Abstract
Background
Differential diagnosis of single-nodule pulmonary metastasis (SNPM) and second primary lung cancer (SPLC) in patients with colorectal cancer (CRC) prior to lung surgery is relatively complex. Radiomics is an emerging technique for image information analysis, while it has not yet been applied to construct a differential diagnostic model between SNPM and SPLC in patients with CRC. In the present study, we aimed to extract radiomics signatures from thin-section computed tomography (CT) images of the chest. These radiomics signatures were combined with clinical features to construct a composite differential diagnostic model.
Method
A total of 91 patients with CRC, including 66 patients with SNPM and 25 patients with SPLC, were enrolled in this study. Patients were randomly assigned to the training cohort (n = 63) and validation cohort (n = 28) at a ratio of 7 to 3. Moreover, 107 radiomics features were extracted from the chest thin-section CT images. The least absolute shrinkage and selection operator (LASSO) regression was used to filter these features, and clinical features were screened by univariate analysis. The screened radiomics and clinical features were combined to construct a multifactorial logistic regression composite model. The receiver operating characteristic (ROC) curves were adopted to evaluate the models, and the corresponding nomograms were created.
Results
A series of 6 radiomics characteristics was screened by LASSO. After univariate logistic regression analysis, the composite model finally included 4 radiomics features and 4 clinical features. In the training cohort, the area under the curve scores of ROC curves were 0.912 (95% confidence interval [CI]: 0.813-0.969), 0.884 (95% CI: 0.778-0.951), and 0.939 (95% CI: 0.848-0.984) for models derived from radiomics, clinical, and combined features, respectively. Similarly, these values were 0.756 (95% CI: 0.558-0.897), 0.888 (95% CI: 0.711-0.975), and 0.950 (95% CI: 0.795-0.997) in the validation cohort, respectively.
Conclusions
We constructed a model for differential diagnosis of SNPM and SPLC in patients with CRC using radiomics and clinical features. Moreover, our findings provided a new assessment tool for patients with CRC in the future.
Keywords
Introduction
As one of the leading causes of tumor-related death, colorectal cancer (CRC) remains a significant public health challenge globally.1,2 Because of the presence of hematogenous metastases, pulmonary metastases (PMs) are common lung malignant lesions in patients with CRC, accounting for approximately 10% to 20% of patients. Resection of PMs is currently thought to improve progression-free survival in patients who can tolerate it. In terms of number, single-nodule pulmonary metastasis (SNPM) can be better surgically resected. Patients with CRC with SNPM are not uncommon in clinical practice.3–7 In contrast to SNPM, second primary lung cancer (SPLC) is a pulmonary malignancy found simultaneously or after the primary tumor.8,9 SPLC in patients with CRC is not rare due to underlying immune deficiencies, gene mutations, carcinogen exposure, lifestyle, or cancer treatment.10,11 Before pulmonary surgery, their differential diagnosis in clinical practice is challenging. To precisely distinguish them, pathomorphological observation and immunohistochemical detection are often achieved by bronchoscopy or percutaneous needle biopsy, 12 while these operations are not applicable in some patients. Therefore, an effective pre-operative examination is required to detect and identify potential metastases early.
In patients with CRC, it is necessary to evaluate lung lesions to confirm initial staging, treatment strategy, and follow-up plan. There is a consensus that imaging of the chest, including X-rays, computed tomography (CT), and positron emission tomography (PET)-CT, is a vital pre-operative and postoperative monitoring tool for patients with CRC.13–16 To date, there have been studies using imaging and clinical features to establish predictive models in the early diagnosis of PMs.17–19 However, it is still tricky for the readout results of CT and PET-CT images to be entirely consistent with the pathological findings. 20 In recent years, radiomics has gradually attracted people's attention. In radiomics, CT, PET-CT, and other images are converted into high-dimensional, mineable data through high-throughput extraction of multiple quantitative features, followed by subsequent data analysis to build predictive models for decision support.21,22 Radiomics has been applied to the prediction of pre-operative staging, the monitoring of distant metastases, and the differentiation of pulmonary nodules in CRC.23–26 Deep learning, combined with radiomics, has been shown to be a valuable tool for evaluating patients with CRC with pulmonary metastases (PMs). 27 There is also a growing body of research focused on using deep learning and related data augmentation algorithms to analyze lung malignancies.28,29
In clinical diagnosis, differential diagnosis of SNPM and SPLC is still relatively complex. Therefore, we extracted the radiomics signatures of lung lesions in patients with CRC and combined the radiomics model with the clinical model to establish a reliable composite differential diagnostic model. The main contributions are as follows:
Radiomics features of CT images were applied to isolated nodules in the lungs of patients with CRC, and it provided a good differential diagnosis between SNPM and SPLC. With models built through multiple machine learning approaches, radiomics can effectively assist in solving this problem. This study also established a clinical image model for comparison and combination of radiomics models. The radiomics model in this study had comparable diagnostic efficacy to the traditional clinical image model. A combined model was constructed by integrating radiomics features with clinical features. The diagnostic efficacy of this model was better than that of existing methods.
Materials and Methods
Patient Selection
This research was a retrospective study. A total of 6401 patients with CRC who received oncotherapy at the First Affiliated Hospital of Soochow University from January 2013 to June 2021 were reviewed. The inclusion criteria of our study were: (1) CRC with histopathological confirmation under surgical resection or colonoscopy; (2) only pathological diagnosis of adenocarcinoma; (3) relevant CT images and medical history were available; and (4) patients with regular re-examination after tumor treatment. The exclusion criteria were as follows: (a) the number of pulmonary nodules > 1 when pulmonary lesions were first detected on chest thin-section CT images; (b) patients with primary cancers other than CRC and lung cancer; and (c) image sequences that could not be sketched due to image quality problems. Eventually, 2 categories of patients (Table 1) were identified: (A) patients with SNPM (n = 66) and (B) patients with SPLC (n = 25). All patient details were de-identified in the study process.
Characteristics of Patients With CRC with Different Pulmonary Nodules.
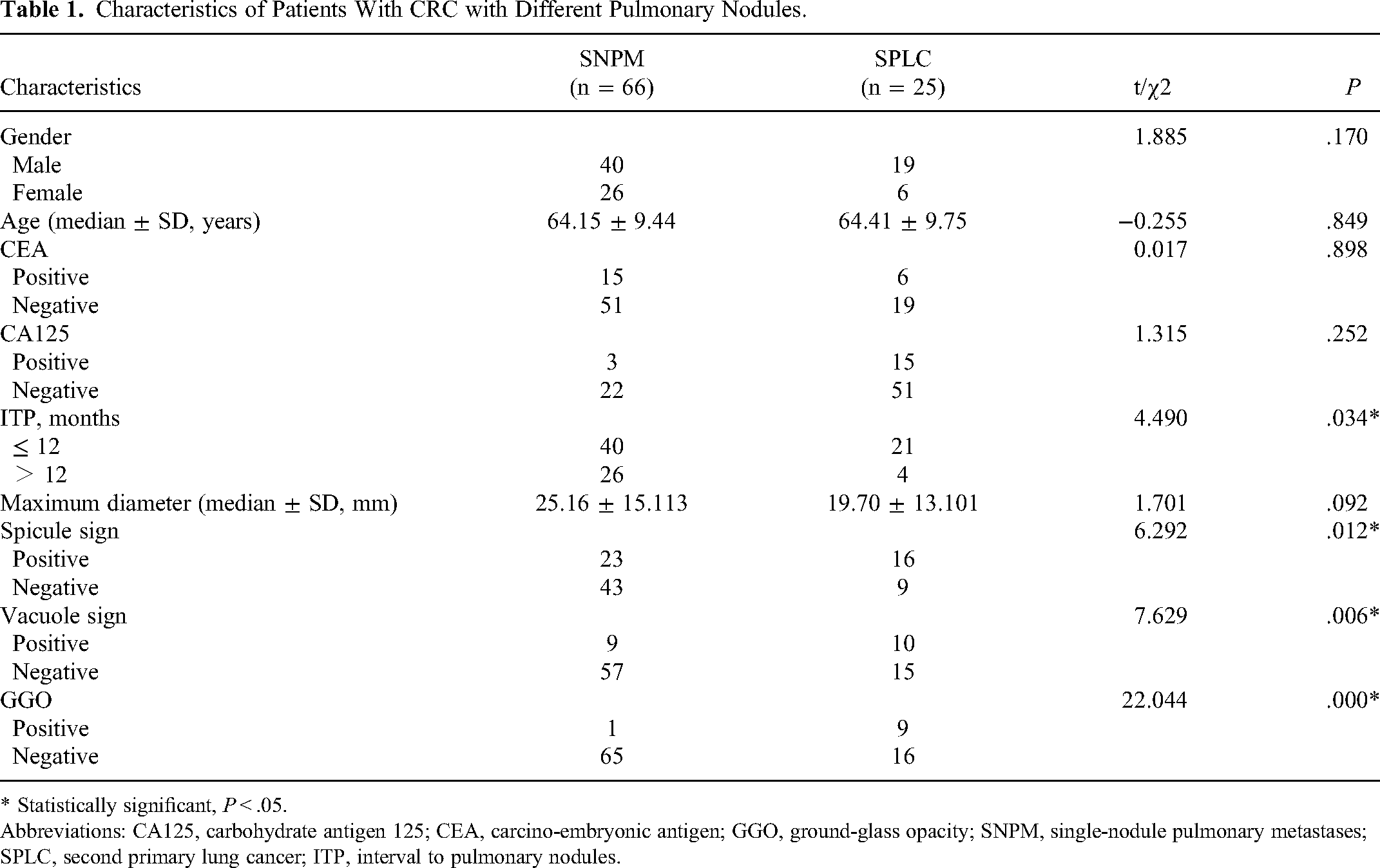
* Statistically significant, P < .05.
Abbreviations: CA125, carbohydrate antigen 125; CEA, carcino-embryonic antigen; GGO, ground-glass opacity; SNPM, single-nodule pulmonary metastases; SPLC, second primary lung cancer; ITP, interval to pulmonary nodules.
Acquisition of Image Data and Clinical Data
All patients underwent chest thin-section CT examinations in the supine position using the GE LightSpeed VCT scanner or GE BrightSpeed Elite 16 scanner (GE Healthcare, Milwaukee, USA). Scans were performed from the lung apex to the lung base using the following parameters: tube voltage 120 kV, tube current automatic mA, helical pitch 0.938, rotation speed 0.6 s, slice thickness, and spacing 5 mm. CT images were reconstructed with a slice thickness of 1.00 to 1.25 mm. The images were reviewed by 2 chest radiologists with more than 10 years of experience together. If there was a disagreement between the 2 radiologists, a third radiologist with 20 years of experience conducted a further review. During this procedure, the following image features were recorded, including maximum lesion diameter, spicule sign, and vacuolation sign. In addition, some clinical data of these patients were collected through the clinical information system, including age, gender, interval to pulmonary nodules (ITP), carcino-embryonic antigen (CEA), and carbohydrate antigen 125 (CA125). The above data were summarized in a table consisting of clinical characteristics.
CT Image Analysis
Chest thin-section CT images were downloaded from the hospital's picture archiving and communication system to an offline workstation for texture analysis. The operator (Y.Y., a graduate student) implemented lesion segmentation using 3D Slicer software (v4.13.0; National Institutes of Health, NIH, USA). The visible boundaries of the lesions were manually outlined based on the regions of interest in each axial tomography. The lesion boundaries were segmented into 2-dimensional regions and then converted into volumes of interest (VOIs) for 3-dimensional (3D) data. The areas of blood vessels, air, normal pulmonary tissue, and surrounding organs were excluded as much as possible in this process. An expert radiologist (DSM, with 20 years of experience in pectoral and abdominal CT) then finally validated the process and revised it. After finishing delineation, the 3D data of VOIs and image sequences (NII format) were transferred to FeAture Explorer software (FAE, V 0.3.6) on Python (3.7.6) (https://github.com/salan668/FAE) for feature extraction and analysis. 30 After preprocessing of data, radiomics features with missing values and noncontiguous variables were excluded. A total of 107 features were extracted finally. The number of radiomics features based on feature classes were as follows and shown in Table 2: (i) 18 for first-order feature, (ii) 14 for shape feature, (iii) 24 for gray level co-occurrence matrix feature, (iv) 16 for gray level size zone matrix (GLSZM) feature, (v) 16 for gray level run length matrix feature, (vi) 5 for neighborhood gray-tone difference matrix feature, and (vii) 14 for gray level dependence matrix (GLDM) feature.
Characteristics of the Training and Validation Cohorts.

Radiomics Feature Selection and Radiomics Model Development
After extraction of features, computer-generated random datasets were used to assign 70% of datasets to the training set and 30% of the datasets to the independent validation set. Consequently, 63 cases were selected as the training dataset (46/17 = SNPM/SLPC), while the other 28 cases constituted the independent validation dataset (20/8 = SNPM/SLPC).
To remove the unbalance of the training dataset, up-samples were conducted by repeating random cases to make positive/negative samples balance. Then, the normalization was applied to the feature matrix (Z score, mean). Since the dimension of feature space was high, the linear models were penalized with the L1 norm or the least absolute shrinkage and selection operator (LASSO), Pearson correlation coefficient, and principal component analysis to select features before building the model. After this process, the dimension of the feature space was reduced, and each feature was independent of the others. Lastly, the analysis of variance, relief, and recursive feature elimination were utilized for feature selections. Multiple methods were used as the classifier, including the support vector machine, linear discriminant analysis, auto-encoder, random forests, logistic regression (LR), LR via LASSO, ada-boost, decision tree, Gaussian process, and naive Bayes. Besides, a 10-fold cross-validation was applied to the training dataset to determine the hyper-parameter (such as the number of features) of the model (Supplemental Figure 1 and Table 1). The hyper-parameters were set according to the model performance on the validation dataset. The above operations were implemented by FAE software and Python 3.0 (https://www.python.org).
The performance of the radiomics models was evaluated by the receiver operating characteristic (ROC) curve analysis, Youden index, and F1 scores. Subsequently, the accuracy, sensitivity, specificity, positive predictive value, and negative predictive value of each model were calculated.
Statistics Analysis in Clinical and Combined Models
The clinical features were age, gender, ITP, CEA, CA125, maximum lesion diameter, spicule sign, vacuolation sign, and ground-glass opacity (GGO). Statistical analyses were performed using SPSS software version 26.0 (SPSS Inc., Chicago, IL, USA) and Medcalc software version 20 (https://www.medcalc.org/). The nominal variables were analyzed by the chi-square test or Fisher's exact test. The continuous variables with abnormal distribution were analyzed by the Mann-Whitney test, while the continuous variables with normal distribution were analyzed by t-test. Moreover, univariate logistic analysis was employed to investigate the discriminative radiomics and clinical features. Multivariate analyses were performed using the logistic regression analysis model in the training and validation cohorts. A P < .05 was considered statistically significant (Figure 1). Based on ground truth data, clinical models were developed in the training and validation sets. The model was manifested by ROC curves, and the differential diagnostic performance of the model was assessed by the area under the curve (AUC). The reporting of this study conformed with STROBE guidelines. 31

The flow diagram of this study. Abbreviation: LASSO, least absolute shrinkage and selection operator.
Results
Patient Clinical Characteristics
Table 1 summarizes the clinical characteristics of the patients in the training and validation cohorts. A total of 91 patients were included in this study. Among the enrolled patients, 66 (72.5%) were patients with SNPM, and 25 (27.5%) were patients with SPLC. Among these patients, there were no statistically significant differences in clinical features between the training and validation cohorts (Table 2). ITP (P = .041), spicule sign (P = .014), vacuolation sign (P = .008), and GGO (P = .001) were significant correlates of PM in the univariate logistics regression analysis (Table 3).
Characteristics of Patients With CRC in the Training Cohort and Validation Cohort.

Abbreviations: CA125, carbohydrate antigen 125; CEA, carcino-embryonic antigen; GGO, ground-glass opacity; SNPM, single-nodule pulmonary metastases; SPLC, second primary lung cancer; ITP, interval to pulmonary nodules.
Extraction and Selection of Radiomics Features
A total of 107 radiomics features were extracted from the CT images (Table 2). Multiple radiomics models were built using different normalization, dimensionality reduction screening, and machine learning methods. Several simple models with good validation efficacy were selected based on the validation results. These models are listed in Table 4. In addition, a cutoff value of 6 features was chosen based on the model's performance, and it was used to compare the performance of radiomics models in different machine learning pipelines (Figure 2).

AUCs of different datasets using 10 machine learning algorithms by different methods. Abbreviations: AUC, area under the curve; ANOVA, analysis of variance; KW, Kruskal-Wallis; PCA, principal component analysis; PCC, Pearson correlation coefficient; RFE, recursive feature elimination.
Multiple Machine Learning Pipelines for Radiomics Models.

Abbreviations: 95% CI, 95% confidence interval; Acc, accuracy; AE, auto-encoder; ANOVA, analysis of variance; AUC, area under the curve; GP, Gaussian process; KW, Kruskal-Wallis; LASSO, least absolute shrinkage and selection operator; LDA, linear discriminant analysis; LR, logistic regression; LRLASSO, logistic regression via LASSO; NPV, negative predictive value; PCA, principal component analysis; PPV, positive predictive value; RFE, recursive feature elimination; Sen, sensitivity; Spe, specificity; Std, standard error; SVM, support vector machine.
Diagnostic Performance of Individual Models
As a frequently used method, the LASSO was chosen to filter features using dimensionality reduction in building the combined model when keeping other processes unchanged. Six radiomics features were screened, and again these were included in the univariate logistics regression analysis, including Glszm_LargeAreaHighGrayLevelEmphasis (P = .336), Gldm_DependenceEntropy (P = .002), Firstorder_InterquartileRange (P = .019), Firstorder_90Percentile (P = .001), Firstorder_Maximum (P = .098), and Shape_Sphericity (P = .000). We obtained 4 significantly associated radiomics features (P < .05) (Table 5), included them in a multifactorial logistic regression analysis along with clinical characteristics, and modeled them accordingly.
Univariate Analysis for Models by Logistic Regression.
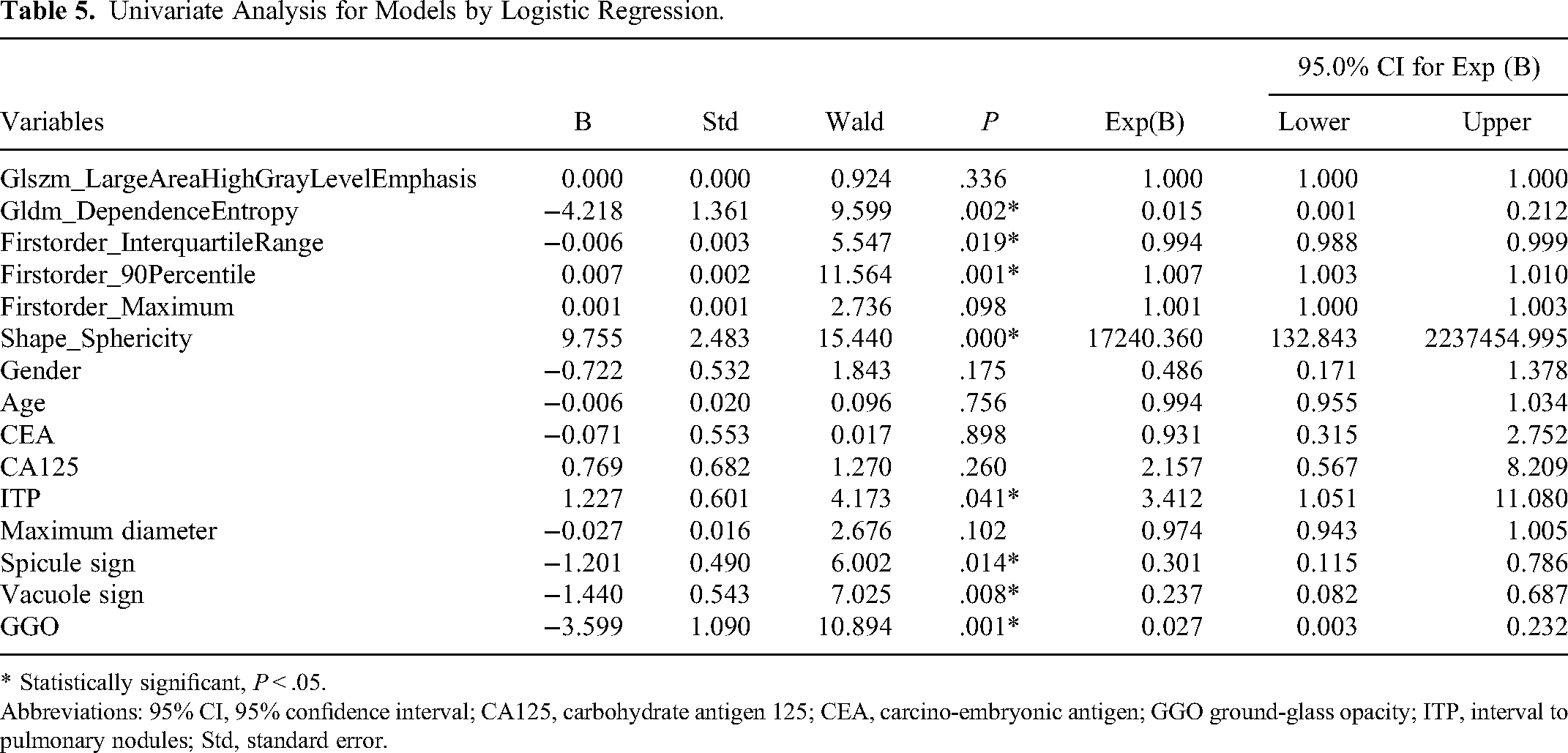
* Statistically significant, P < .05.
Abbreviations: 95% CI, 95% confidence interval; CA125, carbohydrate antigen 125; CEA, carcino-embryonic antigen; GGO ground-glass opacity; ITP, interval to pulmonary nodules; Std, standard error.
Figure 3A and B show the AUCs of ROC curves for 3 models (radiomics, clinical, and combined models) individually in the training and validation cohorts, respectively. In the training cohort, the AUC scores for differentially diagnosing SNPM and SPLC were 0.912 (95% confidence interval [CI]: 0.813-0.969), 0.884 (95% CI: 0.778-0.951), and 0.939 (95% CI: 0.848-0.984) for models derived from radiomics features, clinical features, and combined features, respectively. Similarly, in the validation cohort, the AUC scores of 3 models were 0.756 (95% CI: 0.558-0.897), 0.888 (95% CI: 0.711-0.975), and 0.950 (95% CI: 0.795-0.997), respectively (Table 6).
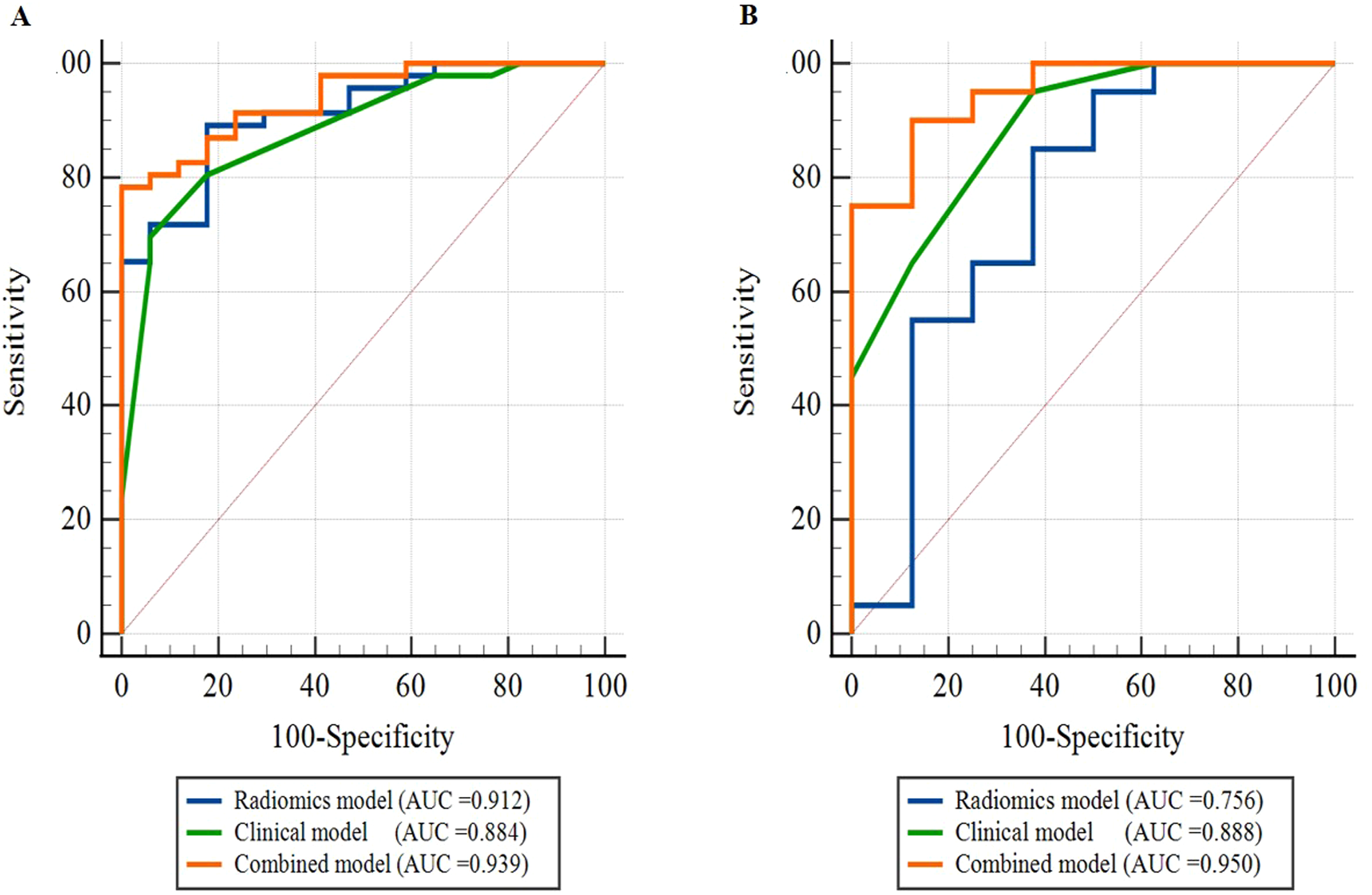
Performance of different models. (A) Training cohort and (B) validation cohort. Abbreviation: AUC, area under the curve.
ROC Analysis for Models by Logistic Regression.

Abbreviations: 95% CI, 95% confidence interval; AUC, the area under the curve; Std, standard error.
In addition, we constructed nomograms based on the results of multivariate logistic regression analysis for the training (Figure 4A) and validation cohorts (Figure 4B). For each patient with CRC with an SNPM in the lung, a corresponding score was obtained based on radiomics and clinical variables. The probability of a diagnosis of SNPM or SPLC could be assessed by calculating the total score.

Nomogram for differential diagnosis of SNPM and SPLC. (A) Training cohort and (B) validation cohort. Abbreviations: SNPM, single-nodule pulmonary metastasis; SPLC, second primary lung cancer.
Discussion
The type of lung lesion for patients with CRC presenting with pulmonary nodules of unknown provenance determines the subsequent surgery and chemotherapy treatment plan. 32 Except for benign lung nodules that require no further treatment, metastatic lung lesions are often resected through minimally invasive surgery. 33 In contrast, primary lung cancer involves complete lung lobe resection, and further mediastinal lymph node dissection is required.34,35 Despite the vital value for postoperative lung surgery,36,37 the differential diagnosis of individual pulmonary malignant nodules has been poorly studied due to the small number of patients compared with the overall number. Radiomics and deep learning models are increasingly being used to evaluate suspicious lung nodules and assist in the decision-making process. For instance, Ren et al have proposed a hybrid framework that enhances various classification models to address the task of lung cancer classification, which has achieved better performance compared to other state-of-the-art methods.28,29 To the best of our knowledge, no studies have combined radiomics features with clinical features to establish a differential diagnostic model between SNPM and SPLC (Figure 5).

Pulmonary malignant lesions in patients with CRC, all located in the right upper lung but presenting differently. The light green overlay represents the actual segmentation area in the figure. (A) Single pulmonary nodule with an irregular shape, multiple spicules, and vacuole signs in a 62-year-old male patient. Postoperative pathology was found to be SNPM. (B) Pulmonary mass lesions with a relatively regular shape in a 73-year-old male patient. Postoperative pathology was SPLC. (C) Nearly circular pulmonary nodule in a 75-year-old male patient, a common manifestation of PMs. Postoperative pathology confirmed SNPM. (D) The single lung nodule was observed as GGO in this image. Postoperative pathological findings in this 63-year-old male patient's lung CRC proved to be SPLC. Abbreviations: CRC, colorectal cancer; GGO, ground-glass opacity; PMs, pulmonary metastases; SNPM, single-nodule pulmonary metastasis; SPLC, second primary lung cancer.
In the present study, we extracted radiomics features from chest thin-section CT images of different natures of malignant lung lesions in patients with CRC and established a radiomics model. The radiomics features in this model included Gldm_DependenceEntropy, Firstorder_InterquartileRange, Firstorder_90Percentile, and Shape_Sphericity. The model had moderate diagnostic efficacy for the differential diagnosis, whether the lung lesions were lung metastases or SPLC. This result was also confirmed by the method of multiple radiomics study cohorts. Compared with our clinical models using the spicule sign, vacuole sign, GGO, and ITP, the 2 had very similar diagnostic efficacy. Furthermore, a composite model combining the 2 was superior to either the clinical model or the radiomics model alone.
With our newly developed clinical model, we believed that the spicule sign and vacuole sign could all be used as diagnostic references to distinguish SNPM from SPLC. This conclusion was consistent with the findings of several previous studies.38–41 Pre-existing research has also concluded that spicule sign, vacuole sign, and GGO are significant factors in the univariate analysis, while spicule sign and vacuole sign are not significant factors in the differential diagnosis of SNPM and SPLC by multivariate analysis. 42 This conclusion is not consistent with some of our findings. The reason might be attributed to the difference in the subjective opinion of the person reading the CT images, further illustrating the necessary introduction of other types of features, such as radiomics, histopathology, immunohistochemistry, and genomics.
In previous studies, ITP is considered an important factor affecting the prognosis of PMs in patients with CRC because it reflects the biology of the tumor. An ITP > 12 months generally represents a worse prognosis (4). In the differential diagnosis of SNPM and SPLC, some scholars have obtained by subgroup analysis that the division of ITP into > 3 and ≤ 3 can help distinguish the 2. However, the time cutoff value taken is still controversial in the known studies. 43 In our present study, ITP was classified traditionally as > 12 (n = 30) and ≤ 12 (n = 61). A univariate analysis was performed to obtain P = .041 (P < .05). Although we considered ITP as a significant independent predictor to identify the 2 and included it in the clinical and combined models, the choice of time cutoff values in ITP needs to be further analyzed in future studies.
Serum CEA is a common tumor marker for monitoring CRC metastasis and recurrence. It may also be relevant for predicting the occurrence of CRC lung metastasis. 44 However, some previous studies have shown that serum CEA is not a significant independent factor in the differential diagnosis of pulmonary lesions. Even in patients with PMs, most patients show negative serum CEA.18,45 Similar results were obtained in our study. This finding indicated that tumor markers, such as serum CEA, might not be effective in the differential diagnosis of PM and SPLC in patients with CRC.
Reproducibility and robustness are vital in radiomics analysis. 46 In the present study, all chest CT images were realized in the same center using almost similar acquisition and reconstruction protocols. In addition to the 10-fold cross-validation, we used different downscaling, screening, normalization, and modeling approaches in our radiomics analysis to support the soundness of this process.
There were several limitations in our study. First, this work was carried out based on a single-center retrospective study, which might lead to potential selection bias. External validation of the model should be conducted with multicenter patient data, when possible, to further evaluate its effectiveness and generalizability. Second, although we had the largest sample size of any known study in this direction, the sample size was still relatively small for machine learning. Prospective trials should be conducted to recruit a sufficient number of patients with CRC with SNPM or SPLC, in order to further evaluate the clinical usefulness of the radiomics differential diagnosis model. Third, the type of clinical information we collected was limited, which might make us unable to uncover potentially significant independent predictors. To improve the generalizability of the combined model, future studies should consider adding information from multiple sources, such as metabolic data from PET/CT, genomics data, and pathomics data.
Conclusions
A radiomics model was developed using machine learning to analyze lung CT images, which demonstrated good performance in differentiating SNPM and SPLC in patients with CRC. The radiomics model exhibited comparable efficacy to the clinical image model that included the spicule sign, vacuole sign, GGO, and ITP. By combining the radiomics model with clinical image features, a composite model was generated, which could enhance diagnostic accuracy and assist in making appropriate clinical decisions for patients with CRC.
Supplemental Material
sj-png-1-tct-10.1177_15330338231175735 - Supplemental material for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer
Supplemental material, sj-png-1-tct-10.1177_15330338231175735 for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer by Yu Yu, Jiaqing Tan, Yi Yang, Bin Zhang, Xiaodong Yao, Shibiao Sang and Shengming Deng in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338231175735 - Supplemental material for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer
Supplemental material, sj-docx-2-tct-10.1177_15330338231175735 for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer by Yu Yu, Jiaqing Tan, Yi Yang, Bin Zhang, Xiaodong Yao, Shibiao Sang and Shengming Deng in Technology in Cancer Research & Treatment
Supplemental Material
sj-pdf-3-tct-10.1177_15330338231175735 - Supplemental material for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer
Supplemental material, sj-pdf-3-tct-10.1177_15330338231175735 for The Differential Diagnostic Value of Radiomics Signatures Between Single-Nodule Pulmonary Metastases and Second Primary Lung Cancer in Patients with Colorectal Cancer by Yu Yu, Jiaqing Tan, Yi Yang, Bin Zhang, Xiaodong Yao, Shibiao Sang and Shengming Deng in Technology in Cancer Research & Treatment
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study is a retrospective study. Only the clinical data of the patients were collected, and the treatment plan of the patients was not intervened. The researchers have tried their best to protect the information provided by patients from disclosing their personal privacy. This study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Soochow University (Approval number: 2022.302).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (No. 81601522), Medical Youth Talent Project of Jiangsu Province (No. QNRC2016749), Gusu Health Talent Program (No. GSWS2020013), Suzhou People's Livelihood Science and Technology Project (No. SYS2019038), Project of State Key Laboratory of Radiation Medicine and Protection, Soochow University (No. GZK1202127), and the open Foundation of Nuclear Medicine Laboratory of Mianyang Central Hospital (No. 2021HYX023 and 2021HYX029).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
