Abstract
Introduction
This study aimed to develop machine learning-based models to predict local recurrence in patients with lung oligometastases receiving stereotactic body radiotherapy (SBRT), using both clinical and radiomic features.
Methods
A total of 80 lung oligometastases from 65 patients treated with SBRT were retrospectively evaluated. Clinical variables and radiomic features extracted from non-contrast planning computed tomography (CT) scans were collected. The dataset was randomly divided into training (70%) and test (30%) sets. Multivariable Cox proportional hazards models were developed to predict local recurrence using three feature sets: clinical only, radiomic only, and combined. Predictive performance was assessed using the concordance index (C-index).
Results
The median follow-up duration was 11.8 months (range, 6.0-31.5), during which local recurrence was observed in 12 out of 80 lesions (15.0%) treated with SBRT. Multivariable Cox proportional hazards models for predicting local recurrence achieved C-index of 0.75 for the clinical model, 0.74 for the radiomic model, and 0.78 for the combined model. The combined model incorporated three features: soft tissue sarcoma histology (HR 7.70, 95% CI 1.65-35.87, p = 0.009), metastasis size (HR 1.07, 95% CI 1.01-1.14, p = 0.036), and Rad-score (HR 4.05, 95% CI 1.58-10.36, p = 0.003).
Conclusion
These findings highlight the potential of machine learning-based models that integrate clinical and radiomic features to predict local recurrence in patients with lung oligometastases undergoing SBRT. Further validation in large, multicenter, and independent cohorts is needed.
Introduction
Oligometastatic disease is defined as 1–5 metastatic lesions for which primary tumor control is not mandatory, but all metastatic sites must be safely treatable. 1 Historically, the primary treatment of this disease has been based on systemic treatments. However, recent studies have shown that combining stereotactic body radiotherapy (SBRT) with systemic treatment confers a survival benefit, and combined therapy has been reported as an effective treatment option for oligometastatic disease. 2
The lung is the most common organ of distant metastasis for many solid tumors and SBRT is frequently used in the treatment of lung oligometastases. 3 SBRT is applied safely and effectively for peripherally located single lung metastases with a maximum diameter of 5 cm. For lung metastases that are centrally located or larger than 5 cm in diameter, the decision to use SBRT is based on the balance of safety and efficacy. In a recently published meta-analysis, the median 1- and 5-year local control after SBRT for lung oligometastases was reported as 90% and 79%, respectively. 4 Primary histology, biologic effective dose (BED), metastasis size, and oligometastatic disease subtype were found as crucial prognostic factors affecting local control.4–6 However, patient and tumor related characteristics alone are insufficient to fully predict recurrence and survival outcomes.
Radiomic analysis, a field of research that derives many quantitative features from medical imaging, is expected to play an important role for personalizing treatment in oncology practice. 7 In this context, radiomics is used across oncology for tumor detection and characterization, staging and early diagnosis, histologic or molecular differentiation, assessment of treatment response, and prognostication.8,9 However, there are few studies evaluating the predictive role of radiomic features in lung oligometastases treated with SBRT.10–14 Furthermore, studies focused on the efficacy of SBRT in terms of local recurrence for lung oligometastases are much rarer. Further research and validation of the results is needed before radiomic features can be incorporated into clinical practice in this patient population.
This study aimed to develop and evaluate machine learning models that integrate clinical and radiomic features for predicting local recurrence in lung oligometastases treated with SBRT. Such models may support risk stratification and enable more personalized treatment planning in future clinical workflows.
Materials and Methods
Patient Population
The study was reviewed and approved by the Akdeniz University Medical Scientific Research Ethics Committee (Approval No: 4, Date: 25.01.2024) and followed the ethical principles of the 1964 Declaration of Helsinki. The ethics committee waived the requirement for obtaining informed consent from individual patients. This waiver was granted due to the retrospective nature of the ethics committee-approved study, the deidentification of all patient information, and the determination that patient consent was not necessary. The reporting of this study conforms to relevant Equator guidelines. 15
This retrospective study included 80 lung oligometastatic lesions from 65 patients who underwent SBRT at our institution between January 2021 and November 2023. All lesions were treated with a biologically effective dose (BED₁₀) of ≥100 Gy. Eligible patients had a limited number of lung metastases, no evidence of polymetastatic disease, and were deemed clinically suitable for ablative radiotherapy. Patients with an Eastern Cooperative Oncology Group (ECOG) performance status greater than 2, or with significant non-malignant comorbidities that could impair treatment tolerance, adherence, or follow-up, were excluded. Additionally, those with a history of prior radiotherapy to the same or overlapping fields were not eligible. These selection criteria were implemented to ensure a clinically homogeneous cohort with sufficient functional reserve to safely receive high-dose SBRT.
The median age was 65 (range, 19-85) years and most of the patients were male (72.3%). All patients had genuine oligometastatic disease, 32 (49.2%) were classified as having de-novo disease and 33 (50.8%) as repeat disease. The three most frequent histologies were non-small cell lung cancer (35.4%), colorectal cancer (15.4%), and soft tissue sarcoma (10.8%). The other 38.4% consisted of 13 different histologies, indicating the heterogeneity of the cohort. The median NLR and PLR values of the patients before SBRT were 2.48 (range, 1.06-9.79) and 143.42 (range, 52.50-378.30), respectively. Patient characteristics are shown in Table 1. Among the 80 lung oligometastases treated with SBRT, 68 were peripheral and 12 were central. The median gross tumor volume (GTV) was 3.1 cc (range, 0.29-63.1), and the median SUVmax was 4.6 (range, 1-18). Detailed lesion characteristics are summarized in Table 2.
Patient Characteristics.

Characteristics of Lung Oligometastases.

Abbreviations: GTV, gross tumor volume; cc, cubic centimeter; SD, standard deviation.
Computed Tomography (CT) Simulation, Segmentation, and Planning
CT simulation was performed using a GE-Discovery scanner (GE Healthcare, USA). The patients were simulated using non-contrast CT images with a slice thickness of 1.25 mm, acquired at 120 kVp and 300 mAs, in the supine position.
Abdominal compression was used in the simulation of 52 patients, and CT images were acquired in three respiratory cycles: normal breathing, inspiration, and expiration. Internal target volume (ITV) was created by combining the GTVs contoured in these three respiratory phases. These patients were treated with helical intensity modulated radiotherapy (TomoTherapy HDA, Accuray, USA). Simulations of 13 patients were made using the breath-hold technique. In these patients, the GTV was contoured in a single phase and no additional margin was given for the ITV. Volumetric modulated arc therapy (VMAT, Elekta Synergy, Sweden) was applied to these patients. Planning target volume (PTV) margins ranged between 3–5 mm.
SBRT dose–fractionation schemes were determined according to institutional protocol and current evidence, considering lesion size, location, and organ-at-risk constraints. Accordingly, lung SBRT was delivered as 48 Gy in four fractions for nine lesions, 50 Gy in five fractions for 55 lesions, and 60 Gy in eight fractions for 16 lesions, consistent with published clinical studies and consensus recommendations.4,16
Image-guided radiotherapy (IGRT) was applied in each fraction. SBRT plans aim for 95% of the PTV volume to reach 100% of the prescribed dose and 99% of the PTV volume to reach a minimum of 90% of the prescribed dose. Because SBRT purposefully allows limited dose heterogeneity within the target to intensify the intratumoral dose and spare adjacent normal tissues, the maximum dose within the GTV was limited to 120% of the prescription, and high-dose regions were confined to the GTV. This constraint aligns with current SBRT practice guidelines, which recommend target maximum doses between 110% and 140% of the prescription. Setting the limit at approximately 120% provides an optimal balance between the biological benefit of a central dose boost, the consistency of planning across treatment techniques (TomoTherapy and VMAT), and the protection of surrounding organs at risk. Published SBRT series have also implemented comparable maximum target doses around 120% as part of validated clinical protocols. 17 The treatment plans for patients undergoing TomoTherapy were calculated using the convolution-superposition algorithm, and those for patients undergoing VMAT were calculated using the Monte Carlo algorithm.
Follow-up and Local Recurrence Definition
Patients were monitored on a quarterly basis for the initial 2 years following the completion of SBRT, at a 6-month interval for the subsequent 3 years, and annually thereafter. To assess local recurrence, progression, and distant metastasis, contrast-enhanced chest and abdominal CT or positron emission tomography (PET) was performed at each follow-up. Treatment response was evaluated by comparing pre- and post-SBRT imaging according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1). 18
To ensure adequate evaluation of treatment outcomes, patients were required to have a minimum clinical and radiographic follow-up of 6 months post-SBRT to be included in this study. Local recurrence was defined as an increase of ≥20% (and at least 5 mm in absolute size) in the sum of the longest diameters of the lesion within the previously irradiated PTV. To distinguish true tumor recurrence from radiation-induced lung injury, imaging findings suggestive of recurrence were reviewed by a multidisciplinary tumor board. When available, histopathological confirmation was also considered. Cases with indeterminate imaging findings or insufficient follow-up to confidently assess recurrence were excluded from the final analysis.
Clinical Parameters
Clinical parameters such as age, gender, ECOG performance status, oligometastatic disease subtype, primary histology, SUVmax, number, size and localization of metastases were collected from hospital records. Primary histology was categorized as soft tissue sarcoma versus other. In addition, the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) measured before SBRT were recorded as markers of systemic inflammation, based on prior studies demonstrating their prognostic relevance in patients treated with radiotherapy.19,20 Given that impaired pulmonary function has been associated with poorer outcomes and higher toxicity risk in patients with lung tumors undergoing SBRT, baseline pulmonary function tests were also evaluated. 21 Parameters included the forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC), the FEV1/FVC ratio, and the diffusing capacity for carbon monoxide (DLCO). All laboratory and physiological measurements were obtained prior to SBRT and used for subsequent analyses.
Radiomic Analysis
Radiomic analysis was based on non-contrast planning CT scans acquired during the SBRT simulation phase. The GTVs used for SBRT planning were defined on the normal-breathing phase CT for TomoTherapy simulations and on the breath-hold CT for VMAT simulations. This protocol-specific region of interest selection was chosen to minimize motion-related variability and to ensure consistency with clinical target delineation practices. Potential protocol-related variability in radiomic features was evaluated by comparing feature distributions between acquisition methods using principal component analysis and standardized mean differences. No systematic protocol-associated shift was observed; therefore, no feature harmonization (eg, ComBat) was applied, and single-vendor acquisition further limited inter-scan variability.
The contours were directly exported from the treatment planning system in DICOM-RT format and imported into 3D Slicer (v4.11, http://www.slicer.org) using the SlicerRT extension.22,23 For preprocessing, images were resampled to 1 mm isotropic voxels using B–spline interpolation for image data and nearest–neighbor interpolation for masks. Intensity discretization was performed using a fixed bin width of 25 Hounsfield units (HU). To preserve the physical meaning of Hounsfield units, we did not apply intensity normalization or histogram matching before discretization. With SlicerRadiomics, an extension of the 3D Slicer software, radiomic features compatible with the Image Biomarker Standardization Initiative (IBSI) were obtained. 24 No additional image filtering beyond the wavelet transform implemented in SlicerRadiomics was used; in particular, Laplacian-of-Gaussian and other non-IBSI filters were not applied. A total of 851 features were automatically extracted for each lesion, including 14 morphological features, 18 first-order statistics features, 75 textural features (gray-level co-occurrence matrix, GLCM, 24 features; gray-level size-zone matrix, GLSZM, 16 features; gray-level run-length matrix, GLRLM, 16 features; neighboring gray-tone difference matrix, NGTDM, 5 features and gray-level dependence matrix, GLDM, 14 features), and 744 wavelet-transform based features.
Radiomic features were assessed for reproducibility prior to further analysis. To evaluate feature stability, an intraclass correlation coefficient (ICC) analysis was conducted on a subset of 20 randomly selected lesions. The same observer performed a second, de novo segmentation of these lesions in a separate session held at least 4 weeks after any prior segmentation of the same cases. The observer was blinded to the initial contours, and the same image phase and preprocessing settings were used. ICC values were calculated using a two-way random-effects, single-measurement model for absolute agreement (ICC [2,1]). 25 Features with ICC ≥ 0.85 were considered highly reproducible and retained for subsequent analyses, whereas features with ICC < 0.85 were excluded.
Machine Learning Analysis
All machine learning and statistical analyses were performed using Python (v3.12.4) with the scikit-survival, lifelines, scikit-learn, numpy, and pandas libraries. A two-sided p-value < 0.05 was considered statistically significant. To evaluate model performance, the dataset was randomly partitioned into training (70%) and test (30%) sets. Feature selection and model development were conducted exclusively within the training set to avoid information leakage. An overview of the radiomic analysis and machine learning pipeline is presented in Figure 1.

Overview of the Radiomic and Machine Learning Pipeline.
Feature Selection
For clinical features, univariable Cox proportional hazards regression was performed. Clinical features with statistically significant associations with local recurrence (p < 0.05) were retained for inclusion in the multivariable Cox model.
For radiomic features, pairwise Spearman correlation analysis was conducted to reduce multicollinearity. From each pair of features with an absolute correlation coefficient greater than 0.80, one feature was removed. Then, radiomic features were subjected to Elastic Net-regularized Cox proportional hazards modeling using the Coxnet survival analysis method. 26
The L1 ratio (0.1-1.0, step 0.1) and alpha (30 values, 10−4 to 10¹, log-spaced) were tuned by 5-fold cross-validation in the training set, and the mean C-index across folds was used for selection. In total, 300 hyperparameter configurations were evaluated (10 L1-ratio × 30 alpha), yielding 1500 internal cross-validation fits (300 × 5-fold) (Figure 2). The best configuration (L1-ratio = 0.7, alpha = 0.16) was chosen, and a single Elastic Net–Cox model was refit on the entire training set at these settings. Features with non-zero coefficients in this refit defined the final Rad-score; no model averaging or ensembling across candidate models or folds was performed.

Elastic Net-Cox Feature Selection for Radiomic Features. Elastic Net-Cox Regularization was Used to Select Radiomic Features Based on C-index Performance.
Rad-Score Definition
The Rad-score was computed as the linear predictor of the single refit Elastic Net–Cox model at the optimal hyperparameters. As shown in Equation (1), the Rad-score is defined as the linear combination of the selected features: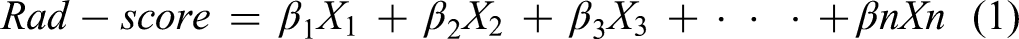
To explore the predictive value of the Rad-score, lesions were dichotomized into high- and low-risk cohorts using the training-set median value as the cutoff. Kaplan–Meier analysis on the test set was then performed to compare local recurrence between the groups, and the log-rank test was applied to assess statistical significance.
Model Development and Evaluation
Models were developed on the training set; model selection and in-sample fit comparisons were performed on the training data. Predictive performance of the locked models was assessed on the independent test set. To evaluate the predictive value of radiomic and clinical features, three multivariable Cox proportional hazards models were constructed: (i) a clinical model including only the clinical features that were statistically significant (p < 0.05) in univariable Cox regression analysis; (ii) a radiomic model including only the Rad-score, computed as a linear combination of selected radiomic features weighted by their Elastic Net–Cox regression coefficients; and (iii) a combined model that integrated both the Rad-score and the statistically significant clinical covariates. As detailed under Feature Selection, the radiomic pathway screened 300 Elastic Net–Cox hyperparameter configurations with 5-fold cross-validation in the training set (1500 internal fits). The clinical and combined models were pre-specified single fits with no further hyperparameter tuning. The Rad-score entered the combined model as a fixed linear predictor.
For interpretability, hazard ratios (HRs), regression coefficients, 95% confidence intervals (CIs), and two-sided Wald p-values were estimated in the training set (combined model). Model fit was compared using the Akaike information criterion (AIC) computed from the training-set fits, with lower values indicating better fit. Predictive performance was quantified on the test set using the C-index with 1000 bootstrap resamples for 95% CIs. 27 Conventional receiver operating characteristic (ROC) curve analysis was performed on the test set using a binary endpoint defined as any local recurrence during follow-up. The area under the curve (AUC) was estimated with 95% CIs based on 1000 bootstrap resamples. Discrimination at a clinically relevant horizon was then evaluated with time-dependent ROC analysis, and the 24-month AUC was reported with 95% CIs using the same bootstrap procedure. Decision curve analysis on the test set quantified net clinical benefit across threshold probabilities of 5%–30%. 28
Results
Local Recurrence
The median follow-up period of the patients was 11.8 (range, 6.0-31.5) months. Local recurrence was recorded in 10 of 65 patients (15.4%) and in 12 of 80 lesions (15.0%) who underwent lung SBRT.
Selected Radiomic Features and Rad-Score
Elastic Net-Cox regression was used to identify the most predictive radiomic features associated with local recurrence in the training cohort. A total of 9 radiomic features were selected for inclusion in the Rad-score (Table 3). The Rad-score for each lesion was calculated as a weighted linear combination of these features, where the weights correspond to the coefficients derived from the Elastic Net-Cox model:
Selected Radiomic Features and β-Coefficients Composing the Rad-Score.

Rad-score = (0.109207×wavelet-HLL_firstorder_Kurtosis) + (−0.074082×wavelet-HLH_firstorder_Median) + (−0.050751×wavelet-HHH_glcm_ClusterShade) + (0.040834× original_gldm_LargeDependenceLowGrayLevelEmphasis) + (−0.034583×wavelet-LLL_gldm_LargeDependenceLowGrayLevelEmphasis) + (0.026144×wavelet-HHL_glcm_ClusterShade) + (0.024653×original_firstorder_Energy) + (−0.006785×wavelet-HLL_firstorder_Skewness) + (−0.000799×original_firstorder_Skewness).
Kaplan–Meier analysis demonstrated a significant difference in local recurrence between high- and low-risk groups stratified by the median Rad-score (Figure 3).

Kaplan–Meier Curves on the test set using the training-set median Rad-score as the fixed cut-off; log-rank p-value reported.
Selected Clinical Features
Among clinical features, soft tissue sarcoma histology (p < 0.001) and metastasis size (p = 0.046) were identified as statistically significant predictors in univariable Cox regression analysis. Kaplan–Meier curves stratified by these clinical features are presented in Figure 4, illustrating their association with local recurrence.

Kaplan–Meier Curves on the Test set Stratified by Significant Clinical Predictors; log-Rank p-Value Reported.
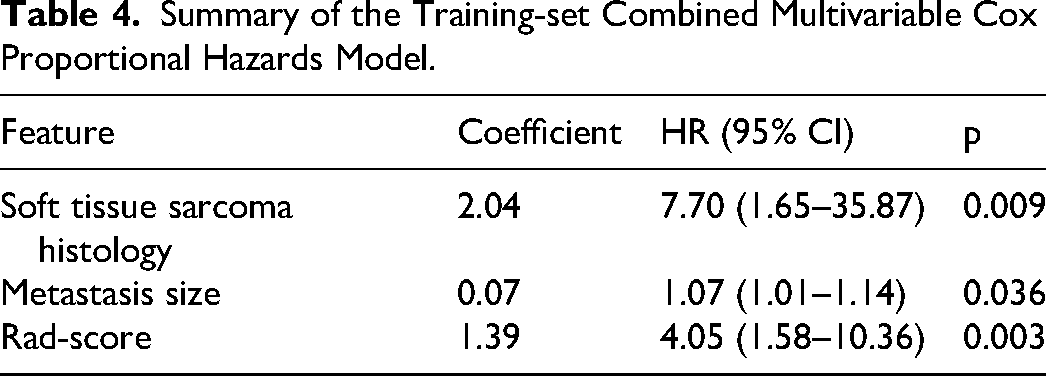
Combined Model Predictors and Estimates
The combined model included three predictors: soft-tissue sarcoma histology (HR 7.70; 95% CI 1.65-35.87; p = 0.009), metastasis size (HR 1.07; 95% CI 1.01-1.14; p = 0.036), and the Rad-score (HR 4.05; 95% CI 1.58-10.36; p = 0.003). All coefficients and hazard ratios were estimated from the training set (Table 4).
Summary of the Training-set Combined Multivariable Cox Proportional Hazards Model.
Akaike Information Criterion
In-sample model fit, summarized by the training-set AIC, also favored the combined model: 32.01 for the combined model versus 42.71 for the clinical model and 46.51 for the radiomic model. These differences correspond to ΔAIC values of 10.70 and 14.50 relative to the lowest AIC, indicating that the combined specification provides a more parsimonious explanation of the data.
Model Discrimination
Multivariable Cox proportional hazards models for predicting local recurrence achieved C-index of 0.75 (95% CI 0.66-0.83) for the clinical model, 0.74 (95% CI 0.64-0.82) for the radiomic model, and 0.78 (95% CI 0.70-0.89) for the combined model (Table 5). Conventional ROC AUCs on the test set were 0.74 (95% CI 0.57-0.89) for the clinical model, 0.73 (95% CI 0.55-0.89) for the radiomic model, and 0.81 (95% CI 0.68-0.92) for the combined model. At 24 months, time-dependent AUCs on the test set were 0.73 (95% CI 0.55-0.88) for the clinical model, 0.71 (95% CI 0.51-0.90) for the radiomic model, and 0.80 (95% CI 0.66-0.92) for the combined model (Figure 5). The superior discrimination of the combined model translated into the highest net benefit across clinically relevant thresholds (5-30%), as shown by the decision curve analysis (Figure 6).

Time-Dependent Receiver Operating Characteristic (ROC) Curves at 24 Months on the Independent Test set for the Clinical, Radiomic, and Combined Models.

Decision curve analysis to show the comparison of model performance.
Model Discrimination.

Abbreviations: C-index, concordance index; CI, confidence interval.
Discussion
Radiomics analysis represents a novel field of research with considerable potential to provide reliable predictions of cancer outcomes.7,29 The primary focus of radiomics analysis studies has been in the field of oncologic imaging. In recent years, a plethora of research has been conducted with the objective of differentiating lesions as malignant or benign, classifying tumor types, predicting adverse effects, treatment response, and survival.29–33 Nevertheless, due to the limitations of radiomic analysis studies on lung oligometastases treated with SBRT, radiomic features are still in the developmental stage in terms of potential clinical applications in this field.
To date, five separate studies have evaluated the predictive role of radiomic features in patients with lung oligometastases treated with SBRT.10–14 In three of these, all histopathologic subtypes were included, and the focus was on predicting treatment response after SBRT.10,12,13 Another study examined the prediction of local recurrence specifically in patients with colorectal cancer. 14 In a recent study, all histopathologic subtypes were included, aiming to predict recurrence-free and overall survival using models based on clinical and radiomic features. 11 To our knowledge, this is the first study to evaluate local recurrence across all histopathologic subtypes using combined clinical and radiomic models. While previous studies have focused on treatment response or survival, none have specifically addressed recurrence prediction in this heterogeneous population.
Cheung et al investigated the predictive role of CT-based radiomic features on treatment response in their study, which included a total of 69 patients, 29 with early-stage non-small cell lung cancer and 40 with lung oligometastases, who underwent lung SBRT. 10 In their study, skewness and root mean squared, which are first-order statistical features describing the distribution of voxel intensities, were found to be statistically significant predictors of treatment response to SBRT. The accuracy of the developed support vector machines (SVM) model was reported as 74.8% and the AUC value for complete treatment response prediction was 0.86. Their study provided evidence that CT-based radiomics features could predict treatment response to SBRT, regardless of tumor histology, primary tumor site, and stage. Furthermore, tumors that respond to SBRT have been demonstrated to exhibit superior OS rates.
Cilla et al evaluated 56 patients who underwent SBRT for lung oligometastases and showed that complete treatment response was associated with four different radiomic features. 13 These radiomic features were surface to volume ratio, skewness, correlation, and gray normalized level uniformity. However, no significant relationship was found between clinical parameters such as age, smoking status, tumor histology and stage, and complete response. In their study, two separate machine learning models, namely logistic regression (LR) and classification and regression tree analysis (CART), were used to predict complete treatment response. In the validation set, the accuracy rate for the developed LR model was found as 64.4% and the AUC value for complete treatment response prediction was 0.70. The accuracy rate for the developed CART model was reported as 75% and the AUC value for complete treatment response prediction was reported as 0.75.
In another study by Cilla et al, radiomics-based discriminant analysis of principal components (DAPC) was used to stratify the treatment response of lung metastases following SBRT. 12 DAPC has been shown to be able to classify 80 lesions into three different clusters based on treatment response depending on the radiomics features. It has been reported that the radiomic features that contribute the most to variation in treatment response are sphericity, correlation, and maximal correlation coefficient.
Fodor et al examined the predictive role of CT-based radiomic features for local recurrence in their study including 38 patients with colorectal cancer who underwent SBRT for lung oligometastases. 14 Four different radiomic features, namely Statistical_Variance, Statistical_Range, GLSZM_zoneSizeNonUniformity, and GLDZM_zoneDistanceEntropy, have been shown to predict local recurrence strongly and independently. Statistical variance, the radiomics feature corresponding to the heterogeneity of HU values within the tumor area, has been reported as the most promising and best performing radiomics feature. In addition, no relationship was found between clinical parameters such as age, primary site, stage and BED10, and local recurrence.
Salazar et al evaluated the predictive role of CT-based radiomic features and clinical features in their study including 111 patients with cancer who underwent SBRT for lung oligometastases. 11 They found that models combining CTTA-derived entropy and FPCA-derived F1 with age achieved moderate predictive performance for recurrence-free survival (C-index: 0.62). Importantly, the inclusion of clinical variables such as lung cancer origin and SBRT indication significantly improved model performance (C-index: 0.67), highlighting the additive value of clinical context. Their findings underscore the complementary roles of radiomic and clinical features in survival modeling, particularly in heterogeneous patient populations.
Prior radiomics studies in SBRT-treated lung oligometastases often modeled treatment response or survival with classification algorithms and rarely integrated robust clinical covariates.10–14 We address these gaps in four ways. First, we model time-to-event local recurrence in a histologically heterogeneous cohort using penalized Cox regression, rather than binary endpoints. Second, we enforce reproducibility (ICC ≥ 0.85) and use IBSI-compliant feature extraction. Third, we prevent information leakage, lock models, and evaluate on an independent test set with bootstrap CIs, reporting both C-index and 24-month time-dependent AUC. Fourth, we quantify clinical utility with decision-curve analysis and compare parsimony using AIC. The combined model achieved higher discrimination (C-index 0.78) and showed a better parsimony-adjusted fit
Given its discrimination on time-to-event outcomes (C-index and ROC AUC) and the observed net benefit on decision-curve analysis, this machine-learning model may support oncologists in treatment planning and clinical management of SBRT-treated lung oligometastases. Despite these encouraging results, several limitations should be noted. This single-center, retrospective study included a small and histopathologically heterogeneous cohort, which may limit statistical power and increase the risk of overfitting. We attempted to mitigate this risk by reducing multicollinearity, using Elastic Net–penalized Cox modeling with 5-fold cross-validation in the training set, and evaluating performance on a held-out test set; nevertheless, the findings should be interpreted with caution. Finally, the models were not validated on an external dataset, and confirmation in larger, multi-center cohorts is needed before clinical implementation. Additionally, competing-risk sensitivity analyses and decision-impact studies are warranted. For transparent deployment, a model card, open code, and a simple risk calculator with site-specific recalibration are recommended.
Conclusion
Machine learning–based models that integrate clinical and radiomic features show potential for predicting local recurrence after SBRT in lung oligometastases. In this cohort, combining clinical variables with radiomic features improved predictive accuracy and may enable more personalized care. Future research should validate these models in larger, independent cohorts and investigate additional biomarkers to further enhance predictive precision. If externally validated, model-derived risk estimates could help identify candidates for dose intensification or margin adaptation within organ-at-risk constraints. They may also prompt consideration of alternative or combined systemic therapies in patients predicted to derive limited benefit from SBRT.
Footnotes
Ethical Approval
This study received approval from the Akdeniz University Medical Scientific Research Ethics Committee (Approval No: 4, Date: 25.01.2024). Ethics approval was obtained, but patient consent was not required. The necessity for obtaining informed consent from individual patients was waived by the ethics committee. This waiver was granted because it is an ethics committee-approved retrospective study, all patient information was deidentified, and patient consent was not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement for This Work
The data that support the findings of this study are available from the corresponding author upon reasonable request.
