Abstract
Urothelial carcinoma (UC) is the most frequent malignancy of the urinary tract, which consists of bladder cancer (BC) for 90%, while 5% to 10%, of urinary tract UC (UTUC). BC and UTUC are characterized by distinct phenotypical and genotypical features as well as specific gene- and protein- expression profiles, which result in a diverse natural history of the tumor. With respect to BC, UTUC tends to be diagnosed in a later stage and displays poorer clinical outcome. In the present review, we seek to highlight the individuality of UTUC from a biological, immunological, genetic-molecular, and clinical standpoint, also reporting the most recent evidence on UTUC treatment. In this regard, while the role of surgery in nonmetastatic UTUC is undebated, solid data on adjuvant or neoadjuvant chemotherapy are still an unmet need, not permitting a definite paradigm shift in the standard treatment. In advanced setting, evidence is mainly based on BC literature and retrospective studies and confirms platinum-based combination regimens as bedrock of first-line treatment. Recently, immunotherapy and target therapy are gaining a foothold in the treatment of metastatic disease, with pembrolizumab and atezolizumab showing encouraging results in combination with chemotherapy as a first-line strategy. Moreover, atezolizumab performed well as a maintenance treatment, while pembrolizumab as a single agent achieved promising outcomes in second-line setting. Regarding the target therapy, erdafitinib, a fibroblast growth factor receptor inhibitor, and enfortumab vedotin, an antibody-drug conjugate, proved to have a strong antitumor property, likely due to the distinctive immune-genetic background of UTUC. In this context, great efforts have been addressed to uncover the biological, immunological, and clinical grounds in UTUC patients in order to achieve a personalized treatment.
Introduction
Urothelial carcinoma (UC) is the most frequent tumor of the urinary system, it may originate from lower (bladder and urethra) or upper (renal pelvis, calyces, and ureter) urinary tract.
Bladder cancer (BC) makes up 90% of UCs and, according to 2022 National Cancer Institute Surveillance, Epidemiology and End Results program, kidney, and renal pelvis cancer represents 4.1% of all new cancer cases in the US, with 79 000 new diagnoses in 2022. 1
Upper urinary tract UCs (UTUC) accounts for 5% to 10% of UCs, with an estimated annual incidence of almost 2 cases per 100 000 persons. 2
Even though BC and UTUC both derive from the urothelium, they arise from separate embryonic tissues, therefore presenting distinct phenotypical and genotypical (genetic and epigenetic) profiles with relevant clinical and therapeutic implications. 3 Moreover, the physiologic function of the upper urinary tract (ie urine collection from the kidney and “transport” of urine, by pressure and gravity, to the bladder) is different from the bladder function (ie reservoir), resulting in different “time of contact” between urine and urothelium. UTUC is associated with specific risk factors including exposure to smoking, aristolochic acid (potentially leading to the Balkan endemic nephropathy, a familial chronic tubulo-interstitial disease with a slow progression to terminal renal failure 4 ), arsenic or occupational carcinogens, analgesic abuse, hypertension, long-standing urinary obstruction, and it is common in patients with Lynch syndrome (LS). 5 From a genetic point of view, recent molecular studies provided evidence of different gene- and protein-expression profiles in BC and UTUC, 6 while in terms of immunopathological features UTUC results predominantly luminal papillary and T-cell deplet. 7
UTUC appears to be more aggressive than BC, and correlates with poor clinical outcomes: two-thirds of UTUC are invasive at diagnosis, with a 5-year specific survival ranging from 50% for pT2/T3 stage to 10% for pT4 stage. 2 Older age, smoking status, poor performance status, obesity, ureteral site or multifocal tumors and delay between diagnosis and surgery are all preoperative factors associated with decreased cancer-specific survival (CSS); postoperative prognostic factors include tumor stage and grade, lymph node (LN) involvement, lymphovascular invasion (LVI), positivity of surgical margins and extensive tumor necrosis. 2
According to the European Association of Urology Treatment Guidelines the gold standard for localized low-risk UTUC is kidney-sparing surgery (KSS), followed by endocavitary treatment with chemotherapeutic agents and/or Bacillus Calmette-Guerin (BCG). Radical nephroureterectomy (RNU) with bladder cuff excision and LN dissection associated with adjuvant platinum-based chemotherapy is the treatment of choice for high-risk UTUC.2,8 Neoadjuvant chemotherapy may be considered. 9
Metastatic disease is treated with systemic therapy. Platinum-based chemotherapy represents the backbone of first-line treatment strategies, while there is no defined therapy for later lines. Recent evidence supports the use of immune checkpoint inhibitors (ICIs), although not allowing any definite conclusion.10,11
In this review, we aim to provide the latest updates about UTUC characteristics, biological and clinical behavior, and therapeutic management, in order to underline its distinctiveness and improve our understanding of this complex and still lethal disease (Figure 1).

Graphical abstract illustrating the main urinary tract urothelial carcinoma (UTUC)-centred fields analyzed in the present review.
Methods
The present work is not intended to be a systematic review, nonetheless we employed the following search strategy and specific keyword combinations to exploit the Medline database through PubMed: “urothelial carcinoma,” “upper urinary tract carcinoma,” “genetics,” “embryogenesis,” “immune microenvironment,” “molecular subtypes,” “prognosis,” “clinical management,” “treatment,” “chemotherapy,” “immunotherapy,” “surgery,” “radiotherapy,” “biomarkers.” No restrictions were placed on the year of publication. Identified original/review articles were examined and the most relevant works were selected according to their level of evidence.
Results
UTUC as a Distinct Tumor Disease
Embryogenesis
Urinary tract development starts in the fifth gestational week by the union of structures from different embryological origins and requires reciprocal interactions between epithelia (epithelial ureteric bud, UB) and mesenchyme.12,13 The UB emerges from the nephric duct (ND), that is the central component of pro/mesonephros, and originates from mesoderm cells of mid-gestation embryos. It eventually forms the collecting duct and ureter of the adult kidney (called the metanephros), while ureter tips induce the formation of nephrons from the circumjacent mesenchyme. 12 Conversely, bladder structure stems from the urogenital sinus, which derives from the urogenital septum and also forms the urethra 14 (Figure 2).

Graphical representation of UTUC embryogenetic process.
To date, 3 classes of secreted proteins are known to be responsible for mesenchymal signals for urothelial development: bone morphogenetic protein 4 (BMP4), retinoic acid (RA), and fibroblast growth factors (FGFs). 15 Forkhead Box F1 (FOXF1) regulates the expression of BMP4 in the ureteric mesenchyme and is, in turn, regulated by the epithelial sonic hedgehog (SHH) signal, whose gene is expressed in the cloaca epithelia. 16 SHH-FOXF1-BMP4 axis controls epithelial differentiation, survival, proliferation and smooth muscle cell differentiation of the surrounding mesenchymal cells. 15 PAX2, PAX8, GATA3, and RET genes appear to be implicated in the regulation of ND morphogenesis.12,17,18
In the view of this complex and multifaceted embryogenetic process, urothelial umbrella cells of the human ureter significantly differ from bladder cells in their uroplakin content, keratin expression pattern, and extracellular matrix-associated proteins.13,19 Moreover, embryogenetic differences between the bladder and ureter are also reflected in distinctive genetic-molecular and immune microenvironmental features of BC and UTUC.
Based on these elements, we speculate that the distinct embryonic origins of UTUC and BC could impact on the specific biological pattern of the neoplasms and their different pharmacological sensitivity
Genomics
Several studies aimed at exploring the molecular background of UTUC in order to better understand the disease and identify specific gene alterations, acting as potential targets for precision medicine.
Some of the strongest evidence emerging from all the published works is that BCs and UTUCs are characterized by a distinct genomic landscape; specifically, although the alterations are usually similar, they present a different prevalence. 20
According to the main findings of several papers, the principal mutated genes in UTUCs are fibroblast growth factor receptor 3 (FGFR3) (20%-74%), chromatin remodeling genes such as KMT2D (27%-65%), KDM6A (10%-94%), STAG2 (7%-60%), CDKN2A (5%-50%), TP53 (0%-47%) and HRAS (2%-13%). The relative percentages are reported in Table 1.7,20–25 Usually, in upper tract carcinoma, the alterations in RTK/RAS/MAPK or FGFR pathways and p53/MDM2 axis, are mutually exclusive. 20
Overview of the Principal Mutated Genes in UTUC.

Abbreviations: FGFR3, fibroblast growth factor receptor 3; KMT2D, Lysine methyltransferase 2D; KDM6A, lysine demethylase 6A; STAG2, stromal antigen 2; CDKN2A, cyclin dependent kinase inhibitor 2A; TP53, tumor protein 53; HRAS, Harvey rat sarcoma virus; LG, low grade; HG, high grade; LS, Lynch syndrome; UTUC, urinary tract urothelial carcinoma.
Conversely, BC has more frequent variations in p53-MDM2 pathway genes, ARID1A, PIK3CA, RB1, ERBB2, and cell cycle pathway-associated genes, the latter especially in metastatic disease.20,22 Mutations in INPPL1 were only observed in BC. 25
BC seems to have a higher median tumor mutational burden 6 and DNA damage repair (DDR) gene mutations, 23 while UTUC has been found to exhibit more microsatellite instability (MSI)26,27 and hypermethylation. 28
Yang et al 25 identified distinct mutational signatures between the 2 sites: BC is often characterized by a mutational signature determining the activation of APOBEC cytidine deaminases (43%), bound to viral infection, retrotransposon jumping or inflammation, whereas UTUC presents more commonly a signature causing deamination of 5-methylcytosine (42%) (Table 1).
Studies inspecting the link between genomic and clinicopathological behavior documented that FGFR3, CREBBP, and STAG2 are typical of low-grade (LG) tumors, while high-grade (HG) UTUCs usually display alterations in TP53, RB1, CDKN2A, and CDKN2B genes, and greater genomic instability20,21,23 (Table 1).
Moreover, significant correlations between gene expression and T stage have been reported: pTa/pT1/pT2 stages have alterations in FGFR3, HRAS, and the RTK/RAS pathway, while TP53, CCND1, ERBB2, ERBB3, and KRAS are typical of pT3/pT4.20,22
Contrasting results on the impact of FGFR3 emerged from the studies conducted by Van Oers et al 29 and Nassar et al, 23 which respectively documented a better survival in patients affected by pT2-pT4 UTUC with FGFR3 mutation, and an association between PIK3CA, EP300, and FGFR3 mutations with poorer survival.
Recently, several works aimed at characterizing gene expression profiles in BC and UTUC, in order to illustrate molecular subtypes, with distinct biological behavior, prognosis and pharmacological sensitivity. With regard to UTUC, a seminal work by Fujii et al 30 identified, through an integrated genomic analysis of 199 UTUC cases, 5 molecular subtypes (hypermutated, TP53/MDM2, RAS, FGFR3, and triple negative [TN]) which correlated with distinct gene expression profiles, tumor location/histology and clinical outcomes. When it comes to BC, Kamoun et al 31 focused on genomic alterations in 7 genes, FGFR3, CDKN2A, PPARG, ERBB2, E2F3, TP53, and RB1, and provided a consensus set of 6 molecular classes, characterized by distinct cells infiltration, oncogenic mechanisms, and clinical features: luminal papillary (24%), luminal nonspecified (8%), luminal unstable (15%), stroma-rich (15%), basal/squamous (35%), and neuroendocrine-like (3%).
The Lynch Syndrome
The hereditary nonpolyposis colorectal cancer Syndrome, known as LS, is an autosomal dominant syndrome caused by germline mutations of mismatch repair (MMR) genes (MLH1, MSH2, MSH6 or PMS2). It is characterized by a higher incidence of neoplasms like colorectal cancer, endometrial or ovarian cancer, tumors of the small bowel, biliary tract, stomach, and pancreas, and also of the skin, brain or genitourinary tract. 32 Specifically, UTUC is the third most frequent malignancy in LS, 33 with a lifetime risk of 8.4%.
For this higher incidence, the current European guidelines recommend screening for LS in patients younger than 60 years old, affected by UTUC. 2
Patients with LS-associated UTUC are usually younger, affected in the ureter site, with lower exposure to tobacco. 24
Donahue and colleagues juxtaposed the genomic background of patients with LS-associated UTUC and the sporadic one, revealing that LS-UTUC was characterized by a higher median number of mutations per tumor and higher frequency of MSI. Specifically, MSI-high LS-UTUC presents more frequent variations in DDR genes, KMT2D, CREBBP, SMARCA4, and ARID1A. Furthermore, alterations in CIC, FOXP1, NOTCH1, NOTCH3, and RB1 resulted nearly exclusive to the LS cohort.
Immune Background
The role of UTUC tumor immune microenvironment (TIME) has been deeply examined both by immunohistochemistry (IHC) and gene-expression analyses. Nonetheless, our knowledge of the immune milieu of UTUC still results incomplete.
Several investigations have delved into the impact of distinct TIME components, mostly embodied by PD-L1 expression and tumor-infiltrating lymphocytes (TILs) density, on patient survival and response to treatment, although without reaching a consensus. Krabbe et al 34 have identified PD-L1 positivity as an independent prognostic factor of favorable outcome, while other studies have documented a significant association between higher tumor PD-L1 expression and shorter CSS 35 or overall survival (OS). 36 Similarly, a recent metanalysis involving 1406 UTUC patients (from 8 retrospective studies) highlighted that PD-L1 overexpression was related to worse survival outcomes in UTUC patients following RNU. 37 Concerning TILs density and functional properties, Nukui et al 38 observed a positive correlation between higher TILs and poorer differentiation, local invasiveness, OS and progression-free survival (PFS) on univariate analysis, while the work by Wang et al 39 upheld the positive prognostic value of high CD8+ TILs in terms of both disease-free survival (DFS) and OS.
In the view of the critical role of TIME in dictating tumor evolution and modulating response to standard-of-care therapies, several strategies have been developed to therapeutically target each TIME component. Currently, advanced TIME-directed therapies have either been clinically approved or are currently being evaluated in trials, including immunotherapies (ie anti-LAG-3, TIM3, TIGIT), antiangiogenic drugs, and agents directed against cancer-associated fibroblasts and the extracellular matrix. 40
To dissect the main features of UTUC immune cell compartments, Robinson and colleagues have analyzed whole-exome sequencing and RNA sequencing data from HG UTUC tumors showing that UTUC phenotype was predominantly luminal papillary and T-cell depleted. 7 Moreover, they designated FGFR3 as a putative regulator of UTUC immune contexture through the attenuation of interferon gamma signalling, thus partly explaining the well-known limited efficacy of immunotherapy and shedding new light on this still uncovered disease.
Clinical Management
Diagnosis and Staging
Hematuria is usually the earliest symptom of UTUC, but the diagnosis may be also incidental. Nearly 7% of UTUC is already metastatic at diagnosis, thus exhibiting systemic symptoms. 2 In the case of hematuria, an abnormal urinary cytology with normal cystoscopy may suggest HG invasive UTUC. 41 In suspicion of UTUC, computed tomography urography (CTU) has the highest diagnostic accuracy, and it is able to identify ureteral wall thickenings, infiltrating lesions or filling defects. Nonetheless, flat lesions, wall invasiveness, and superficial extension of tumors remain difficult to assess. 42
Magnetic resonance urography is usually performed when iodinated contrast is contraindicated. CT of the chest, abdomen, and pelvis is required to evaluate LN involvement and distant metastasis, while a recent retrospective multicentre study on the use of 18F-Fluorodeoxglucose positron emission tomography/computed tomography for the detection of nodal metastasis in 117 surgically-treated UTUC patients reported promising sensitivity and specificity of 82% and 84%, respectively. 43 A flexible ureteroscopy (URS) has to be performed to characterize suspected lesions and take biopsy. 2 Narrow band imaging, optical coherence tomography and confocal laser endomicroscopy are promising imaging technique which may enhance visualization of tumor microarchitecture. 41 URS biopsy has a diagnostic and prognostic role since can inform on tumor grade and stage, key factors in preoperative risk stratification, 42 and also enables the ureteral cytology, thus increasing the sensitivity of the procedure. 2
Nevertheless, unlike bladder tumor, a significant discordance between URS biopsy and final pathology has been reported, 44 since some URS biopsy specimens are so small to preclude any accurate assessment of invasiveness, thus carrying the risk of understaging.42,45 To minimize this eventuality, a flat-wire basket can be used to perform biopsy, increasing procedure accuracy. 46
TNM classification 2017 is the selected staging system. Differently from BC, a subclassification of muscle wall invasion is not performed, due to the thinner muscle layer of upper tract urinary wall. 47
Therapeutic Approaches: Localized Disease
Role of Surgery
The gold standard treatment for HG or invasive nonmetastatic UTUC is RNU with bladder cuff excision and template lymphadenectomy, regardless of tumor location. With similar survival benefits, KSS is the preferred approach for LG or noninvasive distal ureteral tumors. In addition, KSS should be considered in selected patients with a serious impaired renal function or solitary kidney. 2
Nephron-sparing techniques consist mainly of ureteroscopic ablation and segmental ureteral dissection. The endoscopic management is practicable only for patients with low-risk cancer suitable for complete tumor resection. Percutaneous approach, although not routinely employed due to the availability of advanced endoscopic tools, might be pursued in low-risk UTUC of the renal pelvis which are inaccessible or difficult to manage by flexible URS. Complete distal ureterectomy with neo-cystostomy is indicated for low-risk tumors of the distal ureter which cannot be radically removed endoscopically, whereas segmental resection of the proximal and mid ureter is associated with higher failure rates compared to the distal pelvis. Partial pyelectomy or partial nephrectomy are extremely rarely indicated.48,49
Common postsurgical complications include early and delayed hematuria, wound infections, ileus, incisional hernia, and pneumothorax. Ni et al, 50 showed that laparoscopic nephroureterectomy and the open technique offer similar perioperative safety and comparable oncologic efficacy. According to a review by Linehan et al, 51 the major complications of an endoscopic approach may range from potentially life threatening (eg, sepsis or acute renal failure) to less serious. The most frequently reported complications are ureteral stricture or stenosis, hematuria, pain, LG fever, infections, and ureteral perforation.
Differently from the UC of the bladder, biopsy specimens from ureteroscopic staging do not enable an adequate assessment of the depth of infiltration into the upper urinary tract wall. Therefore, in order to obtain a sufficient biopsy specimen, the decision between RNU or KSS is predominantly based on the preoperative imaging combined with the tumor grade. Considering the risk of understaging and undergrading associated with endoscopic management, an intensive follow-up is recommended when kidney-sparing treatment is chosen. 41
There is no oncological benefit for surgery in patients with metastatic UTUC, except for palliative care. 52
Role of Neoadjuvant and Adjuvant Therapy
Based on retrospective studies, neoadjuvant chemotherapy showed promising outcomes compared to RNU alone, with encouraging pathological downstaging and better.2,53–57 However, no randomized prospective trials have been published yet. Chemotherapy regimens adopted in UTUC patients are largely based on those employed for BC. 2
The use of neoadjuvant chemotherapy in UTUC holds various advantages (ie the chance of delivering higher dose of chemotherapy before surgery, testing chemosensitivity of the disease, tumor downstaging), and limitations (including the risk of overtreatment before an accurate pathological diagnosis).58–60 A phase II trial showed a 14% pathological complete response (CR) rate, obtaining a pathological stage minor than ypT1 in more than 60% of patients. 61 A systematic review and meta-analysis reported that the use of neoadjuvant chemotherapy compared with RNU alone, was associated with significantly better outcomes, in terms of pathologic CR rate (≤ ypT0N0M0), which resulted 11%, pathologic partial response (PR) rate (≤ ypT1N0M0) reaching 43%, and a pathological downstaging (cT > pT), documented in 33% of patients. Moreover, neoadjuvant chemotherapy led to higher performance in OS [HR 0.44] and CSS [HR 0.38] 9 (Table 2).
Prospective Studies Assessing Different Neoadjuvant and Adjuvant Regimens in Localized UTUC.
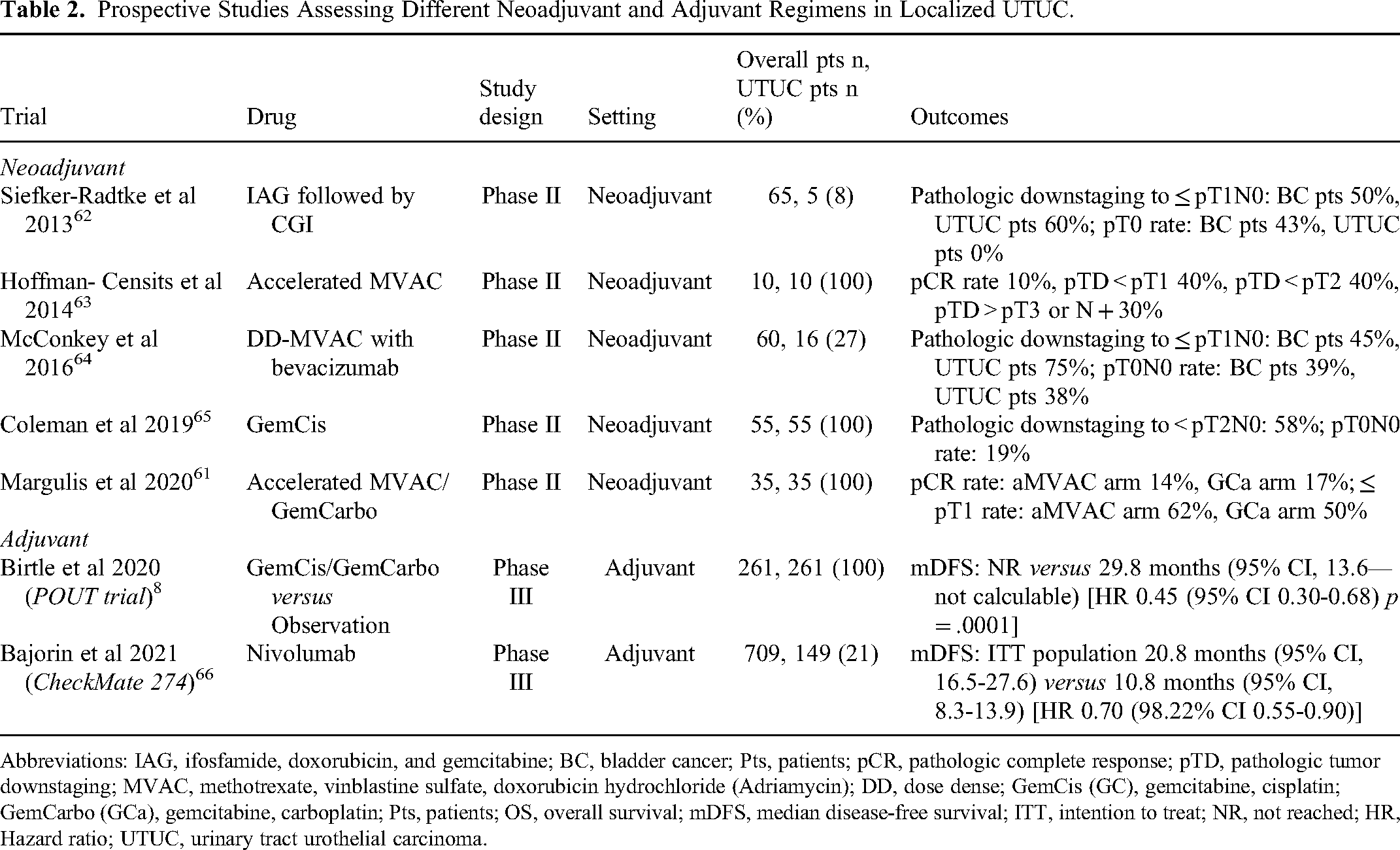
Abbreviations: IAG, ifosfamide, doxorubicin, and gemcitabine; BC, bladder cancer; Pts, patients; pCR, pathologic complete response; pTD, pathologic tumor downstaging; MVAC, methotrexate, vinblastine sulfate, doxorubicin hydrochloride (Adriamycin); DD, dose dense; GemCis (GC), gemcitabine, cisplatin; GemCarbo (GCa), gemcitabine, carboplatin; Pts, patients; OS, overall survival; mDFS, median disease-free survival; ITT, intention to treat; NR, not reached; HR, Hazard ratio; UTUC, urinary tract urothelial carcinoma.
Nevertheless, the evidence for neoadjuvant chemotherapy is at most derived from retrospective or nonrandomized prospective studies, therefore hampering any strong recommendation 9 (Supplemental Table S1).
Conversely, the role of adjuvant chemotherapy in UTUC has been established based on the results of phase III POUT trial, which compared adjuvant gemcitabine-platinum combined therapy (initiated within 90 days after RNU) with surgery alone. 8 Adjuvant chemotherapy resulted in significant DFS benefit [HR 0.45] in high-risk nonmetastatic UTUC (pT2-T4 or N1-3 MO), being also associated with improved metastasis-free survival [HR 0.48], acceptable acute toxicities and only a transient deterioration of patient-reported quality of life. 8 Specifically, 3-year event-free estimates were 71% (95%CI 61-78) and 46% (95%CI 36%-56%) for chemotherapy and surveillance, respectively. Moreover, although not reaching statistical significance yet, chemotherapy conferred a 30% reduction in relative risk of death [HR 0.70, 95% CI 0.46-1.06; P = .09], with a 3-year OS rate of 79% (95%CI 71%-86%) compared to 67% (95%CI 58%-75%) of patient surveillance. 67 Along the same line, in a recent systematic review and meta-analysis, adjuvant chemotherapy versus no chemotherapy, demonstrated benefit in OS (pooled HR 0.77), CSS (pooled HR 0.79) and DFS (pooled HR 0.52). 9
The adjuvant platinum-based chemotherapy is safe. The Pout trial reported a rate of 44% of grade 3 or worse acute treatment-emergent adverse. Patients treated with chemotherapy showed decreases in neutrophils and platelet count, febrile neutropenia, nausea, and vomiting. 8
In the phase III CheckMate 274 trial, involving patients with high-risk muscle-invasive UC, adjuvant nivolumab, compared to placebo, improved DFS in the intention-to-treat population (20.8 vs 10.8 months) and among patients with PD-L1 expression ≥ 1%. 66 In the subgroup analysis, patients with renal pelvis and ureter tumors do not appear to have benefited from adjuvant nivolumab (HR 1.23 95%CI 0.67-2.23 and HR 1.56 95%CI 0.70-3.48, respectively). Updated results demonstrated 12-month DFS rates of 63.5% in nivolumab-treated patients compared to 46.9% in placebo group, 68 although further subgroup analyses mainly focused on UTUC are warranted. On the contrary, the phase III IMvigor010 study, including 54 (6.7%) UTUC patient and evaluating the role of adjuvant atezolizumab versus observation in muscle-invasive UC, did not meet its primary endpoint of improved DFS, 69 thus highlighting the still unclear benefit from adjuvant ICIs in this setting. Nonetheless, the subsequent subgroup analysis conducted by Powles and colleagues reinforced the role of circulating-tumor DNA (ctDNA) as a promising biomarker for molecular residual disease and relapse, and led to the design of the prospective randomized IMvigor 011 trial, testing adjuvant atezolizumab only in ctDNA positive cases.69,70
Despite clear survival benefits deriving from adjuvant chemotherapy, one of the main drawbacks is still the restrained capacity to deliver full doses of cisplatin-based regimens after surgery.2,71,72
All things considered, the evidence for adjuvant chemotherapy appears stronger (with high level of evidence) (Table 2, Supplemental Table S2) than that for neoadjuvant treatment, but data are still insufficient to provide strong recommendations. 9
Role of Radiotherapy in Unresectable Disease
The role of radiotherapy (RT) for UTUC management has not been clearly elucidated, since evidence on salvage/palliative RT in patients with recurrent UTUC is still scarce. 73
RT may offer a safe therapeutic alternative in such patients with nonmetastatic UTUC who cannot tolerate or refuse surgery. 74
Therapeutic Approaches: Advanced Disease
Role of CT in Metastatic Disease
Since limited data are available on management of advanced-stage UTUC, evidence is mainly extrapolated from BC literature and supported by retrospective studies demonstrating that the location of primary tumor (lower vs upper urinary tract) do not significantly affect PFS and OS in patients with metastatic UC treated with platinum-based combinatory approaches.75,76
Platinum-based combination regimens represent the mainstay in first-line treatment, whereas monotherapy is employed as a second- or more-line therapeutic strategy, in detail, single-agent immunotherapy in second-line setting, and enfortumab vedotin in third-line setting.
Specifically, cisplatin-based regimes, such as methotrexate, vinblastine, doxorubicin, and cisplatin or GC (gemcitabine and cisplatin) represent the standard of care in first-line setting and lead to a median OS of around 9 to 15 months.77,78 Carboplatin-based combinations constitute an alternative in patient ineligible for cisplatin due to comorbidities or impaired renal function, although being associated with a short duration of response, poor survival, and high toxicity.79,80
Scarce data are available about the efficacy of other chemotherapeutic agents administered in subsequent lines. Vinflunine plus best supportive care showed a modest, although significant, benefit in OS compared with best supportive care alone, and might be employed as second- or third-line treatment. 81
Role of ICIs and Novel-Targeted Therapy in Metastatic Disease
Immunotherapy was evaluated as first-line treatment, either in combination with chemotherapy, or alone in platinum unfit patients, or as maintenance treatment after platinum-based regimens. The efficacy of atezolizumab-based first-line therapy was evaluated in several trials: as single agent, in phase II, single-arm IMvigor 210 study, 65 which included patients with locally advanced or metastatic UC patients unfit for cisplatin; in combination with cisplatin-based chemotherapy, in phase III, randomized IMvigor 130 trial, involving metastatic urothelial cancer cases.82,83 Phase II, single-arm KEYNOTE 052 trial, assessed the activity of first-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer. 84 Overall, 414 UTUC patients were enrolled in the 3 studies. Overall response rate (ORR) ranged from 22% to 39%. Of note, the combination of atezolizumab with chemotherapy demonstrated a relevant prolongation of PFS compared to chemotherapy alone. 83 Both atezolizumab and pembrolizumab administered as monotherapy in cisplatin unfit patients showed encouraging response rates, survival, and tolerability.82,84
JAVELIN Bladder 100 trial, a phase III study assessing the efficacy of anti-PD-L1 avelumab, as maintenance therapy after first-line platinum-based chemotherapy, documented significantly improved OS in the experimental arm which included 106 patients with unresectable UTUC 85 (Table 3).
Studies Assessing ICIs as First-Line Therapy in Locally Advanced or Metastatic UTUC.

Abbreviations: UTUC, upper tract urothelial cancer; pts, patients; ORR, objective response rate; CI, confidence interval; CT, chemotherapy; mPFS, median progression-free survival; HR, hazard ratio; mOS, median overall survival; ITT, intention to treat; PD-L1, programed death ligand 1; BSC, best supportive care; ICI, immune checkpoint inhibitor.
Concerning the second-line setting, several phase I, II, and III studies tested atezolizumab,86–88 pembrolizumab, 89 durvalumab, 90 avelumab, 91 and nivolumab92,93 in platinum pretreated patients with advanced UC. Among all the above-mentioned compounds, pembrolizumab demonstrated a significant improvement in survival compared to chemotherapy in KEYNOTE 045 trial, in which UTUC patients accounted for 14% 89 (Table 4): in the subgroup analysis the HR for patients with UTUC was better compared with lower tract tumors (HR 0.53 95%CI 0.28-1.01 and HR 0.77 95%CI 0.60-0.97, respectively) . 89
Studies Assessing ICIs as Second or More Line Therapy in Locally Advanced or Metastatic UTUC.

Abbreviations: UTUC, upper tract urothelial cancer; pts, patients; ORR, objective response rate; CI, confidence interval; mPFS, median progression-free survival; HR, hazard ratio; mOS, median overall survival; ICI, immune checkpoint inhibitor.
An increasing number of clinical trials are also evaluating novel targeting agents. Erdafitinib, a tyrosine kinase inhibitor of FGFR1-4, was evaluated in BLC2001, 94 a single-arm, phase II trial, which included a total of 111 metastatic patients, out of which 33 were affected by UTUC, harboring FGFR3 mutation or FGFR2/3 fusion. Median PFS and OS of the overall population were 5.5 and 13.8 months, respectively, with an ORR of 40%. Moreover, Ding et al 95 reported the case of a 67 years old metastatic, chemo-refractory UTUC patient displaying a dramatic response to pembrolizumab in association with erdafitinib.
Among antibody-drug conjugates, enfortumab vedotin, a novel agent targeting the surface protein nectin-4 highly expressed in UC, demonstrated robust antitumor activity in a phase III study including patients previously treated with platinum-based chemotherapy and ICIs. Out of 608 patients enrolled, the primary tumor was localized in upper tract in 205 patients. The subgroup analysis revealed a significant benefit in terms of OS in patients with BC [HR 0.67 (0.51-0.88)], while not reaching statistical significance in patients with UTUC [HR 0.85 (0.57-1.27)]. 96
Finally, numerous trials assessing backbone ICI in combination with chemotherapy, antibody-drug conjugates and tyrosine kinase inhibitors as first- or more-line treatment in UTUC metastatic setting are currently ongoing, underlying the strong biological, immunological, and clinical rationale of personalized therapy in UTUC. 10
Role of RT in Advanced Disease
Among other therapeutic approaches, the role of RT is still marginal, both in metastatic BC and UTUC, being administered mostly with a symptomatic/palliative intent. 97 The current evidence supports the use of RT to rapidly and effectively minimize tumor-induced urinary symptoms, such as hematuria, and demonstrates a similar symptom improvement following hypofractionated palliative RT compared to multifractionated treatments. 97 In addition, as reported by a recent Japanese study, in a selected patient population, consolidative RT (usually more than 50 Gy) may result in long-term disease control and improved quality of life. 98 Finally, although based on a small number of observations, a potential role of stereotactic body radiation therapy in specific cases of oligometastatic UC displaying distant node metastases, has been advised. 99
Prognostic and Predictive Factors
Preoperative stratification into low and high-risk UTUC is fundamental to select patients who are eligible for KSS treatment or RNU, 2 and it is practically based on tumor size, disease focality, cytological and histological grade, histological variants, invasive aspects on CTU, hydronephrosis and history of BC. Other preoperative factors have been demonstrated to impact on prognosis: sessile growth pattern, renal pelvis location, advanced age, too low or too high body mass index, diabetes, and poor performance status confer a worse CSS.2,41,100 Furthermore, smoking exposure and history of BC are associated with a higher risk of intravesical recurrence.2,41
Concerning morphologic classification, it is a well-established notion that pure UCs are characterized by a favorable disease course, while histologic tumor variants are usually associated with advanced stage, multifocality, sessile architecture, necrosis, LVI, and LN metastasis. 101
Several models combining clinical, endoscopic, and imaging features have been developed to improve preoperative risk stratification, although mostly based on retrospective studies with a small sample size.41,45
In the postoperative setting, different nomograms relying on pathological features, have been generated to improve decision-making.41,100 High tumor grade and stage, LN involvement, LVI, and extensive tumor necrosis are linked to worse prognosis.2,102 Furthermore, positive tissue surgical margins, concomitant carcinoma in situ and macroscopic infiltration of peripelvic adipose tissue have been associated with higher risk of recurrence.2,100 Similarly, high expression of immunosuppression proteins such as Tim-3 and PD-1, has been related to bladder recurrence. 103
Among pathological information, solely available after radical surgery, the investigation of specific molecular markers, such as ERBB2 amplification, FGFR3, and TP53 mutation, is known to improve the assessment of oncological outcomes.5,104 Some works have been focused on tissue RNA expression profiles able to subdivide UTUC in molecular subtypes exhibiting distinct clinical characteristics and outcomes.30,45 Moreover, significant prognostic tissue markers, obtained by immunohistochemical analysis on tissue microarrays, have been proposed. In detail, epidermal growth factor receptor/erythroblastosis oncogene B (c-erb), retinoblastoma protein loss, cyclin D1, high molecular-weight cytokeratin, and with E-cadherin have been demonstrated to be variably associated with OS, DFS, and intravesical recurrence-free survival. 36
Beyond the above-mentioned parameters, new prognostic tools are increasingly being explored, especially those that are noninvasive. Analysis of urinary cell-free DNA addressing either genomic or epigenomic alterations, holds great potential as a noninvasive method for the diagnosis of UC, including UTUC, and might also be predictive of prognosis and treatment response. 105
In localized disease, the relevance of ctDNA in identifying patients who may benefit from adjuvant atezolizumab-based immunotherapy has been undoubtedly demonstrated, and the assessment of protein and transcriptomic biomarkers offered new immunologically-relevant insights into the biology associated with ctDNA positivity and atezolizumab efficacy. 70
Regarding the peripheral blood compartment, there has been an increasing interest in inflammatory markers. Several studies documented that high neutrophil to lymphocyte ratio or monocyte to lymphocyte ratio (MLR) and systemic immune-inflammation index (neutrophils * platelets/lymphocytes), were significantly associated with poor OS and CSS.106,107
Finally, regarding clinical/hematological prognostic factors in metastatic UTUC, a recent work conducted by Bersanelli et al 109 provided the first validation of the Sonpavde score 108 (including performance status > 0, hemoglobin < 10 g/dl, liver metastases, time from prior chemotherapy ≥ 3 months) in the UTUC population, proving its efficacy and usefulness for the outcome prediction also in these setting of patients.
However, the identification of reliable prognostic and predictive biomarkers able to improve clinical decision-making in UTUC patients, is still an unmet need.
Future Perspectives
Standard chemotherapy regimens administered according to different schedules, as well as novel targeting and immunotherapeutic agents, are currently exploiting in UTUC patients, Ongoing clinical trials (from https://clinicaltrials.gov/) addressed to UTUC cases, are reported in Table 5.
Actively ongoing studies aimed at assessing different therapeutic combination in locally advanced or metastatic UTUC.

Abbreviations: AE, adverse event; BCG, Bacillus Calmette-Guérin; CSS, cancer-specific survival; CT, chemotherapy; DFS, disease-free survival; DU, distal ureterectomy; EFS, event-free survival; GC, gemcitabine + cisplatin; GFR, glomerular filtration rate; MTD, maximum tolerated dose; MVAC, methotrexate, vinblastine, adriamycin, cisplatin; NA, not available; NACT, neo-adjuvant chemotherapy; ORR, overall response rate; OS, overall survival; pCR, complete pathologic response; PFS, progression free survival; pts, patients; RFS, recurrence-free survival; RNU, radical nephroureterectomy; FGFR3, fibroblast growth factor receptor 3; UTUC, urinary tract urothelial carcinoma.
Conclusive Remarks
In conclusion, UTUC should be considered a unique disease entity in the context of urological malignancies, being endowed with distinctive biologic, genomic, and immune backgrounds which determine a diverse clinical outcome and require tumor-specific management.
Supplemental Material
sj-doc-1-tct-10.1177_15330338231159753 - Supplemental material for Upper Tract Urinary Carcinoma: A Unique Immuno-Molecular Entity and a Clinical Challenge in the Current Therapeutic Scenario
Supplemental material, sj-doc-1-tct-10.1177_15330338231159753 for Upper Tract Urinary Carcinoma: A Unique Immuno-Molecular Entity and a Clinical Challenge in the Current Therapeutic Scenario by Giulia Mazzaschi, Giulia Claire Giudice, Matilde Corianò, Davide Campobasso, Fabiana Perrone, Michele Maffezzoli, Irene Testi, Luca Isella, Umberto Maestroni and Sebastiano Buti in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material (Supplementary Tables S1-S2) for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
