The multivariate analysis revealed that the radiological solid-GGN (hazard ratio [HR], 5.307; 95% CI 1.159-24.310; P = .032) and the lesion number (HR, 4.76; 95% CI 1.88-12.03; P = .001) were independently significant predictive variables for survival as shown in Table 6.
Univariate and Multivariate Analysis for Overall Survival in 283 Patients With Multiple Primary Lung Adenocarcinoma.

Abbreviations: HR, hazard ratio; CI, confidence interval; MIA, minimally invasive adenocarcinoma; pGGN, pure ground glass nodules; No., number; WHO, World Health Organization.
* P value in Cox proportional hazard model.
TNM staging was based on the IASLC eighth TNM Lung Cancer Staging System.
Histologic classification was based on the WHO classification of lung cancer.
Table 7 summarizes the univariate and multivariate analyses of the association of clinicopathologic and CT factors with RFS. In the univariate analysis, RFS was significantly linked with stage I (P = .017), stage III (P = .007), adjuvant therapy (P = .001), the lesion number (P = .003), solid-GGN (P = .024), maximal tumor size (P = .014), invasive adenocarcinoma (P = .043), and tumor homogeneity (P = .044). No relationship was found between RFS and other features. The multivariate analysis showed that the lesion number (HR, 2.41; 95% CI 1.12-5.19; P = .025), stage III (HR, 5.71; 95% CI 1.94-16.81; P = .002), and adjuvant therapy (HR, 2.52; 95% CI 1.24-5.13; P = .011) influenced the RFS.
Univariate and Multivariate Analysis for Recurrence-Free Survival in 283 Patients With Multiple Primary Lung Adenocarcinoma.
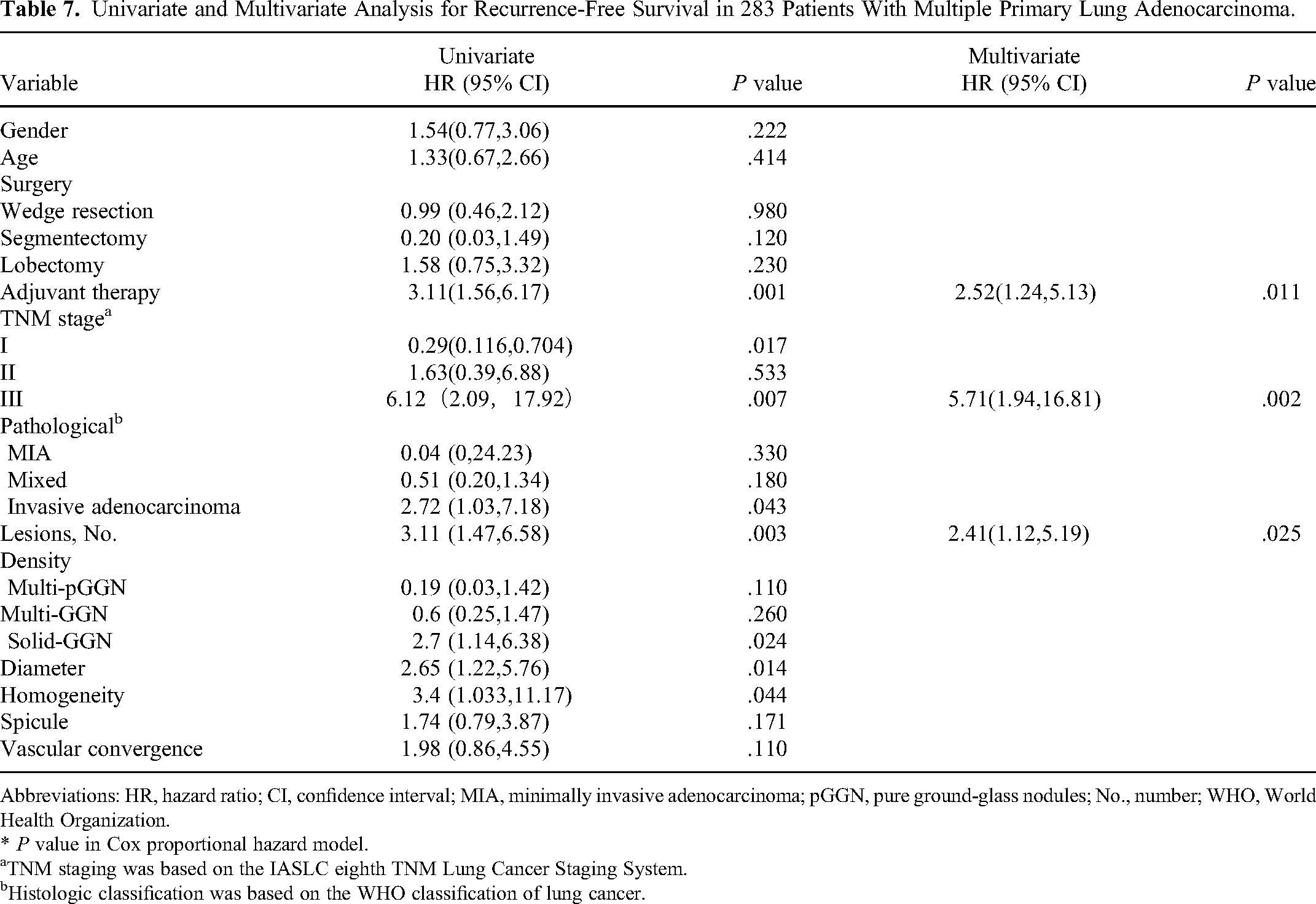
Abbreviations: HR, hazard ratio; CI, confidence interval; MIA, minimally invasive adenocarcinoma; pGGN, pure ground-glass nodules; No., number; WHO, World Health Organization.
* P value in Cox proportional hazard model.
TNM staging was based on the IASLC eighth TNM Lung Cancer Staging System.
Histologic classification was based on the WHO classification of lung cancer.
Figure 3A and B illustrated the OS and RFS of MPLA based on tumor density. The median OS time for solid-GGN was 63 months but could not be estimated for multi-pGGN and multi-GGN. The survival rates at 2 and 3 years were 93.7% and 71.8% for solid-GGN and 99.0% and 88.2% for other groups. The median RFS time for solid-GGN and multi-GGN was 50 months and 51 months, respectively, but it could not be estimated for multi-pGGN.
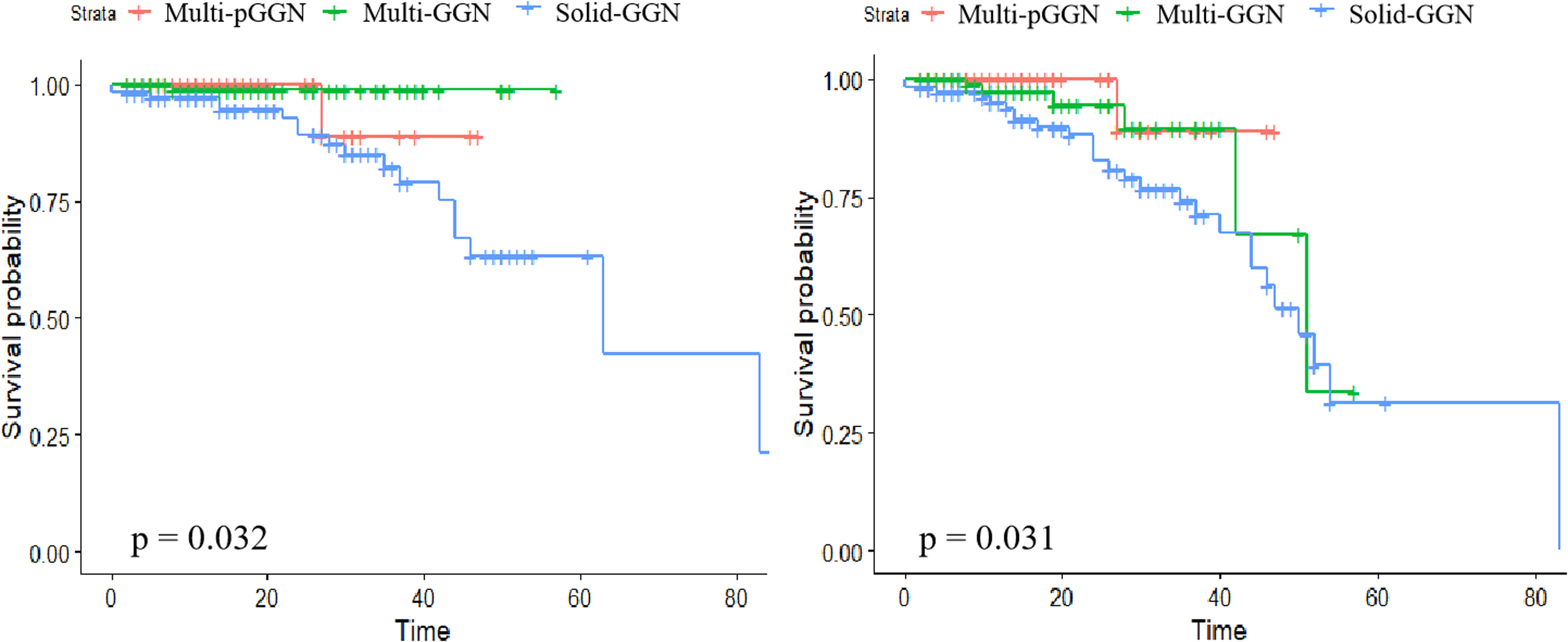
Kaplan-Meier estimates for (A) overall survival and (B) recurrence-free survival of patients, stratified by tumor density.
Figure 4A and B illustrated the OS and RFS of MPLA according to the lesion number. The OS rates for the group with 3 or more tumors at 2 and 3 years were 78.1% and 50%, and those with fewer than 3 tumors were 96.7% and 80%, with median OS time of 83 months. The median RFS time for the group with 3 or more tumors and those with fewer than 3 tumors was 42 months and 52 months, respectively.

Kaplan-Meier estimates for (A) overall survival and (B) recurrence-free survival of patients, stratified by the lesion number.
Discussion
The current study was performed to evaluate the prognostic impact of MPLA. To this end, we evaluated the clinicopathologic features, CT signs, and survival outcomes (OS and RFS) obtained from 283 patients who underwent surgery. Finally, we found that the lesion number was an independent predictor for both RFS and OS, and the solid-GGN was an independent predictor for OS.
In our series, the incidence rate of MPLA was 4.7%, in line with previous studies from 3.7% to 8.0%.8,14–16 Since the lost follow-up rate (5/283, 1.8%) was small, the impact on our results was insignificant. Most patients with MPLA were women and nonsmokers, which were also the characteristics of adenocarcinoma. Patients in stage I with no nodal metastases accounted for most of our study. Overall survival at 2 years had been reported from 61.6% to 89.7% in MPLC.6,15 In this study, patients with MPLA had a slightly higher 2-year OS. Some reasons could explain this finding: First, more than half of the patients were classified as MIA or MIA with invasive adenocarcinoma. Second, most tumors were at the early stage without lymph nodes and distant metastasis. Thus, the prognosis of MPLA was favorable.
It has been reported that there are many risk factors related to the prognosis of MPLC, while the influence of these factors remains controversial. Previous studies7,16 have shown that adjuvant chemotherapy might be an independent predictor of improved survival. Staging is considered a reliable indicator of the prognosis in patients with lung cancer. 17 In our study, stage III and adjuvant therapy were risk factors for RFS in MPLA but not for OS. This may be due to the limited follow-up time. Studies18–22 have also reported inferior oncologic outcomes for segmentectomy compared to lobectomy, and wedge or segmental resection is thought to increase local recurrence. These situations seemed inappropriate to us. We found that the type of resection had no significant impact on survival, consistent with the findings of Tamburini et al. 23 This may be because we investigated the impact of surgical type on prognosis without considering the tumor diameter and location simultaneously.
Among CT features, tumor density is one of the most important factors for predicting survival in solitary lung cancer. 24 In recent studies, this phenomenon can also be observed in multiple lung cancers.25,26 Hattori et al 25 classified tumors into 3 groups based on the extent of GGO and then divided patients with multiple lung cancers into 6 groups. They discovered that pure-solid (PS) + PS group was associated with poor survival. Our study provided strong evidence that the solid-GGN was an unfavorable prognostic factor for MPLA by both univariate and multivariate analyses. One possible explanation may be that the solid-GGN more frequently expressed invasive lung adenocarcinoma and had a higher rate of nodal metastasis than other groups, suggesting an aggressive feature. Additionally, the subtypes of solid tumors were always micropapillary and solid in pathological, 27 associated with the poorest differentiation. These results emphasize that solid-GGN may be an essential risk prognostic factor for MPLA.
In addition, multivariate analysis revealed that the lesion number was also associated with an unfavorable prognosis. In our study, patients with 3 or more tumors had worse OS rates than those with fewer than 3. Yu et al28,29 showed that the 5-year survival for single non-small cell lung cancer was higher than for synchronous lung cancer. Chen et al 30 also found that total nodule number is an independent prognostic factor in resected stage III non-small cell lung cancer. Participants with a lower total nodule number had significantly improved OS than those with higher ones. This implied that the tumor number might visually represent tumor burden in patients and could lead to a poor prognosis. On the other hand, a more significant number of tumors had a higher probability of bilateral distribution. Removing bilateral lung tumors may harm the human body, conferring a worse prognosis. 31
Several studies32–34 have demonstrated that tumor size was one of the most important predictors of survival in patients with MPLC, which concluded that smaller tumor sizes were related to better survival for MPLC. However, our results showed no statistical association between tumor size and patient survival. The reason may be that most tumors in our cohort were at an early stage, so tumors with large sizes were not included.
Several limitations in this study need to be addressed. First, this analysis was based on a small sample from a single institution. Second, only patients who underwent surgery resected were enrolled, which caused selection and information biases inherent. Third, the short follow-up period hampered the OS analysis and limited the validity of the survival analysis. Thus, a larger cohort with a more extended follow-up period is needed. Fourth, as this was a retrospective study, we could not consider tumor diameter and location while discussing the surgical type. We also could not have all patients undergo genotyped testing for comparative analysis with CHA.
Conclusion
In conclusion, this study presented a comprehensive analysis of clinicopathologic characteristics, surgical treatment, CT features, and prognosis of MPLA in patients. Several unique characteristics of patients with MPLA may be used to predict survival. Having solid nodules and lesions ≥3 portend an unfavorable prognosis. This information may be helpful for the prediction of survival and clinical decision-making in future studies.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank all colleagues for helping us during the current study. The authors are also very grateful for all the selfless volunteers who participated in the study.
Authors’ Note
This study was approved by the Ethics Committee of Wuhan Union Hospital (approval number: S377), and the requirement for informed consent was waived by the committee because of the retrospective nature of this study. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
