Abstract
Introduction
Lung cancer is the main cause of cancer deaths around the world in older people. China's smoking rate is rising sharply, and lung cancer incidence is expected to peak. 1 More than 40% of lung cancer cases are adenocarcinoma. 2 Low-dose computed tomography (CT) can detect early pulmonary nodules, which is very important for lung cancer screening and can save lives. 3 However, the false positive rate, unnecessary invasive procedures, and overdiagnosis are still major concerns. 4 Although tissue biopsies are very interesting and useful, there are limitations. For example, a single biopsy may not be representative and may misjudge its genetic complexity due to tumor heterogeneity. 5 More recently, much attention has been paid to circulating biomarkers. Biomarkers in circulating or other body fluids, especially respiratory samples may be used as a key strategy to improve the early diagnosis of lung cancer. A metal oxide semiconductor sensor array-based electronic nose system has been discovered to classify breath samples from healthy controls, patients with lung cancer, and COPD, by identifying the amount of volatile organic compounds. Furthermore, COPD patients with lung cancer have higher levels of alpha 1-antitripsin and neutrophils and lower of cholesterol. These parameters could be potential biomarkers for predicting lung cancer in patients with COPD. 6 Blood cadmium level was found may be a valuable early marker for lung cancer detection, especially in former smokers. 7 Liquid biopsy, such as circulating cell-free DNA, tumor-derived exosomes, circulating tumor cells, tumor-educated platelets, and circulating micro-RNAs is an emerging technology with potential roles in screening and early detection of lung cancer. 8 In fact, in the early stages of lung cancer, the low value or lack of most liquid biopsy components limit their applicability. Larger and more powerful researches are needed to define and verify the role of liquid biopsy in the clinical practice of screening or diagnosing lung cancer. 8 Future lung cancer screening will be greatly improved by the use of spirometry as part of a broader clinical evaluation and by biomarker-based clinical decision making in CT-based lung cancer screening. 9
At present, the main diagnostic serum markers of lung cancer are CEA, CYFRA 21-1, NSE, Progastrin-Releasing Peptide, and Squamous Cell Carcinoma Antigen. However, none of these biomarkers are specific for lung cancer. 10 New biomarkers with higher sensitivity and specificity are urgently needed.
Microtubules are cytoskeleton components associated with a variety of cellular processes, including signal transduction, mitosis, transportation, and motility. Microtubules are composed of α-β tubulin heterodimers.11,12 In some cancers, compared with adjacent healthy tissue, the isotype composition of tubulin is perturbed. Importantly, these aberrant tubulin isotypes correlate with the treatment response and patient outcome. 13 Thus, to improve the clinical use of tubulin-targeted chemotherapeutic agents such as paclitaxel and to develop more effective anti-cancer therapies, the role of tubulin isotypes may be considered beyond their structural function. Mainly due to the lack of tools for assessing the importance of α-tubulin isoforms, and the specificity of tubulin-binding agents for β-tubulin, the importance of α-tubulin proteins has been minimized in the majority of studies in this field. 13 However, with further development of our understanding of tubulin and tumor biology, α-tubulin isoforms, similar to β-tubulin isoforms, are becoming increasingly relevant to the outcome of cancer patients.
There are six isoforms of α-tubulin, and the role of each isoform is unclear.14,15 Among the six isoforms, TUBA1B, also known as tubulin alpha-1b, and K-α1 tubulin, localized on chromosome 12q13.12, is a gap junction protein commonly expressed in most human organs and tissues. There is evidence showing that TUBA1B is associated with poor prognosis of mantle cell lymphoma. 16 Increased TUBA1B expression is also associated with resistance to paclitaxel and poor overall survival in patients with hepatocellular carcinoma. 17 Additionally, hepatocellular carcinoma patients with high TUBA1B expression have a better response to immune checkpoint inhibitors. 18 Furthermore, increased mRNA expression of TUBA1B is observed in thyroid anaplastic carcinomas. 19 Moreover, the differential model of pancreatic ductal adenocarcinoma containing TUBA1B was established with good effect. 20 Differentially regulated proteins were investigated between normal bronchial epithelium and human small cell lung cancer tissues by proteomic analysis and TUBA1B was discovered as one of the proteins that were up-regulated more than 2 fold in small cell lung cancer tissues. 21 To date, there is no study on the clinical significance of TUBA1B in patients with LUAD.
The aim of this research was to detect TUBA1B expression in tissues and sera of LUAD patients and correlate its expression with clinicopathologic characters of LUAD. Finally, we explored the possible role of TUBA1B in the prognosis and diagnosis of LUAD. In our study, we first assessed the differential expression of TUBA1B in tissues, sera of LUAD and normal. Furthermore, we evaluated the relationship between the expression of TUBA1B and the clinicopathology and prognosis of LUAD. Our findings provide new targets for the diagnosis, treatment, and prognosis of LUAD.
Methods
Ethics Approval
All procedures involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). We have de-identified details of all patients. The study was approved by the Ethical Committee of (conceal). The reporting of this study conforms to REMARK guidelines. 22
Tissue Microarray and Immunohistochemistry (IHC)
The tissue microarray, HLugA180Su03, containing 92 LUAD cases, was purchased from Shanghai Outdo Biotech (Shanghai, China) and retrospectively analyzed. The operation time of the patients was January 2008 to July 2013 and the follow-up time was June 2016. Two LUAD tissue samples were mostly necrotic and were not included in the analyses. The remaining 90 LUAD cases contained in the tissue microarray were collected from 49 males and 41 females. Due to incomplete examination and difficulty of information collection, clinical data for some patients presented in the tissue microarray such as tumor size, TNM stage, or lymph node status is absent, resulting in not all of the sum of the numbers being 90. Table 1 summarizes representative clinical data. The mean age of all LUAD patients at the time of surgery was 63 (range: 35–86 years). There were 42 patients with tumor volumes ≥10 cm3 and 35 with tumor volumes <10 cm3. The numbers of patients with T1, T2, T3, and T4 cancers were 23, 36, 10, and 8, respectively. 34 patients were positive for lymph node metastasis, while distant metastasis was noted in one patient. The numbers of patients at stages I, II, III, and IV were 25, 16, 23, and 1, respectively, according to the TNM staging system.
Correlation of TUBA1B Expression with Clinicopathological Characteristics of LUAD Patients.

Statistically significant.
The primary TUBA1B antibody was a rabbit anti-human IgG polyclonal antibody (ab97622, 1:500; Abcam, Cambridge, UK). SPlink Detection Kits (SP-9000, Biotin-Streptavidin HRP Detection Systems) were purchased from Beijing Zhongshan Jinqiao Biotech (Beijing, China). IHC was performed according to the manufacturer's instructions. Briefly, the tissue microarray was deparaffinized and rehydrated. Antigens were recovered by a Citra Plus antigen retrieval solution using an autoclave for 30 min. The 3% hydrogen peroxide was then added to quench endogenous peroxidase activity at room temperature for 15 min. After the slide was blocked by normal serum at 37°C for 40 min, the primary antibody was added and incubated at 4°C overnight. The biotinylated secondary antibody was then added for 40 min at 37°C followed by the avidin-biotin-peroxidase complex for 40 min. The 3, 3′-diaminobenzidine chromogen substrate was added for about 1 min to produce a brownish-yellow reaction product. The microarray was finally counterstained with hematoxylin. All images were captured by a DM4000 microscope (LEICA, Wetzlar, Germany) equipped with DFC450 C camera (LEICA) and 20×, 40× objective lens (LEICA). LAS V4.5 controller software (LEICA) was used for image processing. Two pathologists independently examined all immunostained sections without knowing the patients’ clinical pathology variables. For determining TUBA1B expression, the immunostaining intensity was assessed as 0, 1, 2, and 3. An immunostaining score ≤1 was identified as weak TUBA1B expression and score >1 was identified as strong TUBA1B expression.
Collections of Human Serum Samples
All serum samples were collected from (conceal) from June 2018 to January 2020 and included 55 patients with LUAD before therapy, 35 patients with LUAD after therapy (at least one month after surgery or chemotherapy), 32 age-matched patients with benign lung diseases and 43 age-matched healthy individuals. The inclusion and exclusion criteria for LUAD patients before therapy in this study were as follows: (1) LUAD was confirmed by experienced pathologists through histological examination; (2) sera were collected from patients before any therapy.
The data was obtained from the electronic medical record system and retrospectively studied. Age-matched healthy persons or patients diagnosed with benign lung diseases such as pulmonary infection, chronic obstructive pulmonary disease, and chronic bronchitis were classified as controls. LUAD patients included 57 men and 33 women with an average age of 65 years (range: 42–87 years). The patients with benign lung diseases included 17 men and 15 women with an average age of 66 years (range: 39–87 years). Healthy persons included 19 men and 24 women with an average age of 56 years (range: 38–87 years). Serum samples were collected and stored at −80°C until detection. The serum samples were not conducted for further follow-up investigations.
Enzyme-Linked Immunosorbent Assay (ELISA)
Total protein levels of TUBA1B in serum were measured using a commercially available TUBA1B ELISA Kit (AE12822HU, Wuhan Abebio Science, Wuhan, China). The quantitative sandwich enzyme immunoassay technique was used for all assays. 100 µL of standards or serum samples were added to each well and incubated at 37°C for 2 h. After washing the wells, 100 μL of the biotin-conjugate was added to each well and incubated at 37°C for 1 h. Then, after washing, added 100 μL of streptavidin-horseradish peroxidase and incubated at 37°C for 1 h. After a final washing step, 100 μL of substrate solution was added and incubated at 37°C for 15-20 min in the dark. Finally, when the wells containing the four highest concentration of standards produced a conspicuous blue color, added 50 μL of stop solution. The absorbance of each well was measured within 5 min with a microplate reader set at 450 nm and wavelength correction at 540 nm.
Serum RNA Isolation and Quantitative Real-Time PCR (qRT-PCR) Analysis
The transcriptome levels of TUBA1B in serum of 42 patients with LUAD before therapy, 31 healthy persons were quantified by qRT-PCR analysis. Total RNA was isolated from 200 uL of serum according to the instructions of total RNA extraction kit (R1200, Beijing Solarbio Science & Technology, Beijing, China). RNA was reversely transcribed into cDNA using FastKing gDNA dispelling RT SuperMix kit (KR118, Beijing Tiangen Biotech, Beijing, China). QRT-PCR was done in triplicate. Relative TUBA1B expression was calculated by comparing threshold cycle (Ct) values [2−△△Ct] with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as the internal control. Primers for TUBA1B were: forward, 5’ CACCATCAAAACCAAGCGCA 3’, reverse, 5’ GCACGCTTGGCATA CATCAG 3’.
UALCAN
The network resource UALCAN (http://ualcan.path.uab.edu) from TCGA database can perform in silico verification of target genes. 23 In this study, it was used to explore the transcriptional expression of TUBA1B and its association with clinicopathological features and overall survival in LUAD.
Statistical Analyses
All graphed plots and statistical analyses were performed with softwares Prism 6 (GraphPad, La Jolla, CA, USA) and SPSS 17 (IBM, Chicago, IL, USA). The association between TUBA1B expression and representative clinicopathological factors of LUAD patients was evaluated by the Chi-square test or Yates’ continuity corrected Chi-square test. Intergroup differences of TUBA1B levels in serum were determined by Kruskal–Wallis test and Dunn's multiple comparisons test. Survival curves were carried out by the Kaplan–Meier method and log-rank test was used to compare differences in survival rates between subgroups. The multivariate analysis by Cox proportional hazards model was used to determine predictive factors. P-values < .05 were considered to be significant.
Results
Detection of TUBA1B in LUAD Tissues by IHC
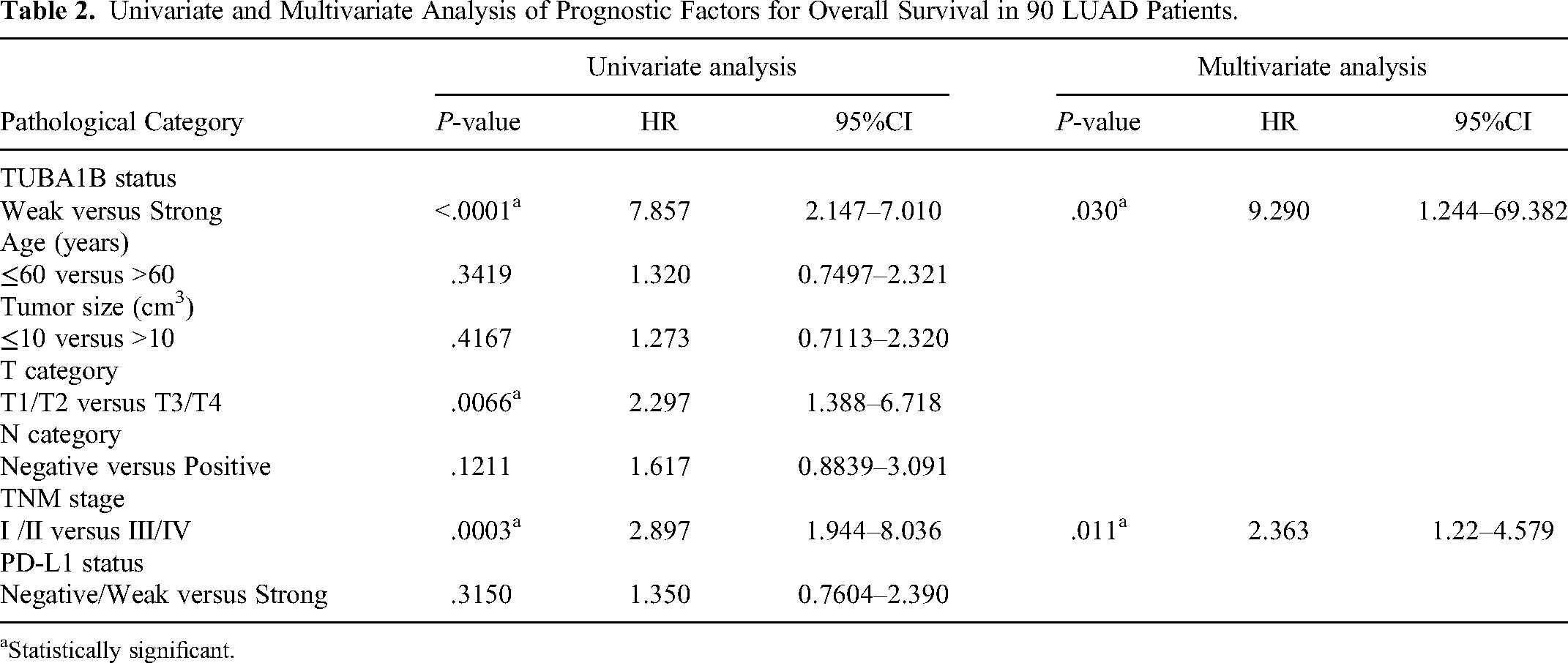
IHC staining indicated that TUBA1B expression was highly expressed in most LUAD tissues and was absent or low in adjacent nontumor tissues (Figure 1A). Positive IHC staining of TUBA1B was found in both the nucleus and cytoplasm of LUAD cells. Table 1 summarizes the association between TUBA1B expression and representative clinicopathological factors. High TUBA1B protein expression was statistically correlated with positive lymph node metastasis (P = .031). Other factors such as age, tumor size, T category, TNM stage, and PD-L1 status, were almost unrelated to TUBA1B expression. The results of Kaplan–Meier univariate analysis indicated that several factors were associated with overall survival of the 90 LUAD patients. These factors included TUBA1B protein expression (P < .0001), T category (P = .0066), and TNM stage (P = .0003) (Table 2). The Kaplan–Meier survival curves showed that compared with LUAD patients with low expression of TUBA1B, the survival rate of patients with high expression of TUBA1B was significantly decreased (Figure 1B). In addition, multivariate analysis revealed that TUBA1B (HR: 9.290, 95% CI: 1.244–69.382, P = .030) and TNM stage (HR: 2.363, 95% CI: 1.22–4.579, P = .011) were two independent predictors of overall survival in LUAD patients (Table 2).

IHC staining of TUBA1B in LUAD on tissue microarray. (A) Representative images of IHC staining of TUBA1B in LUAD and adjacent normal tissues. Intensity of IHC staining in LUAD was assessed as 0, 1, 2, and 3 (IHE DAB coloration, 200×). (B) Kaplan-Meier curves for overall survival of 90 LUAD patients. The overall survival rate of patients with higher expression of TUBA1B was significantly decreased. (C) The overall survival rate of LUAD patients is obviously different between stages I/II and III/IV.
Univariate and Multivariate Analysis of Prognostic Factors for Overall Survival in 90 LUAD Patients.
Statistically significant.
Serum TUBA1B Levels in LUAD Patients
To compare the expression of TUBA1B in the serum of LUAD patients with healthy individuals, serum TUBA1B levels were detected by ELISA before any therapy was initiated. The results of ELISA showed that mean protein levels of serum TUBA1B were significantly elevated in LUAD patients before therapy (17.2 ± 6.1 ng/mL) compared with age-matched healthy persons (4.2 ± 2.6 ng/mL) or patients with benign lung diseases (6.1 ± 3.5 ng/mL) (Figure 2A, P < .0001). To evaluate the performance of TUBA1B protein in distinguishing between LUAD patients and healthy persons, receiver operating characteristic (ROC) curve was performed. According to the ROC, the area under the curve (AUC) was 0.99 (P < .0001, 95% confidence interval 0.98–1.0). At a concentration of 9.3 ng/mL, the sensitivity was 94.6% and the specificity was 97.7% for TUBA1B as a LUAD biomarker (Figure 2B, P < .0001). An ideal tumor biomarker should also be able to predict clinical outcomes. Importantly, soluble TUBA1B levels significantly decreased after effective treatment (Figure 2A, P < .0001). To compare with common clinical biomarkers, we collected the results of CEA, CYFRA 21-1, and NSE which are commonly used in hospital tests and 42 LUAD patients were tested for these three items. The results showed that the positive detection rate of TUBA1B was 92.9%, which was significantly higher than that of the common biomarkers (the highest was 52.4%), showing a great advantage in sensitivity (Figure 2C). However, the mean mRNA levels of serum TUBA1B in LUAD patients were not significantly different from the healthy persons according to qRT-PCR analysis, as shown in Figure 2D. The patient number and power size have been calculated by software PASS. Group sample sizes of 55 and 43 achieve 100.0% power to reject the null hypothesis of equal means when the population mean difference is μ1–μ2 = 17.2–4.2 = 13.0 with standard deviations of 6.1 for group 1 and 2.6 for group 2, and with a significance level (alpha) of 0.025 using a one-sided two-sample unequal-variance t-test.
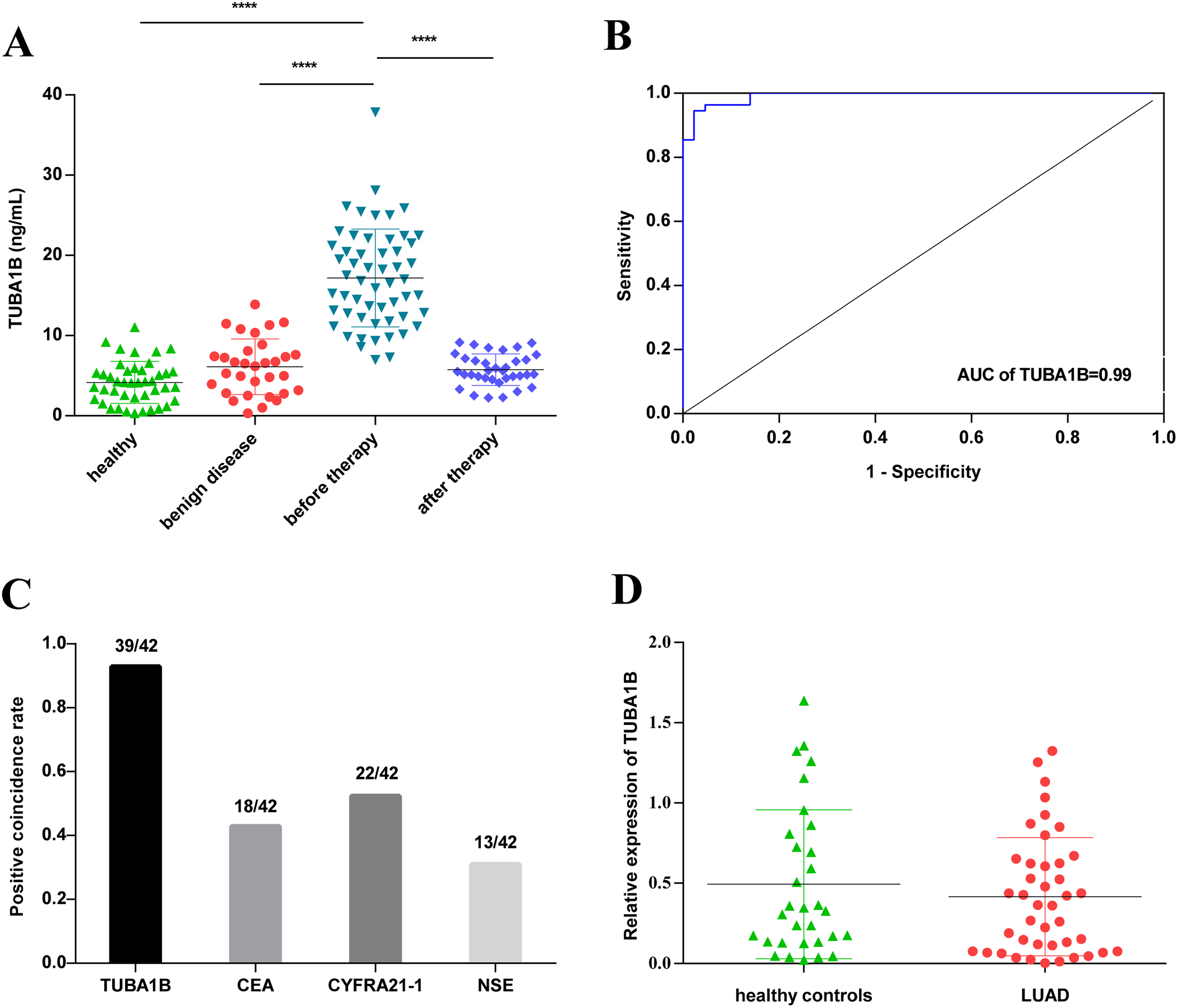
Serum TUBA1B levels in LUAD patients. (A) TUBA1B levels in the serum of healthy individuals (n = 43), patients with benign diseases (n = 32), LUAD patients before (n = 55) and after (n = 35) therapy. The TUBA1B concentration was measured in duplicate with ELISA. (B) ROC curve of data from (A) showed an AUC of 0.99. The cut-off level was 9.3 ng/mL with sensitivity of 94.6% and specificity of 97.7%. (C) The positive coincidence rate of TUBA1B was compared with CEA, CYFRA 21-1 and NSE detected in the same LUAD samples. (D) Serum mRNA levels of TUBA1B in LUAD patients (n = 42) and healthy individuals (n = 31) were analyzed by qRT-PCR. The TUBA1B expression levels were normalized by GAPDH. There was no significant difference in TUBA1B mRNA expression between LUAD patients and healthy individuals. *P < .05; **P < .01; ***P < .001; ****P < .0001.
Validation of Clinical Relevance of TUBA1B in LUAD Patients by UALCAN
The UALCAN analysis showed that TUBA1B is overexpressed in primary LUAD (n = 515) compared to normal lung tissues (n = 59) (Figure 3A). We also observed increased TUBA1B expression in N0, N1, N2 compared to normal tissues (Figure 3B). The results also demonstrated that TUBA1B expression was significantly correlated with the stages (Figure 3C). Similar to these observations, LUAD patients with high expression of TUBA1B (n = 125) showed reduced survival probability (P = .0041) compared with patients with low or moderate expression (n = 377) (Figure 3D).

UALCAN analysis was performed on LUAD samples from the database TCGA. (A) Comparison of TUBA1B expression between normal lung tissues and LUAD tissues. (B) Expression analysis of TUBA1B in different N categories of LUAD. (C) Expression analysis of TUBA1B in different stages of LUAD. (D) Analysis of survival probability between LUAD patients with high TUBA1B expression and low/medium TUBA1B expression. *P < .05; **P < .01; ***P < .001; ****P < .0001.
Discussion
TUBA1B originally discovered as a potential housekeeping protein in research, is related to the microtubule formation. 24 It is well known that microtubules are closely related to the regulation of cell mitosis, signal transduction, transportation, and motility. Therefore, TUBA1B may be related to the progression and invasion of cancer cells, thereby affecting the pathology of tumors.
In this study, the IHC results of the tissue microarray showed a significant increase of TUBA1B expression in LUAD tissues compared to adjacent nontumor tissues. This suggests that TUBA1B might be closely involved in the progression of LUAD. Furthermore, in LUAD patients with lymph node metastasis, the expression of TUBA1B was significantly higher than those without lymph node metastasis. Except for the N category, there was no correlation between TUBA1B expression and age, tumor size, T category, TNM stage, or PD-L1 status. Survival analysis indicated that LUAD patients with higher TUBA1B expression (IHC score 2, 3) had a shorter overall survival. Moreover, multivariate analysis demonstrated that TUBA1B was also an independent prognostic factor for LUAD besides pathology stage. In this study, we further found that serum levels of TUBA1B protein in patients with LUAD were significantly elevated than those in patients with benign lung diseases or healthy individuals. The serum levels of TUBA1B protein in patients with benign lung diseases were similar to those of healthy people, indicating that benign lung diseases such as inflammation, COPD would not lead to the increase of TUBA1B protein in serum, so the diagnostic specificity of serum TUBA1B protein for LUAD was relatively high. The ROC curve demonstrated that the AUC value of serum TUBA1B protein was 0.99 (95% confidence interval 0.98–1.00) with a sensitivity of 94.6% and a specificity of 97.7%. In addition, the positive detection rate of traditional biomarkers CEA, CYFRA 21-1 and NSE in LUAD was up to 52.4%, and that of TUBA1B was 92.9%, showing a significant advantage in sensitivity. The results of UALCAN analysis also verified that mRNA expression of TUBA1B was overexpressed in primary LUAD patients compared to normal lung tissues, and mRNA expression of TUBA1B was significantly associated with N categories and stages. Besides, higher expression of TUBA1B was significantly associated with shorter overall survival in LUAD patients. These results suggest that, besides being a diagnostic marker, TUBA1B may also be a potential prognostic marker.
Although few studies have reported a link between TUBA1B and LUAD, there are evidences that TUBA1B expression is related to poor prognosis in mantle cell lymphoma, 16 hepatocellular carcinoma,17,18,25 and thyroid carcinoma. 19 In Wilms’ tumor, the expression of TUBA1B was identified to be significantly upregulated than that in normal tissues and TUBA1B coexpressed genes were enriched in cell cycle, mismatch repair, DNA replication, spliceosome, and pathogenic Escherichia coli infection pathways. 26 The major tubulin isotypes in both the human non-small cell lung cancer cell line A549 and breast cancer cell line MDA-MB-231 are TUBA1B and TUBB1. 27 Furthermore, CLDN11 inhibited the migration of cancer cells by interacting with TUBA1B and blocking tubulin polymerization in nasopharyngeal carcinoma. 28 In osteosarcoma, TUBA1B was one of the endogenous genes considered to be a key regulator. 29 These findings indicate that TUBA1B may play roles in the occurrence and progression of these cancers. However, there are also investigations that present contradictory findings. For example, low expression of TUBA1B was found to predict relapse among stage II colorectal cancer patients. 30 The expression of TUBA1B was also significantly reduced in metastatic uveal melanomas compared to those without metastasis. 31 The reason for these differences is unclear, and requires further investigations.
Essential roles for TUBA1B in the development and growth of adipose tissue have been proposed. 32 Both retinoic acid-receptor-related orphan receptor-α (RORA) and peroxisome proliferator-activated receptor-α (PPARA) are involved in the regulation of lipid metabolism. By negatively and positively correlating the expression of TUBA1B with PPARA and RORA activation, respectively, the cis-regulatory potential of the TUBA1B silencer, S5, was verified. 33 These two investigations suggest that TUBA1B is involved in lipid metabolism which is closely related to tumorigenesis.
In lung transplant recipients, the post-transplant development of antibodies against TUBA1B correlates with the occurrence of bronchiolitis obliterans syndrome.34,35 Following heart transplantation, autoimmune responses to TUBA1B have also been found to be associated with acute antibody-mediated rejection and vasculopathy. 36 Overall, the results of these studies suggest that anti-TUBA1B may play crucial roles in the pathogenesis of chronic rejection after organ transplantation. The interesting finding was that patients diagnosed with bronchiolitis obliterans syndrome after lung transplant also had exosomes with higher expression of TUBA1B than stable patients. 37 Similarly, Bansal discovered that the presence of TUBA1B in exosomes isolated from lung transplant recipients, which signifies that TUBA1B may dramatically affect transplant biology. 38 Therefore, TUBA1B and anti-TUBA1B may also be used as biomarkers for allograft rejection.
Liquid biopsy such as tumor cells, cell-free DNA, micro-RNAs, tumor-derived exosomes, and tumor-educated platelets in the blood or body fluid is an emerging technology in the early detection of lung cancer. However, the diagnostic performance and clinical applicability remain to be improved. 8 This study shows that TUBA1B has a significant advantage in the sensitivity of diagnosis of LUAD. In addition, these methods are unsuitable for mass screening due to their high cost and complexity. ELISA or other economical and effective methods to detect serum protein TUBA1B may be widely used in the screening of LUAD. E-nose system for volatile organic compound analysis has extraordinary possibilities for distinguishing lung cancer from COPD and controls, and the main advantages are portability, low cost, and stability. 39 But a lot of work needs to be done to make it perfect.
The deficiency of this study lies in the small number of samples. Despite the limitation of the small sample size, the current study is the first to detect the levels of TUBA1B in sera and tissues of LUAD patients and investigate its roles in diagnosing and predicting the prognosis of these patients. This is the first study to find that TUBA1B is elevated in sera and tissues of LUAD patients and high TUBA1B expression is associated with poor prognosis in LUAD patients. TUBA1B has great potential to be a non-invasive prognostic and diagnostic marker for LUAD. Paclitaxel is a drug that mainly targets tubulin. It blocks the cell cycle by inducing microtubule polymerization and inhibiting microtubule depolymerization.40,41 Increased TUBA1B expression has been found to be related with resistance to paclitaxel in patients with hepatocellular carcinoma. 17 Therefore, this drug may be not useful in chemotherapy of TUBA1B overexpressing cancers. Specifically, while the roles of TUBA1B in other cancers and the mechanisms require further detailed investigations, it may be speculated in this study that increased TUBA1B expression in LUAD may be the experimental basis for paclitaxel resistance. Since TUBA1B is present in many tumors, TUBA1B may also exist in the serum of patients with these tumors besides LUAD. Further studies are needed in larger survey samples and longer follow-up times to investigate the possible role of TUBA1B as a broad-spectrum tumor biomarker.
Conclusion
LUAD patients with higher TUBA1B expression had shorter overall survival and protein levels of TUBA1B in serum were obviously elevated in LUAD patients than control. In summary, TUBA1B may be a marker for LUAD diagnosis and prognosis. The study may help refine the group of patients who might benefit from lung cancer screening. Further studies of the mechanisms underlying our observations are warranted.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231178391 - Supplemental material for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis
Supplemental material, sj-docx-1-tct-10.1177_15330338231178391 for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis by Xue-Li Pang, Hong-Fei Du, Fang Nie, Xiang-Gui Yang and Ying Xu in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338231178391 - Supplemental material for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis
Supplemental material, sj-docx-2-tct-10.1177_15330338231178391 for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis by Xue-Li Pang, Hong-Fei Du, Fang Nie, Xiang-Gui Yang and Ying Xu in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-3-tct-10.1177_15330338231178391 - Supplemental material for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis
Supplemental material, sj-docx-3-tct-10.1177_15330338231178391 for Tubulin Alpha-1b as a Potential Biomarker for Lung Adenocarcinoma Diagnosis and Prognosis by Xue-Li Pang, Hong-Fei Du, Fang Nie, Xiang-Gui Yang and Ying Xu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
The authors thank the TCGA for its contribution to human oncology research and the establishment of an open database. The authors also thank the Education Department of Sichuan Province and Chengdu Medical College for their financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the Ethical Committee of The First Affiliated Hospital of Chengdu Medical College (2018CYFYHEC-MS-14, 2019CYFYHEC-MS-14).
Funding
This work was supported by the Education Department of Sichuan Province, China (16ZB0272) and the fund project of Chengdu Medical College in Sichuan Province, China (CYZ18-19)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
