Abstract
Background
According to previous literatures, plasma thioredoxin reductase (TrxR) level was significantly elevated in various malignant tumors and serve as a potential biomarker for diagnosis and prognostic prediction. However, there is little awareness of the clinical value of plasma TrxR in gynecologic malignancies. In the present study, we aim to evaluate the diagnostic accuracy of plasma TrxR in gynecologic cancer and explore its role in treatment surveillance.
Methods
We retrospectively enrolled 134 patients with gynecologic cancer and 79 patients with benign gynecologic disease. The difference of plasma TrxR activity and tumor markers level between two groups was compared using Mann-Whitney U test. By detecting pretreatment and post-treatment level of TrxR and conventional tumor markers, we further assessed the change trend of them with the Wilcoxon signed-ranks test.
Results
Compared with benign control [5.7 (5, 6.6) U/mL], statistically significant increase of TrxR activity was observed in gynecologic cancer group [8.4 (7.25, 9.825) U/mL] (P < .0001), regardless of age and stage. On the basis of receiver operating characteristic (ROC) curves, we found plasma TrxR shows the highest diagnostic efficacy for distinguishing malignancy with benign disease, with an area under the curve (AUC) of 0.823 (95% confidence interval [CI] = 0.767-0.878), in the whole cohort. Besides, patients receiving treatment previously [8 (6.5, 9) U/mL] had a decreased TrxR level relative to treatment-native patients [9.9 (8.6, 10.85) U/mL]. Furthermore, follow-up data showed that plasma TrxR level would be evidently decreased after two courses of antitumor therapy (P < .0001), which is consistent with the downward trend of conventional tumor markers.
Conclusion
Collectively, all these results demonstrated plasma TrxR is an effective parameter for gynecologic cancer diagnosis and concurrently acts as a promising biomarker for treatment response assessment.
Introduction
Gynecologic malignant tumor, a serious threat to women's physical and mental health, includes three major tumors, cervical cancer, endometrial cancer, and ovarian cancer. Other relatively rare malignant tumors, such as tubal and vulvar cancer, also cause unignored distress in marriage and fertility. 1 Cervical cancer is the most common malignancy of female reproductive system. According to the World Health Organization, there are 530 000 new cases and about 311000 deaths each year worldwide, with 80% in developing countries. In recent years, the incidence of cervical cancer has been gradually declining due to the appearance of the human papilloma virus (HPV) vaccine and the widespread availability of cervical cancer screening.2,3 Endometrial cancer accounts for 20% to 30% of malignant tumors of the reproductive system and has been on the rise. The prognosis of endometrial cancer is closely related to age, stage, differentiation, and pathological type. Advanced age and low differentiation are more likely to predict a worse outcome.3,4 Ovarian cancer is the third leading malignancy after cervical and endometrial cancer. With high insidiousness and recurrence rate as well as tricky difficulty in treatment, ovarian cancer mortality ranks the highest among gynecologic tumors, with a 5-year survival rate <45%, and the rate drops to 25% for patients on advanced stage. 5 Consequently, screening and early diagnosis of gynecologic cancer are of great importance.
Currently, there are limited specific and sensitive tumor markers available for the diagnosis and follow up of endometrial cancer. In patients with extra-uterine carcinoma, especially ovarian cancer, carbohydrate antigen 125 (CA125) is the most widely used, and helps to monitor treatment response. However, factors such as inflammation or radiation injury also cause abnormally elevated CA125, whereas in some patients (eg, isolated vaginal metastases) CA125 may not be elevated. Therefore, CA125 may not be an accurate predictor of recurrence without other clinical manifestations. 6 It is less sensitive in the early stages of ovarian cancer and can be nonspecifically increased in a range of benign conditions, like endometriosis and pelvic inflammatory disease.7,8 Due to heavier economic burden and inconvenience, computed tomography, magnetic resonance imaging, and gynecologic ultrasound were tended to be applied as second-line tests in clinical practice. Additionally, imaging examination has limitation to detect the occulted or subclinical lesions. 9 Even though the sensitivity and specificity of a single parameter vary, combined tests would improve the diagnostic accuracy absolutely.
Thioredoxin (Trx), Trx reductase (TrxR), and nicotinamide adenine dinucleotide phosphate (NADPH) form the Trx system, which is crucial to redox homeostasis. TrxR maintains the endogenous substrate Trx protein in a reduced state based on electrons from NADPH. 10 Either in normal or in neoplastic cells, increasing TrxR would be secreted under conditions of oxidative stress and inflammation. In normal cells, TrxR is essential for redox homeostasis and protection against oxidative damage and mutation, and once transformed into malignant cells, TrxR would accelerate tumor growth and progression in turn. 11 TrxR overexpression was observed in a variety of primary malignancies, including gastric, breast, and hepatocellular carcinomas.1213–14 Yoo et al 15 found that knockdown of TrxR1 would result in tumor retraction and metastasis suppression, in addition to a reduction in messenger RNA transcript levels of cancer-associated proteins. Three TrxR isoforms (TrxR1, TrxR2, and TGR) contain a selenocysteine substance that can be used as a target for pharmaceutical therapy, such as gold-containing compounds, platinum compounds, and curcumin analogs, which exert cytotoxic effects through competitive, noncompetitive or reversible mechanism.161718–19
Due to the high incidence and mortality rate of gynecologic cancer, it is urgent to find a convenient and cost-effective method for diagnosis and treatment surveillance . With increasing explorations, a variety of approaches are springing up. Apart from imaging, current hematological examinations mainly depend on conventional tumor markers but not meet the demand. Plasma TrxR was identified as a potential biomarker for various malignant tumors in prior explorations. In order to understand the value of TrxR in gynecologic cancer, we conducted this retrospective study focused on evaluating the feasibility of plasma TrxR in gynecologic cancer diagnosis and monitoring therapeutic efficiency. We expect plasma TrxR to be an alternative indicator in clinical practice.
Materials and Methods
Patients
The eligible patients were consecutively enrolled in this retrospective study from Nanjing First hospital from September 2019 to December 2021. Patients diagnosed as either gynecologic cancer (eg, uterine , ovarian, and tubal cancers) or benign gynecologic disease (eg, uterine leiomyoma, cyst, and chronic inflammation) were based on hematological examination, imaging, and pathological analyses. Patients with chronic system disease and other malignant tumors are excluded. The collected data include patients’ age, stage, plasma TrxR activity, serum level of routine tumor markers, along with the following therapeutic strategies. For patients underwent therapy previously in other organizations, we would also record the past treatment regimens. And we have de-identified all patient details. This study was approved by the ethics committees of Nanjing First Hospital (No. NJFH2019-047, Nanjing, China) and all recruited patients have written the informed consent. The reporting of this study conforms to STARD guidelines. 20
Specimen Collection and Analysis
All patients would receive examinations at the first visit to Nanjing First Hospital. The patients with gynecologic cancer were admitted for hematological test regularly during the period of treatment. In accordance with manufacturer's instructions and prior literatures,2122–23 3 to 5 mL fasting venous blood were collected and centrifugated within 2 h, at 3000 rpm for 5 min. The separated plasma was stored in eppendorf CA125, and CA153 were 8.78 ng/mL, 5 ng/mL, 27 tubes at 2°C to 8 °C. Plasma TrxR activity was determined by spectrophotometry, with commercially approved kits, which were purchased from Clairvoyance Health Technology Co., Ltd. Serum tumor markers were detected by electrochemiluminescence immunoassay and analyzed with Roche Cobas e601 fully automated immunoassay system (Roche Diagnostics). In a single-blind model, performers were unaware of clinical information of samples. The manufacturer recommended that the reference level of plasma TrxR is <4 U/mL in healthy populations. According to expert consensus and previous literatures, the optimal cutoff value of alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), CA199, CA125, and CA153 were 8.78 ng/mL, 5 ng/mL, 27 U/mL, 35 U/mL and 25 U/mL, respectively.2425–26
Statistical Analysis
To understand the difference of plasma TrxR and common tumor markers between malignant group and benign control, we applied Mann-Whitney U test to make a comparison. Receiver operating characteristic curve (ROC) was performed to evaluate the discriminatory power of TrxR for gynecologic cancer, via calculating area under curve (AUC). Based on the Youden index (specificity + sensitivity − 1), we would draw out the critical cutoff value of plasma TrxR. Chi-square test and Fisher's exact test were applied to assess the correlation of TrxR and clinical variables, as well as routine tumor biomarkers. The Wilcoxon signed-ranks test was used to evaluate the change of plasma TrxR and tumor markers after treatment. All statistical tests were analyzed by GraphPad Prism 8.0 (La Jolla) and SPSS 22.0 (IBM) software, with 2-side test, and P < .05 was assumed to be statistically significant.
Results
Patient Characteristics
A total of 213 patients were enrolled in this study, including 134 cases with gynecologic cancer (uterine cancer, n = 80; ovarian cancer, n = 49; others, n = 5) and 79 with benign disease (benign uterine disease, n = 64; benign ovarian disease, n = 13; others, n = 2). In gynecologic cancer group, the median age was 58 years. As shown in Table 1, there exists a great difference of tumor markers between gynecologic malignancies and benign gynecologic disease (CEA, CA125, and CA153, P < .0001), without statistical bias in age (P = .245). The other baseline characteristics, including family history, menarche age, number of pregnancies and births, hormone replacement therapy, and obesity were essentially identical. A significant difference of serum CA125 and CA153 level was observed both in uterine cancer (CA125, P < .0001; CA153, P = .001) (Supplemental Table 1) and ovarian cancer group (CA125, P < .0001; CA153, P = .002) (Supplemental Table 2).
The Characteristics of Gynecologic Cancer and Benign Gynecologic Disease.

Notes: *P < .05.
Abbreviations: AFP, alpha-fetoprotein; CEA, carcinoembryonic antigen; CA, carbohydrate antigen; NA, not available values.
TrxR Activity and Routine Tumor Markers in Gynecologic Cancer and Benign Controls
By detecting plasma TrxR activity and tumor markers level, we attempted to make a comparison between malignant group and benign control. Results were shown as (median [interquartile ranges]). Expectedly, patients with gynecologic cancer had a significantly higher TrxR activity (8.4 [7.25, 9.825] U/mL) relative to benign disease (5.7 [5, 6.6] U/mL) (P < .0001). As well, serum CEA (P < .01), CA199 (P < .05), CA125 (P < .0001), and CA153 (P < .0001) were markedly increased in gynecologic cancer (Figure 1). Both in patients with uterine cancer (8.45 [6.5, 9.975] U/mL) and ovarian cancer (8.3 [7.6, 9.85] U/mL), TrxR activity were also significantly higher than patients with corresponding benign disease. The median plasma TrxR activity in benign uterine group and benign ovarian group were (5.7 [5.125, 6.6] U/mL) and (6.3 [4.45, 6.85] U/mL), respectively. Additionally, some critical biomarkers, such as CEA, CA199, CA125, and CA153 were dramatically elevated in malignancies (Supplemental Figures S1 and S2). Taken together, plasma TrxR activity and common tumor markers are significantly increased in malignant tumors, which indicates they are potentially used for differentiating gynecologic malignancy from benign disease.
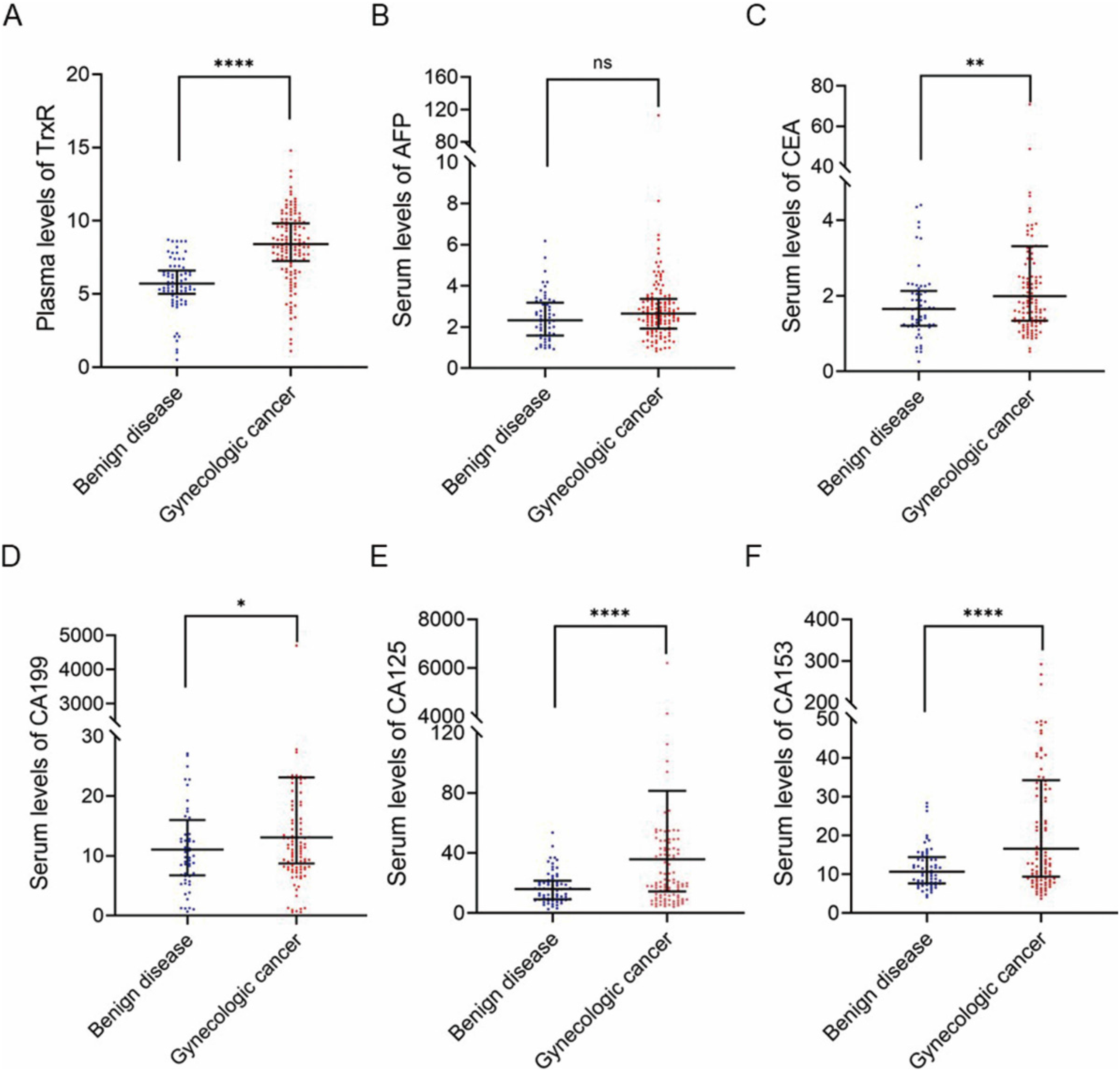
The difference of plasma TrxR activity and routine tumor markers level in benign gynecologic disease and gynecologic cancer. (A) Scatter plot of the distribution of plasma TrxR, (B) serum AFP, (C) serum CEA, (D) serum CA199, (E) serum CA125, and (D) serum CA153 levels in benign controls and gynecologic cancer. The black horizontal lines are median values and interquartile ranges. P values were determined by the Mann-Whitney U test (*P < .05, **P < .01, ***P < .001, ****P < .0001). Abbreviations: AFP, alpha-fetoprotein; AUC, area under curve; CA, carbohydrate antigen; CEA, carcinoembryonic antigen; ns, no statistical significance; TrxR, thioredoxin reductase.
TrxR Activity as a Diagnostic Biomarker for Gynecologic Cancer
Given that plasma TrxR were significantly higher in patients with gynecologic cancer, we further performed ROC analysis to evaluate the diagnostic efficacy of TrxR, by calculating the AUC. Concurrently, we would determine the optimal cutoff value of plasma TrxR for distinguishing malignancies from benign control, based on the maximal Youden Index (sensitivity + specificity − 1). As shown in Figure 2, TrxR displayed the highest AUC (AUC = 0.823, 95% confidence interval [CI] = 0.767-0.878), compared with CA125 (AUC = 0.715, 95% CI = 0.646-0.784), and CA153 (AUC = 0.701, 95% CI = 0.629-0.773). And the critical TrxR value was 7.0 U/mL with a sensitivity of 76.1% and a specificity of 82.3%. Serum AFP, CEA, and CA199 had a slightly weaker diagnostic efficacy, with AUCs of 0.584 (95% CI = 0.497-0.671), 0.64 (95% CI = 0.561-0.72), and 0.613 (95%CI = 0.53-0.696), respectively. More importantly, AUC of a combined panel of TrxR and other tumor markers would be further elevated (AUC = 0.903, 95% CI = 0.861-0.946). As well, TrxR showed a great discrimination ability for uterine cancer (AUC = 0.804, 95% CI = 0.731-0.876) and ovarian cancer (AUC = 0.823, 95% CI = 0.704-0.943). Counterintuitively, the diagnostic capability of other biomarkers was relatively moderate (Supplemental Figures S3 and S4). Anyway, plasma TrxR maintains a high diagnostic accuracy and a combination of plasma TrxR and routine biomarkers would identify malignancy more effectively, relative to TrxR alone or an individual biomarker.

ROC curves of TrxR and routine tumor markers for gynecologic cancer versus benign controls. (A) ROC curves of TrxR, CA125 and the combinations thereof. (B) ROC curves of TrxR, CA153 and the combinations thereof. (C) ROC curves of TrxR, CA125, CA153, and the combinations thereof. (D) ROC curves of TrxR, AFP, CEA, CA199, CA125, CA153, and the combinations thereof. Abbreviations: AFP, alpha-fetoprotein; AUC, area under curve; CA, carbohydrate antigen; CEA, carcinoembryonic antigen; ROC, receiver operating characteristic; TrxR, thioredoxin reductase.
The Correlation of TrxR Activity and Clinical Variables in Gynecologic Cancer
Based on ROC analysis, we calculated the optimal cutoff value of TrxR was 7.0 U/mL according to the maximal Youden Index. Subsequently, we further applied Chi-square test and Spearman's test to authenticate this critical result, and found that there actually exists a great difference between gynecologic cancer and benign group (P < .0001, r = 0.566) when plasma TrxR was set at 7.0 U/mL. Besides, this significant difference was observed both in uterine subgroup (P < .0001, r = 0.51) and ovarian subgroup (P < .0001, r = 0.603) (Supplemental Table 3). Accordingly, a total of 102 patients (76.1%) were defined as high-TrxR activity while the rest 32 patients (23.9%) were low-TrxR activity. Results demonstrated that plasma level of TrxR was highly correlated with some conventional tumor markers, such as CA199 (P = .038), CA125 (P = .012), and CA153 (P = .012), independently of age (P = .105) and stage (P = .259) (Table 2). Similarly, TrxR activity has a close relationship with CA125 (P = .036) and CA153 (P = .026) in uterine cancer (Supplemental Table 4). However, there were too few cases in ovarian cancer group to substantiate the conclusion (Supplemental Table 5). On balance, TrxR setting at 7.0 U/mL was the most favorable for distinguishing gynecologic cancer with benign disease. Concurrently, the close correlation of TrxR level and tumor markers also strongly supports evidence for the optimal threshold.
The Correlation of TrxR and Clinical Variables in Gynecologic Cancer.
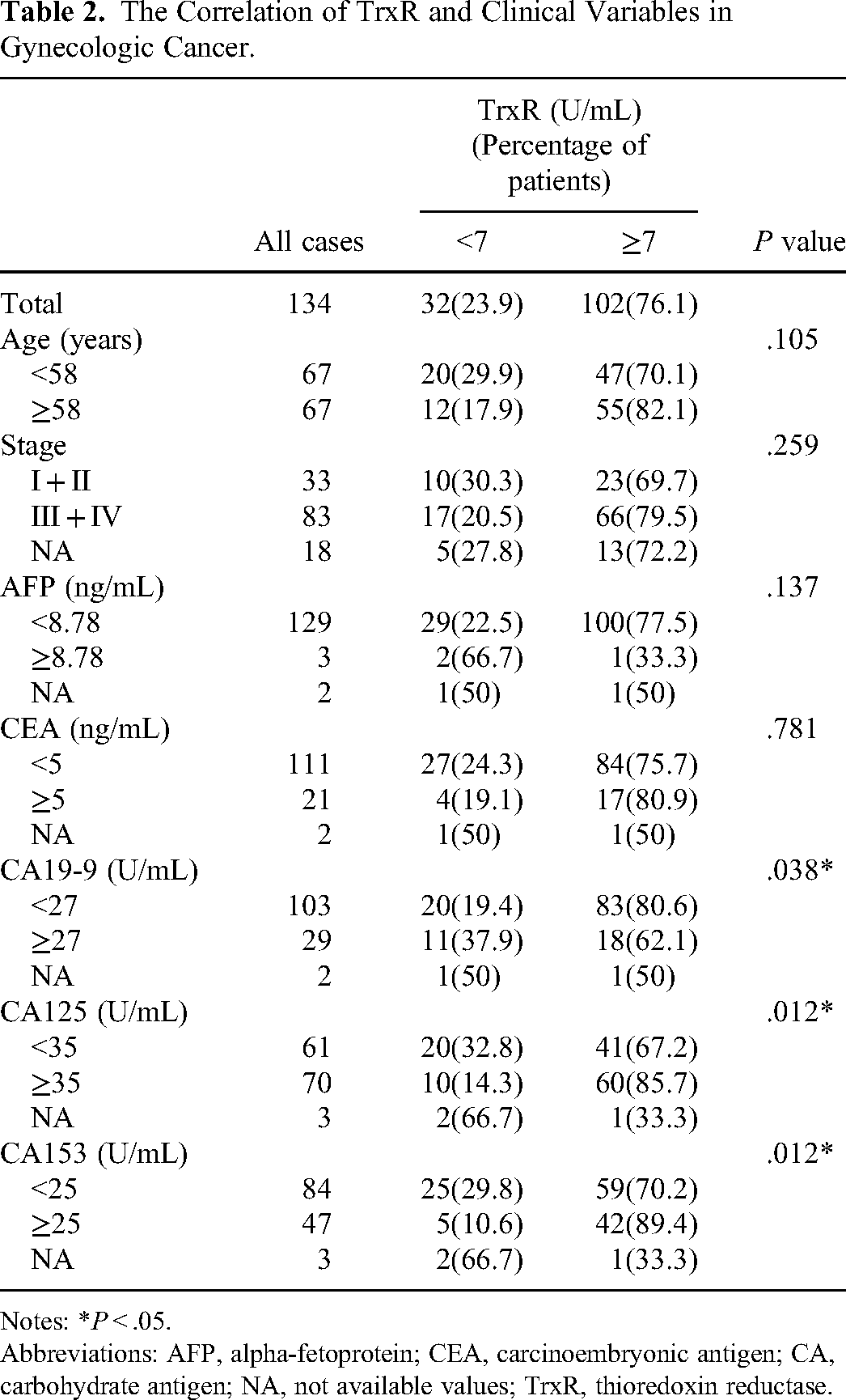
Notes: *P < .05.
Abbreviations: AFP, alpha-fetoprotein; CEA, carcinoembryonic antigen; CA, carbohydrate antigen; NA, not available values; TrxR, thioredoxin reductase.
TrxR Activity as a Biomarker for Assessing Therapeutic Efficacy
The involved 134 patients in gynecologic cancer group included 97 treatment-experienced patients who have received treatment in other organizations previously, and 37 treatment-naïve patients. Either with or without prior treatment, patients with gynecologic cancer had an elevated TrxR level compared with benign disease. In addition, TrxR activity was significantly decreased in patients previously underwent treatment (8 [6.5, 9] U/mL) relative to treatment-naïve patients (9.9 [8.6, 10.85] U/mL) (Figure 3A). In light of prior therapy regimens, treatment-experienced patients could be classified in 3 subgroups, including taxane-platinum, targeted-based therapy, and other strategies like immunotherapy or traditional Chinese medicine. Intriguingly, the lowest TrxR activity was observed in patients adopting targeted-containing regimen (7.6 [4.8, 8.55] U/mL) (Figure 3B). Within 63 patients receiving re-examination of TrxR and routine tumor markers after 2 courses antitumor therapy in Nanjing First Hospital, we found that plasma TrxR was significantly decreased after platinum-based therapy from (8.2 [7.3, 9.1] U/mL) to (6.7 [2.3, 8.3] U/mL), taxane-based therapy from (8.2 [7.4, 9.4] U/mL) to (6.6 [2.3, 8.4] U/mL) and targeted-based therapy from (8.1 [5.6, 8.8] U/mL) to (6.6 [1.5, 7.7] U/mL), with all P value < .001 (Figure 3C). Finally, we further analyzed the change trend of conventional tumor markers and noticed that plasma TrxR, CA199, CA125, and CA153 were all trended downward in the whole cohort. Similarly, CA199, CA125, and CA153 were also decreased significantly in TrxR-decreased cohort and evidently increased in TrxR-increased subgroup (Table 3). To conclude, plasma TrxR would be decreased after corresponding targeted therapy generally. TrxR showed a consistent change trend with routine tumor markers, indicating measuring TrxR level would be a novel approach to monitoring disease.

The correlation of plasma TrxR activity and treatment in gynecologic cancer patients. (A) Scatter plot of the distribution of plasma TrxR in benign controls, treatment-experienced, and treatment-naïve gynecologic cancer patients. (B) Plasma TrxR activity of gynecologic cancer patients based on different therapies. (C) The change trend of plasma TrxR after platinum/taxane-based or targeted therapy. Abbreviation: TrxR, thioredoxin reductase.
The Change of Plasma TrxR Activity and Conventional Tumor Markers After Therapy.

Notes: *P < .05, Wilcoxon signed-rank test.
Abbreviations: AFP, alpha-fetoprotein; CEA, carcinoembryonic antigen; CA, carbohydrate antigen; TrxR, thioredoxin reductase.
Discussion
Early diagnosis and timely treatment are the most effective strategies to improve the survival outcome of patients. As is well known, high-risk HPV (hrHPV) is the leading cause of cervical cancer. HPV screening is therefore vital for the early diagnosis and prevention of cervical cancer.27,28 Nevertheless, the roll-out of screening is very low over the last decades, especially in resource-limited territories. 29 Recent HPV self-sampling techniques identified by convenient, comfortable and private, dramatically increase screening coverage. 30 In addition, new screening methods, such as DNA ploidy analysis, methylation detection and p16/Ki-67 double staining, also facilitate the identification of cervical precancer, which are currently used as triage techniques for HPV-positive patients.3132–33 Unfortunately, there exists no standardized screening test for ovarian cancer and endometrial cancer. Genetic factors play an important role in ovarian carcinogenesis. Identify population at high risk through 3 steps: risk assessment, genetic counseling, and breast cancer susceptibility genes genetic testing.34,35 The risk of ovarian malignancy algorithm, the risk of malignancy index, and OVA1 assays have also been clinically used with improved sensitivity and specificity. A combination of molecular biomarkers (eg, CA125, human epididymis protein 4, CEA, and vascular cell adhesion protein 1) also can evidently improve diagnostic efficiency of ovarian cancer.36,37 More investigation into novel biomarkers, including circulating tumor DNA, microRNA profiling, and DNA methylation signatures are ongoing as well.3839–40 As many as 90% of endometrial cancers would be captured when detection strategies focused on women with postmenopausal bleeding. 41 The follow up of high-risk group currently depends on cervical cytology, pelvic examination, and trans vaginal ultrasound. In addition to these, the emerging radiomics is more objective than the traditional imaging, which extracts high-throughput features from medical images to quantitatively analyze malignant tumors, thereby providing a new insight into the clinical diagnosis and treatment of gynecological cancer.42,43 At present, the biggest problem is that there is no standardization of radiomics research methods. The results obtained by different modeling methods for the same features vary greatly, and different dimensionality reduction methods will also filter out useful key features to varying degrees of errors, so that the application remains at the exploratory phase. Taken together, the search for innovative screening techniques is in full swing and the diagnostic accuracy still needs to be optimized.
TrxR, an important part of Trx system, is involved in diverse physiological and pathological processes, such as inflammation, apoptosis, and redox homeostasis. Researches show that TrxR has a close correlation with neoplasm, parasitic infection, immunodeficiency/rheumatic diseases, circulatory disease, etc.44454647–48 Patients with rheumatoid arthritis have significantly elevated TrxR in synovial fluid and tissues. The gold-containing compounds goldthiosamine and chylranofin, which are clinically used for the treatment of rheumatoid arthritis, are highly potent inhibitors of TrxR. 49 The serum level of TrxR in patients with hypertrophic cardiomyopathy and the acute stage of explosive myocarditis was significantly higher than that of normal individuals, and Trx system may achieve a protective effect against myocarditis by activating nuclear factor kappa B. 50 More importantly, TrxR plays an essential role in certain stage of tumor development or progression. By regulating the balance of redox state of the intracellular and extracellular environment, TrxR can effectively scavenge excessive reactive oxygen species against oxidative stress, thereafter decreasing the risk of carcinogenesis. 51 Meanwhile, once cells become cancerous, high concentration of TrxR would facilitate tumor growth and promote metastasis. 52 It has been revealed that TrxR is overexpressed in a variety of tumor cells and precancerous cells, which is the leading cause of the abnormal proliferation of tumor cells. According to previous literatures, TrxR expression in tumor cells is about 5 to 10 times higher than that in normal tissue.53,54 Soderberg et al 55 found that TrxR can be released into peripheral blood through the tumor cell membrane, suggesting it is reasonable to measure TrxR in peripheral blood. In the following clinical studies, plasma TrxR has demonstrated great diagnostic efficacy in lung, liver, gastric, and prostate cancers.23,5657–58 What's more, plasma TrxR acts as a superior biomarker for evaluating therapeutic response and predicting prognosis in malignancies, compared with conventional tumor markers alone. All these preclinical evidence and clinical data strongly proved that plasma TrxR is a potential biomarker for a wide range of malignancies.
In our study, gynecologic cancer group has a significantly higher plasma TrxR (8.4 [7.25, 9.825] U/mL) than benign control (5.7 [5, 6.6] U/mL) (P < .0001), indicating the elevated level of TrxR in malignant lesions, which is consistent with previous literatures. By establishing ROC curves, we drawn out the AUC of plasma TrxR was up to 0.823 (95% CI = 0.767-0.878). Furthermore, the optimal threshold of TrxR was 7.0 U/mL to distinguish gynecologic cancer with benign disease, with a sensitivity of 76.1% and a specificity of 82.3%. Even though some chronic inflammation and stress conditions can stimulate the body peroxidation, this cutoff value would provide an early warning in tumor diagnosis. In previous explorations, a large-scale study focused on assessing the role of TrxR in nonsmall cell lung cancer recommend 10.18 U/mL as the critical threshold. 57 Another retrospective study showed that TrxR could serve as a potential biomarker for prostate cancer, with the optimal cutoff value of 8.20 U/mL, which is set at 3.85 U/mL for primary liver cancer.56,58 Therefore, it deserves sufficient considerations of tumor heterogeneity when clinicians determine the diagnostic cutoff value. Besides, plasma TrxR has a close correlation with conventional tumor markers, especially with CA125 and CA153 (Table 2). However, routine tumor markers alone have limitations with low sensitivity and specificity. In this study, we noticed the AUC would be evidently improved when TrxR was combined with tumor markers (Figure 2 and Supplemental Figures S3 and S4). This combination panel offers a feasible diagnostic strategy in clinical practice.
Indeed, plasma TrxR level was enhanced both in treatment-naïve and treatment-experienced patients. A majority of treatment-experienced patients have received taxane-platinum regimen before. As mentioned above, platinum compound is a type of cytotoxic agent targeting TrxR. 59 Reasonably, treatment-naïve patients have a significantly higher TrxR activity than those receiving serious lines of therapy (Figure 3A). Within patients receiving re-examinations after two courses of treatment, plasma TrxR levels were generally decreased and the change trend of TrxR and routine tumor markers was consistent (Figure 3C and Table 3). In accordance with previous literature, TrxR overexpression promotes tumor growth by inhibiting the programmed death of tumor cells, stimulating cell proliferation and angiogenesis, and increasing the activity of transcription factors, which also indicates the deterioration of disease.60,61 Deficiency of TrxR would destructible mitochondrial redox environment, damage corresponding signal transduction pathway and subsequently down-regulate expression of vascular endothelial growth factor, which eventually affects tumor proliferation.54,62 Based on this consensus, TrxR is a promising biomarker for monitoring therapeutic efficacy.
There are several limitations in our study. Firstly, the nature of a small-sample, single-center retrospective study does create limitations. Among patients with malignant tumors, plasma TrxR level may be affected by many clinical factors such as tumor-node-metastasis stage, histological differentiation or metastasis but remains disputable.12,21,23 In our study, a majority of gynecologic cancer patients were set on stage III-IV (83 of 134), without adequate samples at earlier stage (33 of 134). Our results demonstrated that plasma level of TrxR was independent of stage (P = .259). Due to the insufficient clinical data, it still needs more efforts to expand the sample size and include more patients with early stage to confirm this conclusion. Additionally, the enrolled patients accepted heterogeneity treatments and the small-sample size mixed with diverse therapeutic regimens, which lead to the difficulty of multigroup refinement. In accordance with the previous literature, taxane-platinum regimen could dramatically decrease plasma TrxR level. Intriguingly, patients experiencing other treatments also have a decreased TrxR activity. More investigations are expected concerning on the chemical interaction between TrxR and targeted agents, immune checkpoint inhibitors as well as other cytotoxic chemotherapy. Secondly, we measured TrxR level in serum, not in histologic specimen. It is still uncertain whether peripheral TrxR level dynamically reflects real-time changes in the tissue. The relationship between peripheral and tissue TrxR levels warrants further investigations. And in the future, the more interesting question would be whether TrxR is pathogenically involved in the development of early recurrence and poor prognosis.
Conclusion
Summarily, TrxR plays an essential role in tumorigenesis and progression. And this is the first clinical study of evaluating plasma TrxR as a biomarker in gynecologic cancer. Final results demonstrated plasma TrxR may be a potential auxiliary for gynecologic cancer diagnosis and treatment effectiveness assessment. Simultaneously, this study brings us inspirations and deliberations: the cutoff diagnostic value of TrxR requires more explorations on basis of different carcinomas, in full consideration of tumor specificity. Next, TrxR is a potential target of antitumor treatment, more novel agents targeting TrxR are eagerly awaited to prevent or reverse chemoresistance. Meanwhile, measuring peripheral blood TrxR has some utility in malignant tumor screening. To assess therapeutic effectiveness and predict prognosis, a combination of imaging and hematological indices remains the leading choice.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231184995 - Supplemental material for Plasma Thioredoxin Reductase as a Potential Biomarker for Gynecologic Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338231184995 for Plasma Thioredoxin Reductase as a Potential Biomarker for Gynecologic Cancer by Yinxing Zhu, MS, Yixuan Hu, MS, Junfeng Shi, MD, Xiaowei Wei, MD, Yaqi Song, MD, Cuiju Tang, MD, PhD, and Wenwen Zhang, MD, PhD in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request. All recruited patients have written the informed consent.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committees of Nanjing First Hospital (approval NJFH2019-047).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Natural Science Foundation of Jiangsu Province, Nanjing Outstanding Youth Fund (grant number 81802667, BK20180133, JQX20009).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
