Abstract
Purpose
To investigate the capability of an Magnetic resonance imaging (MRI) radiomics model based on pretreatment texture features in predicting the short-term efficacy of recombinant human endostatin (RHES) plus concurrent chemoradiotherapy (CCRT) for nasopharyngeal carcinoma (NPC).
Methods
We retrospectively enrolled 65 patients newly diagnosed as having NPC and treated with RHES + CCRT. A total of 144 texture features were extracted from the MRI before RHES + CCRT treatment of all the NPC patients. The maximum relevance minimum redundancy (mRMR) method was used to remove redundant, irrelevant texture features, and calculate the Rad score of the primary tumor. Multivariable logistic regression was used to select the most predictive features subset, and prediction models were constructed. The performance of the 3 models in predicting the early response of RHES + CCRT for NPC was explored.
Results
The diagnostic efficiency of combined model and radiomics model in distinguishing between the effective and the ineffective groups of patients was found to be moderate. The area under the ROC curve (AUC) of the combined model and radiomics model was 0.74 (95% confidence interval [CI]: 0.62-0.86) and 0.71 (95% CI: 0.58-0.84), respectively, with both being higher than the AUC of the clinics model (0.63, 95% CI: 0.49-0.78). Compared with the radiomics model, the combined model showed marginally improved diagnostic performance in predicting RHES + CCRT treatment response. The accuracy of combined model and radiomics model for RHES + CCRT response assessment in NPC were higher than those of the clinics model (0.723, 0.723 vs 0.677).
Conclusion
The pretreatment MRI-based radiomics may be a noninvasive and effective method for the prediction of RHES + CCRT early response in patients with NPC.
Introduction
Nasopharyngeal carcinoma (NPC) has a high incidence in southern China, North Africa, and Southeast Asia. 1 At present, radiotherapy combined with chemotherapy, including induction chemotherapy (IC), concurrent chemotherapy, and neoadjuvant chemotherapy (NC), is an effective treatment method that can significantly improve the survival rate of patients with NPC.2,3 Some researchers found that Although concurrent chemotherapy and radiotherapy (CCRT) is considered the standard treatment for patients with advanced NPC, its efficacy in patients with clinical stage III/IV NPC remains inconsistent, and it significantly increases the toxicity of the treatment. 4 Currently, numerous clinical studies are ongoing to verify if targeted therapy has the potential as a treatment of NPC, and it is expected to become an important part of individualized therapy.
Recombinant human endostatin (RHES) under the brand name of Endostar (ES) is a broad-spectrum anti-angiogenic targeted drug. It reportedly has a multi-target anti-tumor angiogenesis effect. 5 It can directly participate in intracellular signaling pathways and regulate the phosphorylation of endothelial cell surface proteins, such as vascular endothelial growth factor receptor-2. Furthermore, it can inhibit the phosphorylation of nucleolin and the FAK/c-Raf/MEK1/2/p38/ERK1 signaling pathways5–7 and regulate various aspects of the internal tumor microenvironment, such as increasing the pH value inside the tumor and increase the pressure between tissues. 8 It works via the abovementioned pathways to inhibit the migration of endothelial cells that form blood vessels. This inhibits tumor angiogenesis and blocks the nutritional supply to tumor cells, consequently inhibiting tumor proliferation and metastasis. This is the basic principle of action of most targeted anti-tumor angiogenesis drugs, including RHES.
In September 2005, RHES was approved for the treatment of non-small-cell lung cancer (NSCLC) by the State Food and Drug Administration of China. 9 Since then, scholars have reported through clinical experience and empirical evidence that RHES + radiotherapy and/or chemotherapy has an effect superior to that of a single treatment on various malignant tumors, such as metastatic melanoma and gastric cancer.10,11 It also showed a good therapeutic effect and good safety in the treatment of NPC. 12 In mouse xenograft model experiments, RHES was found to enhance the radiosensitivity of NPC cells by reducing VEGF expression. 13 Yang et al. 14 found that RHES can normalize the tumor vasculature, which can alleviate hypoxia. Jin et al. 15 conducted a phase II prospective randomized clinical controlled study (NCT01612286) to explore the clinical efficacy of RHES combined with gemcitabine-cisplatin in the treatment of advanced NPC. They found that this combination can lead to effective tumor regression, control disease progression, and improve prognosis in advanced NPC. Yin et al. 16 retrospectively analyzed the efficacy and safety of CCRT + RHES versus CCRT alone in locally advanced NPC. They found that CCRT + RHES significantly prolonged 3-year progression-free survival (PFS) and distant metastasis-free survival (DMFS) in NPC.
For locally advanced NPC, the prediction of early treatment response is crucial in driving doctor's decision making in terms of selecting and modifying treatment options. However, the selection of the ideal treatment is still ambiguous in clinical practice. Thus, for improving the treatment effect and reducing the economic and psychological burden of patients, effective biomarkers are a hope to be explored to enable the selection of appropriate strategies before treatment. Therefore, the pretreatment detection of patients who are sensitive to RHES + CCRT is important. The prediction of treatment efficacy with the help of a reliable and practical technique before treatment can aid formulation and adjustment of individualized treatment strategies to avoid unnecessary adverse reactions and medical expenses.
However, besides the clinical staging and inaccuracy of treatment techniques, tumor heterogeneity is an important influencing factor causing treatment failure in patients with NPC. As we know, a relationship between radiomics features and tumor biology has been acknowledged for a long time. 17 Radiomics noninvasively extracts high-throughput texture features on medical imaging images using the principles of computer technology. Furthermore, screening these features and constructing models help evaluate tumor heterogeneity and tumor biological characteristics. At the time of this publication, scholars have applied radiomics to the diagnosis of glioma, rectal cancer, and other conditions to carry out many studies, and achieved good results. 17 Some studies have also been conducted on NPC, which revealed that the model constructed based on the texture features of MRI images was instrumental in predicting the treatment response of NPC to intensity-modulated radiotherapy (IMRT), IC, and NC and building a reliable and accurate predictor of tumor treatment response.18–20 A recent multicenter study found that the nomogram based on pretreatment radiomics and clinical factors could predict the prognosis offered by different treatment options to patients with NPC and recommend the best treatment plan accordingly. It has the potential to become a tool to promote personalized treatment of NPC. 21
Therefore, the predictive factor based on the radiomics signature of MR imaging may be a reliable and practical predictor to discriminate responders or nonresponders before RHES + CCRT treatment for NPC to avoid predictably ineffective RHES + CCRT treatment. This study was aimed at investigating an MRI radiomics model based on pretreatment texture features to explore its application value in predicting the early response of RHES + CCRT for NPC.
Materials and Methods
Patient Selection and Clinical Characteristics
Data for this study were obtained from our hospital. In total, 78 patients confirmed as having NPC via biopsy analyses and undergone RHES + CCRT treatment between January 2017 and February 2021 were retrospectively reviewed. The inclusion criteria for the study were as follows: (1) pathological confirmation of NPC; (2) NPC patients with III to IV a stage (According to eighth edition of the American Joint Committee on Cancer/Union for International Cancer Control TNM staging system); (3) patients having undergone no treatment before MRI; (4) patients with a clinical Karnofsky performance score > 70; (5) patients having undergone RHES + CCRT treatment; and (6) patients with stipulated MRI examination times: pretreatment MRI should be completed within 7 days before treatment and MRI after RHES + CCRT should be completed at the end of the treatment. The exclusion criteria of our study were as follows: MRI with significant denture artifacts (n = 2), NPC with stages I–II malignancy or distant metastases (n = 9), and patients with no MRI at the end of the treatment (n = 2). Therefore, 65 of the initial 78 patients were included in the study (Figure 1).

Flowchart of inclusion and exclusion criteria used in the current study.
Data on age and sex of the patients and pathologic type, TN staging, and morphologic size of the lesions were collected as clinical factors. All clinical records and MRI images were collected and recorded by 2 radiologists separately. Any disagreement was resolved through discussion.
This retrospective study was approved by the Medical Ethics Committee, and owing to the retrospective design, the requirement for individual informed consent of this research was waived. All patient details have been deidentified in this study. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 22
RHES Combined with CCRT
CCRT: IMRT was planned and implemented using the Eclipse system (Varian Medical Systems). The target area and dose design of IMRT for NPC were based on the expert consensus of the Radiation Treatment Oncology Organization (RTOG) and reports 50 and 62 of the International Radiation Unit and Measurement Organization (ICRU). The radiation doses of Nasopharynx gross tumor volume (GTVnx), lymph node gross tumor volume (GTVnd), clinical target volume-1 (CTV-1), and clinical target volume-2 (CTV-2) were 70 to 72, 64 to 72, 62 to 64, and 54 to 56 Gy, respectively, 30 to 33 fractions, 5 times a week for 6 to 7 weeks. For concurrent chemotherapy, cisplatin (80 mg/m2) was administered for 3 cycles. A cycle included 21 days. Meanwhile, antiemetic, live-protective, and stomach-protective measures and hydration were carried out.
RHES: All patients received RHES (solubilized in 250 mL of 0.9% normal saline; dosage: 7.5 mg/m2/day) intravenously for the first 10 days of CCRT for 3 cycles. The RHES was provided by Simcere Pharmaceutical Research Co., Ltd.
Criteria for Treatment Response
The tumor treatment response was separately evaluated by 2 doctors according to the Response evaluation criteria in solid tumors (RECIST) 1.1 criteria. 23 They used the maximum diameters of primary tumor measured on CE T1-weighted imaging obtained before and after RHES + CCRT treatment using tools of the picture archiving and communication system (PACS; Carestream). Both radiologists were blinded to patients’ information.
According to the changes observed in the maximum diameters at the 2 time points, the curative effect was identified as follows: (1) complete response (CR), wherein the MRI nasopharyngeal mass has retreated, and the mucosal thickness is <5 mm with no obvious abnormalities noted on nasopharyngoscopy, (2) partial response (PR), wherein a retreat rate of >30% was noted, and (3) stable disease (SD), wherein a retreat rate of <30% was noted. None of the cases in this study showed disease progression. Efficacy evaluation was performed at the end of CCRT. Patients with CR and PR were defined as responders; SD was defined as nonresponders.
MRI Imaging Protocols
All 65 patients underwent routine plain scan and contrast-enhanced MRI examination of the nasopharynx and cervical region using head and neck coils with a 1.5 Tesla Scanner (GE Signa Echospeed; GE Medical Systems). We chose axial T2-weighted imaging with fat suppression (T2WI_FS) and T1-weighted imaging with contrast enhancement (T1WI_CE) for radiomics analyses. In this study, we used the same geometric parameters of T2WI_FS and T1WI_CE; the main imaging parameters were as follows: field of view = 240 mm × 230 mm, matrix = 232 × 219, spatial resolution = 0.25 mm × 0.25 mm × 5.0 mm, slice thickness = 5 mm, spacing between slices = 1 mm, repetition time (TR)/echo time (TE) = 6000/70 ms (T2WI_FS), and TR/TE = 550/8.1 ms (T1WI_CE). The contrast agent used was gadolinium-diethylenetriamine penta-acetic acid (Gd-DTPA, Magnevist; Schering Diagnostics AG) at 0.1 mmol/kg of body weight and an injection rate of 2 mL/s.
Tumor Segmentation and Radiomics Feature Extraction
All the MRI images of the enrolled patients were exported from the PACS system in Digital Imaging and Communications in Medicine (DICOM) format, and we used the ITK-SNAP software (www.itksnap.org, version 3.6.0) for image segmentation. The key points for image segmentation were as follows: removing blood vessels with a diameter of >3 mm inside the tumor and removing secretions and impurities on the surface of the nasopharyngeal mucosa.
Two radiologists who had rich experience in head and neck diagnosis and were blinded to all clinical records of patients delineated the primary NPC layer by layer on the T2WI_FS image and accordingly obtained the three-dimensional volume of interest (VOI) of the primary tumor. Since the geometric parameters of T1WI_CE and T2WI_FS were the same, the VOI was directly copied to the T1WI_CE sequence, and then, the VOI of the tumor lesions on the T1WI_CE images was obtained using the ITK-SNAP software.
In the current study, we used an artificial intelligence software named FeAture Explorer (ver. 0.3.6 on Python 3.7.6) 24 or MRI texture feature extraction. It extracted a total of 144 texture features from the sequences of T2WI_FS and T1WI_CE. The radiomics features extracted included the original, shape, first order, gray-level co-occurrence matrix (GLCM), and gray-level run length matrix (GLRLM). The radiomics features extracted by the 2 doctors were tested for consistency within and between groups. The flow of our study is shown in Figure 2.

Flowchart of building an MRI-based prediction model to predict the efficacy of RHES + CCRT in patients with NPC.
Before using the machine learning model to perform dimensionality reduction analysis on texture features, we used the Z-score method to standardize the mathematical values of all texture features of the patient to eliminate the unit limitation of these texture features.
Statistical Analysis
Statistical analysis was performed using SPSS 20.0 (SPSS Inc.) and R software (version 4.1.0, https://www.r-project.org). For analyzing differences in clinical factors among the groups showing different levels of efficacy, the independent t-test or Mann–Whitney U test was used for numerical variables according to the results of normal distribution of data, and chi-square test was used for categorical variables. Intra-class correlation coefficient (ICC) values with 95% confidence interval (CI) were used to assess the inter-reader agreement of the quantitative measurements. Only the features with an ICC > 0.7 were selected for analysis of radiomics features.
Feature Selection and Radiomics Signature Building
In this study, we set the labeled the responder group as 0 and the nonresponder group as 1. First, we compared the features between the groups using the Mann–Whitney U test. Only the features identified to be significantly different between the 2 groups (P < .05) were selected for the next step of univariate logistic regression analysis. Second, univariate logistic regression was used to explore whether the features were discriminative between the 2 groups. Then, due to the redundancy of the features, we used the maximum relevance minimum redundancy (mRMR) algorithm for dimensionality reduction analysis of texture features that were screened out. After removing redundant and irrelevant texture features using mRMR algorithm, only 8 texture features were finally retained. For each patient, the Rad score of the primary tumor was calculated from the linear combination of texture features selected by mRMR and the weighting coefficient corresponding to each feature. Finally, multivariable logistic regression was used to select the most predictive feature subset, and constructed the prediction models based on clinical factors and the Rad score, including radiomics models (only the Rad score), clinics models (only clinical factors), and combined models (the Rad score combined with clinical factors).
Model Performance Evaluation
In considering the small sample size of the data, we didn’t split the data into training and validation groups, but a 10-fold cross-validation method was used to train and validate machine learning classifiers and prediction models to avoid overfitting of image data. The specific step was to divide the image data into 10 equal parts, taking 9 of them for training and then using the remaining one for verification. The average of the 10 results was used as an estimate of the accuracy of the classifier or prediction model. 25
We developed the receiver operating characteristics (ROC) curves to evaluate and compare the predictive ability of the radiomics model, clinics model, and combined model. Next, to evaluate model performance, we calculated the area under the ROC curve (AUC), sensitivity, accuracy, specificity, positive predictive value (PPV), and negative predictive value (NPV). Finally, we obtained the nomogram, ROC diagram, and calibration curve diagram, which were used to measure the consistency between the predicted RHES response probability and the actual RHES response probability. To evaluate these 3 models, we obtained the goodness-of-fit of the prediction model and the decision curve using the Hosmer–Lemeshow test.
Results
Patient Characteristics
After the RHES + CCRT treatment, 41 of 65 patients (63.0%) were allocated to the responder group and 24 of 65 to the nonresponder group. In this study, the shortest diameter statistically significantly differed between the 2 groups (P = .036). No other clinical factor significantly differed between the groups (P > .05) (Table 1).
Characteristic Differences Between Responders and Nonresponders among Patients with Nasopharyngeal Carcinoma (NPC) were Determined Using the t-Test (age, Longest Diameter, and Shortest Diameter) or chi-Square Test (Classification Variables).

*P < .05.
Radiomics Signature Development
First, using the Mann–Whitney U test, 15 texture features of T2WI_FS were screened out from among 144 texture features extracted from the T2WI_FS and T1WI_CE. More details are shown in Figure 3A. Univariate logistic regression revealed 11 texture features that were discriminative between the responder and nonresponder groups. Then, we used the mRMR to further optimize, and eliminated 3 of these 11 factors, finally retaining 8 texture features that were strongly discriminative between the 2 groups (Figure 3B).

(A) The P value of the 144 texture features was shown on the Manhattan plot. Fifteen texture features above the line y = 1.3 were features with P < .05. (B) The final 8 feature sets were filtered out using the maximum relevance minimum redundancy (mRMR) algorithm.
The images (T2WI_FS)-based Rad score was calculated based on the features for every patient as follows:
Predictive Performance of Models
In this study, the result of the 10-fold cross-validation showed that the mean AUC of radiomics was 0.689. By constructing a combined model based on Rad score and clinical predictors, in this study, we verified the ability and potential advantages of a model constructed based on pretreatment MRI radiomics to evaluate RHES + CCRT treatment response and verified its reliability and stability (Figure 4). The details of performance analysis of the combined model, radiomics model, and clinics model in predicting the efficacy of RHES + CCRT for NPC are shown in Table 2 and Figure 4B.

(A) Constructed nomogram. (B) Receiver operating characteristic (ROC) curves of the clinics, radiomics, and combined models. (C) Decision curve of the nomogram. (D) Calibration curve.
Performance of Clinics, Radiomics, and Combined Models in Assessing RHES + CCRT Response in NPC
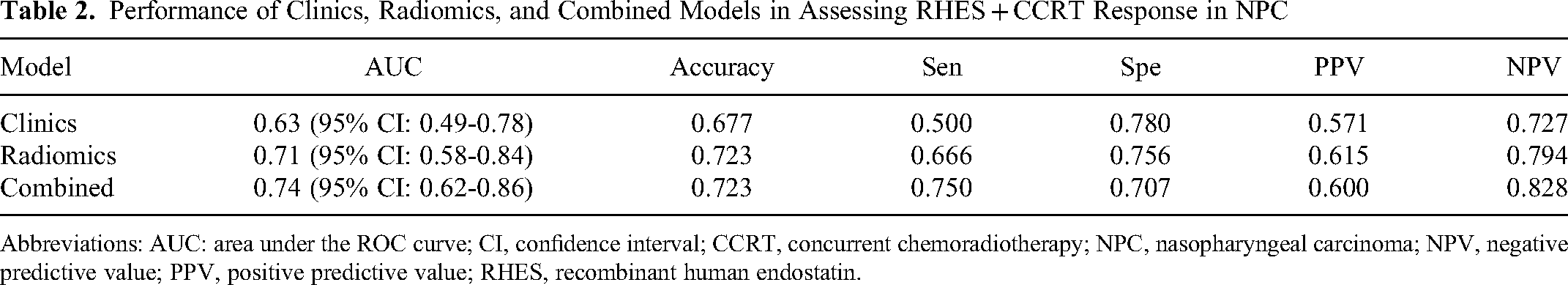
Abbreviations: AUC: area under the ROC curve; CI, confidence interval; CCRT, concurrent chemoradiotherapy; NPC, nasopharyngeal carcinoma; NPV, negative predictive value; PPV, positive predictive value; RHES, recombinant human endostatin.
The results revealed moderate diagnostic efficiency of the combined model and radiomics model in distinguishing between responders and nonresponders. The AUC of the combined model and radiomics model was 0.74 (95% CI: 0.62-0.86) and 0.71 (95% CI: 0.58-0.84), respectively, and was higher than that of the clinics model (0.63, 95% CI: 0.49-0.78]). Compared with the radiomics model, the combined model, which combined imaging texture features with selected clinical factors, showed a marginally better diagnostic performance in predicting RHES + CCRT treatment response. The accuracy of the combined model and radiomics model was higher than that of the clinics model (0.723, 0723 vs 0.677).
The calibration curve of the combined model showed average calibration (Figure 4D), indicating that the combined model can be used to judge the prognosis of patients with an NPC primary tumor after RHES + CCRT treatment with average predictive power, and the sample size needs to be increased in further researches. The decision curve showed that in patients with advanced NPC, the combined model and radiomics model predicted the treatment response to RHES + CCRT better than the clinics model. However, the difference between the combined model and radiomics model was not significant (Figure 4C).
Discussion
In this study, we focused on 65 patients with NPC having undergone RHES + CCRT, among whom 41 (63.0%) were allocated to the responder group and 24 to the nonresponder group. The tumor treatment response of our study was evaluated at the end of the RHES + CCRT treatment. This was different from the previous studies, which remain only about 16% of patients treated with CCRT remained residual tumor 6 months after the treatment. 3 In a study by Yin et al., 16 the CR rates of nasopharyngeal lesions were 53.5% in the RHES + CCRT group 3 months after radiotherapy.
Since the use of RHES for the treatment of tumors is still in phase III clinical trials, there were fewer NPC patients treated with this drug in the last few years in our institution. In addition, this study strictly adhered to the enrollment requirements and included only patients with locally advanced NPC treated with RHES + CCRT. Therefore, the number of cases included in this study was relatively small. Further researches with a larger sample size are needed in the future to overcome such challenges. In the current study, we did not split the data into training and testing groups because of the small sample size of the data. However, 10-fold cross-validation was applied, proving that texture analysis was valuable in discriminating between responder and nonresponder groups and that the result was not due to overfitting. In the study by Li et al., 25 models were trained and validated using stratified 10-fold cross-validation because of the small sample size.
Recently, to predict the early response to treatment in patients with NPC, the following 3 imaging modalities are commonly used: diffusion-weighted MR imaging (DWI), 26 intravoxel incoherent motion diffusion-weighted MR imaging (IVIM-DWI), 27 and dynamic CE MR imaging (DCE-MRI). 28 However, researches on the early response of RHES + CCRT in patients with NPC using these imaging modalities are relatively rare. 29 Based on the principle that radiomics can reflect the heterogeneity of tumors and is related to treatment efficacy and prognosis, our study innovatively established and validated a multi-parameter MRI-based radiomics approach to predict early response to RHES + CCRT in patients with advanced NPC.
In our research, the AUC values of the combined model and radiomics model were 0.74 and 0.71, respectively, and both of them were higher than the AUC of the clinics model (AUC = 0.63). Compared with the radiomics model, the combined model, which combined imaging texture features with selected clinical factors (shortest diameter), showed marginally improved diagnostic performance in predicting treatment response to RHES + CCRT. Notably, the endpoint in this study was clinical treatment response after RHES + CCRT. The model combined radiomics and clinical factors seemed to be more sensitively and closely related to our defined clinical endpoint when compared with radiomics or clinical information alone. It is expected that instead of functional MRI technology, a reliable predictive model can be constructed using conventional sequence texture features to predict the response of RHES + CCRT in patients with NPC in the future.
In our study, even though the baseline shortest diameter was the only aspect that significantly differed between responders and nonresponders, we found the combined model to be better than the radiomics model or clinics model alone. In a retrospective study, a radiomic nomogram was established by combining a radiomics signature with TNM, which showed significantly better prediction of PFS in patients with NPC than TNM alone. 30 One study reported the development of a radiomics nomogram that integrated radiomics signatures from the joint T1-CE, T1-WI, and T2-WI with all the clinical factors. This radiomics nomogram provided a higher concordance index (C-index) in both the training cohort (TC) and validation cohort (VC), suggesting that this model was more accurate than the clinics model or radiomics signature model in predicting IC response in patients with NPC. 31 In another study, scholars found that a model that factored in T1-CE-based uniformity, tumor volume, and the overall stage had better predictive power than a model factoring in either tumor volume or the overall stage in terms of the PFS, and the AUC values were 0.825, 0.659, and 0.616, respectively. 32 Therefore, it can be concluded that the combined model based on radiomics and clinics models better predicted treatment response of patients with NPC to different regimens.
In our study, the finally retained 8 texture features that were strongly related to responder and nonresponder labeling of the patients were calculated from T2WI_FS, including T2WI_FS_GLCM, T2WI_FS_GLRLM, and T2WI_FS_Shape. Those features of multi-parameter MRI described the distribution of voxel intensity within the image as well as represented the heterogeneity of NPC. 33 For example, GLCM-based features reflected tumor roughness and heterogeneity. 34 However, the established model and prediction accuracies varied with different research designs. Simultaneously, the types and numbers of extracted imaging features would differ among different modeling methods. 35 One study focused on 2 different treatments in patients with advanced NPC and compared the performance of radiomics in predicting IC response to both the treatments. In that study, a total of 1188 imaging features were extracted from joint T1-CE, T1-WI, and T2-WI, and the researchers reported that the features extracted from these 3 sequences had good performance in predicting treatment response, with the accuracy of TC and VC being 0.852 and 0.853, respectively. 34 In the study of Zhao et al., the results also showed that a model based on joint T1-CE, T1-WI, and T2-WI had a better prognostic performance in evaluating IC response than a radiomic characteristic model. 31
Our results suggest that the accuracy of combined model and radiomics model for RHES + CCRT response assessment in NPC were better than those of the clinics model (0.723, 0.723 vs 0.677). At the same time, we found that the accuracy of combined model and radiomics model were the same. There was no improvement in the accuracy by combining the imaging texture features with the selected clinical factor. This may be related to the enrolled relatively small sample size in this study, which may affect the final results of our study. Based on the radiomics signature of MR imaging, a predictive factor was developed to preoperatively discriminate between responders or nonresponders scheduled to undergo RHES + CCRT treatment for NPC. This would allow individuals identified as nonresponders to avoid predictably ineffective RHES + CCRT. However, before a radiomics signature can be considered clinically useful and applicable, further external verification and standardized data processing methods are required because the PPV for the combined model was 0.600.
Because of its retrospective nature, our study had some limitations. First, due to the critical inclusion criteria of patients included, we had a relatively small sample size that may affect the final conclusion of the study. Second, this study was a single-center study, which may limit the applicability of our research results to patients from other institutions, and further external validation research is needed to expand this applicability. Third, only anatomical MRI technology was used for research, and functional MRI was not included in the research design. Fourth, our study was based on RECIST 1.1. We delineated tumor response or nonresponse by volume changes before and after RHES + CCRT, which may not be the best indicator for clinical results. Fifth, no statistical methods were used to calculate the sample size selected for this study. Finally, this study did not carry out related studies on the lymph node metastasis of the neck region of NPC at the same time, and this part of the content still has certain challenges.
Conclusion
In conclusion, in this study, the pretreatment MRI-based radiomics may be a noninvasive and effective method for the prediction of RHES + CCRT early response in patients with NPC. The radiomics signature as an MRI-based imaging biomarker may provide a valuable and practical method for promoting personalized treatment for and optimizing the management of patients with NPC.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank members of the Department of Radiology of the First Affiliated Hospital of Guangxi Medical University for technical support. We thank Medjaden Inc. for scientific editing of this manuscript.
Author’s Note
The preprint of our manuscript has been published in Research square.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This retrospective study was approved by the Medical Ethics Committee of First Affiliated Hospital of Guangxi Medical University, China (No. KY-E-294, 2021), and owing to the retrospective design, the requirement for individual informed consent of this research was waived.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Projects for Research and Development of Medical and Health Appropriate Technology of Guangxi Zhuang Autonomous Region (grant number S2018087).

