Abstract
Introduction
Lung cancer is the second most frequently diagnosed cancer type and has the highest mortality in the world, 1 with non-small-cell lung cancer (NSCLC) accounting for 85% of all lung cancer cases. 2 In 2020, according to statistics from 185 countries, it was estimated that the number of diagnosed cases was approximately 2 206 771 (accounting for 11.4% of all cancers), and the number of deaths was 1 796 144 (18.0%), 3 with a 5-year relative survival rate of only 22%. 4 The high mortality rate of lung cancer is related to the high malignancy and late diagnosis of the tumor. More than 60% of lung cancer patients are in the locally advanced stage (stage III) or have metastases (stage IV) at the time of treatment initiation. 5 The 5-year relative survival rate in stage I patients is 57%, while that in stage IV patients drops to 4%. 6 Advanced patients urgently need precise and effective treatments to improve their prognosis.
Although concurrent or sequential chemotherapy with radiotherapy has been a conventional treatment plan for inoperable NSCLC, 7 radiotherapy and chemotherapy have not yet entered the era of precision medicine. In different patients, the sensitivity of tumor cells to treatment and the efficacy of radiotherapy are different. 8 Some patients experience no obvious benefit after receiving radiotherapy. The tumor may still progress locally or even metastasize to distant organs. Some patients may also suffer additional radiotherapy-related adverse reactions (radiation pneumonitis, radiation esophagitis, bone marrow suppression, etc) due to exposure of normal tissues to radiation, resulting in unnecessary burden of medical expenses. At present, response evaluation criteria in solid tumors (RECIST) standards are mostly used in clinics for efficacy evaluation 9 ; this method compares and classifies the changes in tumor and lymph node diameters on computed tomography (CT) images before and after treatment. However, the response of NSCLC tumors to radiotherapy and chemotherapy may be slow, 10 resulting in a delay in the evaluation of efficacy. Furthermore, clinically, there is still a lack of early effective treatment sensitivity predictors to help clinicians evaluate the treatment response of the tumor before radiotherapy and adjust the treatment plan accordingly (such as enhanced chemotherapy or combined targeted or immunotherapy, etc) to improve patient prognosis.
Radiomics is an image analysis technique that enables quantitatively extracted image features from traditional medical images. Radiomics methods show strong predictive performance in the diagnosis and treatment of lung cancer. At present, radiomics methods have been employed to address various questions in the field of lung cancer, especially in NSCLC, including diagnosis and identification,11,12 staging, pathological typing, 13 degree of differentiation, genotyping,14,15 selection of treatment options, toxic side effects, 16 and prognostic evaluation.17,18 The process mainly includes data collection, image segmentation, feature extraction, feature selection, and model construction. 19 In the diagnosis and treatment of lung cancer patients, CT is the preferred imaging examination method, and it is also the most widely used method in radiomics research. As lung tumors present a strong contrast in CT images, including differences in gray value intensity, texture differences, and shape differences of tumors in the image, it can provide guidance for clinical diagnosis and treatment. However, there have only been a few studies on radiosensitivity of NSCLC using CT-based radiomics until now.20,21 In previous studies using radiomics models to predict radiotherapy sensitivity, one study has explored the predictive performance of radiomics models and confirmed that they can accurately predict radiotherapy sensitivity. 21 However, that study lacked the discussion of clinical factors involved. A number of studies have confirmed that the combination of radiomic features (such as intensity, shape, texture, or wavelet) extracted from medical images with clinical parameters can make clinical decision-making more accurate. 22 Therefore, we aimed to develop and validate a prognostic model based on CT radiomics combined with clinical features to predict the radiotherapy sensitivity in patients with inoperable stage III and IV NSCLC.
Materials and Methods
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Kunming Medical University in Yunnan Province (approval number: KYLX202175). Informed consent was waived by the committee because of the retrospective nature of this study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 23 We have de-identified all patient details to ensure the confidentiality of patient information.
Our main study endpoint was the patient's response to radiotherapy. According to the RECIST v1.1 evaluation standard, 9 the local response of the lesion was evaluated as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) by comparing CT images before and after radiotherapy. The radiation-sensitive group was defined as CR + PR, and the non-sensitive group was defined as SD + PD. Model predictive power was evaluated using the AUC.
Overall survival (OS) was defined as the time from the patient receiving radiotherapy to death due to any reason (for subjects who have been lost to follow-up before death, the time of the last medical record was calculated as the time of death).
Results
Baseline Characteristics
A total of 104 patients with NSCLC were included in this study. The clinical information of the patients is shown in Table 1. The overall average age was 57.35 ± 9.554 (range: 31-80) years old, including 89 males (85.6%) and 15 females (14.4%). There were 67 cases (64.4%) of squamous cell carcinoma and 37 cases (35.6%) of adenocarcinoma. More than 70% of patients had a history of smoking. Among them, those in the training set and the test set had no significant differences in clinical baseline characteristics (sex, age, pathological type, smoking history, family history, clinical stage, etc) (Table 1).
Baseline Patient Characteristics.
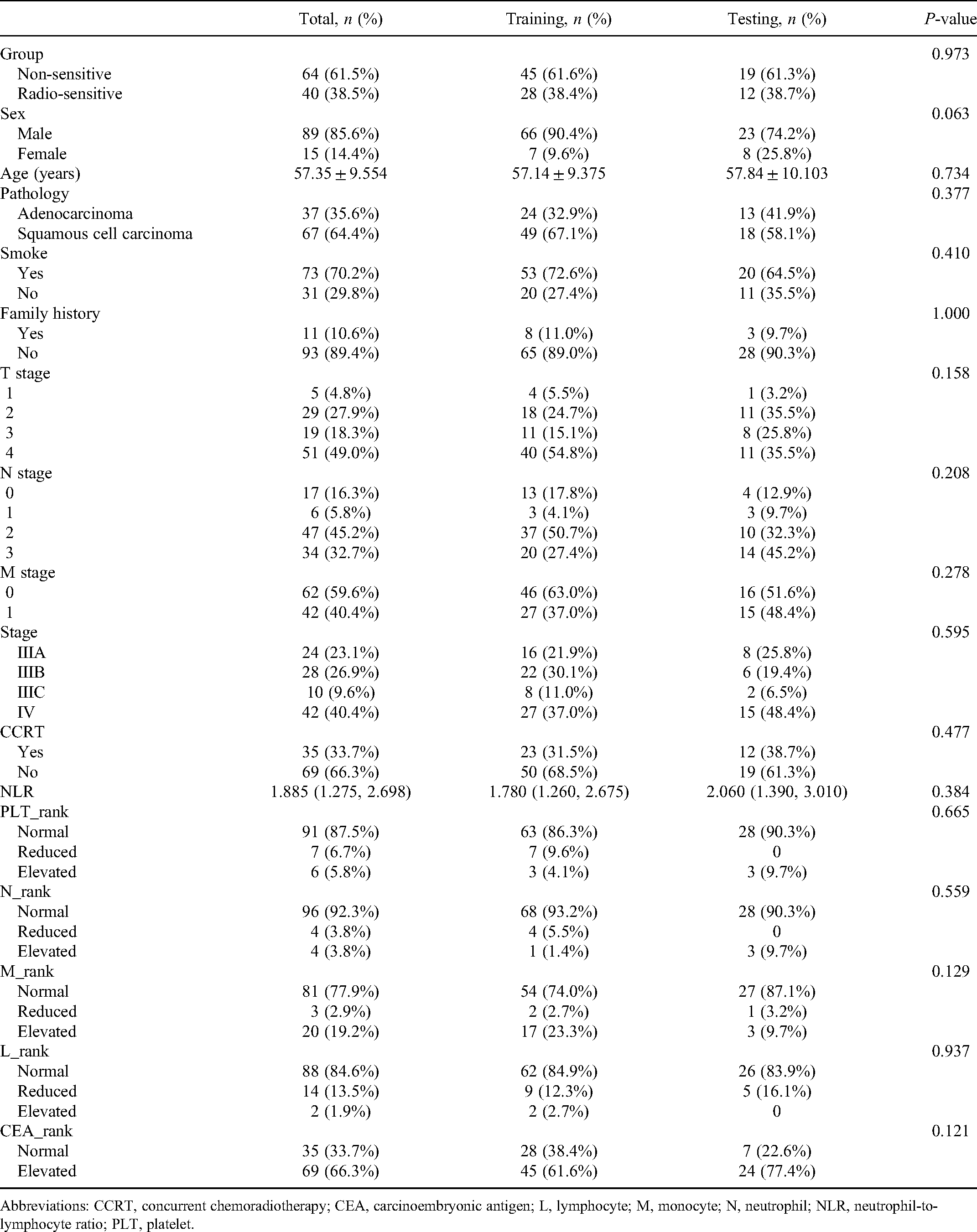
Abbreviations: CCRT, concurrent chemoradiotherapy; CEA, carcinoembryonic antigen; L, lymphocyte; M, monocyte; N, neutrophil; NLR, neutrophil-to-lymphocyte ratio; PLT, platelet.
Radiomics Features
A total of 2460 radiomic features (1228 and 1232 from lung and mediastinal windows, respectively) were extracted from the ROI of each lesion, including first-order features, shape features, and textural features (such as gray-level co-occurrence matrix [GLCM], gray-level size zone matrix [GLSZM], gray-level run length matrix [GLRLM], neighboring gray tone difference matrix [NGTDM], and gray-level dependence matrix [GLDM]).
The performance of the radiomics model based on the 9 radiomics features for predicting radiosensitivity is shown in Table 2. The accuracy of the model in the training, testing, and cross-validation sets was 0.822, 0.871, and 0.740, respectively, and the AUC values were 0.849 (95% confidence interval [CI]: 0.755-0.931), 0.864 (95% CI: 0.683-0.996), and 0.779 (95% CI: 0.655-0.878), respectively. The corresponding ROC curves are shown in Figure 1. The Rad-Score was calculated based on the selected features and their coefficients (Supplemental Appendix A: Rad-Score, Figure 3), and the waterfall chart reflects the performance of the Rad-Score in the prediction (Figure 4).
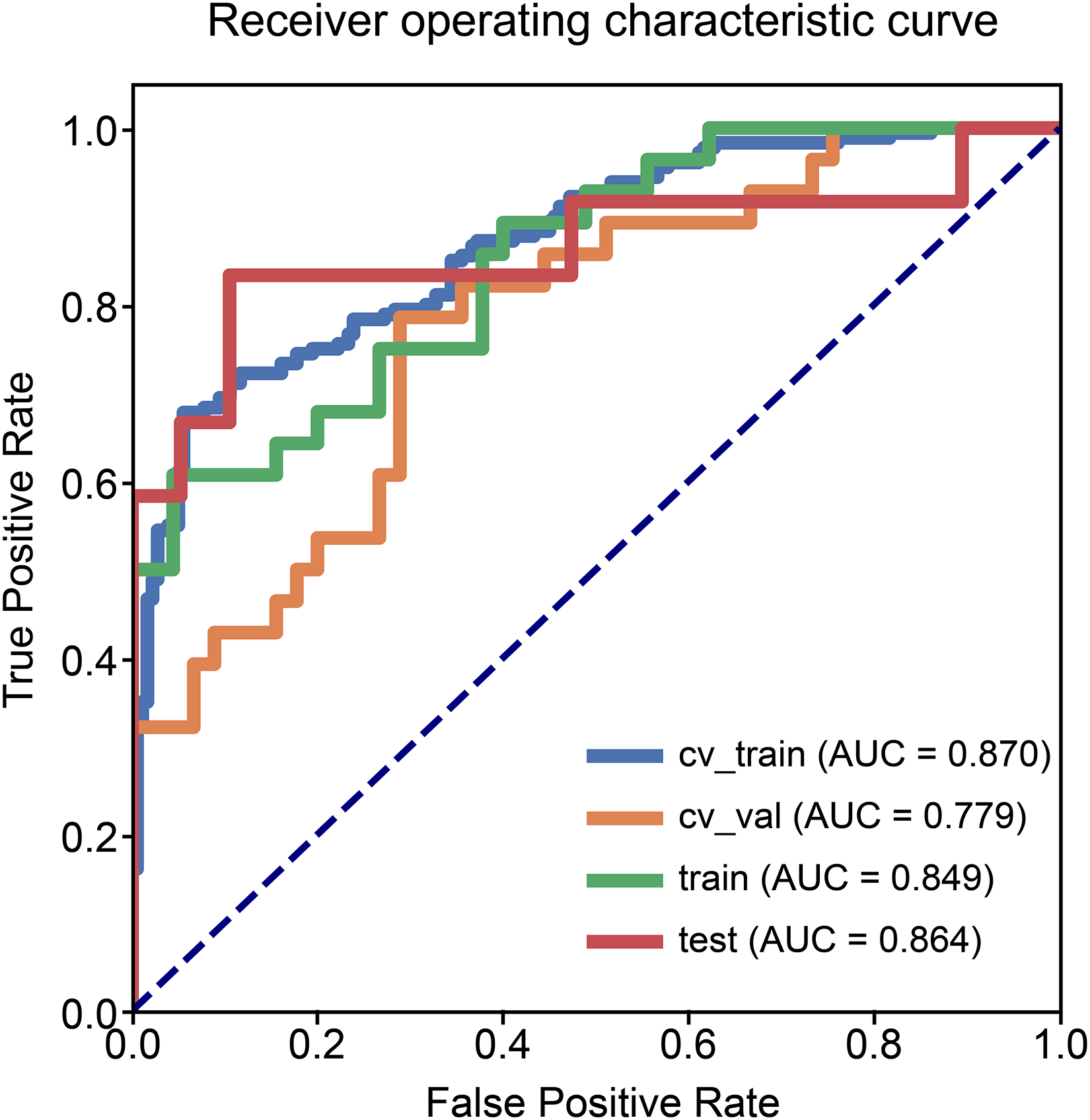
Receiver operating characteristic curves of the radiomics model.

Receiver operating characteristic curves of the combined model.

Nine features selected are presented. The coefficient indicates the feature's contribution to the model.
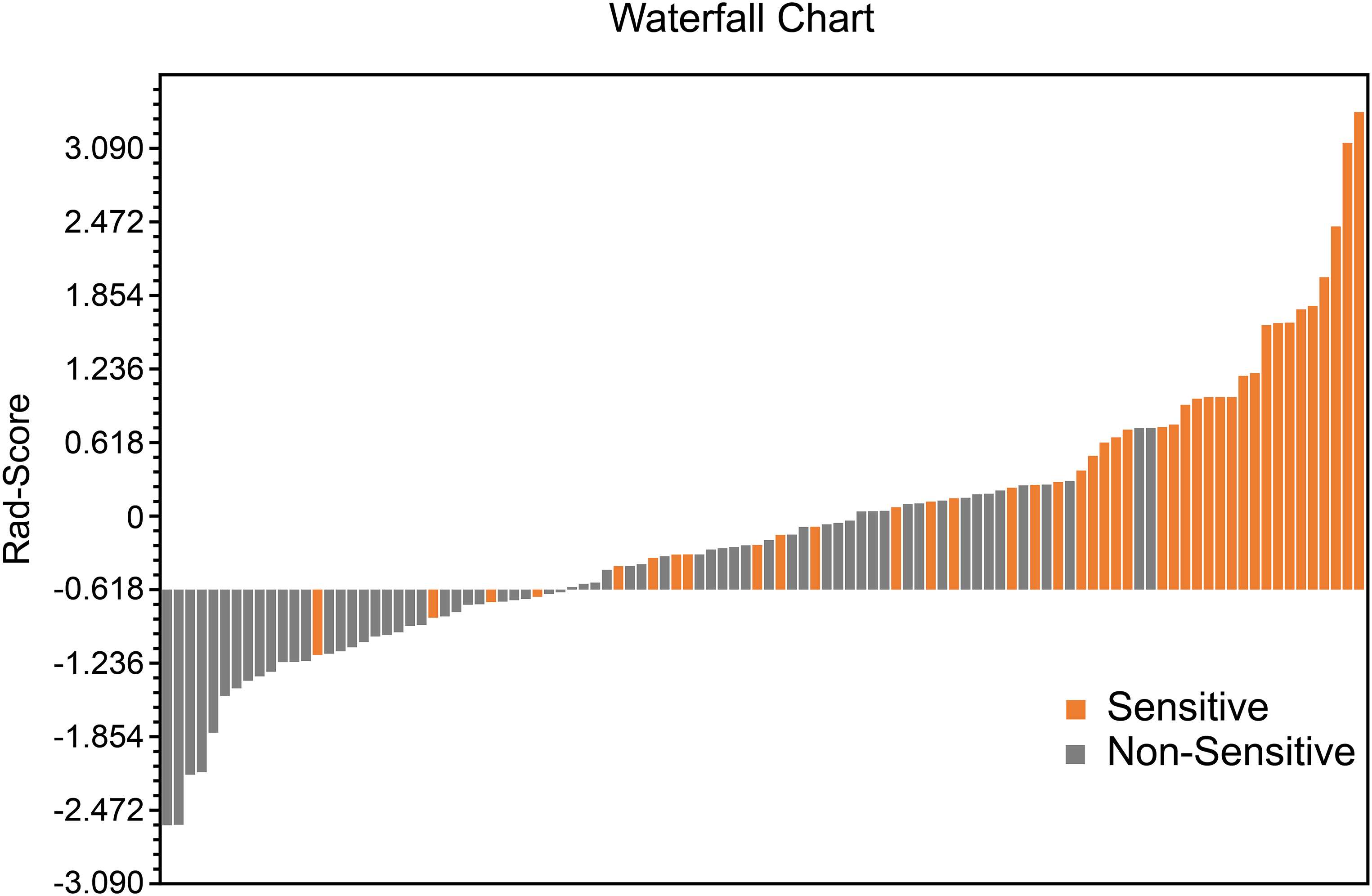
Waterfall chart reflects the performance of the Rad-Score in the prediction of radiotherapy sensitivity.
Statistics in the Prediction.
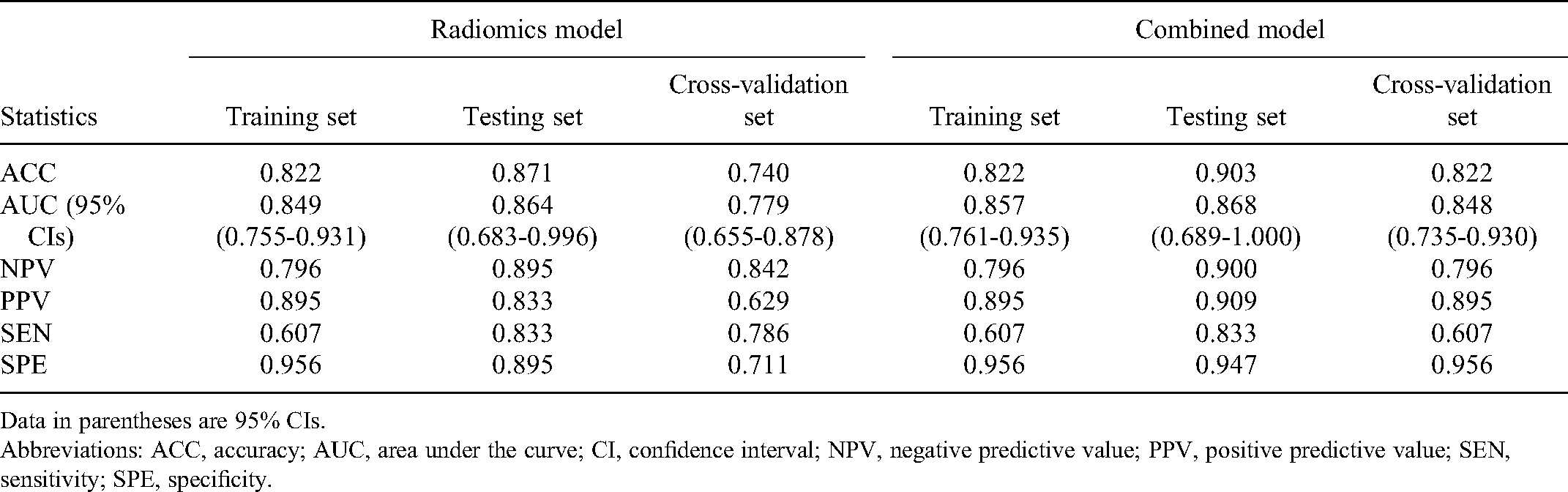
Data in parentheses are 95% CIs.
Abbreviations: ACC, accuracy; AUC, area under the curve; CI, confidence interval; NPV, negative predictive value; PPV, positive predictive value; SEN, sensitivity; SPE, specificity.
Clinical Features
In the univariate analysis of clinical baseline characteristics, we identified several clinical characteristics related to radiotherapy sensitivity: smoking history, clinical stage, concurrent chemoradiotherapy (CCRT), and lymphocyte level status. Among the clinical characteristics, the P-value of the pathological type was greater than 0.15. However, in the actual theory, different types of cells had different responses to treatment; therefore, the pathological types were included in the multivariate logistic regression for analysis. In the multivariate analysis, only the clinical factor of CCRT showed significant differences between the radiation-sensitive and non-sensitive groups (Table 3).
Univariate and Multivariate Analyses of Factors Associated with Radiotherapy Sensitivity.

Data in parentheses are 95% CIs.
Abbreviations: CCRT, concurrent chemoradiotherapy; CEA, carcinoembryonic antigen; CI, confidence interval; L, lymphocyte; M, monocyte; N, neutrophil; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio; PLT, platelet.
Combined Model
As shown in Figure 2 and Table 2, the combined model based on Rad-Score and CCRT had higher AUC values in the training (AUC = 0.857; 95% CI, 0.761-0.935), testing (AUC = 0.868; 95% CI, 0.689-1.000), and cross-validation sets (AUC = 0.848, 95% CI, 0.735-0.930). The accuracy of the testing set and cross-validation set increased to 0.903 and 0.822, respectively (Table 2). In addition, the sensitivity of the cross-validation set of the combined model was reduced compared with the radiomics model, but the specificity of the testing set and the cross-validation set was improved. The calibration plot revealed good predictive accuracy between the actual probability and prediction probability (Supplementary Figure 1). Decision curve analysis (DCA) further demonstrated higher overall net benefit for the combined model compared with the radiomic model (Figures 5 to 7).

The decision curve analyses of the radiomic model and combined model in the training cohort.

The decision curve analyses of the radiomic model and combined model in the testing cohort.

The decision curve analyses of the radiomic model and combined model in the cross-validation cohort.
Survival Analysis Related with Each Factor
In the entire cohort, the median follow-up was 17.0 months (range: 2.0-52.0 months). Univariate and multivariate Cox regression analyses showed that neither concurrent chemotherapy nor Rad-Score had a significant correlation with OS. Univariate analysis revealed that platelet level status was significantly associated with OS (X² = 7.095, P = 0.029). The median survival times in the patients with reduced, normal, and elevated platelet levels were 15.0 months (95% CI: 0.000-30.397), 18.0 months (95% CI: 15.251-20.749), and 6.0 months (95% CI: 0.000-21.603), respectively (Figure 8).

Kaplan–Meier curves of overall survival stratified by platelet levels (P = 0.029).
Discussion
In this study, the radiomics model we established performed well in predicting radiosensitivity in patients with inoperable stage III and IV NSCLC. This result proves that radiomics features have a certain role in predicting the short-term response to radiotherapy in NSCLC. We screened nine features in the radiomics model, and their meanings are explained in the appendix, respectively. Among the selected radiomics features, L_wavelet-HLH_firstorder_Maximum, V_wavelet-HHL_gldm_LargeDependenceLowGrayLevelEmphasis, and V_wavelet-LHH_glszm_SmallAreaLowGrayLevelEmphasis have a higher weight in the radiomics score, indicating that the greater the maximum gray level intensity, the rougher the texture of the tumor, and the better the radiotherapy sensitivity. At the same time, we found that concurrent chemoradiotherapy is also associated with radiosensitivity, and the combined model based on radiomics scores and clinical factors not only improved AUC values, but also demonstrated a higher overall net benefit.
In view of the analysis of clinical factors in this study, CCRT can improve the treatment response to radiotherapy in patients with NSCLC. However, not all patients can tolerate the toxic side effects caused by concurrent radiotherapy and chemotherapy in the clinical treatment process. As a result, patients with poor radiotherapy sensitivity may accept combination therapy, such as combined immunotherapy or targeted therapy, to improve radiation therapy efficiency if they cannot tolerate chemotherapy-related side effects. 29 Among these, the optimized combination of radiotherapy and immunotherapy is an emerging field. Studies have shown that radiotherapy may stimulate or enhance the response to immune checkpoint inhibitors. 30 The combination of immune checkpoint inhibitors and local radiotherapy will improve the control of local and distant metastases and ultimately improve the clinical outcome of patients with oligometastatic cancer. 31 Therefore, the combination of radiotherapy and immunotherapy has certain prospects. Of course, this requires more experimental research to further explore this aspect and validate the effectiveness of this approach. In addition, the advancement of targeted therapy research, especially the development of tyrosine kinase inhibitors (TKIs), has also had a positive impact on therapeutic approaches. Studies have shown that the response to radiotherapy varies depending on the molecular status of tumors, and EGFR-mutated tumors respond well to radiotherapy. 32 Another study on early lung adenocarcinoma found that KRAS mutations were associated with poor local control of SBRT treatment. 33 Studies have also shown that the first-line use of radiotherapy and TKI treatment patterns are associated with better prognosis, 34 while the use of upfront epidermal growth factor receptor-TKI (EGFR-TKI), and deferral of radiotherapy, is associated with inferior OS in patients with EGFR-mutant NSCLC who develop brain metastases. 35 Precise lung cancer treatment includes not only precise radiotherapy technology but also precise targeted and immunotherapy. All of the above will be used to direct further research in the future and to provide help for each patient in choosing a more personalized treatment plan.
In our study, synchronized chemotherapy apparently did not help with survival. This result is quite different from that of many previous studies and meta-analyses in which concurrent chemotherapy improves OS; we believe that a possible reason is that most of the patients in this study had advanced T stage (about 67% of patients with T3-4). For these patients with large tumor volumes and extensive invasion of adjacent tissues and organs (such as the heart, esophagus, large blood vessels, etc.), local treatment is more difficult, increasing the radiation dose cannot preserve important tissues and organs, and reducing the radiation dose cannot achieve radical tumor cure. It is also more difficult to set up a field in the target area of radiotherapy. Therefore, most patients received several cycles of induction chemotherapy before radiotherapy to reduce the tumor treatment volume and normal tissue damage caused by radiotherapy. This is different from the rigorous CCRT in previous studies. Moreover, the results obtained in this study that CCRT after induction chemotherapy did not increase the survival benefit were consistent with the results of previous studies. Ardizzoni found that the addition of single agent taxane given concurrently to radiotherapy failed to significantly improve survival after platinum-based induction in locally advanced NSCLC. 36
In addition, a number of previous studies have discussed the role of the neutrophil-to-lymphocyte ratio (NLR) in the prognostic assessment of NSCLC and have demonstrated that an increase in NLR over the course of radiotherapy had a negative impact on survival. 37 Some studies have shown that NLR is associated with inferior outcomes in localized NSCLC treated with SBRT. 38 In the results of this study, neither the univariate nor multivariate analysis showed a statistically significant impact of NLR. A possible reason is that some patients have received chemotherapy before radiotherapy, and the hematological toxicity of chemotherapy leads to different results of hematological examinations, which causes interference.
In the exploration of factors related to survival prognosis, we did not find a significant correlation between radiotherapy sensitivity and OS. There are several possible reasons worth considering for this observation. One reason for the failure to achieve significant survival-related benefits in patients with radiation-sensitive tumors is the limitation of radiotherapy associated with its side effects. Regarding this limitation, we need to consider whether an individualized dose setting in radiotherapy-sensitive patients is required, which, of course, requires further intensive investigation. In addition, in stage IV patients, death can be caused by metastases.
We found that platelet level status was correlated with OS patients with NSCLC after radiotherapy. Patients with platelet levels higher than normal had a poor prognosis. A number of previous studies have confirmed the existence of thrombocytosis and platelet activation in cancer patients, which can promote the growth, invasion, metastasis, and other malignant behaviors of tumors through a variety of mechanisms39–41 and are closely related to low survival rates. Our research results are consistent with these previous conclusions.
This study also had some limitations: (1) This was a single-center study with a small sample size. (2) The data collection was retrospective, and there may have been deviations in the record of previous medical records. (3) The study excluded patients who have received targeted or immunotherapy before radiotherapy, and there was a lack of research on these two types of patients. (4) The study included patients with NSCLC with different tumor node metastasis stages and failed to conduct a stratified analysis of the different stages.
Conclusions
Radiomic features extracted from CT images combined with clinical features have potential value in predicting radiosensitivity, which may provide more accurate treatment strategy guidance for clinicians treating NSCLC. For radiation-insensitive patients, radiotherapists around the world are actively exploring further possibilities for locally advanced or metastatic cancer treatment to provide a greater advantage to bring more treatment benefits to patients.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221142400 - Supplemental material for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338221142400 for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer by Wenrui Chen, Li Wang, Yu Hou, Lan Li, Li Chang, Yunfen Li, Kun Xie, Linbo Qiu, Dan Mao, Wenhui Li and Yaoxiong Xia in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-3-tct-10.1177_15330338221142400 - Supplemental material for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer
Supplemental material, sj-docx-3-tct-10.1177_15330338221142400 for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer by Wenrui Chen, Li Wang, Yu Hou, Lan Li, Li Chang, Yunfen Li, Kun Xie, Linbo Qiu, Dan Mao, Wenhui Li and Yaoxiong Xia in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-4-tct-10.1177_15330338221142400 - Supplemental material for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer
Supplemental material, sj-docx-4-tct-10.1177_15330338221142400 for Combined Radiomics–Clinical Model to Predict Radiotherapy Response in Inoperable Stage III and IV Non-Small-Cell Lung Cancer by Wenrui Chen, Li Wang, Yu Hou, Lan Li, Li Chang, Yunfen Li, Kun Xie, Linbo Qiu, Dan Mao, Wenhui Li and Yaoxiong Xia in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Kunming Medical University in Yunnan Province (approval number: KYLX202175). Informed consent was waived by the committee because of the retrospective nature of this study. We have de-identified all patient details to ensure the confidentiality of patient information.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (No. 82060558), Yunnan Fundamental Research Projects (No. 202001AS70011), Ten-thousand Talents Program of Yunnan Province (Yunling scholar), Yunnan Provincial Training Special Funds for High-level Health Technical Personnel (No. L-2018001), Yunnan Health Training Project of High Level Talents (No. H-2019074), Kunming Medical University 2021 postgraduate innovation fund project (2021S254).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
