Abstract
Background
Intrahepatic cholangiocarcinoma (ICC) is the second most common primitive liver cancer and is rising in incidence worldwide. Given its low survival and resectability rates, locoregional therapies such as Yttrium-90 transarterial radioembolization (Y-TARE) are increasingly being employed. This retrospective study aim was to confirm and update our previous results about overall survival (OR), safety, and efficacy of Y-TARE in patients with unresectable/recurrent ICC.
Materials and Methods
OS was evaluated as primary endpoint while radiological tumor response at 3 months, based on Response Evaluation Criteria in Solid Tumors (RECIST) 1.1, was considered as secondary endpoint.
Results
Over a total of 49 patients, the overall median survival was 16 months (27-41 months, 95% confidence interval [CI]) from Y-TARE procedure. A significantly longer survival was recorded in naive patients compared to patients previously submitted to any type of liver-directed treatment and radical surgery (18 vs 14 months, P=.015 and 28 vs 14 months, P=.001, respectively). Target lesion and overall objective response for RECIST 1.1 criteria were 64.6% and 52.1%, respectively. Low rates of postprocedural and late complications were recorded.
Conclusions
In unresectable and recurrent ICC, Y-TARE confirms its safety and its potential in increasing OS, especially in naive patients.
Keywords
Introduction
Intrahepatic cholangiocarcinoma (ICC) has been reported to be the second most common primitive liver cancer after hepatocellular carcinoma (HCC). Although a considerably less incidence compared to HCC and a substantial geographical variation due to the exposure to different risk factors and genetic determinants, the incidence of ICC has globally increased, reaching a plateau in the past 10 years and accounting for up to 15% of primary liver cancers.1‐4
Complete surgical resection remains the sole potential curative treatment for ICC. Unfortunately, diagnosis at the early stage of disease has been so far from the exception rather than the rule, accounting for only around 30% of patients admissible for surgical treatment.1,5
The poor prognosis of unresectable ICC is not substantially improved in the last decade, with an estimated median survival of 3 to 8 months.1,4,6 Overall, 5-year survival after resective surgery is approximately 22% to 44%, mainly limited by high and rapid recurrence of ICC, the presence of vascular invasion and positive lymph nodes, an inadequate hepatic functional reserve, and severe comorbidities. 5
Against this background, local ablative and locoregional therapies, such as radiofrequency ablation (RFA) and transarterial chemoembolization (TACE) play an important role in preventing tumor growth and its complications, in relieving symptoms, reducing risk of tumor-related liver failure, and preserving quality of life.7,8
Local ablative techniques such as microwave and RFA are considered the preferred approaches in fewer (possibly up to 5) and smaller ICCs, with a reported median overall survival (OS) ranging from 33 to 38.5 months.9,10 However, local ablation plays a minor role in tumors larger than 5 cm and for those located in the subcapsular region, close to the hepatic hilum or surrounding organs.9‐11 Locoregional therapies, alone or in combination with chemotherapy, have been shown to be of benefit for OS of unresectable ICC.12,13 In patients with multifocal or diffuse liver involvement, TACE has also been demonstrated to be superior to supportive care alone.8,14
At the present time, Yttrium-90 transarterial radioembolization (90Y-TARE) represents a valid therapeutic option for selected patients with unresectable ICC. The 90Y-loaded microspheres act as an internal radiative source with the purpose to deliver a high radiating dose directly to the tumor, minimizing injuries to adjacent liver parenchyma and surrounding tissues.15‐17 The rate of complications may be additionally reduced by performing an accurate patient selection and a complete and precise pretreatment assessment. 15
Safety and efficacy of 90Y-TARE in ICC have been evaluated over time by several studies in which the median OS ranged from 8.7 to 22 months.1,16,18,19
The present study aims to revise and confirm the previous results obtained by our group on safety and efficacy of 90Y-TARE, 16 with special focus on OS of patients with unresectable primary ICC submitted to 90Y-TARE plus standard care.
Materials and Methods
Patients
Medical records of 49 consecutive patients with mass-forming type ICC undergoing 90Y-TARE between 2016 and June 2021 were retrospectively collected and reviewed.
Treatment strategies were preliminary planned by the multidisciplinary tumor board of our Institution after a full investigation of clinical, laboratory, and multimodal imaging features.
Unequivocal unresectable primary or recurrent ICCs were considered suitable for 90Y-TARE if meeting these specific criteria: (1) histologically proven ICC; (2) unresectable primitive ICC or disease relapse/persistence after various medical and surgical liver-directed treatments; (3) Eastern Cooperative Oncology Group (ECOG) performance status score ≤2; (4) total serum bilirubin ≤2.0 mg/dL; (5) granulocyte count ≥1.5×109/L; (6) platelet count ≥50×109/L; and (7) international normalized ratio (INR) ≤1.5.
The following were considered exclusion criteria: (1) gastrointestinal tract shunt fraction unresolvable by coil embolization or (2) estimated lung exposition to a radiation dose >30 Gray (Gy) in a single administration or 50 Gy cumulatively. (3) Child–Pugh class C, (4) extrahepatic life-threatening diseases, and (5) presence of ascites.
The study was conducted in conformity with the ethics guidelines of the Declaration of Helsinki and was approved by our Institutional Review Board. All patients provided a procedural informed written consent. An oral informed consent to participate in the study was obtained from the participants.
The reporting of this study confirms to STROBE guidelines. 20
All patient details were properly de-identified.
Treatment Protocol
All patients eligible for 90Y-TARE underwent pretreatment planning angiogram where tumor feeding vessels, any anatomic arterial variants, and any intratumoral artero-venous shunts were detected. Technetium-99-labeled macroaggregated albumin scanning (99mTc-MAA–SPECT/CT) was consequently performed to detect hepatopulmonary fraction. If suitable, patients were submitted to 90Y resin microspheres (SIR-Spheres; Sirtex Medical) injection under intermittent fluoroscopic visualization, in accordance with the technique previously described.21,22 The amount of injected activity of resin microspheres (activity calculation) was determined, as recommended by the manufacturer, using the partition model method, which is based on the Medical Internal Radiation Dosimetry (MIRD) formalism and 3 compartments: lung, tumor, and normal liver. The prescribed dose was administered with an unilobar distribution according to tumor burden. In case of a bilobar involvement and if considered feasible, a second injection was repeated within the following 30 to 60 days.
Clinical Follow-up and Response to Treatment
A pretreatment anamnestic, clinical and physical evaluation was performed on all patients, as well as a complete specific laboratory profile including liver function, albumin, total bilirubin, complete blood count, coagulation profile, and tumor markers. In all cases, a preliminary radiological evaluation was performed by computed tomography (CT) scan or magnetic resonance imaging (MRI).23,24
After treatment, clinic, laboratory, and radiological follow-up were regularly performed at 1 and 3 months, followed by 3-month evaluations. Retreatment was attempted in each case of incomplete tumor response or progressive intrahepatic disease. Any clinical or biochemical toxicity that occurred after treatment was reported and defined according to the Common Terminology Criteria for Adverse Events of the National Cancer Institute, version 5.0. 25 Any other clinical, laboratory, and imaging information was prospectively acquired and analyzed.
Tumor response was evaluated by expert radiologists in liver imaging using Response Evaluation Criteria in Solid Tumors (RECIST) 1.1.26,27 RECIST 1.1 was preferred to modified RECIST (mRECIST) criteria, as the hypovascular nature and the delayed enhancement characteristics of ICC make response difficult to evaluate with modified RECIST.28,29
The combination of complete response (CR) and partial response (PR) was considered cumulatively as objective response (OR) while disease control (DC) included CR, PR, and stable disease (SD). Radiological response and progression were retrospectively evaluated separately by 2 radiologists with 15 years of pertinent experience in abdominal imaging
Statistical Analysis
OS was evaluated as primary endpoint while efficacy, in terms of radiological tumor response, and safety were considered as secondary endpoints.
Quantitative variables were reported both as mean ± standard deviation and median and range, as applicable. Categorical variables were reported as numbers and percentages. OS was evaluated from the first radioembolization treatment to last encounter or death. Survival data were estimated using the Kaplan–Meier analysis. For each statistical comparison, P values and confidence intervals (CIs) were reported. Univariate analysis between different categories of variables such as performance status, portal vein thrombosis (PVT), prior radical procedure, or systemic chemotherapy and the outcome OS were examined by the log-rank test. P values at <.05 were considered statistically significant. Statistical analysis was performed with SPSS version 22 software (IBM).
Results
As shown in Table 1, baseline demographical and clinical characteristics of 49 patients with unresectable/recurrent ICC were reported and analyzed. Twenty-three patients (47%) were male, in which the majority was ECOG 0 (88%). Hepatic preexisting diseases were unfrequent (cirrhosis 27%, alcoholic disease 10%, cholestatic disease 4%). The most of ICCs were bilobar (57%), with pauci or multifocal distribution (80% in total). Extrahepatic metastases were encountered in 16 cases (33%): the majority of them (27%) had lymphonodal spread (maximum diameter of 3 cm) and 3 cases (6%) had small pulmonary lesions (< 1 cm).
Baseline Characteristics of Patients Before TARE (n=49).

> 37 U/mL.
In addition 16 pts (33%) had non characterizable pulmonary micronodules
Abbreviations: CA: carbohydrate antigen; ECOG: Eastern Cooperative Oncology Group; ICC: intrahepatic cholangiocarcinoma; TAE: transarterial embolization; TACE: transarterial chemoembolization; TARE: transarterial radioembolization.
Eighteen (37%) ICCs had been previously submitted to any type of liver-directed treatment and 12 patients (52%) received systemic chemotherapy before the first radioembolization.
Twenty-two lesions (45%) had recurred after a prior curative-intent surgical procedure (resection or lobectomy), and 13 had been so far submitted to percutaneous ablative (6%) or locoregional treatment (20%).
90Y-TARE Treatment
The principal technical characteristics of the radioembolization procedure and the intraprocedural complications that occurred were reported in Table 2. In particular, treatment targets expressed in terms of targeted liver volume and targeted tumor volume were evaluated. The prescribed median hepatic activity was 1.45±0.56 GBq and the injected tumor dose and lung doses were also reported. No adjunctive administrations were performed.
Characteristics of TARE Procedures (n=49).

BSA: body surface area.
Within 7 days.
Abbreviation: TARE: transarterial radioembolization.
Radiological Response
Complete radiological assessment was performed in 48 (98%) patients since in 1 patient, death occurred before the first radiological evaluation. Overall and target imaging responses at 3 months, based on RECIST 1.1 criteria, are shown in Table 3. Our latest results showed a target lesion OR rate (CR+PR) of 64.6% and an overall OR rate (CR+PR) of 52.1%, both assessed by RECIST 1.1 criteria.
Imaging Responses Based on Overall and Target Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 (49 pts).

Abbreviations: CR: complete response; PD: progressive disease; PR: partial response; SD: stable disease.
Survival
Patients were followed up for a median period of 15 months (range: 1-59 months) from 90Y-TARE and 29 months (range: 3-106 months) from the first diagnosis. At the end of the study, 40 deaths (81.6%) were recorded. The median survival was 16 months (27-41 months, 95% CI) from 90Y-TARE and 34 months (12-20 months, 95% CI) from the first diagnosis.
Baseline, clinical, pathological, and treatment-related factors were evaluated in association with OS from the diagnosis and from the first 90Y-TARE treatment by univariate Cox regression models. These results were summarized in Tables 4 and 5.
Factors Affecting Overall Survival (OS) From Diagnosis: Univariate Analysis.

Dichotomized according to the median value.
Median survival was calculated from the date of diagnoses; log-rank test.
Abbreviations: CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; TARE: transarterial radioembolization.
Factors Affecting Overall Survival (OS) From TARE: Univariate Analysis.
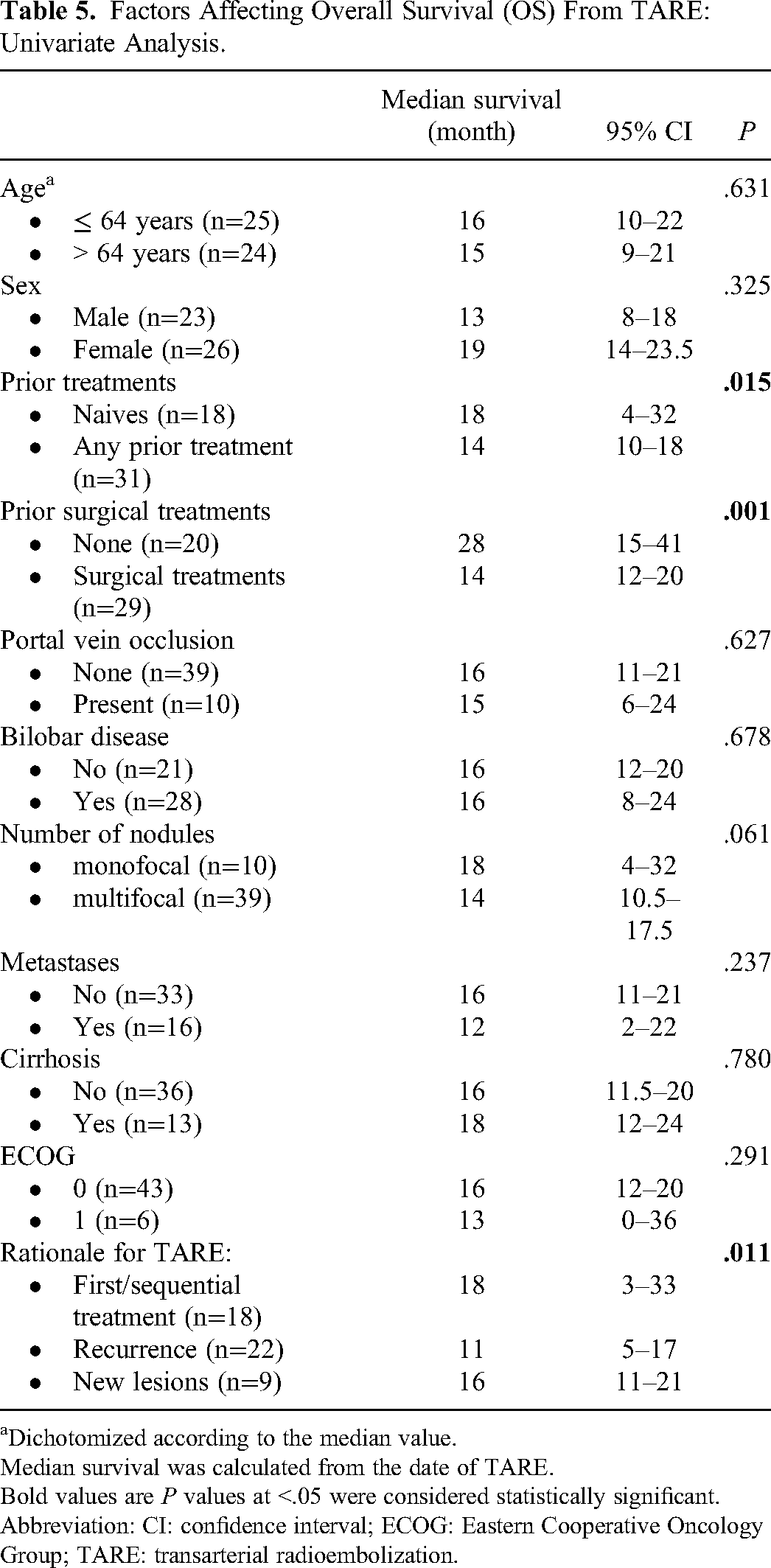
Dichotomized according to the median value.
Median survival was calculated from the date of TARE. Bold values are P values at <.05 were considered statistically significant.
Abbreviation: CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; TARE: transarterial radioembolization.
No statistically significant differences in OS from the time of diagnosis were observed at univariate analysis according to the variables analyzed. Taking into account OS from the first 90Y-TARE, the treatment-naive patients showed significantly higher median survival when compared with those previously treated and compared with those treated with curative-intent surgery (18 vs 14 months, P=.015 and 28 vs 14 months, P=.001, respectively).
After the first 90Y-TARE, 16 patients (32.7%) were submitted to systemic chemotherapy (gemcitabine or gemcitabine plus oxaliplatin) due to disease progression as prescribed by the referring oncologist.
Toxicities
The most frequent adverse events were classified as immediate postprocedural if occurred within 7 days after the procedure and late complications if significant or persistent beyond the mentioned period.
As shown in Table 2, a total of 14 (28.6%) patients experienced 25 postprocedural complications, all characterized by grade 1 of clinical severity. 24 In particular, 11 cases of abdominal pain, 6 cases of postprocedural fever, 6 cases of nausea and vomiting, and 2 cases of fatigue. All the mentioned clinical scenarios reached a complete remission with routine medications.
Late complications included 2 cases (4.1%) of unresolving fever, 3 cases (6.1%) of gastrointestinal ulceration, 3 cases (6.1%) of persistent fatigue, and 1 case (2%) of cholecystitis requiring prolonged hospitalization and conservative treatment. Eight cases (16.3%) of mild ascites were observed within 15 to 90 days after the procedure, and all of them occurred in cirrhotic patients and were resolved by diuretic treatment.
Discussion
Locoregional treatments have largely become a matter of concern in the clinical challenge of management of irresectable or recurrent ICC. A growing interest in TARE with 90Y microspheres is evidenced by the crescent number of clinical studies in the last years, most of them reported improved survival rates.19,29‐31 Despite the increasing literature addressing this issue, the published pooled estimates of post-90Y-TARE survival remain a wide spectrum of results, ranging from a median OS of 8.7 months 32 to longer survival rates showed in Al-Adra et al systematic review including single-arm studies and in several other smaller studies.14,18,33
Even at the present time, as highlighted by Edeline et al., the available results of 90Y-TARE were retrospectively collected and heterogeneous in terms of population of patients enrolled, type of injection delivered, and whether or not any surgical or systemic treatment had been administered prior to first 90Y-TARE. 34 Consequently, the most current results show a heterogeneous median OS ranging from 9 to 22 months. 34 A median OS of 22 months (95% CI, 14-52 months) was reported in the Yttrium-90 Microspheres in Cholangiocarcinoma (MISPHEC) which is, to date, the first prospectively designed trial evaluating the effectiveness of 90Y-TARE in unresectable ICC as first-line treatment in association with chemotherapy.
In our case series, both naïve unresectable tumors and recurrent ICC previously submitted to any other type of locoregional treatment, systemic chemotherapy or liver surgical resection were included. To this must be added the heterogeneity of 90Y-TARE-treated population in terms of natural history of disease at the time of enrolment, defined by the tumor burden and the presence of portal involvement and extrahepatic disease.
The median OS of 16 months reported in our study, even if slightly lower than our previous results, is in perfect alignment with the recent results of MISPHEC trial and seems to confirm the value of 90Y-TARE in unresectable ICC.
As previously suggested by Al-Adra et al., the OS observed is probably negatively influenced by the advanced stages of disease, the poor prognosis, and the progressed time point in the natural history of unresectable ICC. In support of this scenario and as highlighted in our previous series, 16 a significantly longer OS was obtained in patients who did not receive any prior treatment and to curative-intent surgery compared to patients who were naïve at the time of the first 90Y-TARE. Previous chemotherapeutic, resective, ablative, and intra-arterial treatments may both have reduced the amount of functioning parenchyma, both have prolonged the time from diagnosis to first 90Y-TARE, reconfirming the good results of radioembolization as a first-line therapy in unresectable ICCs.
According to the results previously reported, 16 in our series, no association was observed between ECOG performance status and OS from the diagnosis and from the first radioembolization. These results are supported by the better performance status of the patients enrolled compared to the most of other series. The largest percentage of our patients (87.8%) had therefore a performance status of ECOG 0 and none had a performance status of ECOG 2.
Probably due to an insufficient number of cases, no significant decrease in OS was observed when examining the subgroups of patients with portal vein occlusion, bilobar and multifocal disease, and extrahepatic spread. Similarly to the study of Bourien et al, the selection of patients with no major liver involvement (<50%) and very limited extra-hepatic spread mitigates the impact of these factors on survival rates. 35
In the present study, no life-threatening postprocedural and late complications occurred (<Grade 4; no life-threatening events). Overall, postprocedural toxicity was low (28.6% of treated patients) and limited to grade 1 severity. The most frequent late adverse effects were mild ascites, gastrointestinal ulceration, fatigue, persistent fever, and 1 case of acute cholecystitis, in line with those reported in published articles.19,34 Only 1 death was registered within 30 days of treatment. We observed several cases of hepatic dysfunctions, all of which occurred late after treatment and were probably related to disease progression rather than the procedure.
In our study, RECIST 1.1 criteria were preferred for the evaluation of ICC treatment response and posttreatment surveillance was realized using conventional contrast-enhanced CT and MRI, on account of a greater availability, reproducibility, and practicality of these modalities compared to metabolic-based imaging techniques, even if the latter is considered by several studies as earlier and better predictors of response. 36
The optimal imaging response criteria also remain a matter of debate. Some authors have suggested that the change in tumoral 18-fluorodeoxyglucose (18-FDG) activity is more sensitive of treatment response compared to the increase in overall lesion size. 37 Some others have demonstrated how the variable degree of postcontrast enhancement of ICC, differently from hypervascular lesions as HCC, may disadvantage arterial enhancement-based criteria and favor lesion size-based criteria.38,39
The best imaging interval for assessing tumor response to 90Y-TARE is also a controversial issue. In several previous series, the most widely used assessment within 4 weeks after treatment was replaced by the evaluation of posttreatment RECIST 1.1 response at 90 days (3 months), taking into consideration the mechanism of cell damage induced by radioembolization, which becomes manifest over time.40,41 In our study, in agreement with the referring oncologists and radiotherapists, treatment response was evaluated at both 30- and 90-day imaging intervals.
In the present study, several limitations should be encountered. First, the retrospective and single-arm design limits. Second, the contest of single center and a small sample size, which is insufficient for the identification and the analysis of prognostic factors. Other important limitations to be considered are the difficult record of adverse effects, which could have been underestimated due to lacks in patient medical documentation or absent correlation of adverse events to 90Y-TARE procedures. For these reasons, a precise comparison of adverse effects rates with other published results is lacking.
The abovementioned heterogeneity of the study population in terms of patient, tumor, and prior treatment factors is the most important limitation of the majority of published studies regarding 90Y-TARE and limits this series in drawing firm conclusions about treatment efficacy and follow-up.
Nevertheless, our study could be considered highly representative of real-world practice in managing unresectable or recurrent ICC and confirms the previous results achieved in our tertiary referral center. Despite the heterogeneous scenario of ICC, 90Y-TARE has been demonstrated to be safe and beneficial in increasing survival rates, especially in responders at 3 months.
These encouraging results suggest how multicentric randomized controlled trials are necessary to establish the exact role of 90Y-TARE, to compare radioembolization with other intra-arterial strategies, and to further improve the management of unresectable ICC.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study received ethics approval from the ethics committee of the University of Bologna. The Ethics Committee reference number is 193/2021/Oss/AOUBo. Informed written consent was obtained before the procedures were performed and verbal consent was obtained from the study participants for their data to be used and published in this retrospective study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work reported in this publication was funded by the Italian Ministry of Health, RC-2022-2773385 project.
