Abstract
Keywords
Introduction
Total body irradiation (TBI) is an important component of conditioning regimens for patients with acute leukemia undergoing hematopoietic stem cell transplantation (HSCT). 1 While the optimal preparative regimen is a subject of controversy, it is thought that the addition of TBI serves to eradicate chemotherapy-resistant clones as well as disease within sanctuary sites while providing host immunosuppression to decrease the risk of graft rejection. In certain malignant conditions, for young patients with good performance status, the combination of high-dose TBI with myeloablative chemotherapy is preferred. 2 Non-myeloablative regimens with low-dose TBI and reduced-intensity chemotherapy are commonly utilized for older patients or those with poor performance status due to the risk of non-relapse mortality (NRM) with more toxic and intensive regimens. Modern retrospective data suggest TBI-containing myeloablative regimens may improve overall survival (OS) despite increased risk of severe pulmonary toxicity.3,4 Similarly, dose escalation may improve freedom from relapse, though may not improve OS due to increased rates of NRM.5–7
Historically, TBI has been delivered with a 2D planning approach with opposed lateral or anteroposterior-posteroanterior (AP/PA) beams at extended source-to-skin distances (SSD) with custom beam spoiler devices to improve dose homogeneity. Custom blocking is typically used to decrease dose to the lungs and kidneys in efforts to reduce the incidence of acute pneumonitis and late pulmonary and renal toxicities. 8 However, many institutions may not have the resources, facilities, or equipment to deliver TBI with this approach, limiting broad adaptation of TBI-containing conditioning regimens.
Multiple groups have described the use of intensity-modulated radiotherapy (IMRT) for delivery of TBI using both volumetric modulated arc therapy (VMAT) and helical tomotherapy (HT).9–11 This technique is appealing as it allows for more homogeneous dose delivery and decreased dose to organs at risk (OARs) which may facilitate safe dose-escalation to optimize treatment efficacy without increasing the risk of NRM. 9 Due to limitations in the multileaf collimator (MLC) expansion length and jaw size, VMAT plans have historically been delivered with two-field alignments leading to the use of arcs only from the head through the mid-femur followed by a 2D AP/PA approach for the remaining lower extremities.12–15 This change in dose delivery technique potentially allows for significant dose heterogenies within the femur as minor positional changes can result in no coverage or overlapping coverage (Figure 1A). With active marrow of the lower extremities primarily existing within the proximal femur in adults, underdosing of this region may theoretically result in undertreatment of disease and lead to relapse.16,17
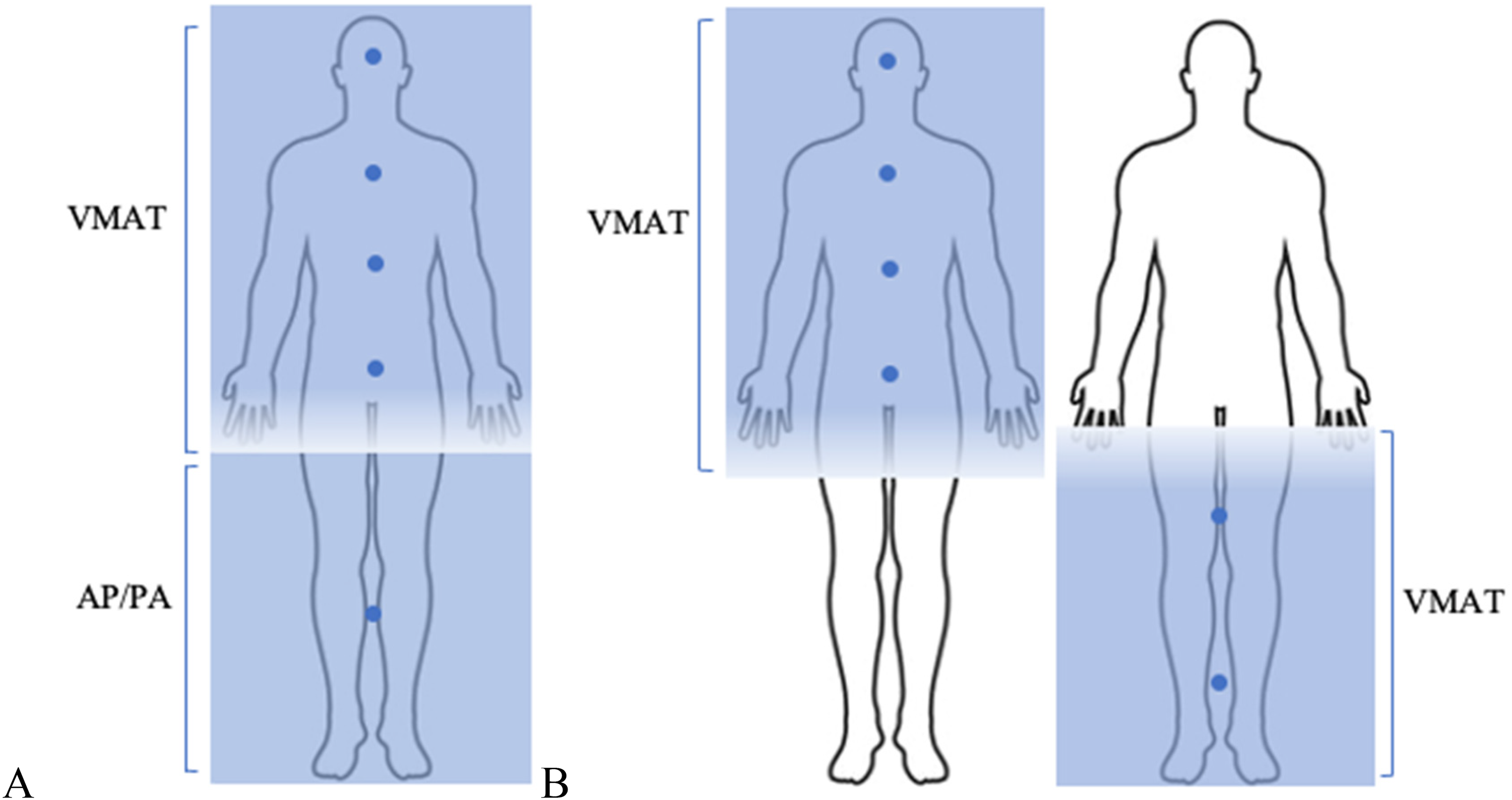
Dose distribution among 2D and VMAT plans.
Some groups have utilized IMRT to not only spare OARs, but to selectively treat only the bone marrow (total marrow irradiation or TMI) or the bone marrow and lymphatics (total marrow and lymphatic irradiation or TMLI) with promising results.18–21 Further, IMRT-based TBI may be less resource-intensive and can be delivered without specialized facilities or equipment, potentially allowing expanded access to this treatment. However, an optimal planning strategy has not been established. In the present study, we describe our institution's technique for high-dose TBI delivered with full-body VMAT and compare dosimetric parameters against patients treated with a previously established HT technique. We also describe patient outcomes of those treated with VMAT using an oropharyngeal mucosal sparing technique that was implemented after two lethal oral mucositis events occurred.
Materials and Methods
Patient Selection
Patients with acute lymphocytic leukemia (ALL) or acute myeloid leukemia (AML) undergoing myeloablative conditioning with high-dose TBI treated with either HT or VMAT at H. Lee Moffitt Cancer Center were retrospectively identified for analysis. Patients were treated between September 17, 2019 and March 25, 2022. The use of HT was discontinued at our institution in 2020 after adaptation of this full-body VMAT technique. All patients met criteria for the eligibility of allogenic transplant as per institutional standard operative procedures. Patients received initial induction chemotherapy and were confirmed to be in remission via bone marrow biopsy and imaging prior to radiation delivery. Patient information was de-identified during data collection. The reporting of this study conforms to STROBE guidelines. 22
Positioning and Immobilization
At a standard source-axis distance (SAD) of 100 cm, the maximum treatment field length is 160 and 120 cm for Tomotherapy and TrueBeam linear accelerators, respectively. Given this limitation, patients were simulated in two positions: first in a head-first supine (HFS) orientation, then in a feet-first supine (FFS) orientation.
Patients treated with HT were simulated in the supine position with arms at their sides. Immobilization was achieved with vacuum cradle device and thermoplastic mask over the head and shoulders. Computed tomography (CT) images were obtained scanning in the HFS direction from the cranial vertex through the midthigh in 5 mm slices. The patient was then rotated 180 degrees and scanned in the FFS direction from feet to midthigh with overlap with the HFS scan.
Patients treated with VMAT were simulated in the supine position with arms at their sides. These patients were positioned on a stereotactic body radiotherapy (SBRT) board using the long SBRT baseplate made by Orfit (Orfit Industries, NV Belgium). A headrest, knee rest, and footrest were added for patient comfort. Then, to achieve immobilization, thermoplastic masks were applied to the head and shoulders, chest and arms, abdomen, and legs and locked onto the SBRT board to create custom-fitted, whole-body mask (Figure 2). The patient then underwent CT simulation in the HFS and FFS positions with 5 mm slice thickness as previously described. A localization isocenter was placed near the base of skull for the HFS scan and near the ankles for the FFS scan. To ensure reproducible alignment, markings were placed on the thermoplastic masks.

Patient simulation.
OARs and Dose Constraints
RayStation treatment planning software (RaySearch Laboratories, Stockholm, Sweden) was used for contouring. Avoidance structures were generated for OARs including the lungs, heart, kidneys, brain, and breasts in female patients. The lungs were contracted 5 mm to generate the lung avoidance structure. The constraints to these avoidance structures are outlined in Supplemental Table 1. After treating 15 patients, an oropharyngeal mucosal sparing technique was adopted with the purpose of reducing morbidity and mortality after two patients experienced Grade 5 mucositis. This OAR was generated by contouring the oropharynx comprising of the oral tongue, floor of mouth, alveolar ridge, buccal mucosa, retromolar trigone, soft palate, posterior pharyngeal wall, base of tongue, glottic mucosal surface, and vallecula and was constrained to a mean dose <7 Gy.
Dose Objectives
Standard high-dose TBI prescriptions were utilized: 1320 cGy was delivered in eight fractions of 165 cGy given twice daily (BID) in 30 patients, and 1200 cGy was delivered in eight fractions of 150 cGy given BID in one patient. The BID fractions were given at least six hours apart to allow for sublethal damage repair. The PTVs were generated differently in VMAT and HT plans as described below. The goal planning parameters are summarized in Supplemental Table 2.
Treatment Planning for VMAT
For VMAT plans, six to eight isocenters were generated in Raystation at the head, thorax, abdomen, pelvis, midthigh, and ankles depending on patient height. In the HFS, four to five isocenters were generated. In the FFS plans, two to three isocenters were generated. The isocenters were placed within 25 cm of each other to ensure sufficient overlap between arcs. Two 365° arcs were planned for each isocenter. To match the dose from the HFS and FFS plans, the HFS treatment planning CT was deformably registered to the FFS treatment planning CT. The HFS dose was deformed, exported to the FFS CT, and added as a beam set to be used as a background dose when optimizing the FFS plan.
VMAT planning was calculated using Monte Carlo in Raystation 11A running on NVIDIA Quadro P4000, with 48 GB VRAM, 32 core Intel Xeon Gold 6254 Processors at 3.1 GHz and 384 GB RAM. The final dose computation with enough histories to achieve 1% uncertainty takes about 200 s with a dose grid set at 3 × 3 × 3 mm3.
Two planning target volumes (PTVs) were created in VMAT plans. A PTVbody was generated to include the whole body with an external contour contraction of 5 mm and subtraction of the OARs with a 5 mm contraction. The dose prescribed to PTVbody was D85%> prescribed dose (Rx). PTVbones was auto-generated using threshold values and included skeletal structures with the purpose of ensuring sufficient coverage of the marrow; this dose was prescribed as D95%>Rx.
The quality assurance (QA) of every individual's plan was performed by medical physicists. IMRT QA of each VMAT plan is performed using SunCHECK (Sun Nuclear Corporation, Melbourne, FL).
Treatment Planning for HT
A single PTV labeled PTVbody was generated to include the entire body excluding OARs. Dose to the PTV was prescribed as D85%> Rx. Planning proceeded as described by Hui et al. 10 Modulation factor and pitch were determined as described by Penagaricano et al. 11 QA for HT plans was performed using a Delta4 phantom and corresponding software (ScandiDos, Sweden).
Treatment Delivery
For VMAT-treated patients, cone beam CTs were acquired for position verification prior to each treatment. VMAT was delivered on a Varian TrueBeam linear accelerator (Varian Medical Systems, Palo Alto, CA) with 6, 10, or 15 MV photons. Energies >10 MV were exclusively reserved for patients with larger body habitus (ie body mass index >30) to obtain a more homogenous dose, achieve prescription goals, and meet OAR constraints. Higher energies typically require fewer MUs. As a result, the contribution from leakage radiation is also reduced. Therefore, the reduction of hotspots and meeting plan constraints outweighed any theoretical concern for the potential increase of secondary cancer risk from neutrons in our adult patient cohort.23–25
For HT-treated patients, megavoltage CT (MVCT) was acquired for position verification prior to each treatment. HT was delivered on an Accuray TomoTherapy device (Accuray Inc. Sunnyvale, California, USA). Patients were seen for on-treatment visits twice during the course of treatment. Patients underwent HSCT one to seven days after completion of TBI. Graft versus host disease (GVHD) prophylaxis was provided for all patients; regimens were selected at the treating transplant physician's discretion.
Endpoints and Statistical Analysis
Patient and treatment characteristics were retrospectively collected via medical chart review. The primary endpoint was to compare dosimetric parameters between patients planned with HT and those with VMAT. OAR mean doses were compared with independent T-tests. The secondary endpoint was the incidence and severity of acute and late toxicities associated with the conditioning regimen and HSCT. Toxicity was determined retrospectively according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Incidence and severity of toxicity were queried from daily progress notes entered into the electronic medical record by medical providers.
Linear regression was performed to test associations between continuous variables. The threshold for statistical significance was set at P < .05. All statistical analyses were conducted using SPSS software (IBM Corp. Released 2021. IBM SPSS Statistics for Macintosh, Version 28.0. Armonk, NY: IBM Corp).
Results
Patients
Thirty-one patients with acute leukemia were treated with high-dose TBI using VMAT or HT at our institution; patient characteristics are summarized in Table 1. The median follow-up was 9.6 months (95% confidence interval 6.6-12.5). Treatment-associated toxicities included nausea, diarrhea, oral mucositis, dermatitis, and GVHD which are summarized in Table 2. No patients experienced pneumonitis.
Patient Characteristics.

Abbreviations: KPS, Karnofsky performance status; B-ALL, B-cell acute lymphocytic leukemia; T-ALL, T-cell acute lymphocytic leukemia; AML, acute myeloid leukemia; CR, complete response; GVHD, graft versus host disease; MTX, methotrexate; MMF, mycophenylate mofetil; PTCy, post-transplant cyclophosphamide; TBI, total body irradiation; VMAT, volumetric modulated arc therapy.
Highest Grade Acute Toxicity.

Abbreviations: GVHD, graft versus host disease.
There were four cases (12.9%) of NRM with three deaths (9.7%) occurring within 100 days of transplant. Two of these were due to Grade 5 oral mucositis in which these patients developed severe aspiration pneumonia with hypoxemia requiring endotracheal intubation, bacterial sepsis, and eventual multi-organ system dysfunction; deaths occurred on days +15 and +21. One patient experienced respiratory failure due to multifocal fungal pneumonia resulting in death on day +21. One patient experienced a Grade 5 gastrointestinal bleed on day +242 which was the result of GVHD. Disease relapse occurred in three patients (10.3%) on days +212, +526, and +625. There was no disease relapse in those treated with oropharyngeal mucosal sparing and no deaths due to disease relapse at the time of this report.
Treatment Plans
VMAT was used in 26 (83.9%) patients while HT was used in five patients (16.1%). The mean lung dose achieved with VMAT was significantly lower than with HT (7.4 vs 7.7 Gy, P = .009). There were no significant differences among mean doses to the heart, kidney, and breast nor among the maximum dose to the brain. The goal D85% to the PTVbody was achieved in both VMAT and HT plans. PTVbones was created only for VMAT plans to ensure adequate marrow coverage; the D95% to PTVbones was achieved. Comparison of the dosimetric parameters is summarized in Table 3.
Comparison of Dosimetric Parameters.
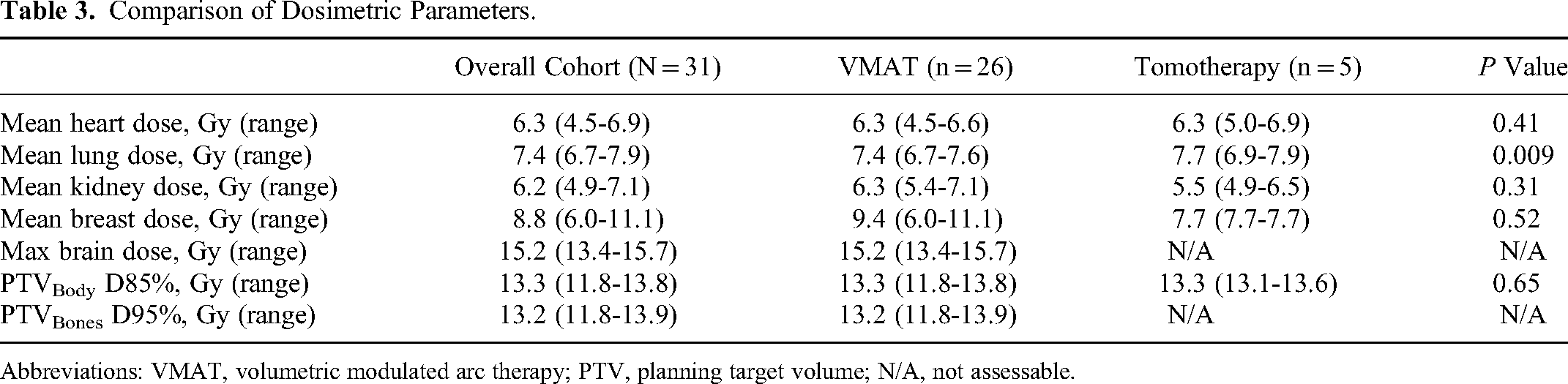
Abbreviations: VMAT, volumetric modulated arc therapy; PTV, planning target volume; N/A, not assessable.
Oral Mucosal Sparing
After the treatment of 15 patients, we witnessed two deaths attributed to Grade 5 oral mucositis. As a result, we adopted an oropharyngeal mucosal sparing technique. Sixteen patients (51.6%) received oropharyngeal mucosal sparing which was delivered via VMAT. The mean dose to the this mucosa was successfully reduced in the patients treated with this technique (6.9 vs 14.1 Gy, P < .001). There was no significant difference in oral mucositis among all grades of toxicity in those treated with and without this technique (P = .22); however, no further Grade 5 mucositis toxicities were observed after implementation of the oropharyngeal mucosal sparing technique (Supplemental Table 3). As all plans met PTV prescription coverage and OAR dose tolerance limits, the head-to-head comparison of all OARs between these treatment subgroups was not performed.
Discussion
To our knowledge, the present study provides the first description of this full-body utilization of VMAT for high-dose TBI in HSCT conditioning. This method allows for more individualized dose tailoring, delivery of a homogenous dose, higher superficial doses without the need for bolus, reduced setup complexity, the ability to selectively spare OARs, and the possibility of safe and tolerable dose escalation without violation of dose constraints.
Previously reported VMAT-based TBI techniques use arcs for treatment of the head and neck, torso, upper extremities, abdomen, and pelvis and use AP/PA beams for the lower extremities.12–15,26 This field arrangement leads to steep dose cut-offs, thus creating a risk of significant dose heterogeneities including both under and over dosing the femur where the two fields meet. This could pose risk of undertreatment of disease within the femur.16,17 Our treatment technique utilizes overlapping arcs throughout the entire body which significantly reduces potential dose heterogeneities when contending with two-field alignment (Figure 1).
Our literature review revealed one other study (n = 7) that used VMAT throughout the entire body rather than AP/PA fields to the lower extremities for high-dose TBI delivery. 27 The technique described also used two fields, however, more isocenters (nine to fifteen) were utilized, patients were treated only with 6 MV photons, and the lungs and kidneys were the only consistently contoured OAR. Additionally, bolus was applied to the head, back, sternum, shinbones, and knees to increase superficial dose. Our method met all dose goals including those to superficial targets, in part, because of the creation of a second PTV specifically for the bones. Arc therapy itself can provide higher dose to superficial sites without the need for bolus due to the multiple angles of beam entry that allow for dose buildup prior to reaching target. 28 Elimination of bolus reduces resource utilization, set-up time, and complexity.
HT is another method of delivering whole-body IMRT-based TBI that has been previously described. Here, we have shown that full-body VMAT can meet the same dose objectives as HT-delivered treatments. The only statistically significant difference in dosimetric parameters was that the lungs received a lower mean dose with VMAT when compared to HT (7.4 vs 7.7 Gy, P = .009). With pneumonitis being a major cause of TBI-related morbidity and mortality, this finding is indeed interesting and has potential for clinical importance.29,30
Oral mucositis is multifactorial entity related to radiation, chemotherapy, GVHD prophylaxis, and acute GVHD that occurs in up to 75% of patients undergoing high-dose chemoradiation for HSCT; when severe, patients may require patient-controlled opioid analgesia and feeding tubes and are at risk for nutritional deficiencies, aspiration, and infection.31–34 Our institution adopted an oropharyngeal mucosal sparing technique with the purpose of preventing radiation-induced oral mucositis after observing two patient deaths attributed to severe oral mucositis. Using VMAT, lower mean doses were successfully achieved within the oropharyngeal mucosa (6.9 vs 14.1 Gy, P = .001) in sixteen patients without sacrifice of PTV coverage. No statistical difference among toxicity was reached at the time of this report; importantly though, no further Grade 5 toxicities occurred after implementing this technique. Statistical significance was likely not reached due to the low power of this study and merits further exploration.
With lower doses obtained within the lungs, oropharynx, and oral cavity, there is opportunity to continue exploring OAR-sparing in TBI to reduce doses to other radio-sensitive organs that may result in clinically significant toxicity. For example, commonly experienced toxicities in our cohort included nausea (58.1%) and diarrhea (61.3%). These and other toxicities could be mitigated through VMAT-based TBI to selectively spare the stomach and small bowel. The capacity of selective dose avoidance with VMAT raises the question as to which structures should be irradiated from a clinical perspective, a concept that has begun to be explored through approaches such as TMLI and TMI.10,16–19 Fundamentally, VMAT-based TBI allows for selective OAR sparing that is readily accessible and realistically achievable at any institution with a VMAT-capable linear accelerator.
We recognize there is a theoretical risk of providing insufficient dose to tumor sanctuary sites along the oropharynx when performing mucosal sparing and when excluding breast tissue in females. We do not have evidence to suggest higher relapse rates but acknowledge that a median follow-up of 9.6 months is insufficient to fully assess for disease relapse, and longer follow-up is required. However, other institutions that have investigated TLMI and TMI which spares more tissue than our oropharyngeal mucosal sparing technique have had acceptable incidences of disease relapse.19–21,35
We also acknowledge that the overall cohort and comparator HT group is relatively small and thus limits the power of this study. Additionally, TBI is not solely responsible for the observed morbidity and mortality; conditioning chemotherapy, donor type, CMV status, and GVHD prophylaxis also contribute to toxicity including mucositis. Delving into these factors is beyond the scope of this study but may be considered in a future study.
Lastly, we understand there is a theoretical risk of late secondary malignancies due to increased neutron contamination with the use of IMRT. Energies >10 MV were reserved for patients with larger body habitus to improve dose homogeneity, meet dose constraints, and lower hot spots when necessary. We felt these benefits outweighed our concerns for the potential increased risk of secondary malignancy in these select circumstances.23–25
Conclusions
Ultimately, by describing our institution's unique full-body VMAT plan in detail, we hope that more institutions may consider this method in conditioning for HSCT as it does not require equipment for shielding as do 2D plans, eliminates potential dose heterogenicity at the mid-femur that can occur when contending with two-field alignment, does not appear inferior to HT, is potentially capable of providing lower lung doses when compared to HT, can be used to selectively spare OARs without compromise of PTV coverage leading to less TBI-induced morbidity and mortality, and can be implemented at any facility with a VMAT-capable linear accelerator. As centers begin to explore IMRT-based TBI treatment strategies, we also strongly recommend sparing of the oral mucosa and close monitoring for severe acute toxicities.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231180779 - Supplemental material for Feasibility and Toxicity of Full-Body Volumetric Modulated Arc Therapy Technique for High-Dose Total Body Irradiation
Supplemental material, sj-docx-1-tct-10.1177_15330338231180779 for Feasibility and Toxicity of Full-Body Volumetric Modulated Arc Therapy Technique for High-Dose Total Body Irradiation by Emily Keit, Casey Liveringhouse, Nicholas Figura, Joseph Weygand, Maria L. Sandoval, Genevieve Garcia, Julia Peters, Michael Nieder, Rawan Faramand, Farhad Khimani, Sungjune Kim, Timothy J. Robinson, Peter A.S. Johnstone, Jose Penagaricano and Kujtim Latifi in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
None.
Authors’ Note
Author responsible for statistical analysis Casey Liveringhouse, Email: Casey.Liveringhouse@moffitt.org
Data Statement
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
Ethical approval to report this case series was obtained from the University of South Florida's institutional review board under IRB # 00021984 which was approved on 5/7/2015 and renewed on 4/16/2019.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
