Abstract
Breast cancer is one of the leading causes of cancer-related deaths in women worldwide. Circular RNAs (circRNAs), a novel class of endogenous noncoding RNA with a covalently closed continuous loop that lacks the 5′-cap structure and the 3′-poly A tail, are more stable than linear RNAs and less susceptible to degradation by nucleases. CircRNAs are widespread in multiple mammalian genomes and have been detected in various tissues, cells and body fluids. Increasing evidence shows that abnormal expression of circRNAs is involved in the development of a variety of diseases, including breast cancer. Numerous studies have explored the potential of circRNAs as biomarkers in various malignant tumors. In this review, we aim to provide a comprehensive overview of the latest advances in circRNAs as promising biomarkers in the early diagnosis, prognosis, molecular type, metastasis and drug resistance of breast cancer.
Introduction
With 2.3 million new cases per year, breast cancer accounts for 11.6% of all cancer cases in 2022, ranking second in global cancer incidence and the fourth leading cause of cancer death worldwide. It is the most common cancer and the leading cause of cancer death among women, posing a serious threat to women's health. 1 At present, neoadjuvant chemotherapy combined with surgery and postoperative systemic therapy have become the accepted strategies for the treatment of breast cancer. 2 However, breast cancer patients are prone to drug resistance, tumor metastasis and recurrence, and the prognosis of breast cancer patients is not satisfactory.3,4 The 5-year survival rate for patients diagnosed with stage I breast cancer is close to 100%, while the 5-year survival rate for patients diagnosed with stage IV breast cancer drops rapidly to 26%.5,6 The 5-year survival rate for patients with breast cancer without metastasis is as high as 80%, compared to only about 27% for patients with metastasis. The most common metastatic sites of breast cancer are ipsilateral axillary lymph nodes. The most distant metastatic organs are bone (50%-65%), lung (17%), brain (16%) and liver (6%), while metastasis to other organs (such as spleen, kidney or uterus) is relatively rare. 7 Patients with brain metastases have the worst prognosis, with a median survival of only 4-6 months. 8 Patients with triple-negative breast cancer (TNBC) have a greatly increased probability of brain metastases, about 30%-50%, 9 which significantly shortens their survival. Obtaining new clinical tools for early and accurate diagnosis of breast cancer is the key to improve the prognosis for breast cancer patients.
In recent years, with the development of genomics and technologies like liquid biopsy, the understanding of non-coding RNAs (ncRNAs) has been continuously enhanced. NcRNAs are functional transcripts that have limited or no protein-coding potential, mainly including microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs). They have emerged as promising biomarkers for various diseases, particularly cancer.10,11 These RNA molecules are stable in body fluids such as blood, making them suitable for non-invasive liquid biopsy. 12 MiRNAs are 21-23 nucleotides long, single stranded, generated endogenously noncoding RNA molecules that regulate gene expression by binding to target mRNAs. 13 Some miRNAs are dysregulated in breast cancer and have the potential to serve as biomarkers. Recent studies have confirmed that serum miRNA-1 may serve as a promising noninvasive biomarker for predicting the treatment response in breast cancer patients receiving neoadjuvant chemotherapy. 14 lncRNAs are single-stranded and longer than 200 nucleotides. They can regulate messenger RNAs (mRNAs) stability, translational efficiency, and interfere with post-translational modifications of proteins, playing critical roles in gene regulation and exhibiting differential expression in many cancers. 15 Nowadays, lncRNAs are emerging as crucial biomarkers in the diagnosis and prognosis of various cancers, including breast cancer. 16 For example, LINC00960 is related to triple-negative breast cancer (TNBC) viability, colony formation, organotypic growth, cell migration, and the induction of cell death, affirming LINC000960 as an unfavorable prognostic biomarker promoting TNBC pathogenesis. 17
Unlike linear RNAs (miRNAs, lncRNAs, etc), circRNAs have neither a 5 ‘cap structure nor a 3’ tail structure, but are formed of covalent closed rings.18,19 CircRNAs with covalent ring structure are not easily degraded by exonuclease and have higher structural stability than linear RNA. CircRNAs are widely distributed across tissues, peripheral blood, exosomes, and other body fluids (such as gastric juice, saliva, and urine). 20 Furthermore, with 10 times more diversity in circRNAs variants compared with linear RNA variants, and combined with their high stability, circRNAs are equipped to influence every stage of oncogenesis. 21 Numerous circRNAs are dysregulated in breast cancer and manifest multiple crucial functions by altering metabolism, immune regulation, angiogenesis, epithelial-mesenchymal transition (EMT), cell cycle regulation, apoptosis etc, and thus involving in breast carcinogenesis, progression and drug resistance of breast cancer, thus highlighting their tremendous potential as biomarkers (Figure 1A).22,23 For example, circCARM1, derived from breast cancer stem cells (BCSC), played an important role in breast cancer cell glycolysis by sponging miR-1252-5p which regulates PFKFB2 expression, thus influencing cell migration. 24 CircGSK3β is highly expressed in breast cancer cell lines and promotes breast cancer cell proliferation, migration, invasion, and immune evasion, by interaction with miR-338-3p and subsequent regulation of PRMT5 and PD-L1. 25 Homo sapiens (has)_circ_0000515 was overexpressed in breast cancer tissues. It bound to miR-296-5p, which, in turn, specifically targeted CXCL10. Hsa_circ_0000515/miR-296-5p/CXCL10 axis regulated tumor cell proliferation, angiogenesis, and inflammation in vivo. Inhibition of hsa_circ_0000515 suppressed breast cancer progression by interrupting hsa_circ_0000515-mediated degradation of miR-296-5p. 26 It not only serves as a potential biomarker for breast cancer but also provides a target for the future treatment of breast cancer. CircEZH2 could promote EMT of breast cancer in a CXCR4-dependent manner via upregulated KLF5, which was shown to be upregulated in liver metastases in breast cancer and predicted worse prognosis in breast cancer patients. 27 The functional mechanisms of circRNAs mainly involve serving as miRNA sponges, protein binding, transcriptional regulation as well as encoding proteins and peptides (Figure 1B), which play significant roles in regulating different molecular pathways. 28 A systematic understanding of the functions of circRNAs and their underlying functional mechanisms is conducive to the discovery of specific biomarkers and the exploration of new treatment approaches, thus inspiring cancer management.

The functions and functional mechanism of circRNAs in breast cancer. (A) CircRNAs manifest multiple functions by altering metabolism, immune regulation, angiogenesis, epithelial-mesenchymal transition (EMT), cell cycle regulation and apoptosis, and thus involve in breast carcinogenesis, progression and drug resistance of breast cancer. Moreover, their potential as biomarkers has been recently highlighted. (B) The functional mechanism of circRNAs. (a) Acting as miRNA sponge, circRNAs are rich in numerous miRNA binding sites and can competitively bind to miRNAs. (b) By binding to proteins, circRNAs affect the functions of RNA-binding proteins (RBPs) and the interactions among RBPs. (c) CircRNAs can upregulate the transcription through interacting with the RNA polymerase II (RNA Pol II) complex. (d) CircRNAs can be translated into proteins. The figure is drawn by Figdraw.
In recent years, researchers have conducted extensive studies on the function and functional mechanism of circRNAs in various malignant tumors, providing a variety of potential biomarkers and therapeutic targets for researchers and clinicians. Coupled with their stability and high specificity, circRNAs have unique advantages over traditional tumor markers, including differentiating between benign and malignant diseases, predicting tumor risk and progression, monitoring treatment sensitivity, and early detection of cancer.
To identify relevant studies, we searched the literature on NCBI PubMed with the keywords “(CircRNAs) AND (Breast cancer)” as the screening criterion. Articles with clinical samples published between 2020 and 2024 that exclude basic research were selected and carefully read.
Here, we briefly review the functions of circRNAs in breast cancer and the underlying functional mechanisms, to provide clinicians and researchers with a comprehensive overview of the latest advances in circRNAs acting as biomarkers for breast cancer research regarding diagnosis, prognosis, molecular type, metastasis and drug resistance. We hope to inspire further research on circRNAs to identify ideal biomarkers and therapeutic targets.
CircRNAs as Markers for Early Diagnosis of Breast Cancer
More and more studies have found that circRNAs are abnormally expressed in the blood and tissues of breast cancer patients, suggesting that this abnormal expression of circRNAs may be useful as a diagnostic detection indicator for early breast cancer (Table 1). Homo sapiens (hsa) _circ_0005046 and hsa_circ_0001791 are highly expressed in breast cancer tissues with AUC of 0.77 and 1, respectively, which could distinguish breast cancer and adjacent tissues well. The AUC of hsa_circ_0001791 is 1, indicating that hsa_circ_0001791 has a high diagnostic value for breast cancer. 29 Circ_0007255 is up-regulated in serum of breast cancer patients with an AUC of 0.7748. 30 Studies have shown that in breast cancer tissue, circWHSC1, 31 circRREB1, 32 circ_0009910, 33 circZCCHC2, 34 circCAPG, 35 circDNAJC11 36 and circPFKFB4 37 were up-regulated. Hsa_circ_0069094, 38 hsa_circ_0079876, 38 hsa_circ_0017650 38 and hsa_circ_0017536 38 were significantly up-regulated in breast cancer plasma, and circPRMT5 39 was significantly up-regulated in breast cancer serum.
CircRNAs in Breast Cancer Diagnosis.

BCT: breast cancer tissues, BCP: breast cancer plasma, BCS: breast cancer serum, BCPB: breast cancer perpheral blood, NA: not applicable.
The diagnostic value of a single circRNA is limited, and the combined application of multiple markers could improve the diagnostic value. Omid-Shafaat R et al. found that circ-ELP3 was highly expressed while circ-FAF1 was decreased in the serum of breast cancer patients, with the AUC of 0.733 and 0.787, respectively. The sensitivity and specificity of circ-ELP3 and circ-FAF1 were 65%, 64% and 77%, 74%, respectively, but the combined application of circ-ELP3 and circ-FAF1 improved the diagnostic efficacy of breast cancer, with AUC up to 0.891, sensitivity and specificity of 96% and 62%. 40 When circ_0000745, circ_0001531 and circ_0001640 were combined in whole blood for the diagnosis of breast cancer, the AUC was 0.9130. 41 Studies have shown that in breast cancer tissues, the expression of hsa_circ_103110, hsa_circ_104689 and hsa_circ_104821 is down-regulated, however, the expression of hsa_circ_006054, hsa_circ_100219 and hsa_circ_406697 were up-regulated. The effect of the combined model was better than that of the single indicator. 42
At present, numerous studies have compared circRNAs expression before and after surgery, and found that the expression level of circRNAs decreased or increased significantly after breast tumor resection. For example, the expression levels of circ-ELP3 in postoperative plasma were significantly reduced compared with those in preoperative plasma. 40 The expression levels of circ-FAF1 in plasma after operation were significantly higher than those before operation. 40
CircRNAs as Markers to Evaluate the Prognosis of Breast Cancer
The expression level of circRNAs in breast cancer is closely related to the pathological characteristics and survival of breast cancer patients, and could be used as a potential marker to predict the prognosis of breast cancer (Table 2). Studies have shown that the overall survival (OS) of breast cancer patients with high expression of circHMCU, 43 exosomal circSIPA1L3, 44 circRREB1, 32 circATP2C1/has_circ_0005797, 45 has_circ_0000069, 46 circDNAJC11, 36 circ_0008717, 47 circPFKFB4, 37 circPTK2, 48 circ-EIF6/hsa_circ_0060055, 49 circRBMS3 50 and circANKS1B 51 was short, while breast cancer patients with low expression of circRNA-CREIT 52 was short. TNBC patients with high expression of hsa_circ_102229, 53 circWHSC1, 31 circCAPG, 35 circ_0001006, 54 circTBC1D14 55 and circWAC 56 had significantly shorter OS than TNBC patients with low expression. The expression of circWSB1 is up-regulated in breast cancer tissues, and the higher the expression level of circWSB1, the shorter the survival time of patients and the higher the recurrence rate. 57 Breast cancer patients with high expression of circ_0044234 58 and TNBC patients with high expression of hsa_circ_001783 59 had short disease-free survival (DFS). Breast cancer patients with high expression of circCDYL, 60 circ_0004674, 61 hsa_circ_0067842 62 and low expression of circ-LARP4 63 had short OS and DFS. Clinical data from patients with human epidermal growth factor receptor 2 (HER2) -positive early breast cancer treated with trastuzumab show that patients with high expression of circCDYL2 have shorter OS and DFS. 64 The expression level of has_circ_0006528 in the tissues of stage III breast cancer patients is higher than that of early-stage breast cancer patients. The relapse-free survival (RFS) and OS of patients with high expression level of has_circ_0006528 were significantly lower than patients with low has_circ_0006528 expression. 65 The expression of circPAPD4 is down-regulated in breast cancer tissues, and breast cancer patients with low circPAPD4 expression have an unfavorable RFS compared with patients with high circPAPD4 expression. 66 Breast cancer patients with high circ_0001667 67 expression and TNBC patients with low circDUSP1 68 expression showed poor survival rates. Breast cancer patients with high circPRMT5 expression showed short OS and progression-free survival (PFS). 39
CircRNAs in Breast Cancer Prognosis.
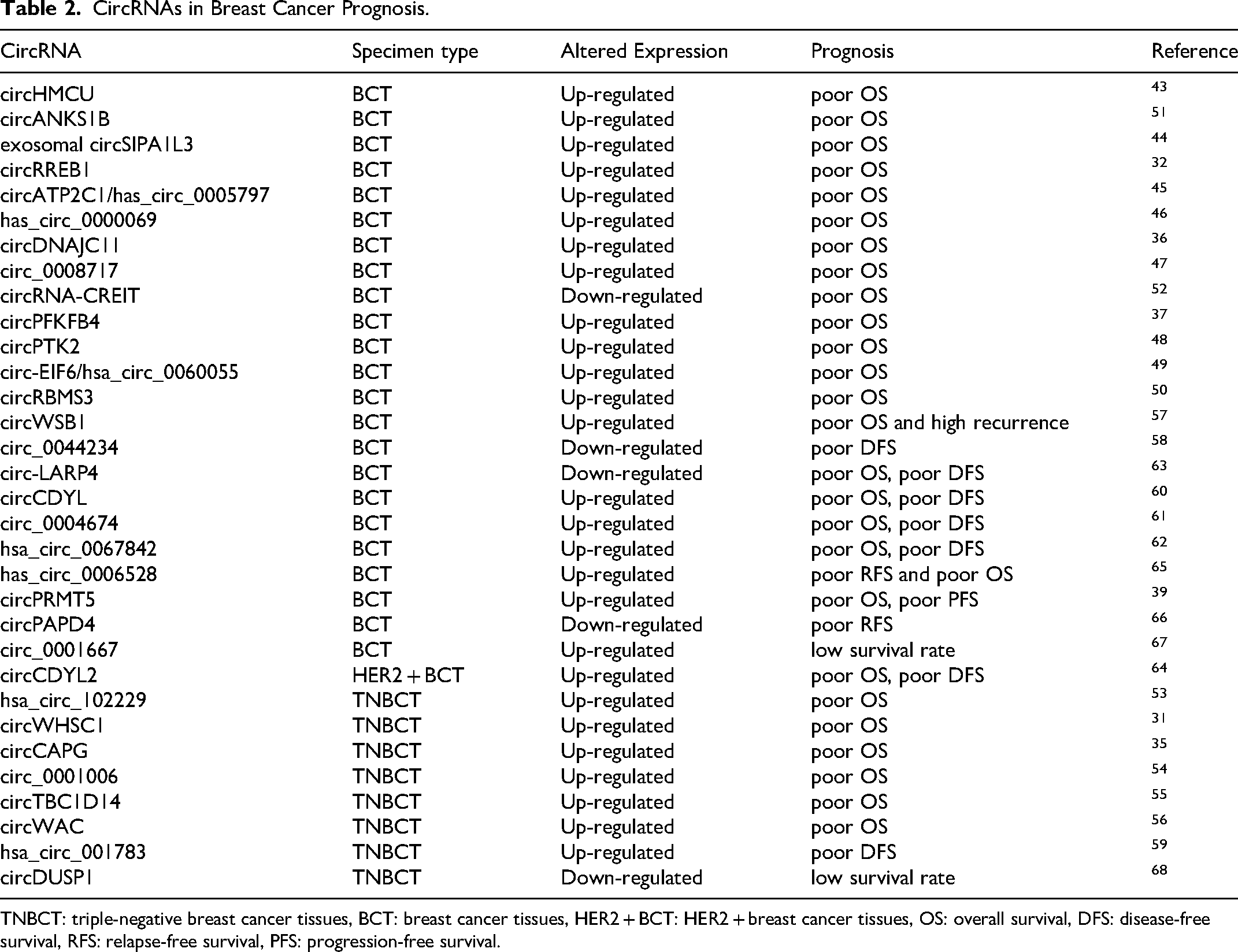
TNBCT: triple-negative breast cancer tissues, BCT: breast cancer tissues, HER2 + BCT: HER2 + breast cancer tissues, OS: overall survival, DFS: disease-free survival, RFS: relapse-free survival, PFS: progression-free survival.
CircRNAs can Distinguish Between TNBC and non-TNBC
The subtype of triple-negative breast cancer (TNBC) is characterized by the absence of three specific receptors: human epidermal growth factor receptor 2 (HER2), progesterone receptors (PR) and estrogen receptors (ER), 69 which account for about 15%-20% of all breast cancers. 70 This subtype is associated with poor prognosis, limited treatment options and higher recurrence rates, 71 with more than 50% of patients relapsing within 3-5 years of diagnosis 72 and the median OS currently treated being 10.2 months. 73 CircRNAs have different expression levels in different molecular types of breast cancer, and could be used as markers to distinguish different molecular types, especially in distinguishing TNBC from non-TNBC (Table 3). Compared with non-TNBC tissues, hsa_circ_0006220, 74 circ_0044234, 58 circ_FOXO3, 75 circNR3C2 76 and circ_0000977 77 were significantly down-regulated in TNBC tissues. Compared with luminalA/B and HER2-positive molecular types, the expression level of circ_0044234 in TNBC is the lowest, and the difference is the greatest between the TNBC with the luminal A type breast cancer. 58 There was no significant difference in the expression of circCD44 in non-TNBC and corresponding adjacent tissues, while in TNBC and corresponding adjacent tissues, circCD44 was up-regulated in TNBC tissues, indicating that circCD44 may be a diagnostic marker of TNBC. 78 CircBRAF, 79 circZCCHC2, 34 circANKS1B, 51 has_circ_001783 59 and circRPPH1/has_circ_000166 80 were significantly up-regulated in tumor tissues of TNBC patients.
CircRNAs in Distinguishing Molecular Types.

TNBCT: triple-negative breast cancer tissues.
CircRNAs as Markers to Predict Breast Cancer Metastasis
Lymph node metastasis is the most common mode of breast cancer metastasis, and some circRNAs can be used as markers to predict lymph node metastasis (Table 4). Circ_0000160, 81 circ-FOXO3 75 and circDUSP1 68 were significantly down-regulated in breast cancer tissues with lymph node metastasis, and hsa_circ_0000091 was significantly down-regulated in plasma of patients with lymph node metastasis. When plasma hsa_circ_0000091 combined with ultrasound was used to diagnose lymph node metastasis of breast cancer, the diagnostic accuracy was increased. 82 CircWHSC1 83 and circZCCHC2 34 are highly expressed in breast cancer tissues with lymph node metastasis, and the expression of circEGFR in breast cancer lymph node metastasis tissues is higher than that in non-metastatic tissues. 84
CircRNAs in Breast Cancer Metastasis.
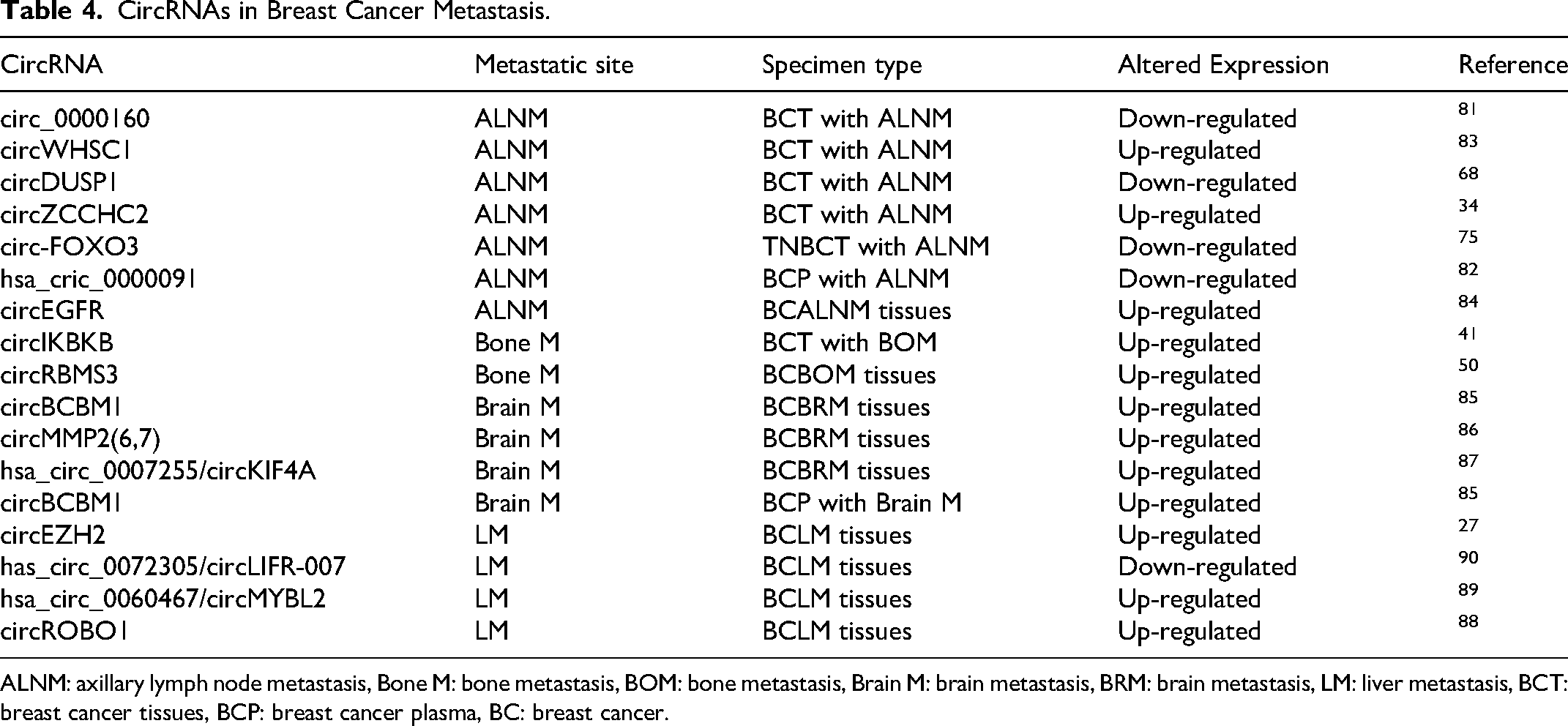
ALNM: axillary lymph node metastasis, Bone M: bone metastasis, BOM: bone metastasis, Brain M: brain metastasis, BRM: brain metastasis, LM: liver metastasis, BCT: breast cancer tissues, BCP: breast cancer plasma, BC: breast cancer.
Differential expression of circRNAs is also present in breast cancer patients with and without distant metastases. CircIKBKB is not detected in normal breast tissues, and its expression level is upregulated in breast cancer tissues. Moreover, it is higher in metastatic compared to non-metastatic cases. Identifying the expression of circIKBKB in patients with breast cancer is helpful for diagnosis and treatment of patients with breast cancer complicated with bone metastases. 41 CircRBMS3 is highly expressed in breast cancer bone metastases and may be a potential marker for predicting bone metastasis in patients. 50
CircBCBM1 is significantly upregulated in both breast cancer brain metastases tissue and plasma. 85 CircMMP2(6,7) is stepwise upregulated from breast cancer tissue without brain metastases, breast cancer tissue with brain metastases to brain metastases tissue of breast cancer. 86 The level of hsa_circ_0007255/circKIF4A is elevated in brain metastases, and it holds promise as a new biomarker for the diagnosis and treatment of TNBC brain metastases. 87
CircEZH2, 27 circROBO1 88 and hsa_circ_0060467/circMYBL2 89 were significantly up-regulated in breast cancer liver metastasis tissues. Has_circ_0072305/CircLIFR-007 was significantly down-regulated in liver metastasis tissues. 90
CircRNAs as Markers to Assess Sensitivity to Drug Therapy
Drug therapy for breast cancer is effective, but long-term drug therapy may lead to drug resistance, which is unfavorable to the prognosis of breast cancer patients (Table 5). Doxorubicin (ADR) is a broad-spectrum antitumor drug that inhibits DNA and RNA synthesis. Circ_0006528, 91 circ_0001667, 92 circ_0044556 93 and circATXN7 94 are all up-regulated in ADR-resistant breast cancer tissues and cell lines.
CircRNAs in Breast Cancer Drug Resistance.

BCT: breast cancer tissues, BCS: breast cancer serum, ADR: doxorubicin, PTX: paclitaxel, TAM: tamoxifen.
Paclitaxel (PTX) is a common chemotherapy drug for breast cancer. However, with the passage of treatment time, the efficacy and effect of PTX will decline, and breast cancer patients who receive PTX treatment for 6-10 months will develop resistance to PTX.95,96 Circ-RNF111, 97 circ_0069094 98 and circ-ABCB10 99 are up-regulated in PTX-resistant breast cancer tissues. CircABCB1 is highly expressed in breast cancer compared to normal tissues, and is even more expressed in docetaxel-resistant patients than in docetaxel-sensitive patients. 100
Tamoxifen (TAM) is an estrogen receptor modulator and is a postoperative adjuvant therapy mainly for patients with ER/PR receptor-positive breast cancer. The expression of circTRIM28 101 and circ_UBE2D2 102 was significantly increased in tamoxifen-resistant tissues, and the expression of circ_UBE2D2 was also increased in exosomes derived from breast cancer resistant cells.
Trastuzumab significantly improves the prognosis of HER2 positive breast cancer patients, and consequently, trastuzumab resistance greatly hinders the treatment of HER2 positive breast cancer patients. Circ-β-TrCP was significantly increased in the serum of trastuzumab resistant subjects, and the circ-β-TrCP derived β-TrCP-343aa protein promoted trastuzumab resistance by elevating expression of NRF2 and antioxidant genes. 103 Circ-BGN 104 and circCDYL2 64 were significantly overexpressed in trastuzumab resistant tissues, and HER2-positive patients with high circCDYL2 expression had a higher recurrence rate after trastuzumab treatment. 64
Conclusion and Prospect
The incidence of breast cancer has been increasing year by year, which is a serious threat to women's health. As an emerging class of non-coding RNA, circRNAs with a covalently closed circular structure are more stable than linear RNAs and less susceptible to degradation by nucleases. This makes them more stable in body fluids, and therefore suitable for liquid biopsy. Detecting circRNAs through body fluids is non-invasive, which enhances patient comfort and compliance. Moreover, circRNAs expression is dysregulated in breast cancer, playing a crucial role in the carcinogenesis, progression, and drug resistance of breast cancer through various mechanisms, and thus circRNAs have emerged as promising biomarker candidates for breast cancer diagnosis, prognosis, molecular subtyping, metastasis, and drug resistance.
The clinical application of circRNAs as markers of breast cancer still has several problems. Despite promising results from preclinical and exploratory studies, circRNAs have not been extensively validated in large-scale clinical trials. The insufficient clinical validation hinders their translation into routine clinical practice for breast cancer diagnosis, prognosis, and monitoring treatment responses. More large-scale clinical trials are needed to validate their sensitivity, specificity, and reliability. Another limitation is that circRNAs detection is costly and not fully standardized, and there may be consistency issues in results between different laboratories. This requires further technical optimization and the establishment of standardized protocols. Moreover, the functional role of specific circRNAs in breast cancer remains largely unexplored. While some circRNAs have been implicated in key oncogenic processes, the exact mechanisms through which they influence tumor behavior and treatment responses are not yet fully understood. In the future, more active research should be carried out on more precise mechanisms of circRNAs in breast cancer. Additionally, the ideal diagnostic strategy is often multimodal, combining various methods to enhance accuracy. For example, circRNAs can be used in conjunction with mammography and other imaging techniques (such as ultrasound and mammography) to provide a more comprehensive diagnostic picture. CircRNAs have shown immense potential as biomarkers in breast cancer, and their further exploration in clinical trials and other applications will be essential to fully realize their value in the future.
At present, many efforts have been made to address these challenges, such as the emergence of new, highly sensitive detection methods. For example, a small number of clinical samples indicate that, different from quantitative polymerase chain reaction (qPCR), the tetrahedral DNA framework (TDF) sensor does not require an additional target amplification step involving multiple enzymes and procedures. Therefore, it exhibits the advantages of simplicity, ease of operation and versatility, and is expected to become a new detection method. 105 Some circRNAs have entered clinical trials, and it is expected that their unique diagnostic and therapeutic value will be seen in clinical practice in the future. The ongoing clinical trials and studies will help to further explore their clinical applicability and validation. For instance, hsa_circ_0001785 and hsa_circ_100219 may potentially be used as diagnostic and prognostic biomarkers for human breast cancer in the future (NCT05771337, https://clinicaltrials.gov/study/NCT05771337). Furthermore, with the emerging functions and mechanisms of circRNAs in breast cancer, they can not only serve as biomarkers but also as therapeutic targets. For example, anti-sense oligonucleotide (ASO)-targeting circPVT1 inhibits ERα-positive breast cancer cell and tumor growth, re-sensitizing tamoxifen-resistant ERα-positive breast cancer cells to tamoxifen treatment. Thus, circPVT1 may serve as a diagnostic biomarker and therapeutic target for ERα-positive breast cancer in the clinic. 106 Recent studies highlighted the therapeutic potential of exploiting tumour-specific circRNAs in the development of effective antigen-based cancer vaccines, including peptide vaccines and circRNA vaccines. It suggests that vaccination utilizing tumour-specific circRNAs may serve as an immunotherapeutic strategy against malignant tumours. 107 Currently, research on the circFAM53B-219aa dendritic cell vaccine treatment has entered clinical trials (NCT06530082, https://clinicaltrials.gov/study/NCT06530082).
In conclusion, more and more studies have shown the value of circRNAs as tumor biomarkers and potential therapeutic targets. This review focuses on the role of circRNAs as tumor biomarkers in breast cancer diagnosis, prognosis, molecular types, metastasis and drug resistance, highlighting the significant implications for future circRNAs research. Although further research is needed, the promise of circRNAs as tumor biomarkers for breast cancer is promising.
Footnotes
Acknowledgements
We would like to thank Dr Michael N Routledge (University of Leicester) and Prof. Yun Yun Gong (University of Leeds) for language editing and valuable comments.
Author Contributions
YW and KZ conceived the idea. LX, YH, LD, ZQ, SC, and YD searched the literature, designed and draw all the figure and tables. YW and KZ supervised the whole work and revised the manuscript. LX and YH wrote the paper. All authors read and approved the final manuscript.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
Since this is a review article, there are no new datasets generated or analyzed. All relevant data and information cited in this review are available in the original research articles referenced throughout the manuscript. For any additional inquiries or access to specific data sets, readers are encouraged to refer to the cited studies.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Considerations
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation (No. ZR2021MH045), and Special Funds for Scientific Research on Breast Diseases of Shandong Medical Association (No. YXH2021ZX058).
